Abstract
Cerebral ischemia results in damage to neuronal circuits and lasting impairment in function. We have previously reported that stimulation of
INTRODUCTION
Cerebral ischemia damages neuronal circuits and results in lasting impairments, 1 with survivors exhibiting varied levels of recovery. To date, drug interventions have primarily focused on minimizing cell death. These therapies, however, have not translated into clinical practice, leaving neurorehabilitation as the only practical option to enhance brain remapping and improve lost functions. Transfer of neuronal activity from damaged to functionally related and preserved tissue within the brain can occur on both sides of the brain after unilateral stroke.2–4 The processes involved in remapping or rewiring the brain after injury include neurogenesis, sprouting of new connections, and synaptogenesis. 4 Imaging of nerve cells within tissue surrounding the stroke (peri-infarct region) has showed extensive synaptic remodeling after brain injury and stroke.3,5 Consistent with these findings we have recently shown that pharmacological interventions targeting the peri-infarct cortex after a stroke hold great promise and afford substantial improvements in functional outcomes.6,7 Importantly, these studies highlight not only a need to increase neuronal activity, but also a need for trophic factor support via the release of brain-derived neurotrophic factor (BDNF). 6 Further, BDNF has been highlighted as a key regulator of rehabilitation-induced recovery after stroke. 8
A number of studies have now provided compelling evidence that neural plasticity and cognitive function can be enhanced by both BDNF and positive AMPA receptor modulation.9,10 The ampakine, CX1837, has attracted considerable attention, as it is a positive allosteric modulator of AMPA receptors that under homeostatic conditions stimulates and increases BDNF mRNA and protein levels.11,12 We have recently showed that CX1837 enhances poststroke functional recovery in young animals.
6
We have also shown that CX1837-mediated functional recovery occurred in a BDNF/TrkB-dependent manner, a finding that has been confirmed by Massa
As aging alters the responsiveness of the cortex, and how tropic factors may act, it is likely that cotreatments will afford greater recovery after stroke in aged populations. As stroke preferentially affects the aged, we asked the question, whether treatment with either BDNF or CX1837 be equally effective in aged mice as they are in young, or whether combined treatment be required. As multiple signaling pathways could potentially be involved in regulating motor recovery after stroke, we also aimed to explore which signaling pathways are being activated after stroke in following CX1837, BDNF, and a combination of the two together.
MATERIALS AND METHODS
Animals and Surgical Procedures
All procedures described in this study were performed in accordance with guidelines on the care and use of laboratory animals set out by the University of Otago, Animal Research Committee and the Guide for Care and Use of Laboratory Animals (NIH Publication No. 85–23, 1996). Focal stroke was induced by photothrombosis in aged (22 to 24 months old) male C57BL/6 mice weighing ~ 30 to 33 g as previously described.6,7,17 Under isoflurane anesthesia (2% to 2.5% in O2) mice were placed in a stereotactic apparatus, the skull exposed through a midline incision, cleared of connective tissue and dried. A cold light source (KL1500 LCD, Zeiss, Auckland, New Zealand) attached to a x40 objective giving a 2-mm diameter illumination was positioned 1.5 mm lateral from bregma and 0.2 mL of Rose Bengal solution (Sigma-Aldrich, Auckland, New Zealand; 10 g/L in normal saline, intraperitoneally) was administered. After 5 minutes the brain was illuminated through the intact skull for 15 minutes.
Mice were housed under a 12-hour light/dark cycle with
A calmodulin kinase II
In Vivo Drug Dosing
CX1837 (1 mg/kg) was dissolved in 30% hydroxypropyl
A hyaluronan/heparan sulfate proteoglycan biopolymer hydrogel (HyStem-C, BioTime, Alameda, CA, USA) was used to locally deliver recombinant human BDNF (100
The pan-Akt inhibitor, GSK-690693, was dissolved in dimethyl sulfoxide and then diluted 1:1 in 0.9% saline. GSK-960693-filled ALZET-1002 pumps (BioScientific, NSW, Australia) were implanted subcutaneously between the scapula 5 days after stroke and replaced every 2 weeks to maintain steady-state plasma levels of GSK-690693. The concentration in one minipump, 10 mmol/L, delivers ~ 400
Behavioral Assessment
Animals were tested once on both the grid-walking and cylinder tasks, 1 week before surgery to establish baseline performance levels. For all of the studies, animals were tested on weeks 1, 2, 4, and 6 after stroke at approximately the same time each day at the end of their dark cycle. Behaviors of the animals were scored by observers who were masked to the treatment group in the study as previously described.6,7,17
Brain-Derived Neurotrophic Factor Enzyme-Linked Immunosorbent Assay
Tissue was collected from around the stroke site from stroke+vehicle and stroke+CX1837 (1 mg/kg) treated groups 7 days after stroke. Cortical tissue collected included the stroke core plus 1 mm of tissue that surrounded the infarct core, and flash frozen on dry ice. To measure the release of BDNF from the hydrogel, tissue was collected at 7, 14, 28, and 42 days after stroke from both vehicle and BDNF impregnated hydrogel.
Equal volumes of tissue were homogenized in 100
Quantification of Phosphorylated Proteins using the Bio-Plex Suspension Array System
The Bio-Plex suspension array system (Bio-Rad Laboratories) enables simultaneous quantification of multiple phosphorylated and total proteins in each well of a 96-well plate. In this system, an antibody directed against the desired target protein is covalently coupled to internally dyed beads with a fluorescence whose wavelength is specific for each target protein. The bead-coupled antibodies are allowed to react with lysate samples containing the target proteins. Then biotinylated detection antibodies specific for different epitopes of the proteins are added to the reaction, followed by an addition of streptavidin-phycoerythrin. A dual-laser, flow-based microplate reader system (Bio-Plex 200, Bio-Rad) detects the internal fluorescence of the individual dyed beads and the signal intensity on the bead surface. The relative abundance of the each target protein is reported as the ratio of fluorescence among the wells. In the present study, the abundance of phospho-mitogen activated protein kinase kinase (MEK) 1, phospho-extracellular signal-regulated kinase (ERK)1/2, phospho-CREB, phospho-c-Jun N-terminal kinase, phospho-p38 mitogen activated protein kinases, phospho-Akt, were simultaneously quantified in stroke samples and quantified against standards supplied by the manufacturer. The relative abundance of the target protein was calculated as fluorescence intensity – background fluorescence.
Peri-infarct tissue was collected from stroke+vehicle treated (control,
Statistical Analysis
All data are expressed as mean ± s.e.m. For behavioral testing, differences between treatment groups were analyzed using two-way ANOVA with repeated measures and Newman–Keuls' multiple pair-wise comparisons for
RESULTS
CX1837 Improves Motor Recovery after Stroke in Aged
We have previously shown that boosting AMPA receptor-mediated BDNF release after stroke results in an improvement in motor function in young mice. 6 As the majority of strokes occur in aged mice, we wanted to assess whether the BDNF-inducing ampakines, CX1837, was also effective in aged mice.
Mice were given a stroke that affects both primary and sensory motor cortex and received behavioral testing for 6 weeks after stroke. Stroke causes mice to exhibit limb use deficits for at least 6 weeks after the infarct, with increased foot faults on a grid-walk task, and an increased use of the ipsilateral forelimb to the stroke in spontaneous use on the cylinder task (Figure 1).
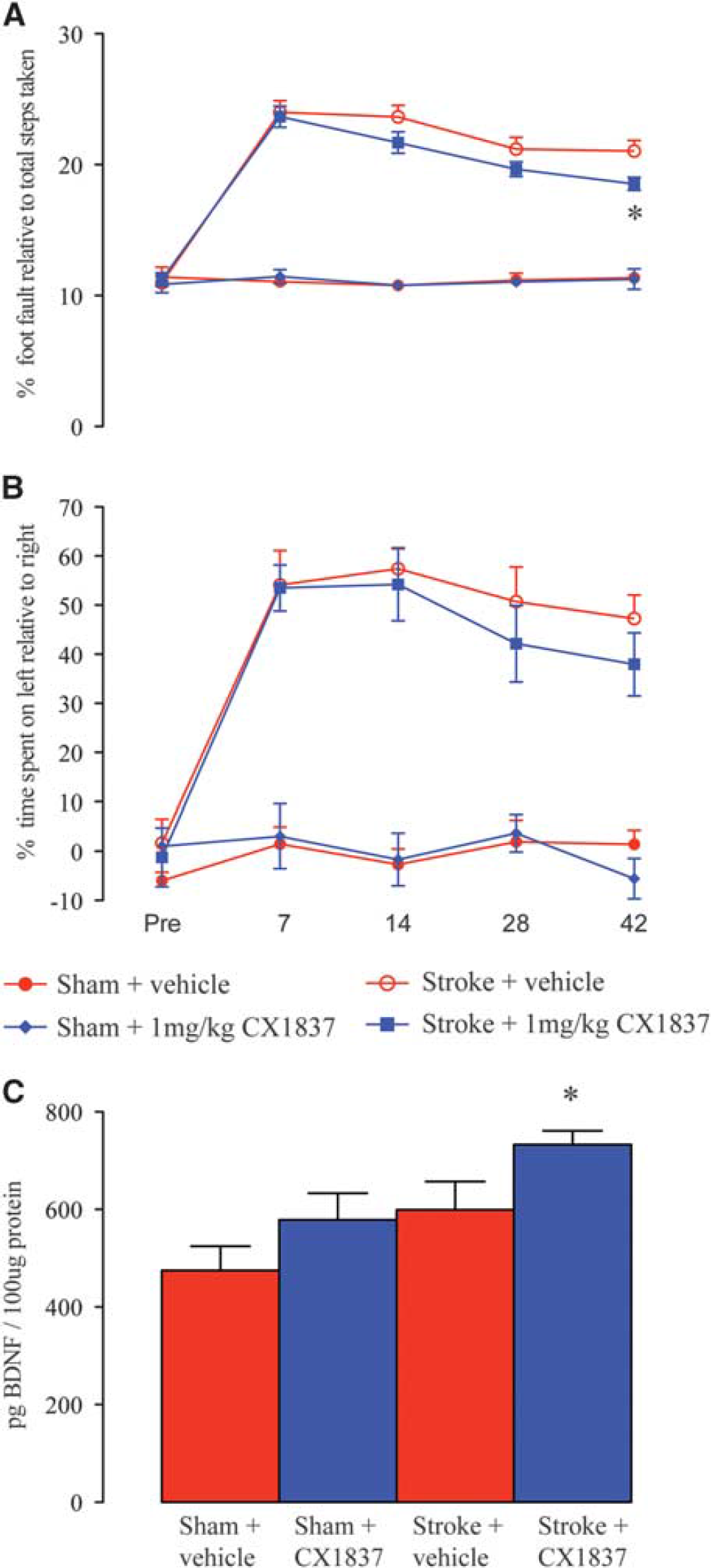
Behavioral recovery in the presence of the high-impact ampakine, CX1837. Behavioral recovery after stroke was assessed on grid-walking (
To test the effect of AMPAR-mediated BDNF release on motor functional recovery after stroke, we administered CX1837 starting 5 days after stroke in 2-year-old mice. CX1837 crosses the blood–brain barrier when given systemically, increases excitatory signaling and stimulates an increased release in BDNF.6,11,12 CX1837 (1 mg/kg, intraperitoneally twice daily) promotes a mild gain of function in the impaired forelimb of the grid-walking task (Figure 1A;
To test that 1 mg/kg CX1837, similar to what was observed in young,
6
stimulated an elevation in BDNF, peri-infarct tissue samples were taken to measure BDNF levels. Stroke induced a small yet nonsignificant increase in BDNF levels in the peri-infarct cortex compared with control cortex at 7 days after stroke (
Local Hydrogel Delivery of Brain-Derived Neurotrophic Factor Improves Motor Recovery after Stroke in Aged Mice
We have previously reported that the BDNF-inducing ampakine, CX1837, may promote motor recovery after stroke via the enhancement of BDNF expression. 6 Further, others have reported that BDNF can enhance motor recovery after stroke, although this is dependent on BDNF reaching a threshold level.8,19 Based on our observation that CX1837 may not be stimulating BDNF levels to reach a certain threshold, we aim to test whether local delivery of BDNF via hydrogel infusion into the stroke cavity could enhance functional recovery.
We and others have previously shown that small and large proteins as well as the BDNF receptor decoy, TrkB-Fc, can be released from hydrogels over weeks and influence recovery of motor functions after stroke.6,17,18 However, to confirm that the BDNF (100
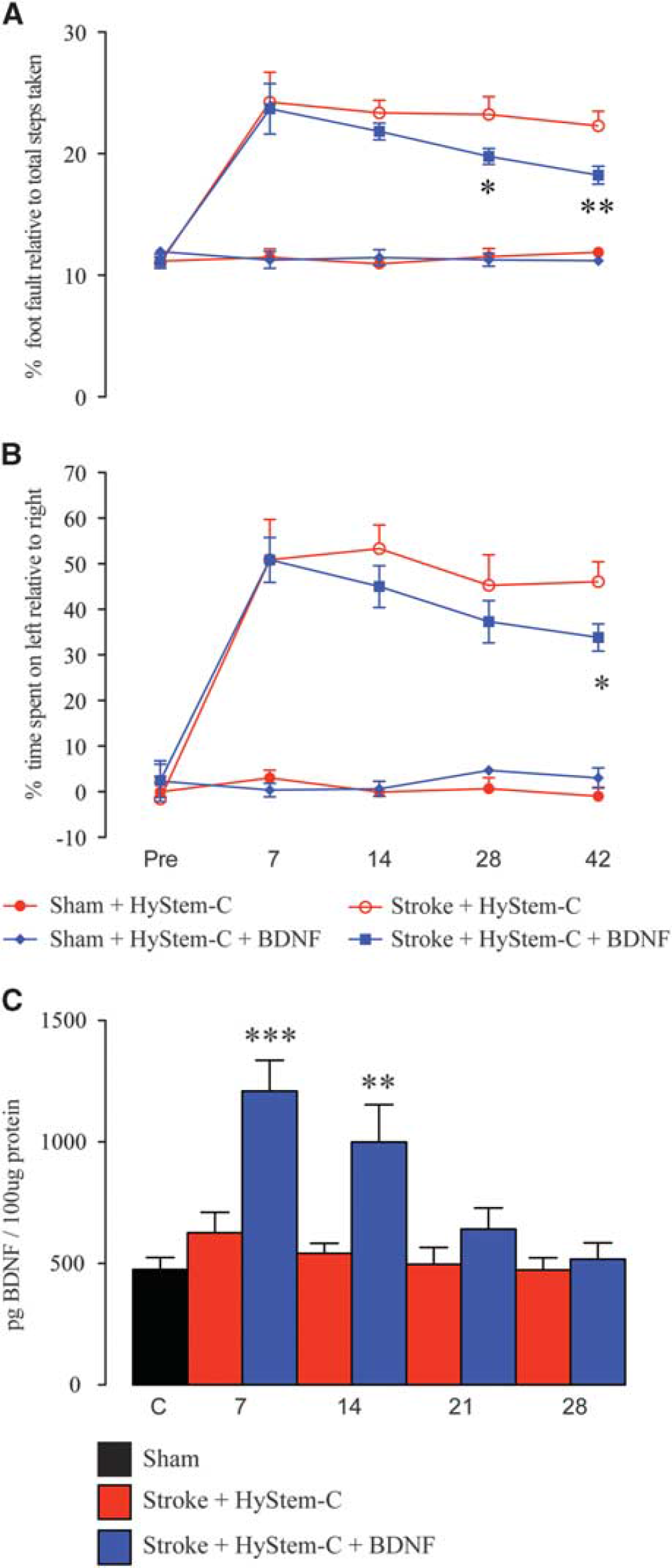
Behavioral recovery in the presence of local hydrogel delivery of brain-derived neurotrophic factor (BDNF). Behavioral recovery after stroke was assessed on grid-walking (
To test the effect of local hydrogel release of BDNF on motor functional recovery after stroke, we infused either BDNF or vehicle impregnated hydrogel into the stroke cavity from 5 days after stroke. Stroke+vehicle impregnated hydrogel animals show marked impairments for at least 6 weeks after stroke on both the grid-walking and cylinder tasks (Figures 2A and 2B, respectively). Animals that received the BDNF impregnated hydrogel showed a mild gain of function in the impaired forelimb of the grid-walking task at weeks 4 and 6 after stroke (Figure 2A;
Combined Hydrogel Delivery of Brain-Derived Neurotrophic Factor and Systemic Administration of CX1837 Markedly Improves Motor Recovery after Stroke in Aged Mice
Previous studies have shown that BDNF levels need to reach a certain threshold to see significant functional recovery in rats. 19 Based on the findings in Figure 1, it is likely that BDNF levels did not reach this threshold or that this threshold is shifted in the aged mice. Further, data presented in Figure 2 indicate that in addition to BDNF being released into and elevated in peri-infarct tissues, an activity component may also be required to enhance recovery. Therefore, to test this we have combined both therapies: local BDNF released from a biopolymer hydrogel and systemic administration of CX1837.
Mice that received combined treatment showed marked improvements on the grid-walking task from week 2 after stroke (Figure 3A;
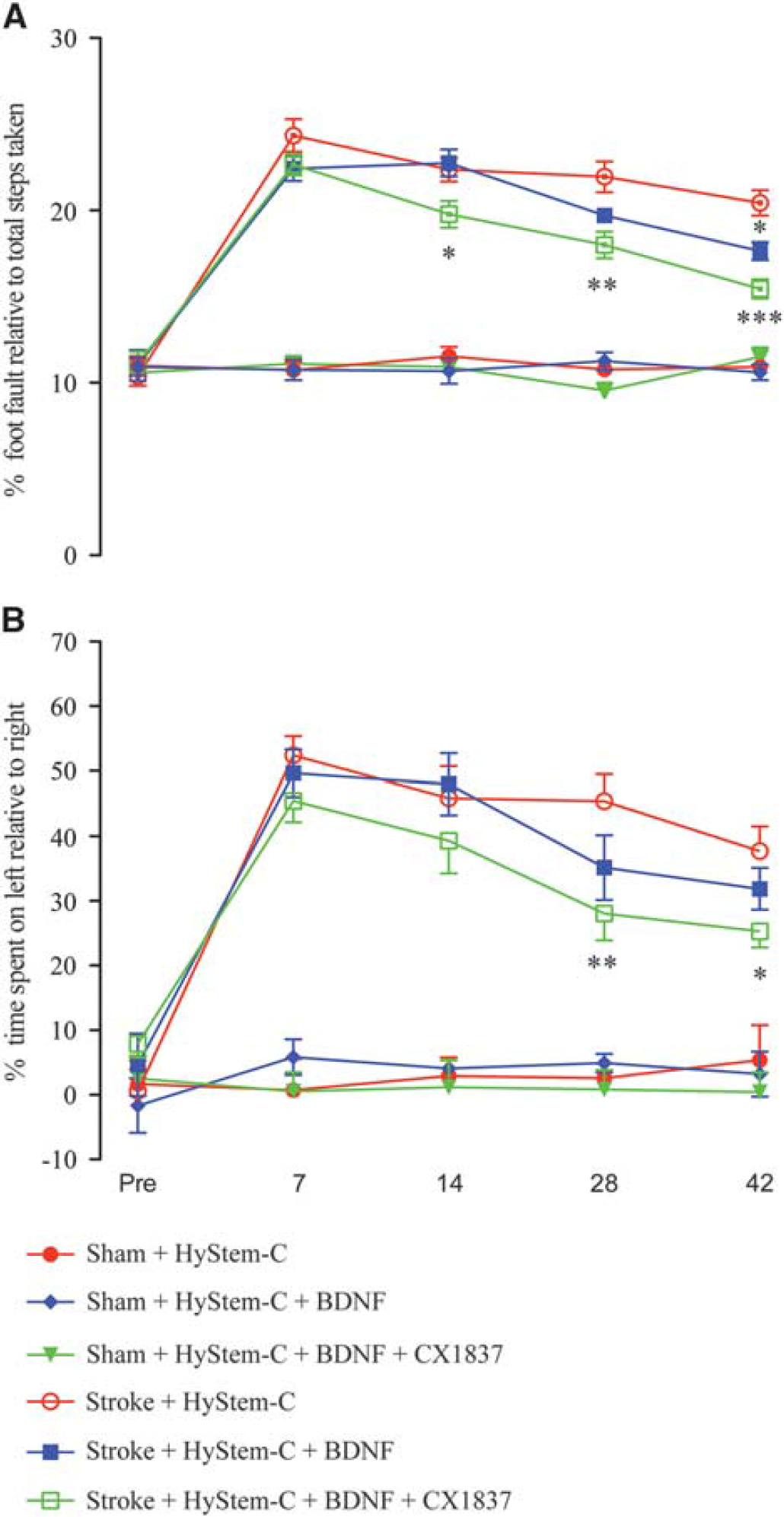
Behavioral recovery in the presence of both local hydrogel delivery of brain-derived neurotrophic factor (BDNF) and CX1837. Behavioral recovery after stroke was assessed on grid-walking (
Combined Hydrogel Delivery of Brain-Derived Neurotrophic Factor and Systemic Administration of CX1837 Alters Intracellular Signaling Proteins
Binding of BDNF to the tropomyosin receptor kinase receptor has been shown to activate and phosphorylate multiple downstream signaling pathways. To help understand which signaling pathways could be involved in the functional recovery associated with the combined CX1837 administration and BDNF biopolymer delivery, peri-infarct tissue was collected and assessed by Bio-Plex suspension array system for phospho-specific changes in signaling pathways.
Assessment of p-p38 MAPK (Figure 4A) and p-JNK (Figure 4B) activity levels showed no significant differences across any of the treatment groups (
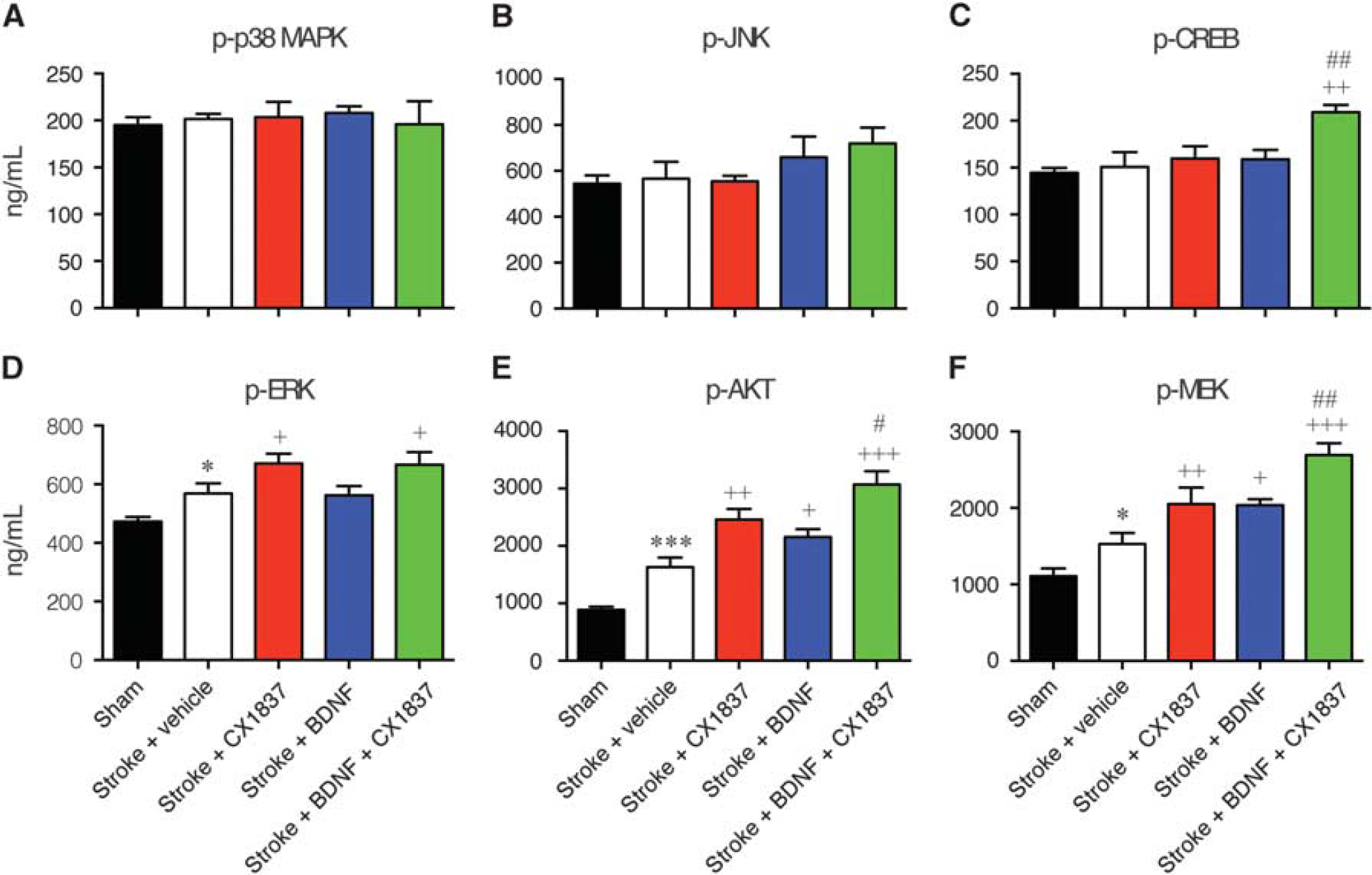
Local hydrogel delivery of brain-derived neurotrophic factor (BDNF) and CX1837 alters intracellular signaling pathways. Changes in intracellular signaling pathways were assessed in peri-infarct cortex from sham (black bars), stroke+vehicle (white bars), stroke+CX1837 (red bars), stroke+BDNF (blue bars), and stroke+CX1837+BDNF (green bars). The peri-infarct cortex was isolated 2 weeks after stroke (9 days after starting treatment) to assess changes in p-p38 MAPK (
Neuron-Specific Deletion of CREB Partially Blocks the Combined Brain-Derived Neurotrophic Factor and CX1837-Induced Improvements in Motor Recovery after Stroke
Brain-derived neurotrophic factor–TrkB activation can promote neuronal synaptic activity via the activation of CREB, a key element in the formation of long-term memory and learning. 20 Increased BDNF levels are associated with enhanced CREB activity. 21 Combined treatment with CX1837 and local hydrogel delivery of BDNF resulted in a phosphorylation of CREB. To test the hypothesis that increased CREB activation contributes to improved recovery of motor function, we bred mice where we had selectively deleted CREB from forebrain neurons, CKC-CREBloxP/loxP mice.
Wild-type littermate controls, CKC-CREB−/–, that received combined (local BDNF released from a biopolymer hydrogel and systemic administration of CX1837) treatment showed marked improvements on the grid-walking task from week 2 after stroke (Figure 5A;
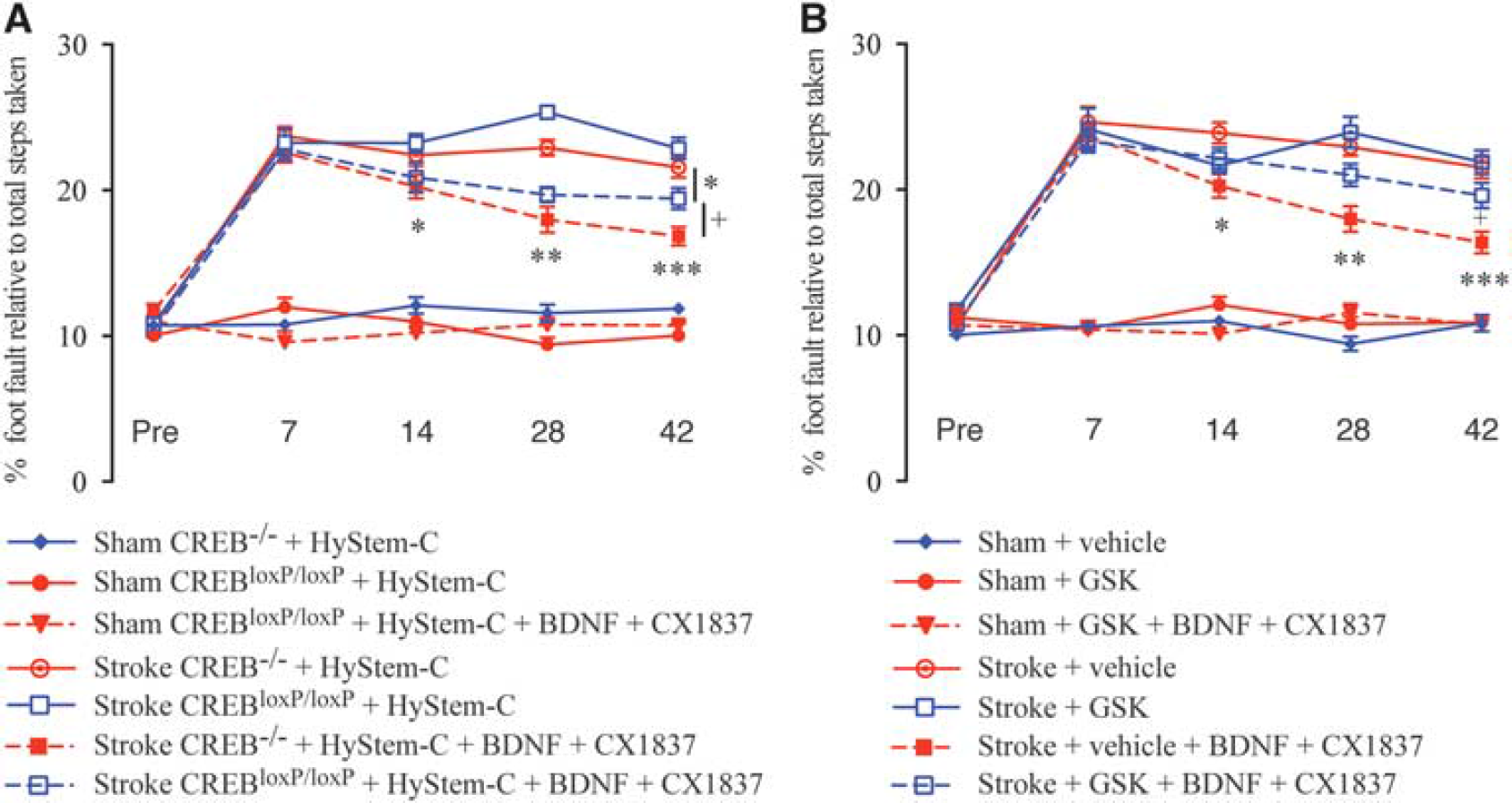
Blocking CREB or AKT signaling blocks poststroke functional recovery after local hydrogel delivery of brain-derived neurotrophic factor (BDNF) and administration of CX1837. Assessment of potential neuronal signaling pathways associated with combined CX1837 and BDNF-induced behavioral recovery after stroke was assessed in CKC-CREBloxP/loxP mice (mice where CREB has been deleted from neurons;
Systemic Administration of the pan-AKT Inhibitor, GSK-690693, Blocks the Combined Brain-Derived Neurotrophic Factor and CX1837-Induced Improvements in Motor Recovery after Stroke in Aged Mice
Significant work has been undertaken and shown that AKT signaling plays a major role in multiple physiologic processes. Activation and phosphorylation of PI3K/AKT signaling promotes neurite initiation, outgrowth, and growth cone stability,22–24 as well as contributing to growth and branching of dendrites. 25 In addition, exercise has been shown to facilitate improvements in cognitive function via phosphorylation of AKT and CREB signaling and BNDF expression.26–28 Based on these data, we hypothesized that blocking the combined BDNF and CX1837-induced phosphorylation of AKT using the pan-AKT inhibitor, GSK-690693, would impair the recovery trajectory.
Mice that received combined (local BDNF released from a biopolymer hydrogel and systemic administration of CX1837) treatment showed marked improvements on the grid-walking task from week 2 after stroke (Figure 5B;
DISCUSSION
Although nearly 80% of strokes occur in individuals aged 65 and over, the majority of preclinical studies highlighting improved functional recovery after stroke have been performed using young adult animals. This is a significant issue as it relates to the development of translational therapies; with numerous studies having identified that recovery mechanisms in aged are significantly different to those of younger animals.18,29,30 Despite, the potential implications concerning successful translation, there have been surprisingly few studies that evaluate the efficacy of pharmaceutical interventions in aged animals, particularly in the context of stroke recovery. As we had previously shown the benefits of CX1837 in younger animals we wished to determine whether the compound has the same globally beneficial effects in aged (24 months old) mice.
We have previously shown that CX1837 can markedly ameliorate functional deficits in younger animals in a BDNF/TrkB-dependent manner. 6 Extending on this work, in the current study we have identified that systemic delivery of CX1837 also enhances functional recovery in aged animals. Importantly, however, the magnitude of the effect was considerably less than what we had previously observed in younger animals (49% gain of function in young versus 26% gain of function in aged at 6 weeks 6 ). Consistent, with our observations of moderate behavioral improvement we observed that BDNF levels in CX1837-treated aged mice were also not as high as previously reported in younger animals. This dynamic relationship between BDNF and recovery has been previously identified by other authors to be critical, 8 with BDNF levels not only need to be increased, but also need to reach a critical threshold in order for recovery to be evident. 19
As the more modest levels of recovery observed in aged animals could have been attributed to age-related deficits in BDNF production, we next investigated whether localized delivery of BDNF could enhance the effects of CX1837. Our results here indicated that local hydrogel delivery of BDNF alone was able to enhance functional recovery in aged mice (36% gain of function at 6 weeks). Notably this gain of function is less than what we have previously reported after CX1837 treatment in young mice. 6 Interestingly, when BDNF and CX1837 were combined we observed marked improvements in motor function (57% gain of function at 6 weeks) that is greater than what we report after CX1837 treatment alone in young mice. The most probable explanation for our current findings is that successful recovery in aged animals requires an enhancement of cortical excitability (provided by CX1837) in combination with a significant elevation in BDNF levels.
Previous studies have shown that interventions starting from days 3 to 7 after stroke can aid in improving functional recovery in both animals,6,7,17,18 and humans. 31 Recent studies in rodents have shown that this recovery is most likely because of acivitydriven changes in BDNF. 19 In the present study, we failed to see any significant elevation in BDNF levels after stroke, indicating that the endogenous neuroplastic mechanisms involved in improving motor functions in young, are lacking or impaired in aged mice. Further, treatment with cx1837 resulted in a smaller increase in BDNF levels compared with previous published data in young mice (75% increase in young versus 54% increase in aged mice). 6 The findings in the present study are consistent with previous studies reporting that BDNF levels decrease with age. 32 In addition, the BDNF threshold required to achieve recovery is not obtainable with CX1837 treatment alone and coadministration with BDNF is required.
Previous studies have shown that TrkB ligands including BDNF can activate both PI3K/AKT and ERK1/2 signaling pathways. 33 We show here that combined administration of CX1837 and local hydrogel delivery of BDNF, resulted in a synergistic increase in phosphorylation of AKT, MEK, and CREB in aged (22 to 24 months old) mice, with p-AKT levels showing the greatest change. The PI3K/AKT signaling pathway plays a central role in regulating cell growth, proliferation, and survival under physiologic and pathologic conditions. 34 In addition, in cultured hippocampal neurons, PI3K/AKT signaling has been shown to be involved in axonal sprouting,22–25 which is an important mechanism underlying poststroke functional recovery.17,18,35 Further, recent work has also shown that modulation of GSK-3 after stroke can enhance axonal outgrowth. 36 This is a critical observation as in the present study we observe an increase in both p-AKT and p-CREB, which would highlight the AKT/GSK-3/CREB signaling pathway as being critical for mediating functional recovery after combined treatment with CX1837 and BDNF in aged.
Disruption to BDNF–TrkB signaling leads to a decrease in transcription of CREB, which is critical for processes of synaptic plasticity and learning. 37 Importantly, previous studies have shown that administration of the TrkB ligand, LM22A-4, binds and actives downstream signaling pathways including AKT and ERK, 13 and can ameliorate motor impairments in the model of both traumatic brain injury and stroke.13,14 Consistent with this, we also report that multiple signaling pathways are activated in response to CX1837 and BDNF treatments, and include an increase in p-AKT, p-ERK, and p-MEK. As CREB deletion only partially blocked the recovery afforded by combined treatment of CX1837 and local BDNF delivery, other signaling pathways, in addition to the proposed AKT/GSK-3/CREB pathways, are likely to be playing a role in recovery. For instance, previous studies have shown that exercise, which stimulates the release of BDNF, activates the PI3K/Akt/mTOR. 38 Consistent with this pathway in poststroke recovery, ischemic postcondition has been shown to activate the AKT/mTOR pathway and stimulate an increase in axonal outgrowth, as indicated with an increase in GAP43 expression in peri-infarct regions. 39 To this end the full complement of signaling pathways that play a role in poststroke recovery, or their involvement in mechanisms associated with recovery, still remain to be full elucidated.
The current set of experiments show that combined therapy with systemic administration with CX1837 and local delivery of BDNF is required to afford the greatest improvement in functional recovery after stroke in aged animals. We also show that this recovery is occurring through several key-signaling pathways with a significant amount of recovery occurring via the AKT/GSK-3/CREB pathway, which is also consistent with other pathologies such as Alzheimer's disease to improve cognitive function. 40 The current set of studies support the need for further development and investigation of ampakine and BDNF treatments for stroke recovery. Finally, we highlight a critical signaling pathway (AKT/GSK-3/CREB) that is involved in age-related functional recovery after stroke. This latter represents a promising direction for future work directed at developing targeted drug therapies that can be translated into clinical settings.
AUTHOR CONTRIBUTIONS
ANC designed the experiments, prepared the figures, and wrote the paper. ANC, KP, MN, FRW, and EKG performed the experiments. ANC and EKG analyzed the data. All authors reviewed and edited the manuscript.
DISCLOSURE/CONFLICT OF INTEREST
ANC is on a patent that was filed through UCLA on the use of hydrogels to deliver growth factors. All other authors declare no conflict of interest.
Footnotes
ACKNOWLEDGMENTS
The authors thank Cortex Inc for generously providing the ampakine and BioTime Inc for supplying the hydrogel.
