Abstract
In a rat embolic stroke (eMCAO) model, the effects of 100% normobaric hyperoxia (NBO) with delayed recombinant tissue plasminogen activator (tPA) administration on ischemic lesion size and safety were assessed by diffusion- and perfusion (PWI)-weighted magnetic resonance imaging. NBO or room air (Air) by a face mask was started at 30 mins posteMCAO and continued for 3.5 h. Tissue plasminogen activator or saline was started at 3 h posteMCAO. Types and location of hemorrhagic transformation were assessed at 24 h and a spectrophotometry hemoglobin assay quantified hemorrhage volume at 10 h. In NBO-treated animals the apparent diffusion coefficient/PWI mismatch persisted during NBO treatment. Relative to Air groups, NBO treatment significantly reduced 24 h infarct volumes by ~30% and ~ 15% with or without delayed tPA, respectively (
Introduction
Despite extensive research and numerous clinical trials, recombinant tissue plasminogen activator (tPA) within 3-h of stroke onset remains the only approved therapy for acute ischemic stroke (AIS). This treatment is used in only approximately 2% of AIS patients in most hospitals because of numerous contraindications to treatment and most importantly a narrow therapeutic time window (Kleindorfer et al, 2008). However, it is well recognized that early reperfusion leads to smaller ischemic lesion volumes and favorable clinical recovery in AIS patients (Christou et al, 2000; Hacke et al, 2004). It has been suggested that rigorous patient selection based on imaging criteria and endovascular approaches to reperfusion may be promising strategies to extend the treatment window (Fisher and Ratan, 2003; Smith et al, 2005). However, these approaches have the common disadvantage that they require patient transport to a suitable hospital for advanced neuroimaging before treatment.
Given tissue hypoxia's critical role in the primary and secondary events leading to cell death after cerebral ischemia (Zauner et al, 2002), several experimental studies investigated the potential benefits of oxygen therapy to improve outcome after focal cerebral ischemia (Flynn and Auer, 2002; Henninger et al, 2007; Kim et al, 2005). Although these studies indicated that treatment with 100% normobaric hyperoxia (NBO) may be an effective stroke therapy, important issues remain unaddressed. Although most AIS result from disruption of cerebral blood flow (CBF) as a consequence of either
The main objectives of this study were to assess the early initiation of NBO in conjunction with delayed (3 h) tPA therapy on
Materials and methods
Animal Preparation
All procedures used in this study were performed in accordance with and after approval by our institutional review board and all experiments were performed in a blinded, randomized manner. To alleviate pain, animals received 0.05 mg/kg subcutaneous buprenorphine immediately as well as 6 h after the end of anesthesia. Male Sprague-Dawley rats (
Study Design
The study consisted of three different experiments.
Macroscopic, Qualitative Assessment of Intracerebral Hemorrhage
In experiments 1 and 3 at 24 h after embolization, brains were removed, cut into 1.5 mm coronal sections, and ICH was assessed. Intracerebral hemorrhage, identified as blood evident at a macroscopic level, was evaluated on the unstained brain sections. Three major types of hemorrhage were identified (Figure 1): hemorrhagic infarction type 1 (HI-1, punctate petechiae within the infarcted area characterized by isolated small red marks within the tissue that did not extend through the tissue as a blood vessel would); hemorrhagic infarction type 2 (HI-2, confluent petechiae within the infarcted area, grossly visible as red speckling of an area, usually surrounded by soft infarcted tissue but without space occupying effect); parenchymal hematoma (PH, a large homogenous mass of blood within the tissue). The number of slices showing each type of hemorrhage was calculated for each animal in the experimental groups.
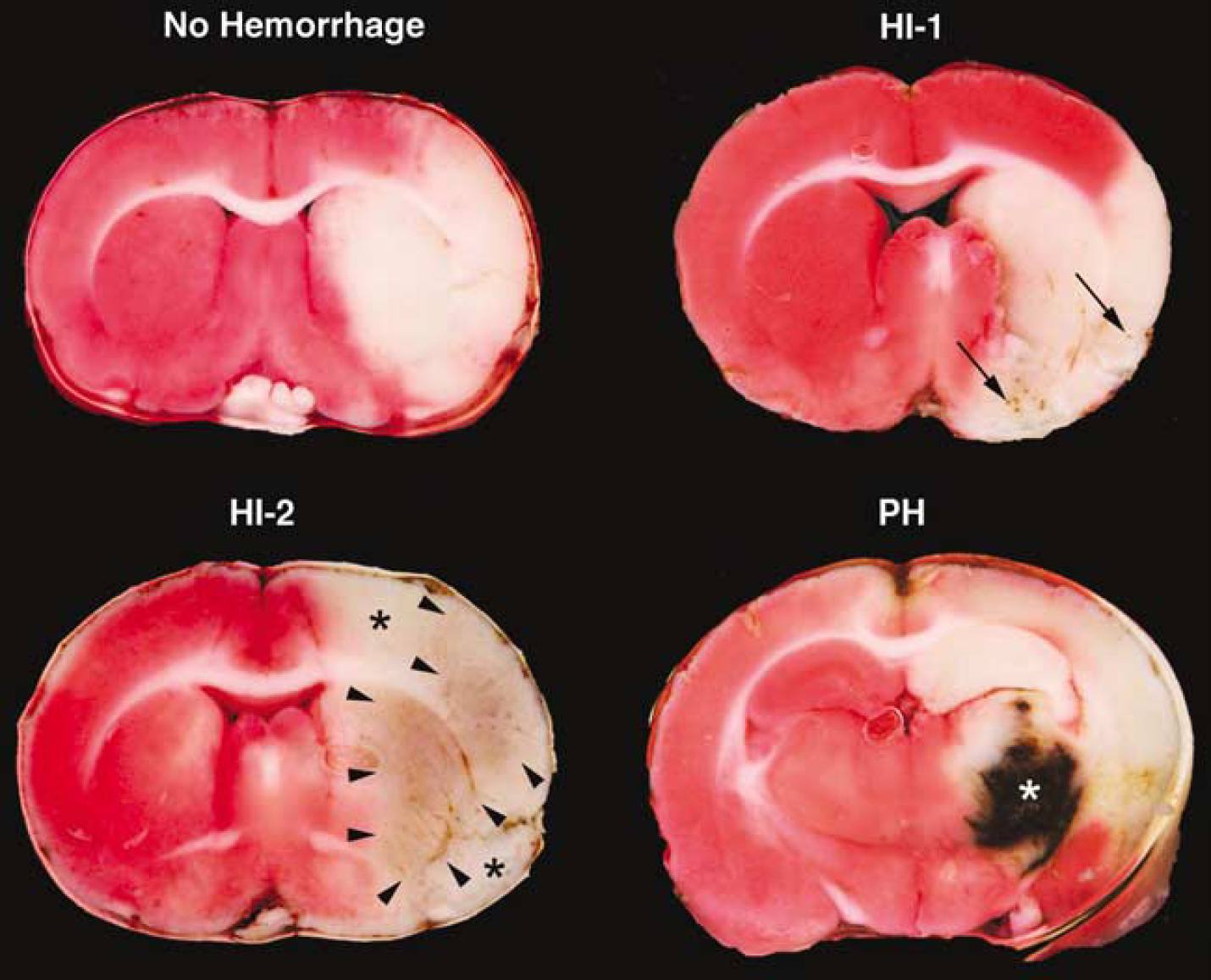
Representative TTC section indicating location and classification of intracerebral hemorrhages: hemorrhagic infarction type 1 (HI-1, black arrows denote individual petechiae), hemorrhagic infarction type 2 (HI-2, borders indicated by black arrowheads); parenchymal hematoma (PH, white asterisk, this animal died prematurely after the end of the MRI session). Less than 10% of all animals did not display any type of hemorrhage. Note that the periphery of infarcted areas typically did not display hemorrhages (black asterisks).
Assessment of Infarct Size by 2,3,5-Triphenyltetrazolium Chloride Staining
In experiment 1 after assessment of macroscopic cerebral hemorrhage, brain sections were stained with 2,3,5-triphenyltetrazolium chloride (TTC). Care was taken to cut the brains corresponding to the MRI slices. Infarct volume was measured and corrected for edema as previously described (Meng et al, 2004). Animals dying prematurely between 16 and 24h after stroke onset were included in this data analysis (Henninger et al, 2006b).
Spectrophotometry
In experiment 2, 6 h after the end of tPA infusion, animals were electively killed and transcardially perfused with saline to wash out intravascular blood. Then brains were removed and ICH volume was quantified spectrophoto-metrically (Dijkhuizen et al, 2001). Briefly, hemispheric brain tissue was homogenized in phosphate-buffered saline (total volume = 3 mL) for 30 secs, followed by 1 min sonication on ice, and 30 mins centrifugation (13,000g). Next, 1.6mL Drabkin's reagent (Sigma, St Louis, MO, USA) was added to 0.4 mL aliquots. After 15mins, optical density was measured at 540 nm with a spectrophotometer (Spectronix 3000; Milton-Roy, Rochester, NY, USA) to calculate the hemoglobin concentration. The hemorrhage volume was calculated based on the relationship between optical density and hemoglobin concentration.
Magnetic Resonance Imaging Measurements
Magnetic resonance imaging experiments were performed on a 4.7 T/40 cm horizontal magnet equipped with a Biospec Bruker console (Billerica, MA, USA), and a 20 G/cm gradient insert (ID = 12 cm, 120 ms rise time). A surface coil (ID = 2.3 cm) was used for brain imaging and an actively decoupled neck coil for perfusion labeling (Meng et al, 2004). Animals were imaged at 20 (baseline), 45, 60, 90, 120, 150, 180, 210, 240, and 270 mins posteMCAO. Three ADC maps were separately acquired with diffusion-sensitive gradients applied along the
Calculation of In Vivo Lesion Size
Images were analyzed using QuickVol II (http://www.quickvol.com/; Henninger et al, 2007). Quantitative CBF and ADC maps and their corresponding threshold-derived lesion volumes were calculated as described previously (Meng et al, 2004). The viability thresholds were 0.53 × 10−3mm2/s for ADC and 0.3 mL/g per mins for CBF (Meng et al, 2004).
Region of Interest Analysis
Four region of interests (ROIs; each 4 × 4 pixels) were manually defined at all imaging time points in the ipsilesional hemisphere on the ADC and initial CBF maps that were coregistered with the TTC results, and compared with corresponding contralesional areas. The position of these ROIs remained constant over time and described four possible tissue states: subcortical (core ROIs 1 and 2) and cortical (penumbral ROIs 3 and 4) areas with or without clear accumulation of extravascular blood on the TTC-stained brain sections, respectively. These ROIs were selected to allow for quantitatively ascertaining ADC/CBF-tissue signatures predictive of hemorrhagic transformation because previous clinical and experimental studies indicated that the duration and severity of ischemia may determine the risk for thrombolysismediated hemorrhage (Dijkhuizen et al, 2001; Kidwell et al, 2002).
Statistical Analysis
Data are presented as mean±standard deviation unless otherwise stated. Within- and between-group differences in the CBF- and ADC-lesion volumes as well as physiologic values were statistically evaluated with two-way repeated measures analysis of variance
Results
Animal Exclusion
In experiment 1, one animal in the NBO/tPA group had a subarachnoid hemorrhage and was replaced. Two additional animals from this group were excluded from the TTC -analysis of infarct size (but included in all other analyses) because of premature death after the end of the MRI session. Notably, both animals had PH-type hemorrhages in subcortical regions. One animal developed a mean arterial blood pressure increase to 170 mm Hg at ~ 190 mins after embolization (10 mins after start of tPA infusion) and a large PH was noted on subsequently obtained MR images at 210 mins (Figure 1).
Physiologic Parameters
There were no differences in physiologic parameters between corresponding groups of the three experiments (not shown) and thus data were pooled for presentation purposes. Glucose, electrolytes, and pH did not differ between time points and groups and remained within the normal physiologic range (not shown). In agreement with previously reported data, 100% NBO increased PaCO2 and PaO2 levels at 90 and 180 mins compared with Air/saline animals (Table 1). Relative to the Air groups as well as baseline, there was a modest, but statistically significant increase in mean arterial blood pressure in NBO-treated animals at 90 and 180 mins (
Physiologic parameters
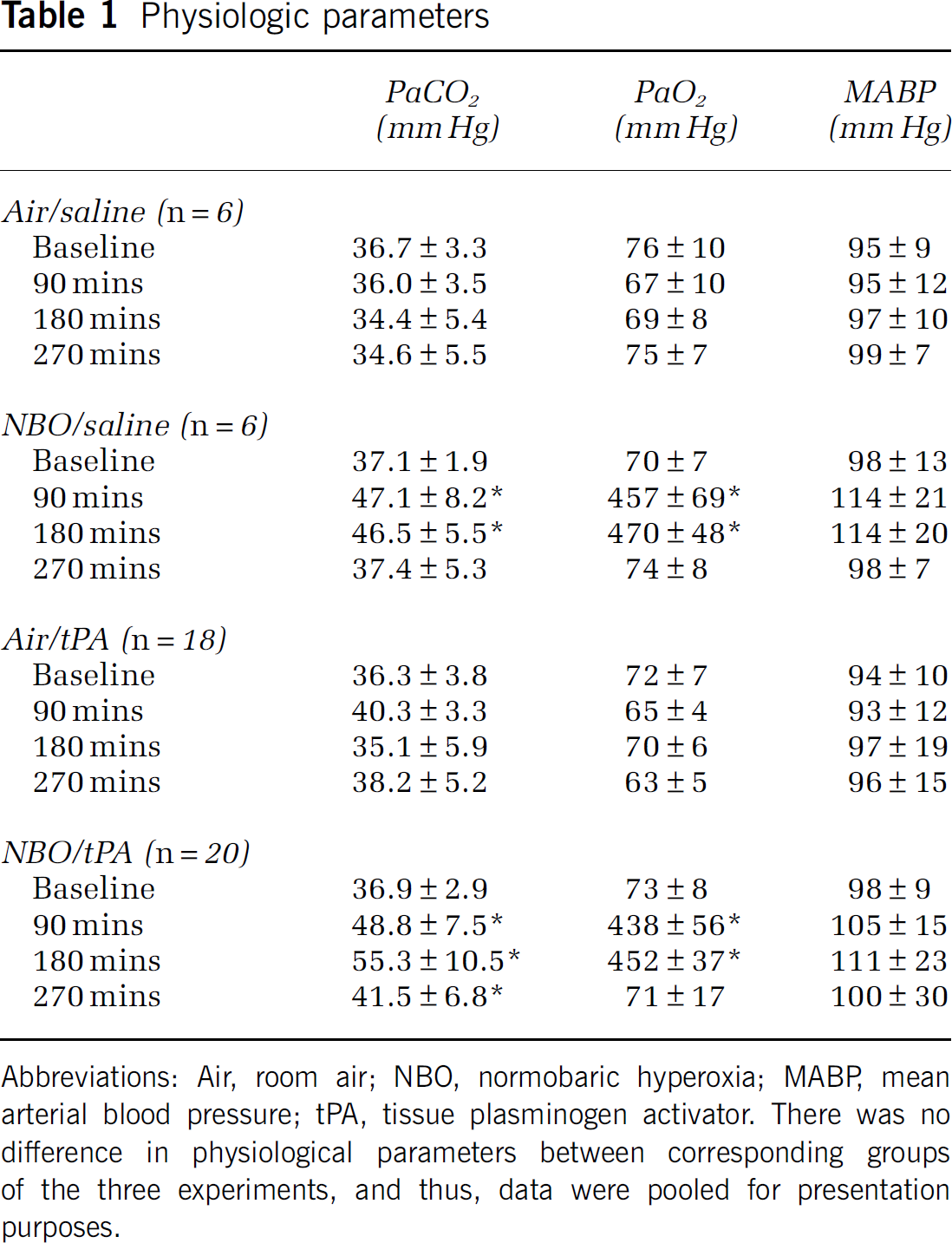
Abbreviations: Air, room air; NBO, normobaric hyperoxia; MABP mean arterial blood pressure; tPA, tissue plasminogen activator. There was no difference in physiological parameters between corresponding groups of the three experiments, and thus, data were pooled for presentation purposes.
Infarct Volume
In experiment 1 (Figures 2 and 3) corrected infarct volumes were 370±16 mm3 (Air/saline), 318±25 mm3 (NBO/saline), 340±58 mm3 (Air/tPA), and 241±37 mm3 (NBO/tPA), respectively. These were significantly smaller in both NBO groups as compared with the Air/saline group. In addition, the NBO/tPA group had a significantly smaller mean infarct volume compared with the Air/tPA groups (Figure 3). Lastly, there were no significant differences in relative swelling of the ipsilesional hemisphere between experimental groups on TTC staining (
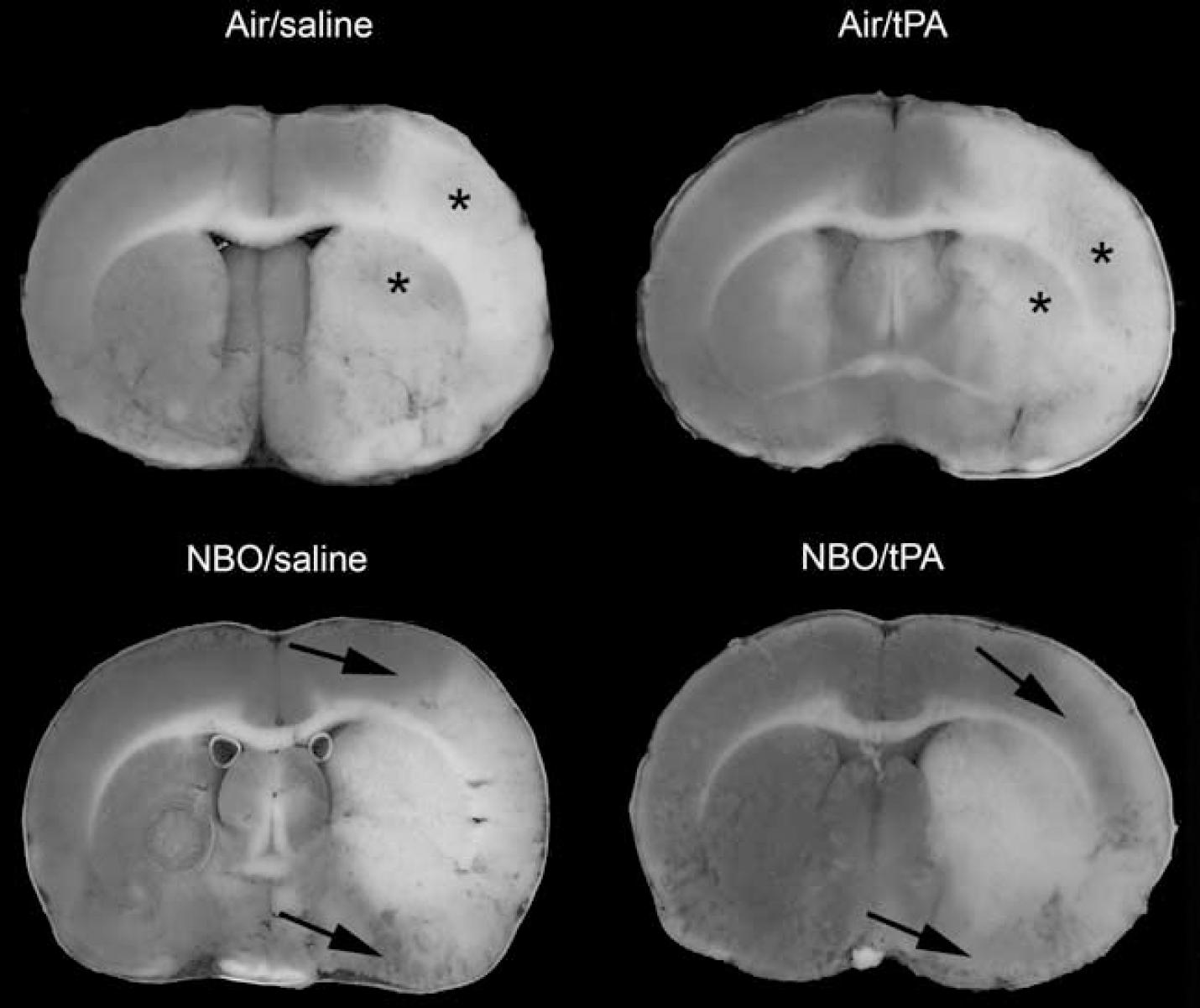
Representative TTC-stained brain slices obtained 24 h after eMCAO from animals subjected to MR-imaging. Black arrows indicate tissue salvage in the periphery of the ischemic lesion of NBO-treated animals. Asterisks denote HI-2 type hemorrhages.
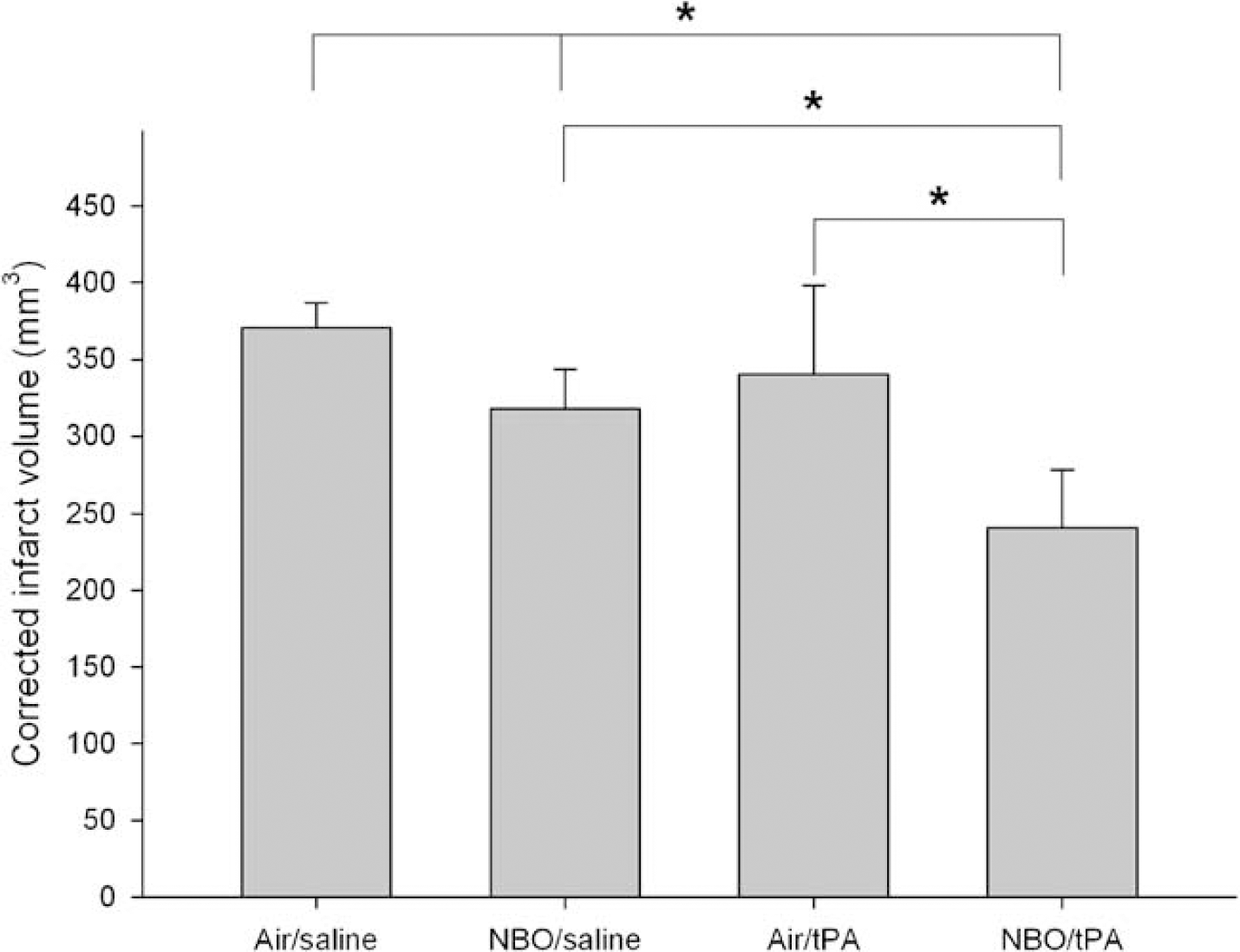
Attenuation of edema corrected infarct volumes in experiment 1 at 24 h posteMCAO (
Hemorrhagic Transformation
Figure 1 shows representative TTC stains with the defined hemorrhage types. Table 2 presents the location and frequency of the different hemorrhagic subtypes observed in experiments 1 and 3. No hemorrhages were observed outside the infarcted tissue. Hemorrhage was observed in subcortical structures (caudate putamen, thalamus, hippocampus) corresponding to the ischemic core as well as in the peripheral, cortical zones of the infarct. However, though NBO/tPA animals had a tendency towards fewer cortical versus subcortical hemorrhagic infarctions (
Qualitative hemorrhage analysis
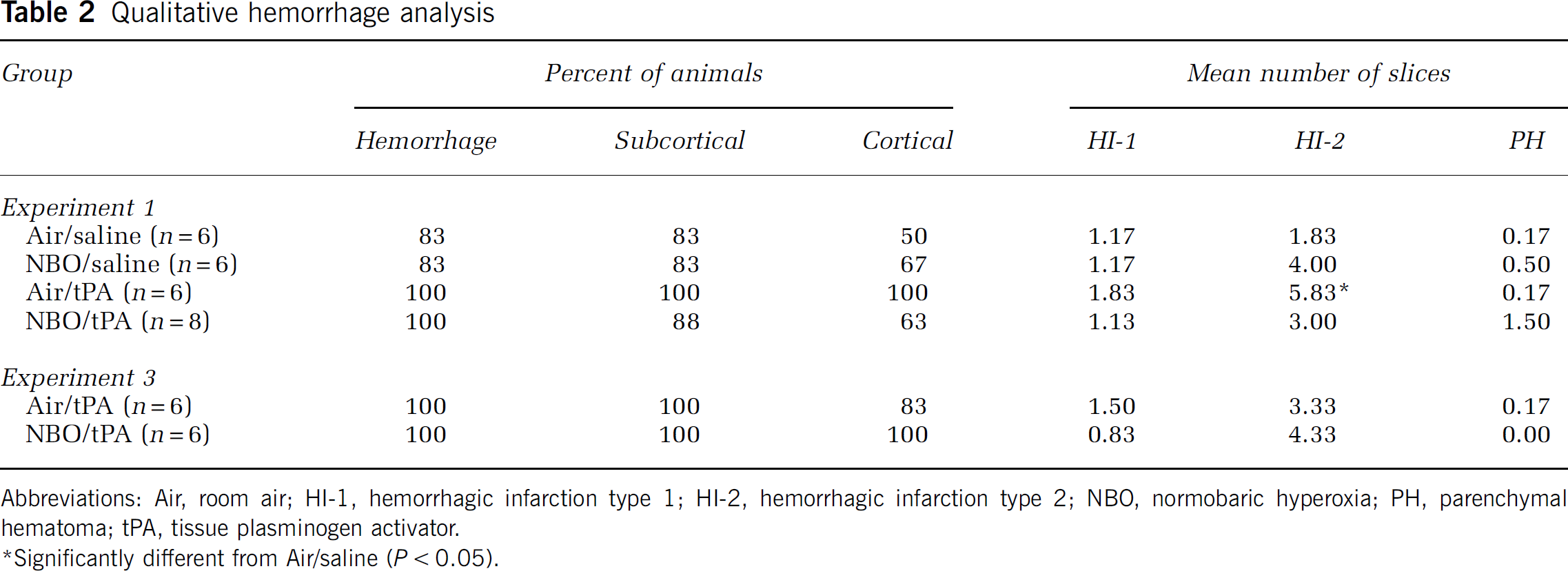
Abbreviations: Air, room air; HI-1, hemorrhagic infarction type 1; HI-2, hemorrhagic infarction type 2; NBO, normobaric hyperoxia; PH, parenchymal hematoma; tPA, tissue plasminogen activator.
Significantly different from Air/saline (
Spatiotemporal Evolution of Apparent Diffusion Coefficient- and Cerebral Blood Flow-Derived Lesion Volumes
Figure 4 summarizes the spatiotemporal evolution of threshold-derived ADC- and CBF-lesion volumes. CBF-lesion volume did not differ between groups and remained relatively constant over time before tPA infusion. In contrast to Air-treated animals, ADC-derived lesion volume stopped growing after the start of NBO and was significantly smaller than in Air/saline animals from 120 to 150 mins (
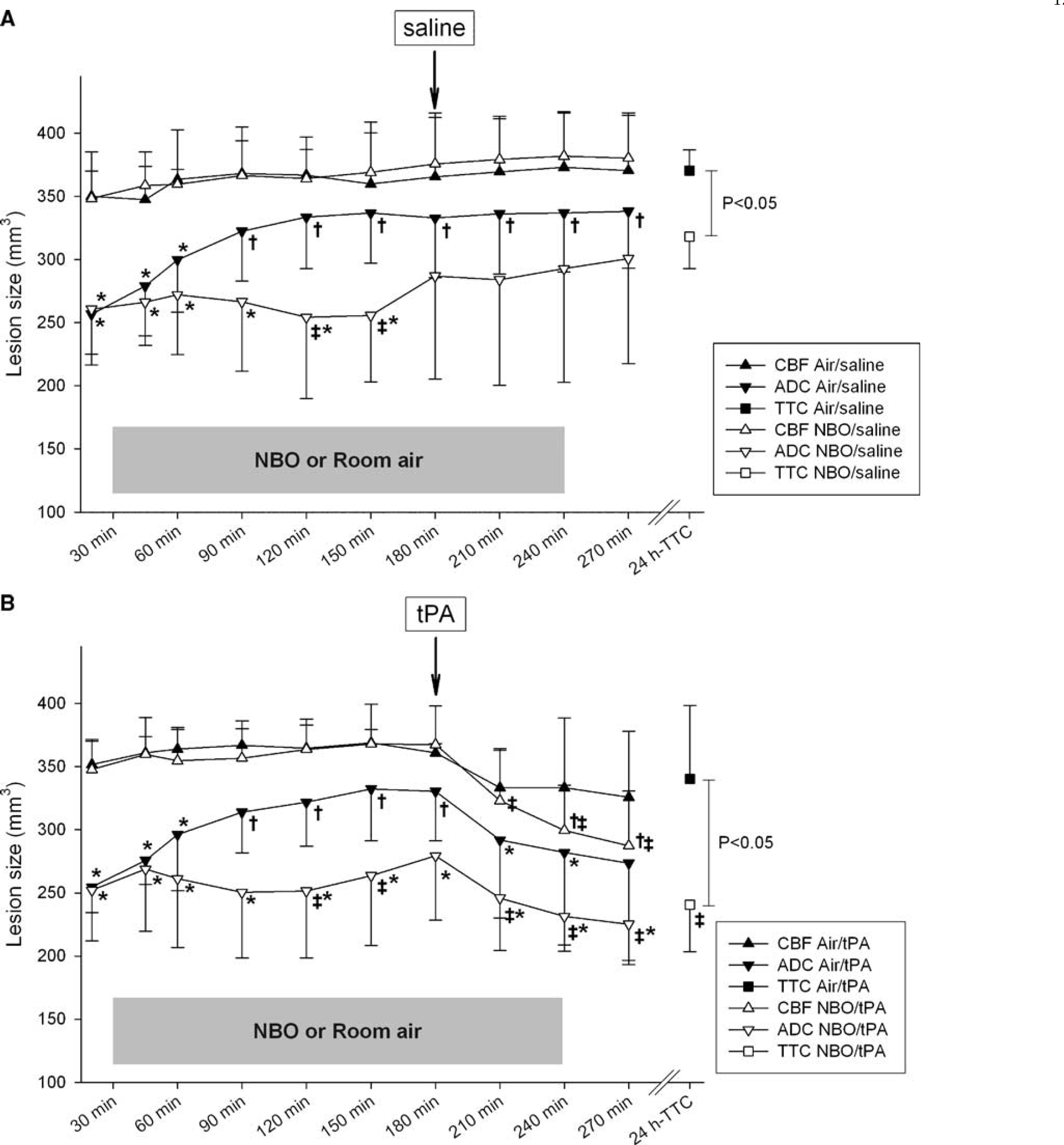
Spatiotemporal evolution of threshold-derived apparent diffusion coefficient (ADC)- and cerebral blood flow (CBF)-lesion volumes of normobaric hyperoxia (NBO)-treated and room air-treated control (control) rats. Note that data shown for group NBO/tPA includes the results from the two animals excluded from subsequent TTC analysis. Further, though statistical comparisons were made between all groups, for clarity's sake graphs
Quantitative CBF Values within Region of Interests
Region of interests characterized as types 2 and 4 (no hemorrhage) were identifiable in all animals. Region of interests 1 and 3 were present in three and five (Air/saline), five and four (NBO/saline), six and six (Air/tPA), and eight and four (NBO/tPA) animals, respectively. Apparent diffusion coefficient and CBF did not significantly differ between contralesionai ROIs over the course of the experiment (
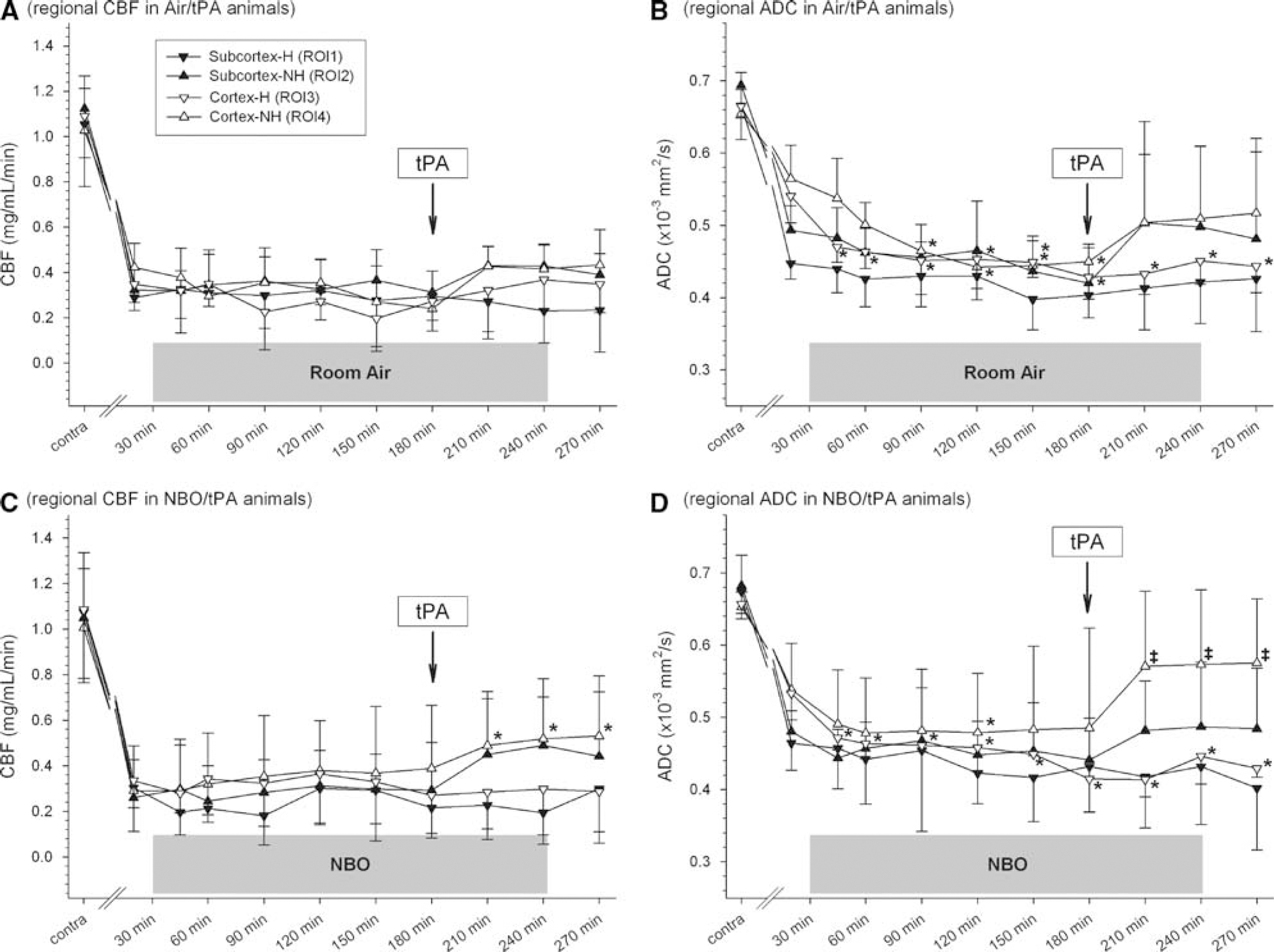
Regional cerebral blood flow (CBF;
Quantitative Apparent Diffusion Coefficient Values within Region of Interests
There were no significant differences in ADC between ROIs within groups Air/saline and NBO/saline at corresponding time points (
Discussion
Extending the time window for successful reperfusion is an important priority to increase the number of AIS patients who might be treated. Supplemental NBO is believed to have therapeutic potential for AIS treatment for several reasons: NBO therapy is readily available, easy to administer, noninvasive, can be initiated promptly after stroke onset (e.g., by paramedics), is tolerated for relatively long periods without acute sequelae, and may ‘freeze’ the penumbra based on MR criteria and potentially allow for the initiation of reperfusion at later time points (Henninger et al, 2006a; Pagano and Barazzone-Argiroffo, 2003; Singhal, 2007). Although the capability of NBO to preserve the ischemic penumbra and reduce ischemic lesion volume was shown previously, no prior study assessed the interaction between NBO and tPA (Flynn and Auer, 2002; Henninger et al, 2007; Kim et al, 2005; Singhal et al, 2005). Such information is paramount as available data indicates that NBO's efficacy is reduced if definite reperfusion cannot be achieved (Henninger et al, 2007; Kim et al, 2005; Singhal et al, 2005). In addition, there are potential safety concerns that delayed thrombolytic therapy might be associated with greater hemorrhagic side effects (Cocho et al, 2006; Dijkhuizen et al, 2001). By using various MRI techniques to evaluate the relationship between ischemic cerebral damage and ICH after combination therapy of NBO and delayed thrombolysis with tPA in a rat eMCAO model, this study enhances our knowledge about these issues. Tissue plasminogen activator was administered 3 h after eMCAO, which is beyond the previously documented time at which a significant ADC/CBF mismatch is measured in the used model (Henninger et al, 2006b). This late tPA treatment combined with early initiation of NBO was shown to reduce the extent of ischemic injury
In agreement with previous experimental studies showing reduced final lesion sizes in transient, mechanical reperfusion stroke models, NBO significantly preserved the ADC/PWI mismatch that could be salvaged by delayed tPA-mediated reperfusion (Flynn and Auer, 2002; Henninger et al, 2007; Kim et al, 2005). However, lesion volume reduction with NBO was less impressive than previously reported for the combination of tPA and mechanical reperfusion in the suture model (Henninger et al, 2007). As noted previously, the acute beneficial effects of short-term NBO alone cannot be sustained with permanent ischemia (Henninger et al, 2007; Kim et al, 2005). Hence, the finding of greater tissue protection in the suture model is not a surprising finding given the less extensive vessel occlusion as well as instantaneous and more complete reperfusion after mechanical reperfusion (Henninger et al, 2007, 2006b).
Rigorous AIS patient selection for delayed i.v. tPA treatment within the 3 to 6 h time window based on the PWI/DWI mismatch showed beneficial effects of the treatment on day 90 outcomes with an acceptable rate of ICH (Schellinger et al, 2007). Although we observed a significant reduction in the initial CBF- and ADC-lesion volumes after tPA administration in Air animals, this effect was not sustained at 24 h. This lack of a treatment effect with delayed tPA alone in our model may be explained by secondary ADC-declines with subsequent tissue infarction, as previously described and suggests that delayed tPA alone may be ineffective when the mismatch volume is small or modest (Kidwell et al, 2000; Li et al, 2000).
The development of ICH after using i.v. tPA remains the most important complication of this therapy in clinical practice. Recent studies do not suggest that the risk for symptomatic ICH appears to increase when treatment is initiated beyond 3 h from stroke onset in selected patients (Davis et al, 2008; Schellinger et al, 2007). Although free oxygen radicals have been shown to be implicated in brain injury and blood-brain barrier dysfunction after cerebral ischemia/reperfusion (Heo et al, 2005) several experimental studies did not show enhanced oxidative stress or its surrogate markers with oxygen therapy (Agardh et al, 1991; Henninger et al, 2006a, 2007; Kim et al, 2005; Liu et al, 2006; Schabitz et al, 2004; Singhal et al, 2002b; Sunami et al, 2000). In concordance with these prior studies, we did not find evidence of increased ICH or swelling after NBO therapy. In fact we observed a trend towards reduced amounts of hemorrhage both qualitatively and quantitatively with NBO and delayed tPA as compared with delayed tPA and Air. Further validation studies are clearly needed to assess NBO's potential to decrease tPA-associated hemorrhage. Regardless, extensive evaluation of hemorrhagic risk was prompted by the two fatal ICH events that occurred in experiment 1 with delayed tPA and NBO, one of which was related to a substantial rise in blood pressure shortly after initiation of tPA treatment. The extensive analyses of ICH risk appear to assuage concerns about the potential of NBO and delayed tPA to increase the rate of intracerebral bleeding.
In conclusion, this experimental study provides initial evidence that NBO can be safely combined with delayed i.v. tPA to extend the potential therapeutic time window for such treatment. This observation could have clinical relevance. It suggests that NBO could potentially be initiated prehospital or at arrival to extend and enhance penumbral survival in ischemic stroke patients. Penumbral imaging should then be performed to show the presence and extent of the ischemic penumbra and in patients showing a reasonable amount of penumbra delayed tPA therapy might then be useful. This approach will have to be tested in carefully designed and implemented clinical trials but NBO as a possible penumbral extender has substantial appeal because of its simplicity, wide availability and reasonable safety profile.
Footnotes
Acknowledgements
Funding sources not involved in the writing of the article or in the decision to submit the article for publication. Tissue plasminogen activator was a kind gift of Genentech Inc. The authors thank Eng Lo, Yoshihiro Murata, and Meghan Shea for their help with these experiments.
The authors report no conflicts of interest.
