Abstract
Ciliary neurotrophic factor (CNTF) is neuroprotective against multiple pathologic conditions including metabolic impairment, but the mechanisms are still unclear. To delineate CNTF effects on brain energy homeostasis, we performed a multimodal imaging study, combining
Keywords
INTRODUCTION
The cytokine ciliary neurotrophic factor (CNTF) exhibits well-established neuroprotective effects against a large variety of pathologic conditions, including excitotoxicity and metabolic impairment.1,2 It has been tested in clinical trials for Huntington's disease, 3 amyotrophic lateral sclerosis, 4 and diseases of the retina. 5 However, the mechanisms involved in CNTF neuroprotection remain largely uncharacterized.
Ciliary neurotrophic factor is known to directly activate catabolic pathways in peripheral organs such as the liver, muscle, and adipose tissues, and to regulate food intake and energy expenditure in the hypothalamus. 6 Besides these metabolic effects, CNTF is a potent activator of astrocytes. We previously reported that astrocyte activation by CNTF alters their metabolic profile, leading to protective effects on neurons exposed to glycolytic inhibition. 7
Here, we aimed to further delineate CNTF metabolic effects on the intact brain. We performed a multimodal imaging study combining proton magnetic resonance spectroscopy ( 1 H-MRS), high-performance liquid chromatography (HPLC) analysis and brain mapping of glutamate (Glu) with Chemical Exchange Saturation Transfer (gluCEST) on rats injected with a lentiviral vector encoding CNTF.
MATERIALS AND METHODS
Injection of Lentiviral Vectors
We used self-inactivated lentiviral vectors that encode either the human CNTF gene (‘lenti-CNTF’) or the β-galactosidase gene (‘lenti-LacZ’). 8
Two-month-old male Sprague Dawley rats (Charles River, France) were injected with lentiviral vectors as described previously, 9 and analyzed between 1.5 and 3 months after injection (CNTF effects being stable for at least 6 months 8 ). To avoid bias associated with lateralization, both combinations of injections (lenti-LacZ in the left striatum and lenti-CNTF in the right striatum, or the opposite) were performed.
Housing and experiments were performed in strict accordance with the European Community regulations (Directive 2010-63/EEC) and French regulations (Code Rural R214/87-130). Experimental procedures were approved by a local ethics committee registered by the French Research Ministry (committee #44, approval #10-057).
Proton Magnetic Resonance Spectroscopy
Proton magnetic resonance spectroscopy ( 1 H-MRS) experiments were performed on a horizontal 7 T Agilent scanner (Palo Alto, CA, USA). We used a volume coil for radiofrequency transmission and a quadrature surface coil for reception.
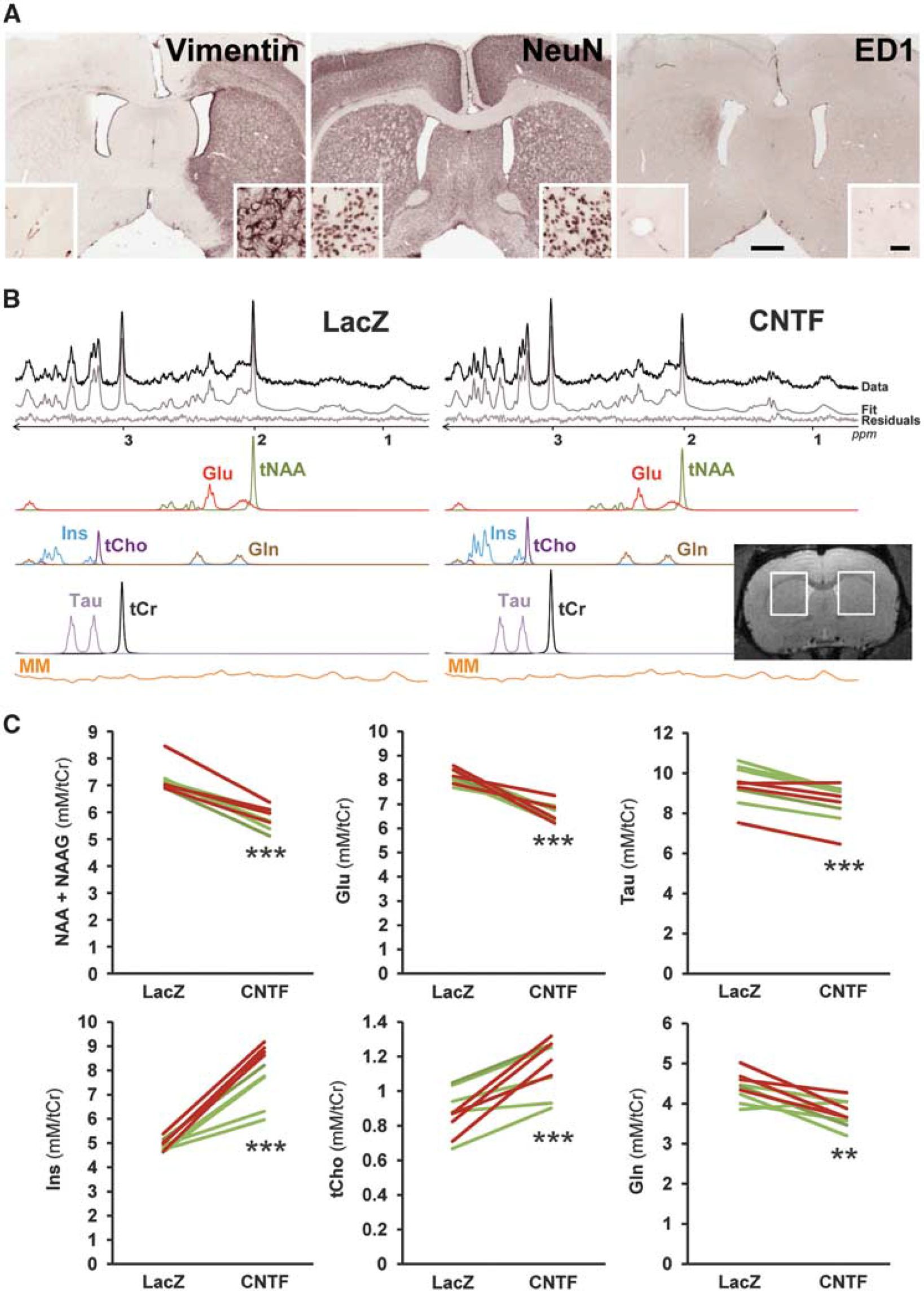
Ciliary neurotrophic factor (CNTF) remodels striatal metabolite concentrations as measured by proton magnetic resonance spectroscopy ('H-MRS). (
Metabolite concentrations were quantified with LCModel. 10 A macromolecule spectrum was acquired by metabolite nulling and included in LCModel basis set. Seven metabolites were reliably quantified (Cramér-Rao lower bounds ≥ 5%). Metabolite concentrations were normalized to total creatine, set at 8 mmol/L.
Chemical Exchange Saturation Transfer Imaging of Glutamate The gluCEST imaging was performed on a horizontal 11.7 T Bruker scanner with a volume coil for radiofrequency transmission and a quadrature surface coil for reception (Bruker, Ettlinger, Germany). The ‘Mapshim’ routine was applied in a voxel encompassing the slice of interest and a B1 map was also acquired to correct for inhomogeneities of the saturation pulse. A gluCEST image centered on the injection sites was acquired with a 2D fast spin-echo sequence preceded by a frequency-selective continuouswave saturation pulse (300 × 300 μm2 in-plane resolution, 1 mm slice thickness) with echo time/repetition time = 6/5,000 ms, 10 echoes and effective echo time = 30 ms. The saturation pulse was applied during 1 second with a 5-μT B1 amplitude. Two images were acquired with saturation pulse applied either at +3 ppm (Msat(+3 ppm), the center of CEST peak for Glu) or at a symmetrical frequency relative to bulk water (Msat(-3 ppm)). The gluCEST contrast was calculated pixel by pixel, as a percentage, by the equation: 100 X (Msat(-3 ppm) – Msat (+3 ppm))/Msat(-3 ppm).
High-Performance Liquid Chromatography Analysis Six rats were killed with a lethal dose of pentobarbital 4 months after lentiviral vector injection. Each striatum was rapidly dissected out, weighted, and frozen in isopentane. Samples were stored at −80°C until HPLC analysis coupled with UV detection of
ELISA Measurement of Human Ciliary Neurotrophic Factor
The striatum of five lenti-CNTF rats and four sham-operated rats was rapidly dissected out on ice, homogenized in PBS with 1% Triton X-100 and protease inhibitors (Roche, Basel, Switzerland). After centrifugation (20,000 g, 15 minutes, 4°C), the supernatant was collected and after fivefold dilution, analyzed by ELISA with the Quantikine ELISA Human CNTF immunoassay kit in reference to standards, according to the manufacturer's instructions (R&D Systems, Minneapolis, MN, USA). 12
Immunohistochemistry
Immunohistochemistry was performed on the brain of paraformaldehyde-perfused rats as described previously, 8 with the following antibodies: ED1/CD68 (1:500; Serotec, Raleigh, NC, USA), NeuN (1:2,000; Millipore, Billerica, MA, USA), and vimentin (1:2,000; Calbiochem, La Jolla, CA).
Statistical Analysis
Results are expressed as mean ± SEM. Paired
RESULTS
Lentiviral Vector-Mediated Expression of Ciliary Neurotrophic Factor in the Rat Striatum
We first measured the levels of human CNTF by ELISA, 5 weeks after infection with lenti-CNTF in the rat striatum. Ciliary neurotrophic factor levels were 2.39±0.53 ng/mg total protein in lenti-CNTF injected rats, while it was below 3.10-4 ng/mg total protein in sham-operated rats.
To further evidence CNTF production in the rat striatum, we performed immunostaining for vimentin, a marker of reactive astrocytes known to be induced by CNTF.8,9 Astrocytes overexpressed vimentin in a large part of the striatum injected with lenti-CNTF (28.47±4.80 mm3, Figure 1A).
We also checked that lentiviral injection or transgene expression had no detrimental effect on striatal cells. There was no detectable change in NeuN staining and no immunoreactivity for ED1/CD68, a marker for activated microglia (Figure 1A), as already reported.8,9
Ciliary Neurotrophic Factor Induces Multiple Changes in Metabolite Concentrations
Rats injected with lenti-LacZ in one striatum and lenti-CNTF in the contralateral striatum were imaged using a 7 T magnet, 1.5 to 3 months after infection. There was no detectable change in
There was a significant increase in the concentrations of two metabolites enriched in glial cells: myoinositol (Ins, +61±7%,
The Decrease in N -Acetyl-Aspartate and N -Acetyl-Aspartyl Glutamate Concentrations Is Confirmed by High-Performance Liquid Chromatography Analysis
To measure NAA concentrations by an alternative method, we performed HPLC analysis coupled with UV detection on striatal samples. This method allows the separate measurement of NAAG, which is otherwise combined with the main NAA peak on the 1 H-MRS spectrum.
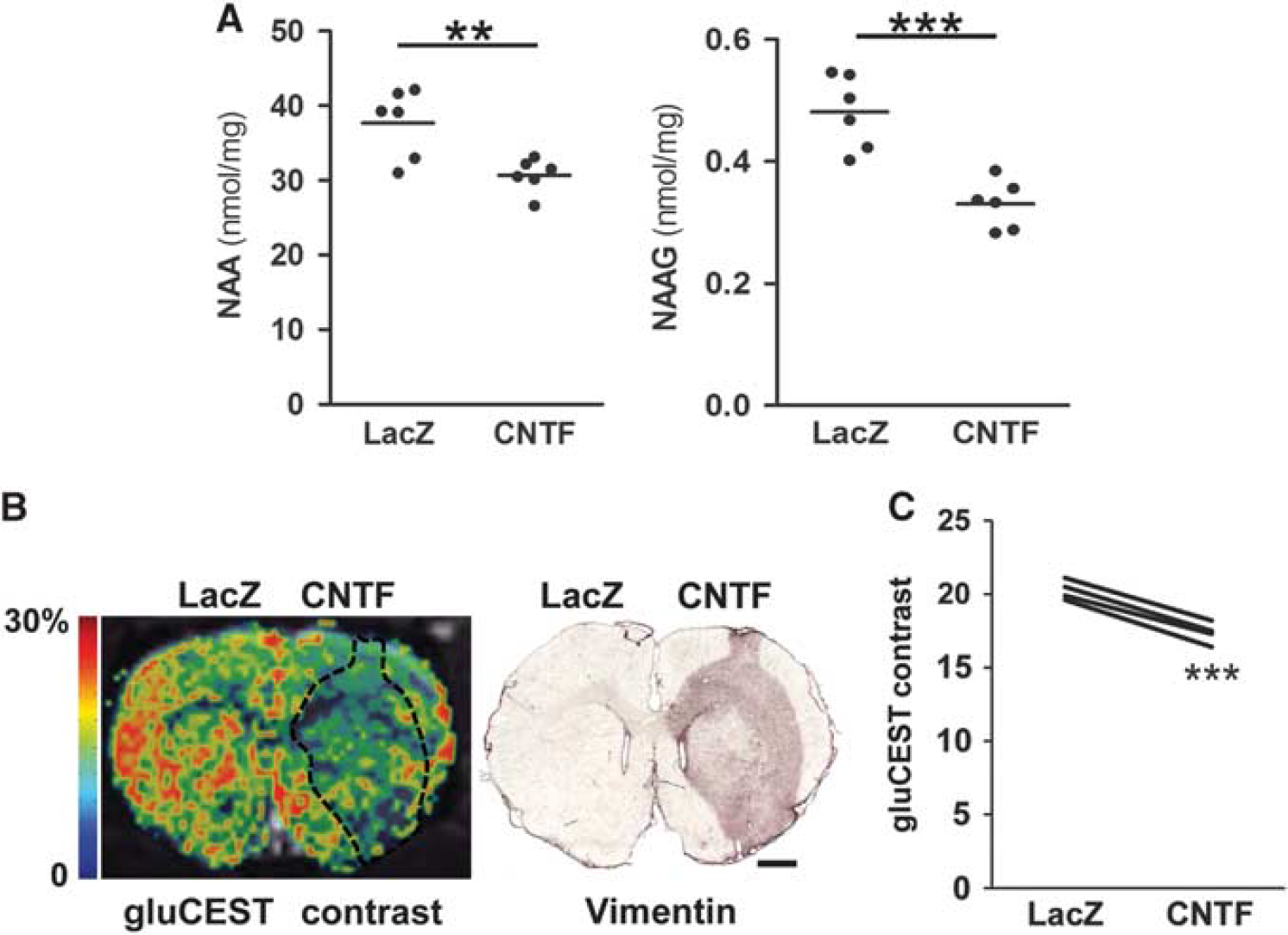
High-performance liquid chromatography (HPLC) analysis confirms decreased NAA and NAAG levels with ciliary neurotrophic factor (CNTF) and chemical exchange saturation transfer of glutamate (gluCEST) imaging reveals lower Glu levels in the striatum displaying CNTF-activated astrocytes. (
Chemical Exchange Saturation Transfer of Glutamate Imaging Reveals a Decrease in Glutamate Levels that Matches the Volume of Astrocyte Activation
To confirm the decrease in Glu levels with CNTF and to map its spatial extension
DISCUSSION
Our study shows that CNTF triggers complex changes in striatal metabolism, affecting metabolites enriched in astrocytes as well as those enriched in neurons. The significant decrease in Gln levels shows that CNTF does not globally increase the concentrations of glial metabolites, but rather modulates the relative abundance of specific brain metabolites. By alternating the side of lentiviral vector injections, we ruled out possible bias due to spectroscopic localization or lateralization. In addition,
1
H-MRS results were validated by two alternative methods. The decrease in NAA+NAAG concentrations was confirmed
Ciliary neurotrophic factor is a neurotrophic cytokine with demonstrated protective effects against multiple brain insults including excitotoxicity1,12 and metabolic impairment.2,7 Ciliary neurotrophic factor neuroprotective effects were reproduced in nonhuman primates, opening the path to clinical trials for amyotrophic lateral sclerosis, 4 diseases of the retina 5 and Huntington's disease. 3 Therefore, it is rather unexpected that CNTF reduces neuronal metabolite levels in the rat striatum. Indeed, decreased tNAA levels are classically interpreted as reflecting neuronal death or at least dysfunction in 1 H-MRS studies. 14 However, using the same strategy to overexpress CNTF in the rat striatum, we previously showed that CNTF does not alter the expression of many neuronal proteins, 8 does not modify the spontaneous electrophysiologic activity of striatal neurons, 15 and confers significant neuroprotection against excitotoxicity. 15 This was consistent with two independent studies based on lentiviral vectors,12,16 in which intrastriatalal CNTF levels were in the ng/mg protein range, as measured in the present study. Other delivery modes (adenoviral gene transfer 2 and encapsulated cells releasing CNTF 17 ) confirmed the protective effects of CNTF against striatal lesions. The levels of CNTF achieved with these different strategies are not directly comparable to our ELISA measurement. Encapsulated cells are known to produce CNTF less efficiently due to their low survival rate after implantation. 18 This may explain the limited beneficial effects observed in a phase I clinical trial for HD patients, 3 although this strategy was very efficient in primates. 19 Considering the strong evidence for CNTF neuroprotective effects in the striatum, our results show that decreased tNAA and Glu levels may occur independently of neuronal dysfunction, and may instead be associated with significant neuroprotective effects.
Overall, CNTF induces a complex reorganization of striatal metabolism. Accordingly, we previously showed that CNTF activates the AMP kinase, a master regulator of energy metabolism,
7
and that CNTF alters the metabolic profile of astrocytes.
7
Given the importance of energy homeostasis for proper neuronal function, it is tempting to speculate that such metabolic plasticity may participate in CNTF neuroprotective effects, as demonstrated in an
The molecular mechanisms mediating CNTF effects on neuronal metabolites remain to be determined; they may be direct or indirect. Indeed, both NAA and Glu metabolism involve complex interactions between neurons, astrocytes but also oligodendrocytes. 20 The facts that (1) CNTF acts primarily on astrocytes and (2) there is a strong spatial correlation between reactive astrocytes and decreased Glu levels observed by gluCEST, suggest that astrocytes may be involved in the remodeling of striatal metabolism.
Overall, we show that the neurotrophic cytokine CNTF induces significant metabolic plasticity in brain cells. Such metabolic signature would be classically interpreted as neuronal dysfunction or death in 1 H-MRS clinical studies. Instead, our results raise the intriguing possibility that it may indicate ongoing compensatory mechanisms.
AUTHOR CONTRIBUTIONS
MACS, JF, YB, LBH, MG, AB, GA, DH, JV, and CE performed experiments, MACS, JF, JV, and CE designed research, analyzed data and wrote the manuscript, EB, GB, and PH contributed reagents/materials and edited the manuscript.
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
