Abstract
Idiopathic rapid eye movement sleep behavior disorder (RBD) is a risk marker for subsequent development of neurodegenerative parkinsonism. In this study, we aimed to investigate whether regional cerebral metabolism is altered in patients with RBD and whether regional metabolic activities are associated with clinical measurements in individual patients. Twenty-one patients with polysomnogram-confirmed RBD and 21 age-matched healthy controls were recruited to undertake positron emission tomography imaging with [18F]fluorodeoxyglucose. Differences in normalized regional metabolism and correlations between metabolic activity and clinical indices in RBD patients were evaluated on a voxel basis using statistic parametric mapping analysis. Compared with controls, patients with RBD showed increased metabolism in the hippocampus/parahippocampus, cingulate, supplementary motor area, and pons, but decreased metabolism in the occipital cortex/lingual gyrus (P < 0.001). RBD duration correlated with metabolism positively in the anterior vermis (r=0.55, P = 0.01), but negatively in the medial frontal gyrus (r=-0.59, P = 0.005). In addition, chin electromyographic activity presented a positive metabolic correlation in the hippocampus/parahippocampus (r=0.48, P = 0.02), but a negative metabolic correlation in the posterior cingulate (r=-0.61, P = 0.002). This study has suggested that region-specific metabolic abnormalities exist in RBD patients and regional metabolic activities are associated with clinical measures such as RBD duration and chin electromyographic activity.
Keywords
INTRODUCTION
Idiopathic rapid eye movement sleep behavior disorder (RBD) is a parasomnia characterized by the loss of normal skeletal muscle atonia during rapid eye movement (REM) sleep with prominent motor activity and dreaming. Patients with RBD are at substantial risk for the future development of neurodegenerative disorders, particularly synucleinopathies such as Parkinson's disease (PD), dementia with Lewy bodies (DLB), and multiple system atrophy.1–4 Imaging studies with single photon emission computed tomography (SPECT) and positron emission tomography (PET) have attempted to detect abnormal functional alterations in RBD patients predictive of evolution toward the onset of synucleinopathies. Reduced striatal binding of radioligands such as 123I-FP-CIT and 11C-DTBZ was reported to reveal subclinical nigrostriatal dopaminergic damage in RBD patients before the appearance of PD.5–8 Cerebral blood flow (CBF) studies with SPECT also showed some similarities in the distribution of impaired brain perfusion between patients with RBD and those with synucleinopathies as a consequence of localized neural degeneration.9–11
18F-fluorodeoxyglucose (FDG) PET has been a valuable imaging method for studying neurodegenerative disorders in the past two decades. Differences in regional metabolic activity could be measured between parkinsonian patients and healthy subjects by using a voxel-to-voxel search.12–14 By contrast, network analysis of FDG PET images was useful in discerning abnormal functional connectivity in PD and atypical parkinsonism.15–17 These complementary techniques have proved to be useful in describing the physiopathology of synucleinopathy disorders at both the local and system levels, and therefore provide specific biomarkers in the assessment of disease progression, modification, and treatment response. 18
FDG PET has also been extended to detect abnormalities in the prodromal stage of synucleinopathies and to predict potential phenoconversion toward subsequent development of a neurodegenerative disease. Indeed, a recent investigation reported regional differences in metabolic activity at the major nodes (i.e. pons and putamen) of a PD-related covariance pattern in RBD patients. 10 Of importance, our research group has just identified a spatial covariance pattern in patients with RBD by using the network analysis of FDG PET images from a cohort of RBD patients and healthy controls. 19 Although this pattern was designed to reveal RBD-related metabolic alteration at a system level, it did not provide valuable information concerning metabolic differences and clinical correlates in specific cerebral regions. In the current study, we aimed to map such areas in the same set of FDG PET images by using voxel-to-voxel searches on a whole-brain basis. Furthermore, we explored the relationships between clinical indices and relative metabolic values on a voxel basis.
MATERIALS AND METHODS
Subjects
Twenty-one RBD patients (male/female 17:4; age 65.0 ±5.6 (mean±s.d.) years) were recruited in this study from 38 patients who were sequentially diagnosed with RBD at the Sleep and Wake Disorders Center, Department of Neurology, Huashan Hospital, Shanghai, China. The others were excluded because of their unwillingness to undertake FDG PET. Mean RBD duration of the enrolled patients was 5.7 ± 3.5 years at the time of PET scanning. All patients participating in this study met the RBD diagnostic criteria of the Second edition of the International Classification of Sleep Disorders: (1) Presence of REM sleep without atonia on polysomnography (PSG) during REM stage. (2) At least one of the following is present: (a) sleep-related injurious or disruptive behaviors by history and (b) abnormal REM sleep behaviors documented during PSG monitoring. (3) Absence of electroencephalograph epileptiform activity during REM sleep unless RBD can be clearly distinguished from any concurrent REM sleep-related seizure disorder. (4) The sleep disturbance is not better explained by another sleep disorder, medical or neurologic disorders, mental disorders, medication use, or substance use disorder.
To rule out neurologic conditions, all patients underwent a standard neurologic examination followed by a further test for parkinsonism by a senior specialist of movement disorders. The Unified Parkinson's Disease Rating Scale motor rating (UPDRS III, items 18 to 31) was used as an index of parkinsonian signs. These patients had no evident parkinsonism (UPDRS motor score 2.4 ±2.1; range 0 to 7) and took no psychotropic medications including antidepressants and anxiolytics. All the patients exhibited normal cognitive abilities for memory, language, executive, visuospatial functions, and attention according to the commonly used clinical criteria based on Mini-Mental State Examination score (28.7 ± 1.2; range 26 to 30). 20
This study also included 21 healthy subjects who were age- and gender-matched to the patients with RBD (male/female 17:4; age 62.5 ±7.5 years). They had no history of neurologic or medical disorders and each had a normal neurologic examination. These subjects did not undergo PSG, but all tested negative by RBD Single-Question Screen. 21
This study was approved by the Ethics Committee of Huashan Hospital in Shanghai, China. The study was carried out in compliance with the Code of Ethics of the World Medical Association and the standards established by the author's Institutional Review Board and granting agencies. All subjects or a legally responsible relative gave informed consent to take part in the study before undergoing PET scanning.
Polysomnographic Evaluation
All patients with RBD were confirmed by overnight audio-video-PSG recordings in our sleep laboratory. Measured PSG variables included the following: electroencephalograph with six-channel scalp electroencephalograph montages (F3/F4, C3/C4, O1/O2 referred to the contralateral mastoid), electrooculography, and electromyography (EMG) (submental, left lower limb, and right lower limb). Airflow was measured using a nasal cannula and mouth thermocouple. Thoracoabdominal movement was measured by inductance plethysmogram, and arterial oxygen saturation (SpO2) by finger pulse oximetry. Sleep stage was manually scored according to the standard criteria, 22 with the exception that REM sleep was scored on the basis of electroencephalograph and electrooculogram only. During REM sleep, the presence of phasic submental EMG activity (>15%) and/or tonic EMG activity (>30%) was used to determine REM sleep without atonia stage. 22 The PSG parameters for this RBD cohort have been published previously. 19
FDG PET Scanning
All subjects were asked to fast for at least 6 h, but had free access to water before PET imaging. PET scans were performed with a Siemens Biograph 64 PET/CT (Siemens, Munich, Germany) in a three-dimensional mode. A computed tomography transmission scan was first performed for attenuation correction. The emission scan was acquired between 45 and 55 minutes after intravenous injection of 185 MBq of 18F-FDG. As no arterial blood sampling was taken in this clinical imaging protocol, we used radioactivity images to measure changes in relative regional glucose metabolism. All studies in patients and normal individuals were performed in a resting state in a quiet and dimly lit room. All patients and normal individuals were monitored via cameras and remained awake during the course of uptake and scanning procedure.
Image Processing
Imaging data were processed by using Statistical Parametric Mapping (SPM5) software (Wellcome Department of Imaging Neuroscience, Institute of Neurology, London, UK) implemented in Matlab 7.4.0 (Mathworks, Sherborn, MA, USA). Scans from each subject were spatially normalized into Montreal Neurological Institute (MNI) brain space with linear and nonlinear three-dimensional transformations. The normalized PET images were then smoothened by a Gaussian filter of 10 mm full-width at half-maximum over a three-dimensional space to increase signal-to-noise ratio for statistic analysis. The set of resulting images in both RBD and healthy subjects described above had previously been used for identifying the RBD-related brain network. 19
Data Analysis
PET imaging data were analyzed by using the SPM5 software as described previously. 23 To characterize metabolic activity in RBD patients compared with controls, we performed a group comparison by using a two-sample t-test according to the general linear model at each voxel. Mean signal differences over the whole brain were removed by analysis of covariance in each individual subject. We also used a multiple regression analysis to determine the relationship between cerebral metabolic values and clinical measures (i.e., RBD duration and chin EMG activity) in patients with RBD. These two variables were entered as covariates into the model together along with age and UPDRS scores to eliminate the interaction between these factors.
To evaluate the results, we set the peak threshold at P < 0.001 (uncorrected) over whole brain regions with an extent threshold of 60 voxels (corresponding to a tissue volume of 480 mm3). For a stricter criterion, we also highlighted clusters that survived a family-wise error correction at P < 0.05. Significant regions were localized by Talairach-Daemon software (Research Imaging Center, University of Texas Health Science Center, San Antonio, TX, USA). The SPM maps for altered glucose metabolism and clinical correlations were overlaid on a standard T1-weighted magnetic resonance imaging (MRI) brain template in stereotaxic space.
To quantify metabolic changes in specific regions, we used a 4-mm radius spherical volume of interest (VOI) centered at the peak voxel of clusters that were significant in the SPM analyses. We then calculated the relative cerebral glucose metabolic values (i.e., globally adjusted) in patients and normal individuals with the ScanVP software (Version 5.9.1; Center for Neuroscience, the Feinstein Institute for Medical Research, Manhasset, NY, USA).
Statistic Analysis
Differences in regional metabolic values between RBD patients and normal controls were assessed by using two-sample t-tests. In addition, correlations between regional metabolic values and corresponding clinical indices in RBD patients were assessed by computing Pearson's correlation coefficients. All analyses were performed using the SPSS software (SPSS, Chicago, IL, USA) and considered significant for P < 0.05.
RESULTS
Characteristics of Relative Brain Activity in Patients with RBD In comparison with healthy controls, patients with RBD showed increased metabolism bilaterally in the superior frontal gyrus, supplementary motor area (SMA), cingulate (BA 24), hippocampus/parahippocampal gyrus, insula, inferior parietal lobule, and the pons, and in the left superior temporal gyrus (BA 39), right precuneus (BA 31), and right cerebellar tonsil, associated with decreased metabolism in the bilateral occipital cortex and lingual gyrus (BA 17, 18), right superior temporal gyrus (BA 22), and left transverse temporal gyrus (BA 41) (Figure 1A and Table 1).
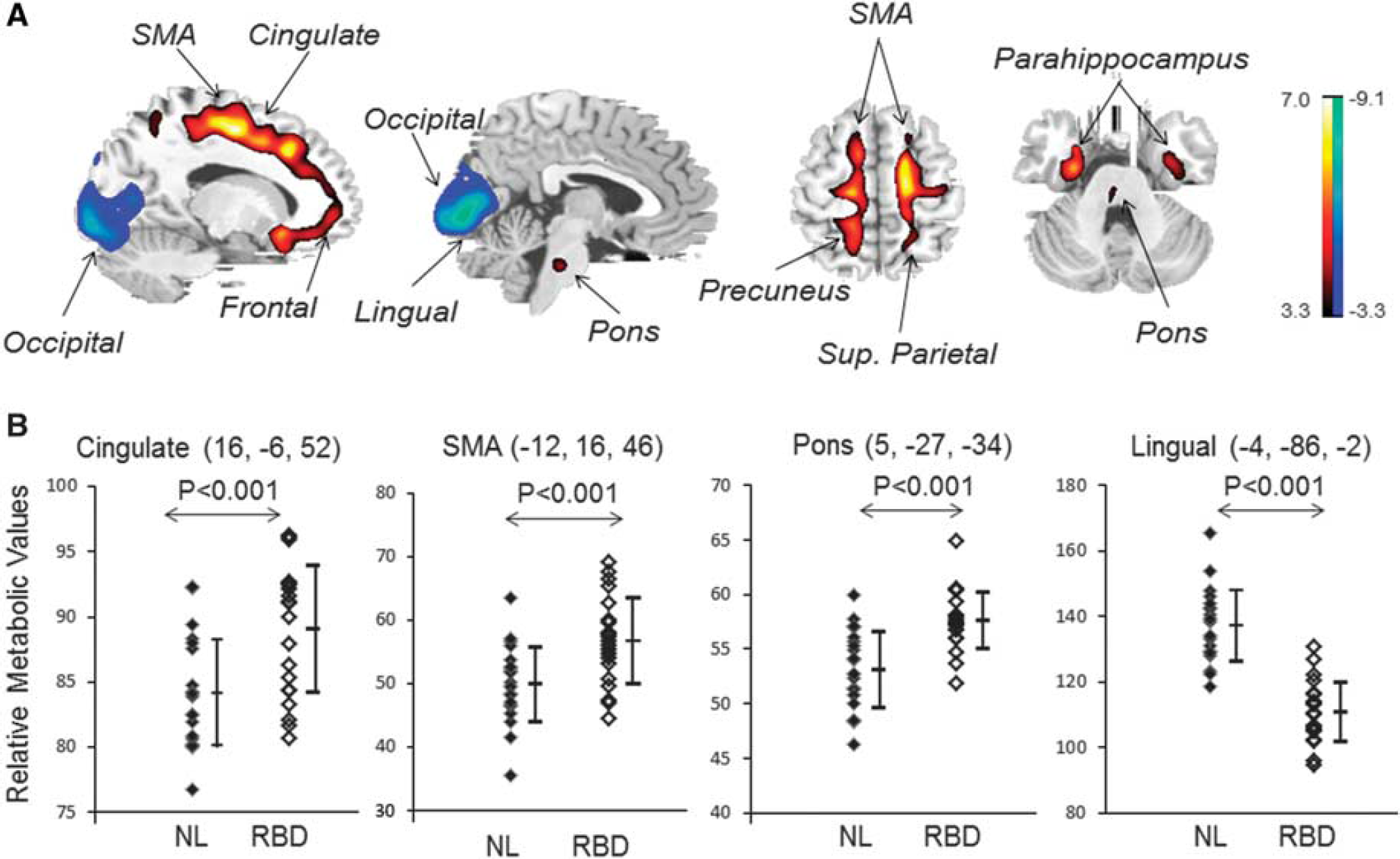
Brain regions with relative abnormal metabolism in patients with idiopathic rapid eye movement sleep behavior disorder (RBD). (
Brain regions with significant metabolic differences in idiopathic RBD patients compared with normal subjects
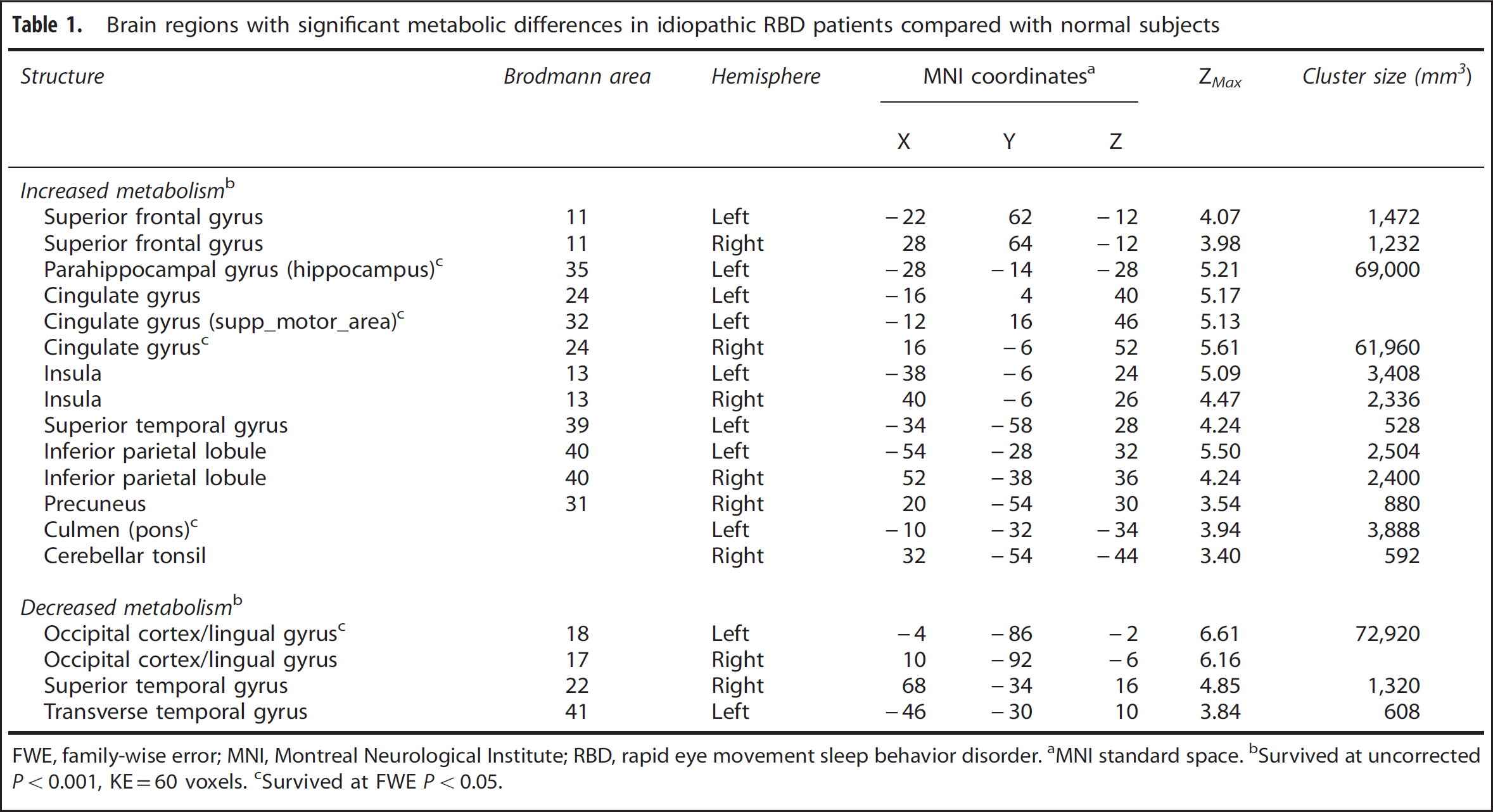
FWE, family-wise error; MNI, Montreal Neurological Institute; RBD, rapid eye movement sleep behavior disorder. aMNI standard space. Survived at uncorrected P < 0.001, KE = 60 voxels. cSurvived at FWE P < 0.05.
Figure 1B showed the differences in relative metabolic values between the RBD and control groups in the four major functional regions (Figure 1A). The increased metabolic values in the VOIs centered at the right cingulate, left SMA, and right pons displayed significant group differences between patients and healthy individuals (P < 0.001). Similarly, the decreased metabolic value in the VOI centered at the left lingual gyrus also showed significant group discrimination between patients and controls (P < 0.001).
Correlation of Clinical Indices with Relative Brain Activity in Patients with RBD
The regression analysis yielded a positive correlation of RBD duration in patients with metabolic activity in the left posterior cingulate (BA 31), right precuneus (BA 7), and right anterior vermis. In contrast, RBD duration correlated negatively with metabolic activity in the bilateral frontal gyri, right anterior cingulate (BA 32), left inferior temporal gyrus (BA 20), insula (BA 13), postcentral gyrus (BA 3), and right culmen (Figure 2A and Table 2). The sample plots for the two major regions from the regression analysis (see Figure 2A) were given in Figure 2B. The duration in RBD patients correlated significantly with relative metabolic values in the right anterior vermis (r=0.55, P = 0.01), as well as in the right medial frontal gyrus (r=-0.59, P = 0.005).
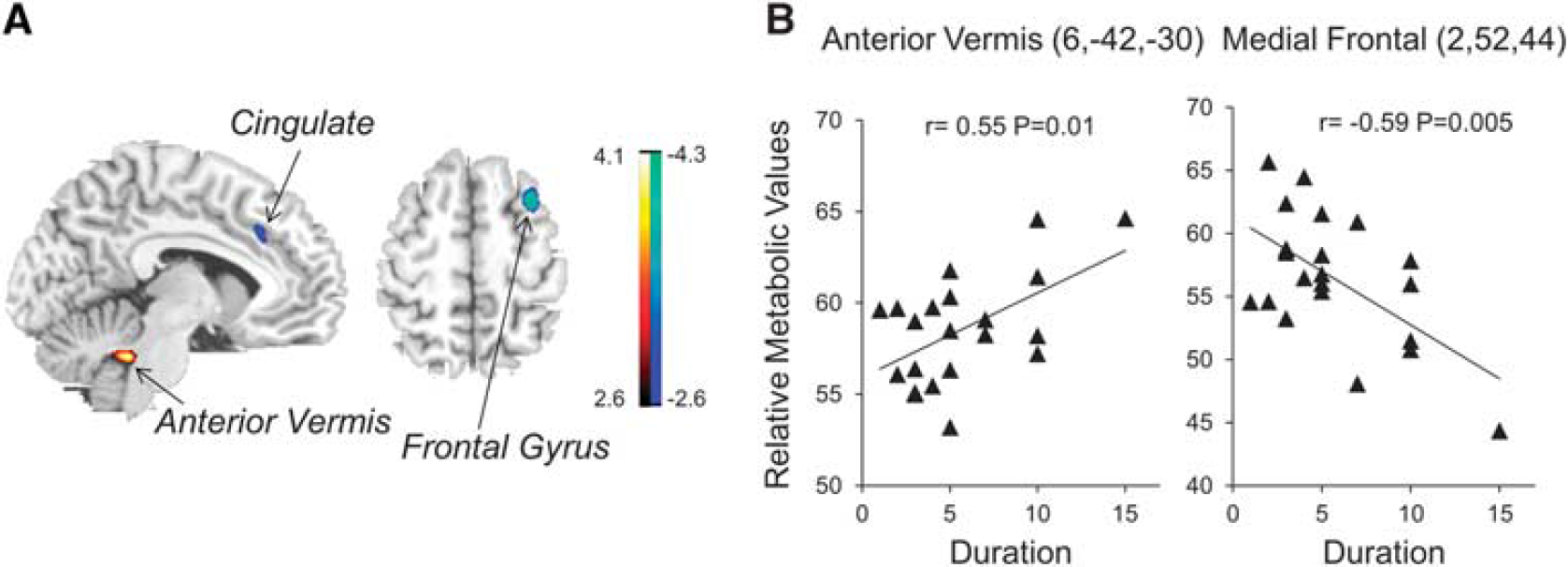
Brain regions correlated with duration in idiopathic rapid eye movement sleep behavior disorder (RBD) patients. (
Brain regions with significant clinical correlation in idiopathic RBD patients using multivariate regression
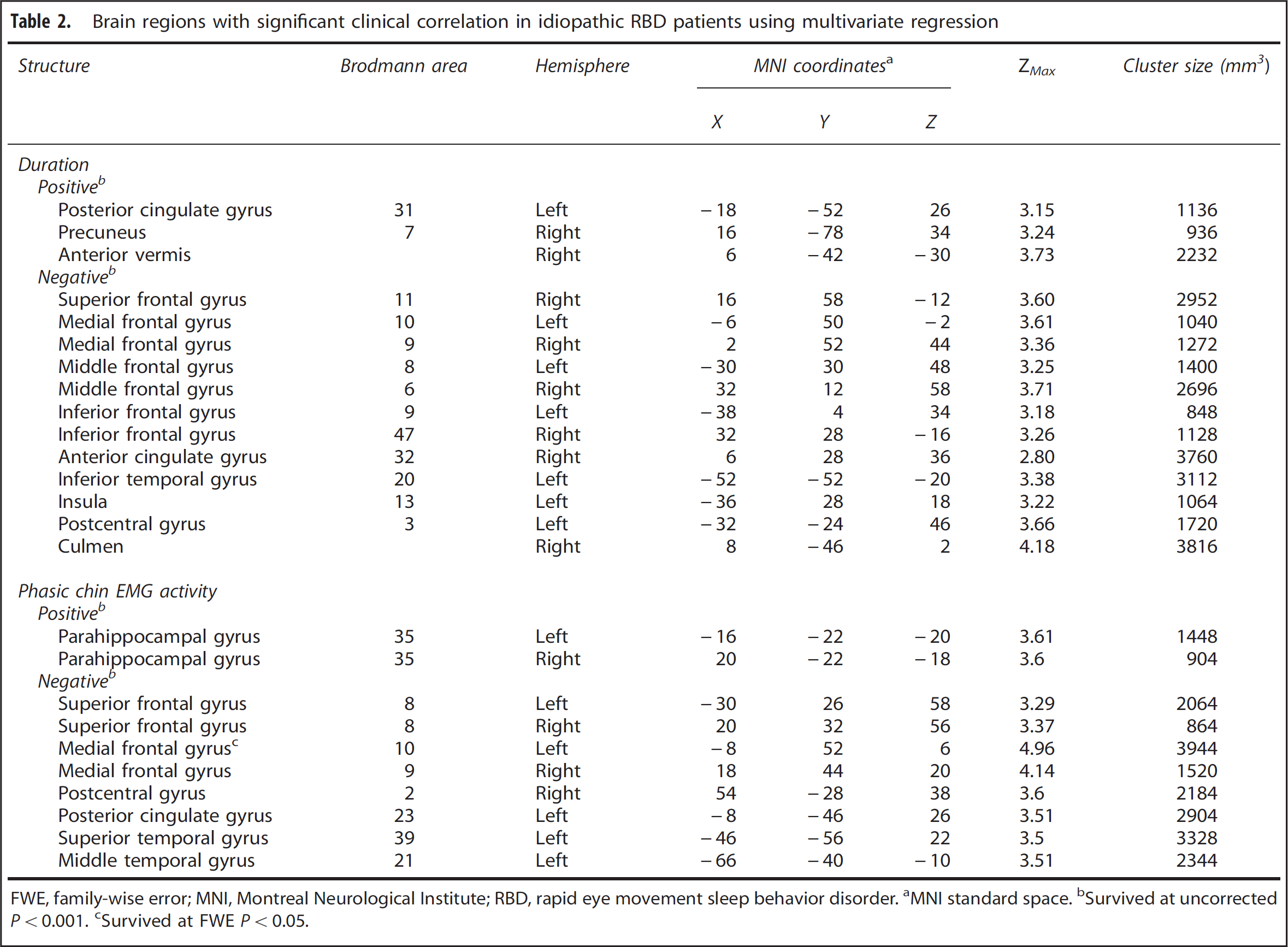
FWE, family-wise error; MNI, Montreal Neurological Institute; RBD, rapid eye movement sleep behavior disorder. aMNI standard space. bSurvived at uncorrected P < 0.001. cSurvived at FWE P < 0.05.
We also observed a positive correlation between phasic chin EMG activity and metabolic activity in the bilateral parahippocampal gyrus, but negative correlations in the bilateral frontal gyrus, right postcentral gyrus, left posterior cingulate (BA 23), and temporal gyrus (BA 21, 39) (Figure 3A and Table 2). The sample plots for the two corresponding regions (see Figure 3A) were presented in Figure 3B. The phasic chin EMG activity in RBD patients correlated significantly with relative metabolic values in the left parahippocampus (r=0.48, P = 0.02), as well as in the left posterior cingulate (r = −0.61, P = 0.002).
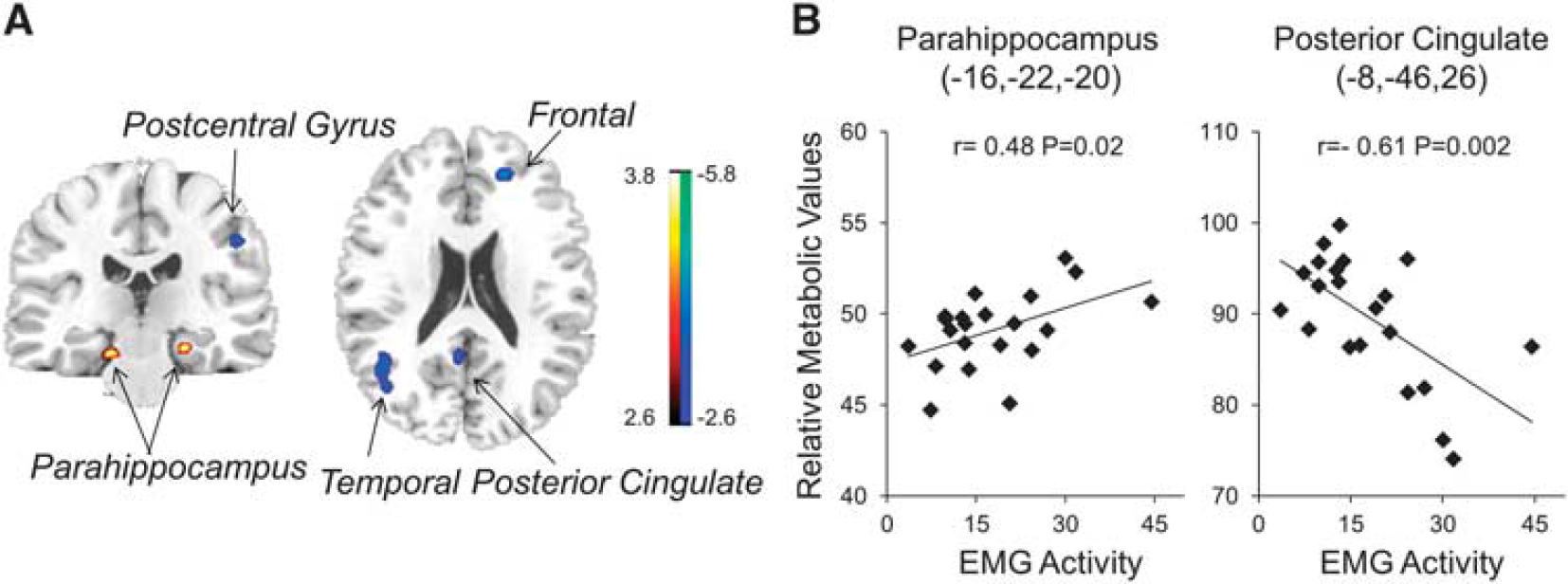
Brain regions correlated with phasic chin electromyography (EMG) activity in idiopathic rapid eye movement sleep behavior disorder (RBD) patients. (
DISCUSSION
This article describes the first functional imaging study to map changes in regional glucose metabolic activity on a whole-brain basis in the same derivation cohort of PSG-confirmed RBD patients described previously. 19 We found that RBD patients had abnormal metabolic activity in both cortical and subcortical regions relative to normal controls. The significant group differences in these brain regions were confirmed by post hoc VOI analyses of relative metabolic values. Multiple regression analysis in the patients showed significant correlations between regional metabolic values and individual clinical indices such as RBD duration and chin EMG activities. These results further showed the potential of FDG PET in the assessment of disease progression and subsequent evolution on a regional basis.
Metabolic Differences Between RBD Patients and Healthy Subjects The principal finding of this study is the localization of disease-related metabolic abnormalities in RBD patients compared with normal controls. The regions of abnormal metabolism seen in our study were in accordance with the prominent areas reported in other functional imaging studies. Pons and hippocampal gyrus, for example, were observed consistently with elevated CBF by using perfusion SPECT, 11 particularly in the RBD patients who subsequently developed PD and DLB. 24 Altered microstructures, volumes, and densities of the brainstem and/or hippocampus, as well as thinning in cortical thickness, were also described in MRI studies using diffusion tensor imaging and voxel-based morphometry.25–28 As neurons in the subcoeruleus areas of the brainstem have proved to be responsible for REM sleep atonia in patients with RBD, it is not unexpected that pontine dysfunction was observed in both the current and the previous imaging studies.
Although we found no changes of putamen activity in the current study, previous studies described increased CBF or glucose metabolism in this region.10,11 Additionally, altered nigrostriatal connectivity was observed in RBD patients by using resting-state functional MRI. 29 One possible reason for the absence of metabolic alteration in the putamen in this study might be the relative short RBD duration of our recruited patients. As the putamen is commonly recognized to be associated with motor dysfunction (e.g., limb tremor) in clinical parkinsonism, our result may simply suggest that these patients are in the mild stage of RBD. However, whether metabolic abnormalities of the putamen exist in advanced RBD patients before phenoconversion to parkinsonism remains to be determined by longitudinal studies.
The current study also revealed some new areas with abnormal metabolic activity in the cortex. The metabolic activity increased in the SMA, cingulate, and superior frontal gyrus along with the insula and inferior parietal lobe, but decreased in the occipital cortex, lingual gyrus, and temporal gyrus. The increased perfusion in the SMA was also reported during an RBD episode in a patient by SPECT imaging, 30 and these results further suggest that elevated locomotor drive may be behind the clinical manifestation of human RBD. 1 Of note, the increased perfusion in the cingulate seen in the RBD patients at baseline was previously found to be predictive of eventual phenoconversion to PD. 24 Conversely, the reduced metabolic activity evident in the occipitotemporal regions might be related to impaired color vision based on a regional perfusion study. 11 The different observations between the present study and the existing metabolic or perfusion studies might be explained by the methodological factors described below.
It may be worth considering the possible role of compensation in RBD-related brain network. The controlling of normal muscle atonia during REM sleep involves a specific circuitry that originally projects from sublaterodorsal tegmental nucleus within the brainstem. It has been proven that descending neurons from sublaterodorsal tegmental nucleus has an important role in inhibiting skeletal motoneurons, and therefore prevents the occurrence of abnormal motor behavior during REM sleep; RBD pathogenesis is likely to stem from the breakdown of this circuitry. 31 The relative hypermetabolism observed in RBD patients in the pons and the wide range of neocortex in this study may represent the overactivity of descending projections from sublaterodorsal tegmental nucleus as an early compensatory mechanism to regulate abnormal muscle atonia. It is expected that this effect might become weaker with the progression of RBD towards the onset of any particular form of synucleinopathies.
Correlation Between Regional Metabolism and Clinical Features One unique feature of this study is that we delineated the cerebral metabolic areas associated with RBD duration and phasic chin EMG activity in RBD patients. Interestingly, the positive correlations of metabolic activity in the posterior cingulate gyrus with RBD duration had been reproduced by those of corresponding regional perfusion with deficit in color vision. 11 It is noteworthy that a positive correlation was also observed between phasic REM density and metabolic changes in the hippocampus/parahippocampal region, a major area involved in the pathophysiology of RBD because of correlations observed between its local perfusion and impaired motor, olfactory, and color discrimination function.11,24 These findings in the cingulate and hippocampal regions substantiated the involvement of limbic circuits in support of RBD as a risk factor for cognitive decline. Indeed, severity of REM atonia loss has been reported to predict impending parkinsonism in RBD patients. 32 In this vein, elevated metabolic activity in the hippocampus/parahippocampus may be a harbinger of the neurodegenerative process in RBD, leading to clinical parkinsonism, given that increased baseline hippocampal perfusion points to the development of presumed synucleinopathies. 24 Nevertheless, the accuracy of this prediction has to be further verified by follow-up studies in our RBD sample.
Some other cerebral areas were also found to be in association with each of these two clinical indices in this study, especially those showing negative metabolic correlations in the frontal and temporal cortices. The latter were generally compatible with the decreased perfusion and corresponding cognitive correlations described previously in patients with RBD. 11 Although a multiple regression design was applied to circumvent the interaction between clinical factors in the analysis, we still observed an overlap of cerebral regions correlating with these two indices (e.g., the frontal gyrus, postcentral gyrus, and temporal gyrus). One potential explanation could be that, as excessive muscle activity increased with RBD duration in patients, 33 the resulting overlap might arise from the synchronization of underlying pathophysiology between two clinical factors. Moreover, we found that the metabolic values in the anterior and posterior aspects of cingulate correlate negatively with RBD duration and chin EMG activity, respectively. Why this relationship occurs is currently not known, but decreased glucose metabolism was reported in the anterior and posterior cingulate gyri in normal adults with dream enactment behaviors. 34 These findings point out a possible link to DLB given that the temporal cortex and posterior cingulate are known to be metabolically deficient in DLB.
It is helpful to discuss several issues for the study looking into clinical correlation with regional glucose metabolism. Although RBD duration and EMG chin activity represent clinically related indices, these two variables were not correlated in our RBD cohort. The regression analysis showed similar and different clinical correlates between these two variables on a regional basis. However, the correlations were moderate overall with only one region survived after correction for the effect of multiple comparisons. These results were likely because of the relatively short duration, narrow range of chin muscle activity, and limited sample size in our RBD patients. Whether our findings could be reproduced still need to be explored further in a large sample with a longer disease duration and wider distribution in muscle activity on an individual level. Nonetheless, these imaging results showed that RBD duration and EMG chin activity are partially overlapping, but they may also serve as different descriptors for assessing unique features of RBD.
Metabolic Alterations and Markers of α-Synucleinpathy
RBD is considered to represent a clinical risk marker for the subsequent development of α-synuclein diseases, especially for PD and DLB. 2 Regional metabolic abnormality in PD has been reported in several independent cohorts, and was characterized by hypermetabolism in the basal ganglia, sensorimotor areas, pons, and cerebellar cortices, associated with hypometabolism in the parietooccipital and prefrontal cortices.12–14 Although the cortical metabolism showed a few similarities between PD and RBD based on the result reported in this study, it is worth noting that pontine hyperactivity acted as a key metabolic feature in both disorders. This finding was in accordance with the hypothesis of Braak staging, 35 which described the first appearance of Lewy bodies in the pontine tegmentum in PD, associated with subsequent attainment of other cerebral regions as disease evolved. Further support was provided by increased metabolic activity and perfusion of local pons in patients with RBD, with greater perfusion in the patients who later phenoconverted to PD/DLB in comparison with their nonphenoconverter counterparts.10,24 Metabolic studies in DLB also shared some findings with the current study, such as hypometabolism in the occipital areas. This region, which is specialized in color vision, might explain the dysfunction of color discrimination observed in both patients with RBD 36 and those with DLB. 37
Comparison with Other Studies and Methodological Issues
Using the same set of imaging data used in the present study, we have recently described an RBD-related covariance pattern (RBDRP) characterized by increased metabolic activity in the pons, thalamus, medial frontal and sensorimotor areas, hippocampus, supramarginal and inferior temporal gyri, and posterior cerebellum, associated with decreased activity in occipital and superior temporal regions. 19 This pattern presented a high degree of overlap to the analogous PD-related covariance pattern. 17 In addition, network expression of RBDRP discriminated individual patients from healthy controls and revealed a possible evolution process from this disorder on the way to the development of PD.
Although we observed high similarities between the topography of RBDRP in the previous study and the distribution of RBD-related regional metabolic activities in the current study, it is important to note that these results portray very different measures of brain function. Indeed, the prior study concerned the derivation of a brain network using spatial covariance analysis of variance information of glucose metabolism measured in FDG PET images. By contrast, the current study reported the regionally specific differences in mean value of glucose metabolism using SPM. It has been shown that the expression of a brain network can be measured prospectively in new subjects for group classification on an individual case basis and the network approach has a higher sensitivity compared with SPM in detecting localized changes in neurodegenerative disorders. 38 The latter is supported by the fact that metabolic changes observed in some regions such as the thalamus in the RBDRP were absent in the present study. Nevertheless, the findings from the current study provided complementary information to the previous study and helped support and explain the elevated RBDRP activity in the RBD subjects as a group. 19 In general, the higher the similarity between the regional metabolic abnormality in a single patient and the topography of a brain network, the greater the subject score of that brain network in the single patient.
It is necessary to understand the discrepancies between this work and other studies in light of some variations and limitations inherent in the experiment. The major factor may be attributed to the differences in patient samples and imaging methodology. Indeed, the recent study with FDG PET measured brain metabolic changes in a set of predefined regions in a smaller number of patients with similar RBD duration, 10 whereas the other studies used SPECT imaging to measure brain perfusion in a comparable number of patients with longer RBD duration.10,11,24 Although perfusion SPECT and metabolic PET are supposed to measure the same neural activity in healthy brains, CBF and glucose metabolism may differ from each other under diseased conditions, particularly after medications acting on the central nervous systems. 16 Moreover, only relative values of CBF or metabolism were quantified in all these studies owing to clinical constraints, although there might be more statistic power if absolute CBF or metabolism was calculated with the collection of arterial blood sampling.39–40
We also need to point out that brain metabolism of our RBD patients was measured with FDG PET in wake state but not in REM stage, such as in the previous metabolic or perfusion imaging studies. It was conceivable that assessment of metabolic activity in REM sleep might provide different results. To our surprise, we found that most areas with significant metabolic alterations in waking patients were also associated with eye movements in healthy controls during REM sleep. 39 Similarly, a CBF study with PET also observed significant changes in the activity of limbic and paralimbic areas (e.g., the hippocampus/parahippocampal gyrus, cingulate, insula, and temporal pole) in healthy subjects in the REM stage. 40 Although the potential function of limbic/paralimbic activation during REM sleep remains unclear, it is phenomenologically consistent with the autonomic activation and lability observed in this particular stage, and could be related to the rich emotional content of dreams reported during this sleep stage. 40 The accordant findings from the present and other studies of waking RBD patients or healthy subjects in REM sleep provided further support that the regions with metabolic abnormalities we observed in relation to RBD did not occur by chance.
CONCLUSION
In this study, we substantiated the distribution of characteristic regional metabolic activity in RBD using FDG PET imaging and voxel-based brain mapping analysis. We also found significant correlations between abnormal metabolic values and different clinical indices of RBD duration and chin EMG activity. The abnormal metabolic substrates provided further support in light of RBD as a risk factor for developing neurodegenerative parkinsonism. Because of preliminary nature of the current work at baseline, cross-sectional studies in other independent cohorts with larger sample sizes would be necessary to confirm the reproducibility of abnormal RBD-related regional metabolic activities and their clinical correlates. Furthermore, follow-up studies would also be carried out in our patient cohort to clarify the relationships between metabolic activities and their longitudinal changes in disease progression and eventual clinical outcomes of RBD patients.
Footnotes
JG contributed to the analysis and interpretation of data, as well as the manuscript drafting and revision. PW, HY, and HZ contributed to the acquisition of data. SP contributed to the analysis and interpretation of data. YG and CZ contributed to the conception and design of the study. DE, YM, and JW contributed to the manuscript revision and the final approval of the version to be published.
The authors declare no conflict of interest.
