Abstract
Homocysteine (Hcy) is a risk factor for vascular dysfunction. High levels of Hcy may result in vascular injury accelerating atherosclerosis leading to ischemia. After ischemia, endothelial progenitor cells (EPCs) migrate from bone marrow to repair damaged sites either through direct incorporation of EPCs or by repopulating mature endothelial cells. This study looks into the relationship between increased Hcy in patients with cerebrovascular disease (CVD) and EPCs. Some patients with hyperhomocysteinemia were treated with B vitamins to evaluate if the treatment reverses the elevated Hcy and its impact on their EPC levels. EPCs were treated with Hcy to determine the
Introduction
A direct relationship exits between an increasing concentration of homocysteine and the risk of cardiac disease, stroke, and venous thrombosis (Haynes, 2002; Hackam and Anand, 2003; Kaplan, 2003; Perna et al, 2003). In older populations, modest increases in homocysteine level have been associated with an increased risk of cardiovascular disease (Haynes, 2002; Wald et al, 2002; Hackam and Anand, 2003; Kaplan, 2003). However, the mechanism for the vascular injury is not yet fully understood but is likely multifactorial (Wald et al, 2002; Hackam and Anand, 2003; Perna et al, 2003; Refsum et al, 2004). Possible mechanisms include stimulation of free radical formation (Kanani et al, 1999; Weiss et al, 2003), nitric oxide-mediated endothelial dysfunction (Stamler et al, 1993), platelet activation (Fryer et al, 1993), hypercoagulability (Harpel et al, 1992), vascular smooth muscle proliferation (Southern et al, 1998), and dysfunction of flow-mediated brachial artery reactivity (Kanani et al, 1999). It is known that endothelial dysfunction predisposes a patient to atherosclerosis, vasoconstriction, and thrombosis (Probstfield et al, 1993). For example, forearm endothelial dysfunction is associated with an increased risk of cardiovascular events. Coronary endothelial dysfunction also predicts cerebrovascular events, demonstrating the systemic nature of the atherosclerotic process (Targonski et al, 2003). Normally there is a strong inverse correlation between the number of endothelial progenitor cells (EPCs) and Framingham coronary risk score (FCRS), where higher EPC numbers are correlated with improved endothelial function (Hill et al, 2003).
EPCs originate in the bone marrow and are precursors to mature endothelial cells with distinct characteristics, and have the ability to be mobilized in response to vascular trauma or tissue ischemia (Verma and Anderson, 2002). A recent report suggests that the source of endothelial cells in vasculogenesis in tumor cells may be periendothelial in origin instead of bone marrow (Purhonen et al, 2008). However, there are several reports that indicate bone marrow origins of EPC in ischemiainduced vasculogenesis (Asahara et al, 1999a, b), and this process is promoted by cytokine and vascular endothelial growth factors released at the site of injury (Asahara et al, 1999a; Gill et al, 2001). These cells subsequently migrate to the site of injury and differentiate into mature endothelial cells, promoting reendothelialization and neovascularization. The incorporation of EPCs has been showed at sites of carotid artery endothelial injury that was surgically induced as well as after cerebral (Hess et al, 2002), myocardial (Verma and Anderson, 2002), and limb ischemia (Werner et al, 2003).
Indirectly, both homocysteine and EPC levels appear to be predictive of vascular risk, albeit with opposite response. However, to date there is no direct comparative study performed between these two variables at the clinical level. In the present study, we examined whether increasing concentration of serum homocysteine can be correlated to reduction in EPCs. Furthermore, we studied the cause for the inverse relationship between Hcy and EPCs and found that apoptotic pathways are involved in homocysteine-mediated death of EPCs. We also examined if the homocysteine-mediated apoptosis of EPCs can be prevented by B vitamins.
Materials and methods
Ethics
This study was approved by the Research Ethics Review Board of the University of Alberta. Informed consent was obtained in writing from all the subjects.
Subjects
Three sets of subjects were recruited representing a spectrum of cerebrovascular risk: (1) subjects with acute ischemic stroke admitted to the inpatient neurology ward; (2) subjects with stable ischemic stroke (more than 1 month after event); (3) healthy controls with no history of stroke or cardiovascular disease.
Homocysteine Measurement
Homocysteine was measured in blood samples from patients after at least 12 h of fasting, using direct chemiluminescent immunoassay method as described elsewhere (Rasmussen and Moller, 2000). In patients with acute stroke, it was measured within the initial 24 h of presentation and in all stable patients at 30 days or longer after the vascular event.
Cell Culture
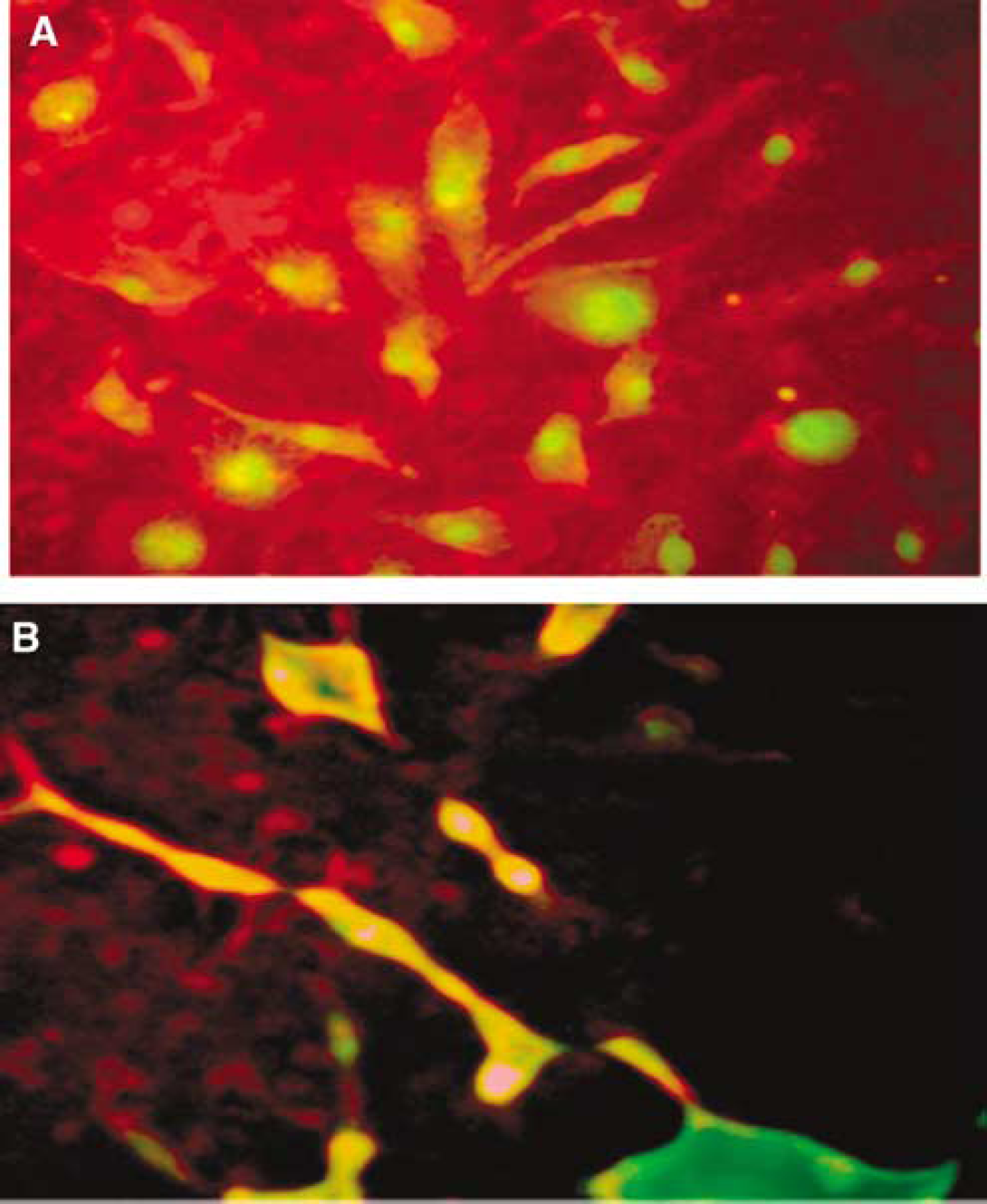
EPCs were isolated and cultured according to methods described and published earlier (Rasmussen and Moller, 2000; Hill et al, 2003). In brief, mononuclear cells (MNCs) were collected from peripheral blood using Ficoll (Sigma, St Louis, MO, USA) density gradient at room temperature (RT). MNCs in a buffy coat layer were collected, washed 2 × with phosphate-buffered saline (PBS) followed by Medium 199 (Invitrogen, Carlsbad, CA, USA). The M199 is supplemented with 20% fetal bovine serum, 100 U/mL Penicillin G (Sigma) and streptomycin sulfate (Sigma). EPCs were cultured M199 at 37°C with 5% CO2. Preplating was performed in a six-well (5 × 106 per well) human fibronectin-coated plate (Becton Dickinson, Franklin Lakes, NJ, USA). Final plating was performed in 24-well (1 × 106 per well) fibronectin-coated plate/cover slip. Medium was changed every third day and colonies were counted on the seventh day of final plating. EPC colonies were counted manually in each well of an individual sample. An EPC colony consists of a central cluster of rounded cells with surrounding radiating thin, flat cells. To avoid bias, colonies were counted by two different observers who were unaware of patient's clinical profile. The isolated EPCs were then cultured on serum-free medium for 24 h before treatment with homocysteine.
EPC Characterization by FACS
EPCs were analyzed by flow cytometry using 100 mLof mononuclear cell suspension. Direct immunofluorescent staining of the cells was performed using CD34-Allophycocyanin (20 μL; Jackson Laboratories, Bar Harbor, ME, USA) and CD31-Phycoerythrin (20 μL; Serotec, Raleigh, NC, USA) antibodies. Immunofluorescence was measured immediately after the completion of staining and the data were analyzed using CellQuest software (Becton Dickinson). CD34+-CD31+ cell counts were obtained from each sample after the measurement of 10,000 events in the lymphocyte gate. The cytometer was calibrated and the lymphocyte gate was setup using unstained mononuclear cells.
Caspase Assay
Activated caspases (−3, −8) were detected by caspase staining kits (BioVision, Exton, PA, USA). This assay uses a caspase inhibitor of respective caspases conjugated to sulfur rhodamine/fluorescein as the fluorescent marker and observed under microscope. Treated and untreated EPCs (1 × 106 per well) were cultured in 24-well plate on fibronectin-coated cover slips. After 24 h incubation EPCs were washed with PBS and then 1 μL dye-conjugated caspase inhibitor diluted in 500 μL M199 was added to each well. After 1 h incubation at 37°C with 5% CO2 cells were washed 3 × with supplied wash buffer (500 μL). Numbers of cells with activated caspases were visualized under fluorescence microscope and counted (number per field). Data were presented as mean number of caspasepositive cells.
Cytochrome c Detection
Cytochrome
Inhibitor Study
The cells were treated for 24 h with Z-IETD-FMK (10 μmol/L), a caspase-8 inhibitor, or Z-VAD-FMK (10 μol/L), global caspase inhibitor, in the incubation media, before the caspase assay described earlier. This was performed to determine the role of specific caspases in the induction of apoptosis by homocysteine.
Quantitative Assay
Microscopic images were converted into quantitative data by dividing a cover slip into four quadrants. Two fields were selected from each quadrant plus two fields from the center, totaling 10 fields. Microscopic images were captured by a charged coupled device (CCD) camera (Qimaging, Pleasanton, CA, USA) and analyzed by Openlab 4.0.1 software (Improvision, Waltham, MA, USA). Positive cells were counted for each field.
Statistical Analysis
Descriptive statistics are expressed as means±s.d. and median range for continuous variables and as percentages for categorical variables. Univariate analysis was performed by using analysis of variance (ANOVA), Kruskal—Wallis H- and Pearson's χ2-test where appropriate. Spearman's ρ-test was used to examine the relationship between EPC levels and fasting homocysteine levels and FCRS. Wilcoxon's signed rank test was used to compare the homocysteine level and EPC colony counts between baseline and 12 weeks. General linear model (GLM) procedure was used to relate age, sex, cigarette smoking, hypertension, diabetes, total cholesterol levels, creatinine level, C-reactive protein, 3-hydroxy-3-methylglutaryl-CoA (HMG-CoA)-reductase inhibitor use, FCRS, and type of subject (healthy controls, stable, and acute) to EPC levels. All variables with (
Results
Human EPC Characterization
EPCs were characterized by the formation of colonyforming units after 7 days of incubation. FACS was performed using CD34 and CD31 antibodies (Lapergue et al, 2007). Here it is important to emphasize that the identification of EPCs are still controversial. Several markers have been used for EPC characterization (Vasa et al, 2001; Ghani et al, 2005), however for our study we have followed the method described by Hill et al (2003) therefore, CD34 and CD31 double-positive cells were considered as EPC for this study.
Homocysteine is Inversely Correlated with EPCs in Patients with Hyperhomocysteinemia
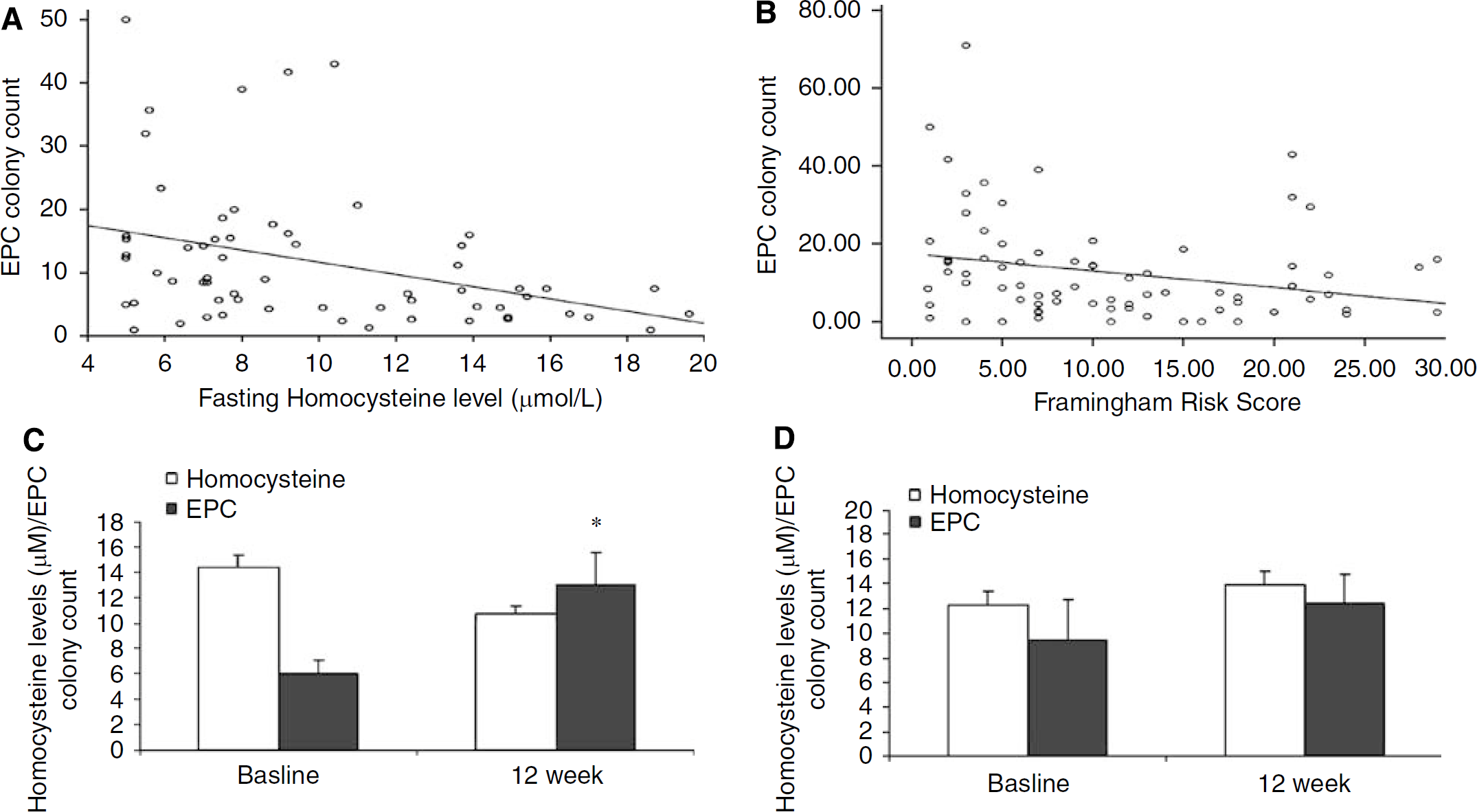
There were a total of 94 subjects comprising 60 men (63%) and 34 women (37%). The mean age was 63.5 years (s.d.±12.86). Seventy-seven of the subjects had a history of cerebrovascular disease. Prevalence of cardiovascular risk factors for the sample is provided in Table 1a. Twenty-nine subjects (30.5%) were on HMG-CoA-reductase inhibitor treatment, all of whom were in the group with stable cerebrovascular disease.
Cerebrovascular risk factors in study population (
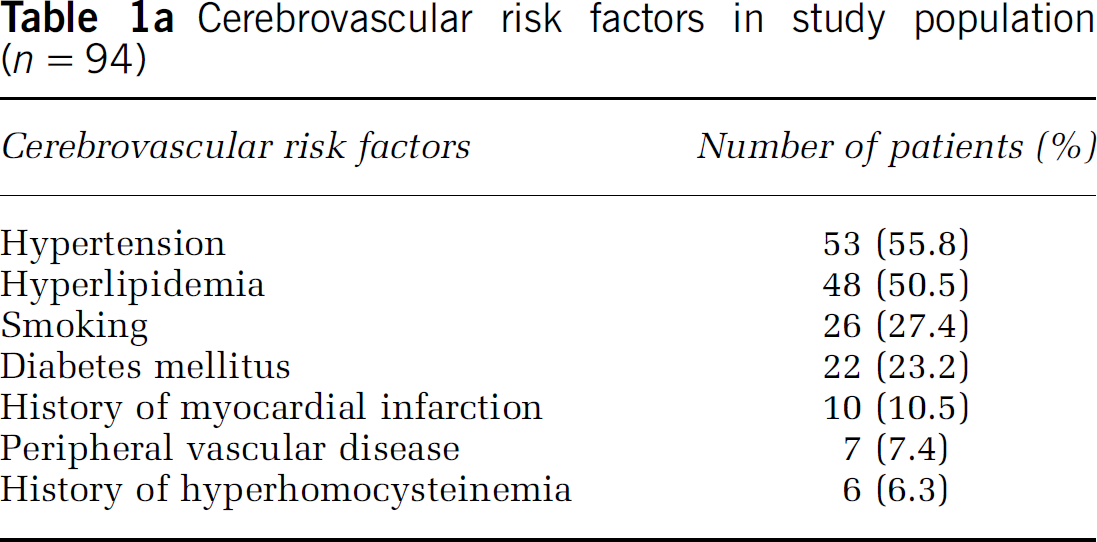
Among the patients studied there was an inverse relationship between EPC colony counts and fasting homocysteine (ρ = −0.415;
Clinical data of the patients recovering from stroke. (
Univariate regression analyses of various risk factors and colony counts
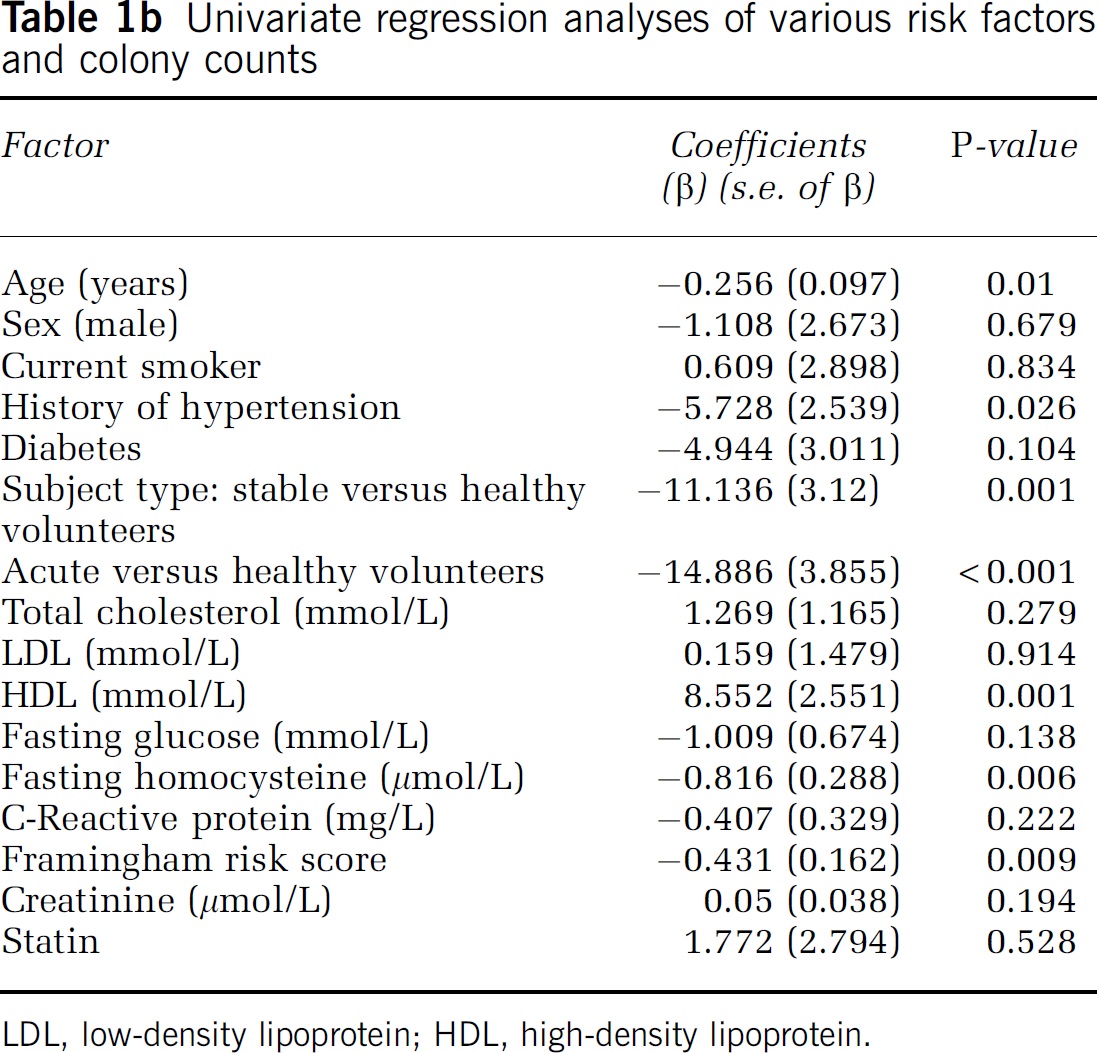
LDL, low-density lipoprotein; HDL, high-density lipoprotein.
Predictors of colony counts form multiple regression analyses
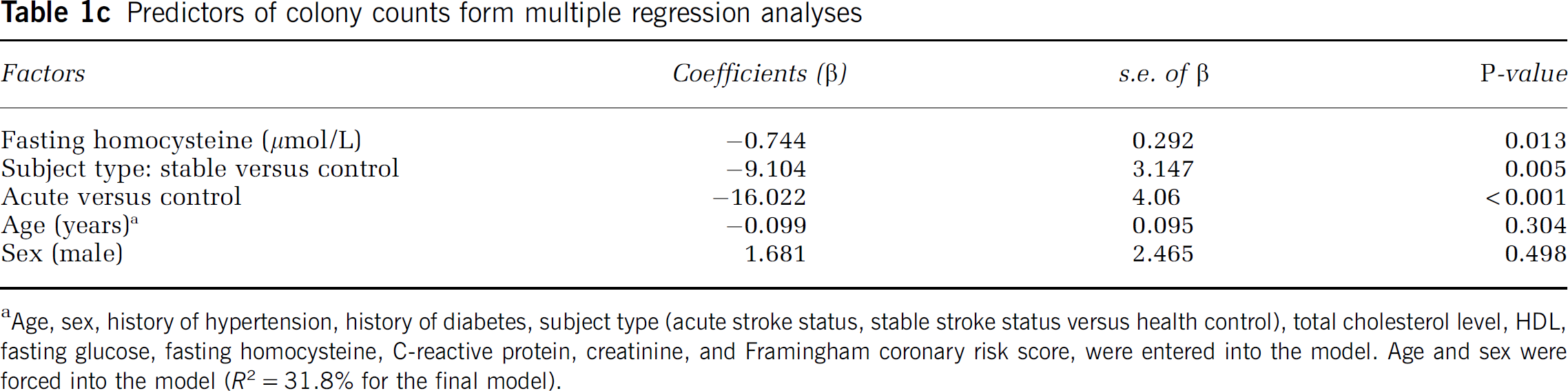
Age, sex, history of hypertension, history of diabetes, subject type (acute stroke status, stable stroke status versus health control), total cholesterol level, HDL, fasting glucose, fasting homocysteine, C-reactive protein, creatinine, and Framingham coronary risk score, were entered into the model. Age and sex were forced into the model (R2 = 31.8% for the final model).
There were 12 patients with homocysteine levels above 10 μg/L (74 μmol/L) who were treated with B vitamins. In six patients the treatment resulted in a decrease in homocysteine by 4 μmol/L or more. In the remainder, there was no decrease in the homocysteine levels with the treatment. Successful treatment resulted in a significant increase in EPC colony counts from 5.56±2.9 to 15.54±8.98 (
Homocysteine Induces Activation of Caspases 8 and 3 and Release of Cytochrome c in EPCs
To determine the effects of homocysteine on EPCs we looked at the apoptosis in the EPC. We saw that homocysteine is able to induce apoptosis in the EPC after 24 h treatment at 200 μmol/L (Figure 2). Furthermore, at the intracellular level our
Induction of apoptosis in EPC by Homocysteine. (
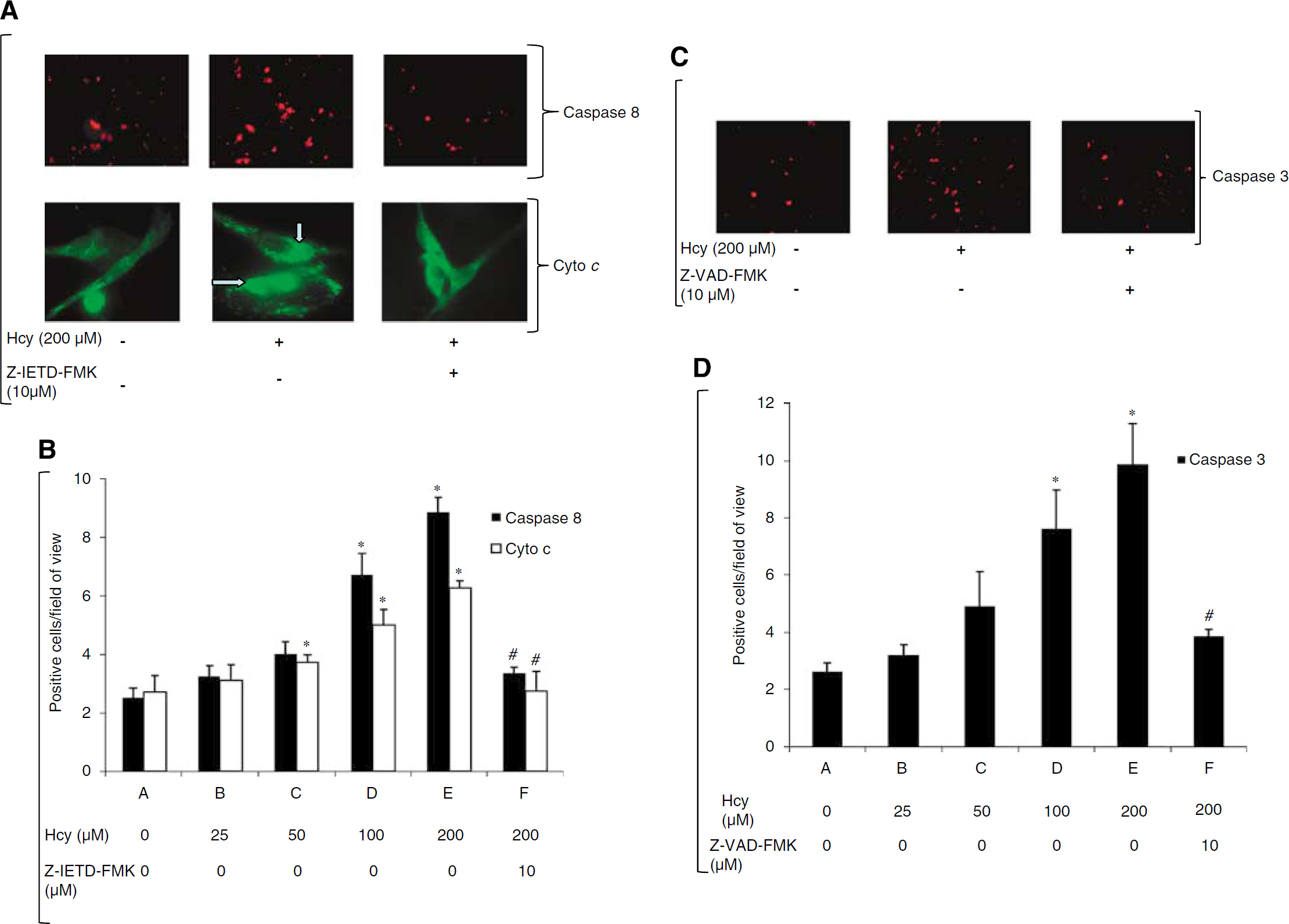
(
Subsequently, we measured caspase-3 expression in EPCs after homocysteine treatment of EPCs,
Vitamin B6 and B9 Impairs Caspase-3 Activation in Homocysteine-Induced EPC Apoptosis
Similar to the result shown above homocysteine (200 μmol/L)-treated EPCs show a significant increase in the number of caspase-3-positive cells compared with control EPCs (control versus 200 μmol/L Hcy: 3.30±0.78 versus 9.48±1.64,
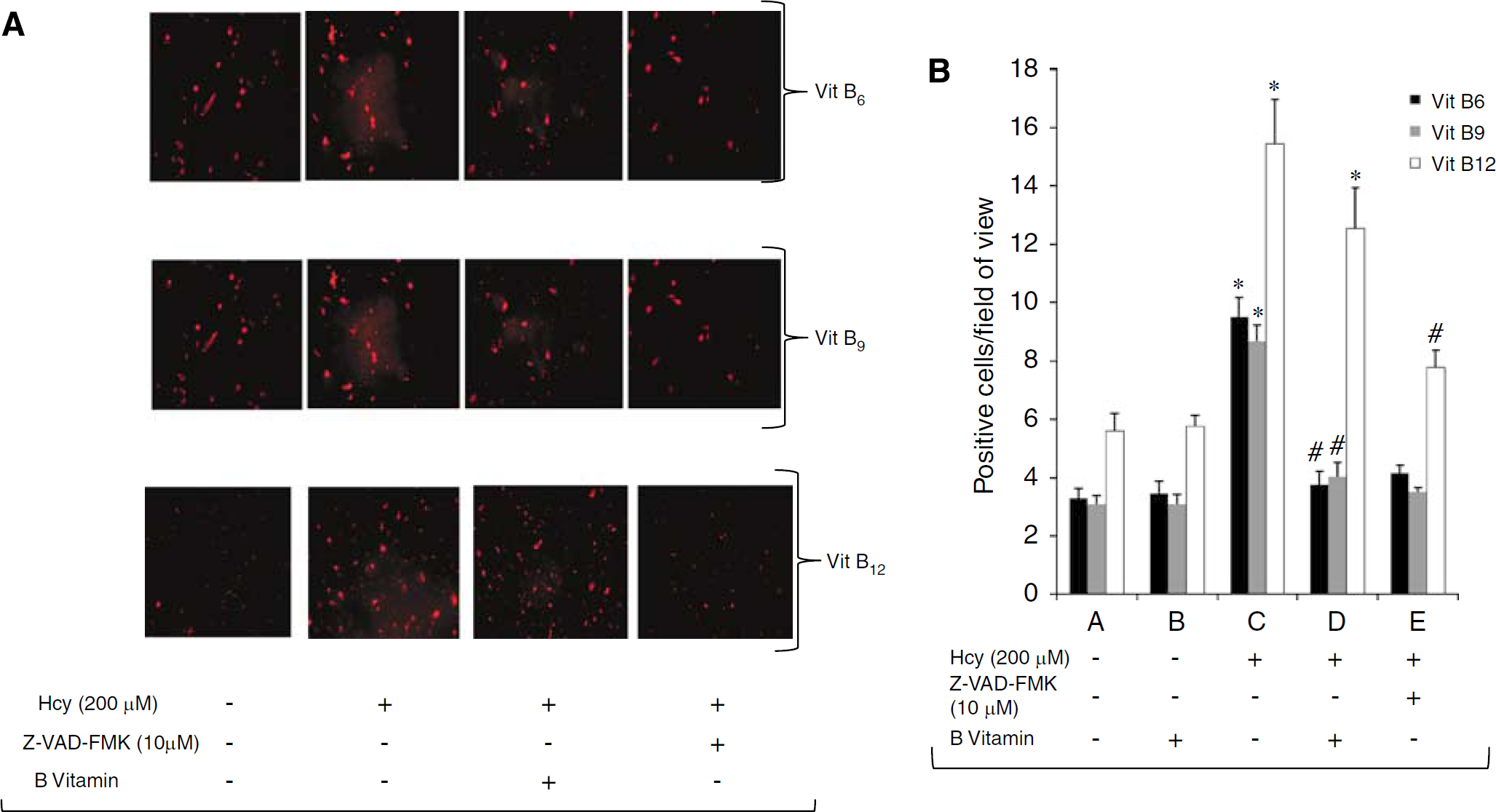
Effect of vitamin B on homocysteine-induced activation of caspase-3 in EPCs. (
Interestingly, vitamin B12 treatment (8 ng/mL) did not affect homocysteine-mediated increase in caspase-3 activation compared with the just homocysteine-treated group (12.55±3.41 versus 15.45±3.71,
Discussion
Homocysteine is a sulfur-containing amino acid, synthesized during metabolism of methionine. It decomposes either by remethylation to methionine or
The effects of homocysteine on endothelium function have been studied extensively (McCully, 1996; Chen et al, 2004; Noor et al, 2007).
Having seen a correlation between hyperhomocysteinemia and EPC level, we wanted to determine the direct effect of homocysteine on EPCs. Homocysteine has been reported to decrease EPC numbers in cell culture (Chen et al, 2004). However, there are no reports on the underlying mechanism of homocysteine-induced cytotoxicity in EPCs. We have reported earlier that that homocysteine induces the apoptotic pathway in EPCs (Noor et al, 2007). At the intracellular level we found the caspase-8 pathway was most affected by homocysteine treatment of EPCs and there was 3.5-foid increase in caspase-8 with 200 μmol/L homocysteine. Caspase-8 belongs to family of cysteine proteases and is involved in apoptosis. It is activated either by the prototypic CD95 (Fas) apoptosis pathway (Nagata and Golstein, 1995) or by the tumor necrosis factor related apoptosis inducing ligand (TRAIL)-R1 pathway (Wiley et al, 1995) leading to an increased release of cytochrome
In our clinical study we also found that B vitamin treatment reduced the homocysteine level in some patients. Interestingly, there was a concomitant increase in the EPC colonies in those patients (Figure 1D). Therefore, to ascertain the role of therapeutic effects of vitamin B treatment, we examined the effects of vitamins B6, B9, and B12 on homocysteine-induced apoptosis,
Together, our data show that increasing concentrations of serum homocysteine are associated with decreasing levels of circulating EPCs and that the homocysteine-induced EPC reduction may be due to apoptosis through caspase-8-mediated release of cytochrome
Footnotes
Acknowledgements
The authors are grateful to Abdul Salam for statistical analyses. The study was funded by Heart and Stroke Foundation of Canada.
