Abstract
The objective of this article was to study the reproducibility and effect of levodopa on dopamine transporter function measurements using 2β-carbomethoxy-3β-(4-[18F]-fluorophenyl)tropane ([18F]CFT) positron emission tomography (PET). Seven de novo patients with Parkinson's disease (PD) were studied twice, before and after three months of levodopa medication. Eight healthy volunteer subjects participated in the reproducibility study. The [18F]CFT PET scan was done twice with an interval of approximately 2.5 months. The regions of interest (anterior and posterior putamen, caudate nucleus, and cerebellum) were drawn on individual magnetic resonance imaging (MRI) images, matched with the PET images, and copied onto the PET images. The [18F]CFT uptake was calculated as the region–cerebellum:cerebellum ratio at 180 to 210 minutes. Three-month levodopa treatment in PD patients had no significant effect on [18F]CFT uptake in any striatal subregion between the two PET scans. In PD patients, the percent change from baseline was 4.1% in the anterior putamen, 1.9% in the posterior putamen, and 4.0% in the caudate nucleus. No significant differences in [18F]CFT uptake between the first and second PET scan in any striatal subregion occurred in healthy controls. The intraclass correlation, indicating the reproducibility of the PET scan within subjects, was 0.94 for the anterior putamen, 0.86 for the posterior putamen, and 0.91 for the caudate nucleus. The percent change from baseline was 4.0% in the anterior putamen, 1.1% in the posterior putamen, and 2.8% in the caudate nucleus. Long-term levodopa treatment in PD patients had no effect on the [18F]CFT uptake in the striatum and the test–retest reproducibility was very high. These findings confirm [18F]CFT as a suitable ligand to monitor progression of PD.
Progressive loss of nigrostriatal dopaminergic neurons is the basic pathophysiologic mechanism of Parkinson's disease (PD). Positron emission tomography (PET) or single photon emission computed tomography (SPECT) has demonstrated this reduced dopaminergic function in vivo using 6-[18F]fluoro-L-dopa (FDOPA;Boyes et al., 1986; Brooks et al., 1990) and dopamine transporter ligand [123I]2β-carbomethoxy-3β-(4-iodophenyl)tropane ([123I]β-CIT;Innis et al., 1993) and 2β-carbomethoxy-3β-(4-[18F]-fluorophenyl)tropane ([18F]CFT) (Rinne et al., 1999).
Positron emission tomography and SPECT are increasingly used in studying the progression of nigrostriatal dopaminergic hypofunction and the neuroprotection in PD. There are a few PET studies using FDOPA as a ligand (Vingerhoets et al., 1994; Leenders et al., 1998; Morrish et al., 1998; Nurmi et al., 1999). In these studies, the rate of annual decline in tracer uptake was approximately 8% to 10% in the putamen and 3.5% (Morrish et al., 1998) or 6.6% (Nurmi et al., 1999) in the caudate nucleus, or, using a different method of analysis, 1.7% in the total striatum (Vingerhoets et al., 1994). With [123I]β-CIT, an annual rate of decline of 11.8% was found in the total striatum (Marek et al., 1998), and a [18F]CFT PET study showed an annual rate of decline of the baseline mean of 13.1% in the putamen and 12.5% in the caudate nucleus (Nurmi et al., 2000).
In studying the progression in PD it is important to know the test–retest reproducibility of PET or SPECT measurements in the same individuals when doing serial scannings. In a study with [123I]β-CIT SPECT, the test–retest variability of striatal V3” was 16.8% ± 13.3% for PD patients and 12.8% ± 8.9% for healthy controls (Seibyl et al., 1997). The measure V3” is directly related to the binding potential defined as number of binding sites divided by the dissociation constant (Bmax/Kd).
Another important issue is medication because patients are usually without antiparkinsonian medication at the first scan, whereas patients are on various medications, primarily levodopa treatment, at the time of the subsequent scan. For ethical reasons, it is impossible to study the progression in PD for a longer period without allowing the patients their medication. With [123I]β-CIT SPECT, no significant difference among PD patients in dopamine transporter ligand uptake was found before and after 4 to 6 weeks treatment of L-dopa and carbidopa or L-selegiline (Innis et al., 1999). In a [123I]β-CIT SPECT study with baboons, infusion of a high dose of L-dopa had no effect on the dopamine transporter ligand binding (Laruelle et al., 1993).
The purpose of the current study was to evaluate the usefulness of a dopamine transporter PET ligand, [18F]CFT, in monitoring progression and treatment effects in PD. Therefore, the authors studied the effect of levodopa treatment on [18F]CFT uptake and the normal variation of test–retest [18F]CFT PET in healthy controls.
MATERIALS AND METHODS
Subjects
The authors studied 7 de novo patients with PD (6 women, 1 man). Patients were diagnosed and treated at the Department of Neurology, University of Turku, Finland. The patients had at least 2 of the 3 cardinal features of PD: resting tremor, rigidity, or hypokinesia. In addition, no atypical features (Hughes et al., 1993) were seen. The disability of the patients was evaluated by the motor part of the Unified Parkinson's Disease Rating Scale (UPDRS). The mean UPDRS motor score was 31.1 ± 4.8 (mean ± SD, range 23 to 38). The Hoehn and Yahr stage was two for all of the patients. The mean age of the patients was 66.6 ± 8.5 years (mean ± SD) with a range of 51 to 73 years, and the mean duration of the symptoms (±SD) was 2.5 ± 0.9 years (range 1.2 to 3.7 years) at the time of first PET scan. The patients were not taking antiparkinsonian medication at the time of the first PET scan. Levodopa treatment was started thereafter with a favorable effect. The patients were scanned twice, before medication and after three months of levodopa treatment. The patients were given levodopa and carbidopa or levodopa and benserazide and the dose of levodopa was raised individually, that is, 200 mg to 400 mg per day at the time of the second PET scan. The levodopa treatment was stopped at least 6 hours before the PET scan.
Eight healthy volunteer subjects (5 women, 3 men) with a mean age of 54.5 ± 9.3 (range 40 to 70) years and no history or evidence of neurologic or psychiatric diseases were evaluated in the reproducibility study. The subjects were scanned twice approximately 2.5 months apart.
In addition to the PET scan, all patients underwent a 1.5 Tesla MRI, which was interpreted by an experienced neuroradiologist and no abnormalities were found. The MRI scan was used in the region of interest analysis (see Regions of interest).
The current study was approved by the Joint Ethical Committee of Turku University and Turku University Central Hospital. A written informed consent form was signed by each subject.
Radiochemistry
[18F]CFT (or [18F]WIN 35,428) (Haaparanta et al., 1996) was synthesized by electrophilic fluorination of 2β-carbomethoxy-3β-(4-trimethylstannylphenyl)tropane (Bergman et al., 1997). The precursor was dissolved in 700 μL Freon-11 and 100 μL acetic acid, after which high specific radioactivity electrophilic [18F]fluorine was bubbled through the mixture. The [18F]CFT formed was isolated with preparative HPLC, the eluent evaporated, and the residue dissolved in a saline and phosphate buffer solution. The electrophilic [18F]fluorine was synthesized from [18F]fluoride according to the method developed at the authors' laboratory (Bergman and Solin, 1997). The radiochemical purity exceeded 98% and the specific radioactivity at the end of synthesis was 12.5 ± 3.1 GBq/μmol.
PET imaging
Positron emission tomography scans were performed with the GE Advance PET scanner (General Electric Medical Systems, Milwaukee, WI, U.S.A.) in the two-dimensional scanning mode. Briefly, the transaxial spatial resolution full width at half maximum at a radius of 10 cm in midplanes was approximately 5 mm axially and transaxially. The subjects were positioned in the scanner with three-dimensional laser alignment with reference to the orbito-meatal line. On average, at the first PET scan, 156 ± 27 MBq, and at the second PET scan, 144 ± 33 MBq of [18F]CFT was injected intravenously corresponding to a mean injected mass of 4.8 μg (SD = 0.8, range 3.4 to 6.1 μg). A 60-minute PET scan was performed 150 minutes after injection.
Regions of interest
The regions of interest were identified and drawn on the head of the caudate, anterior and posterior putamen, and cerebellum on both hemispheres on 1.5 Tesla MRI images that were matched with the PET images using the surface fitting method (Pellizari et al., 1989) and resliced according to PET images using trilinear interpolation. The root mean square misfit of MRI–PET fitting was 1.83 mm (Pellizari et al., 1989). The heads of the caudate and anterior and posterior putamen were located on two consecutive planes. The regions of interest were then copied onto the PET images that had been matched with PET images as described above. The average of the radioactivity concentrations of the anterior and posterior putamen and caudate nucleus in these two planes was then calculated. The cerebellum was used as reference in the analysis because it contains a negligible amount of dopamine and dopamine receptors. The uptake of [18F]CFT was calculated as the regioncerebellum: cerebellum ratio at 180 to 210 minutes (Laakso et al., 1998; Rinne et al., 1999). The means of the corresponding left and right hemisphere regions on interest values were averaged before statistical analyses were performed.
Statistics
Descriptive statistics, means, and standard deviations were used to assess subjects' age, duration of symptoms, and the different regions of interest (caudate and anterior and posterior putamen).
The intraclass correlation coefficients for each region were calculated for PD patients and healthy controls to evaluate the reliability of the replicated measurements. Standard deviations within subjects in both groups were estimated with a linear model of two repeated measurements (scan1 and scan2). In addition, percent changes calculated as the difference between scan1 and scan2 measurements divided by the scan1 measurement were used to quantify the magnitude of the variation within the subjects.
Repeated measurements analysis of variance with treatment as a between-factor and region as a within-factor was used to analyze the changes between scan1 and scan2 measurements. Bonferroni-adjustement was applied when necessary to overcome the multiplicity problem. All statistical analyses were performed with PROC MIXED in SAS (Version 6.12; SAS Institute, Cary, NC, U.S.A.) with subjects treated as a random effect. For the covariance structure, a compound symmetry was selected. The estimation was performed with the restricted estimation maximum likelihood method. A two-sided P value < 0.05 was considered statistically significant.
RESULTS
Effect of levodopa
In PD patients, the mean [18F]CFT uptake ratio was reduced in the anterior putamen to 42.5% (P < 0.0001), in the posterior putamen to 26.6% (P < 0.0001), and in the caudate to 67.3% (P = 0.001) from the control mean ratio. The absolute [18F]CFT uptake ratio values at the time of the first and second PET scan are shown in Table 1. In PD patients, the 3-month levodopa treatment had no significant effect on dopamine transporter function because the [18F]CFT uptake was not significantly changed in any striatal subregion between the two PET scans (P = 1.0 in the anterior and posterior putamen and in the caudate nucleus, after Bonferroni-adjustement to multiple comparisons). The intraclass correlation, indicating the reproducibility of the PET scan within subjects, was high at 0.97, 0.95, and 0.96 for the anterior putamen, posterior putamen, and caudate nucleus, respectively. The high intraclass correlation indicates that the variation within subjects is small compared with the variation between subjects. The percent change calculated as the difference between scan1 and scan2 measurements divided by the scan1 measurement ranged from 1.9% to 4.1%. In PD patients, the scan-to-scan intrasubject standard deviation varied from 3.7% to 6.3% (Table 1). Fig. 1 shows individual [18F]CFT uptake values in PD patients and controls at scan1 and scan2 in striatal subregions.
Reproducibility measurements of [18F]CFT uptake in the anterior and posterior putamen and caudate nucleus in patients with Parkinson's disease and controls
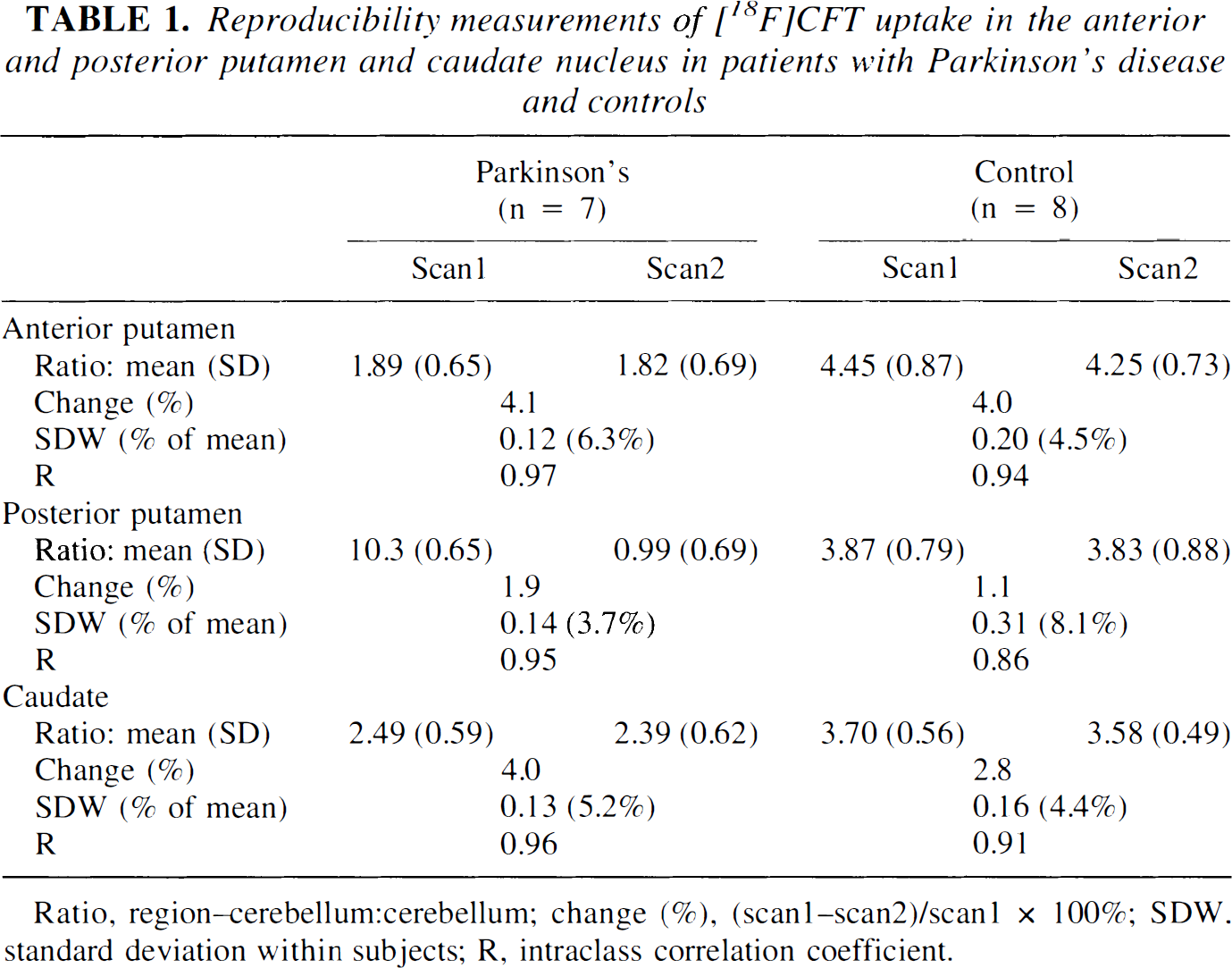
Ratio, region–cerebellum:cerebellum; change (%), (scan1–scan2)/scan1 × 100%; SDW, standard deviation within subjects; R, intraclass correlation coefficient.
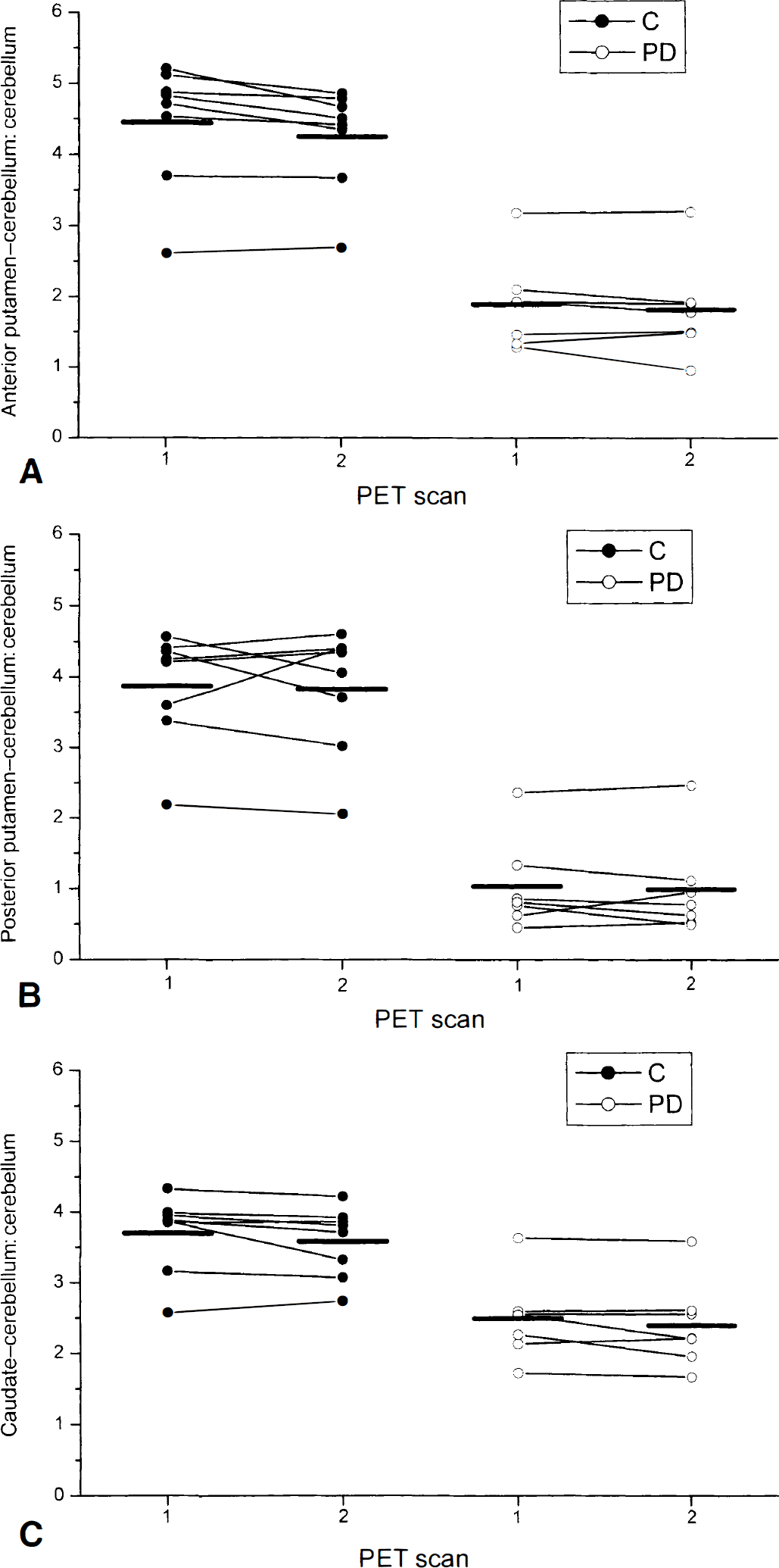
The [18F]CFT uptake values calculated as the region–cerebellum:cerebellum ratio at scan1 and scan2 in 7 patients with Parkinson's disease and 8 controls.
The mean motor UPDRS score decreased from 31.1 (SD = 4.8) at scan1 to 21.1 (SD = 5.0) at scan2 during the 3-month levodopa treatment. The difference (mean = 10.0, SD = 3.1) between the motor UPDRS scores at scan1 and scan2 was highly statistically significant (P = 0.0014, one-sample t-test).
Reproducibility
In healthy controls, there were no significant statistical differences in [18F]CFT uptake between the first and second PET scans in any striatal subregion (P = 0.09, 1.0, and 0.57 in the anterior putamen, posterior putamen, and caudate nucleus, respectively, after Bonferroni-adjustement to multiple comparisons). The reproducibility of [18F]CFT uptake values was also calculated using the intraclass correlation coefficient, which was 0.94 for the anterior putamen, 0.86 for the posterior putamen, and 0.91 for the caudate nucleus, indicating good reproducibility. The percent change calculated as the difference between scan1 and scan2 measurements divided by the scan1 measurement varied from 1.1% to 4.0%. In healthy controls, the within-subject standard deviation values ranged from 4.4% to 8.1% (Table 1).
The repeated measurements analysis of variance showed no significant difference in the change of [18F]CFT uptake ratios between the two scans between PD patients and healthy controls in any striatal subregion (P = 0.75). This further indicates that levodopa treatment had no significant effect on [18F]CFT uptake in PD patients.
DISCUSSION
The current study shows good reproducibility of dopamine transporter PET ligand [18F]CFT uptake measured by the region–cerebellum:cerebellum ratio. The high intraclass correlation coefficient found indicates that the variation within subjects is small compared with the variation between subjects. This was true for healthy controls and PD patients. Moreover, the small percent change between the two PET scans, and the low within-subject standard deviation (scan-toscan intrasubject standard deviation) indicate good reproducibility.
The authors found no significant effect of 3-month levodopa therapy on [18F]CFT binding in any striatal subregion in patients with PD. In a previous [123I]β-CIT SPECT study, PD patients were given a subchronic treatment of 4 to 6 weeks of L-dopa and carbidopa and no significant effect of L-dopa treatment on dopamine transporter ligand binding was found in the striatum (Innis et al., 1999). These findings in PD patients are in agreement with animal studies. In a [123I]β-CIT SPECT study with baboons, intravenous infusion of L-dopa had no acute effect on [123I]β-CIT binding in the striatum (Laruelle et al., 1993). In addition, in rats, 2 weeks of treatment with L-dopa and benserazide did not affect striatal dopamine transporter binding evaluated by [3H]CFT (Vander Borght et al., 1995). However, this finding in rats with intact nigrostriatal system does not necessarily reflect the situation in PD patients. The current results extend the previous findings by showing that even the 3-month levodopa therapy had no significant effect on dopamine transporter function studied by [18F]CFT PET. Taken together, these findings indicate that in studies of progression of PD the decline found in dopamine transporter ligand uptake is not because of the levodopa effect. However, it is possible that changes may take place even after 3 months of treatment; but this is unlikely because levodopa already would have achieved the steady-state level after 6 weeks of treatment (Innis et al., 1999). In addition, the levodopa dose that the authors' administered to their patients was small, but was titrated according to clinical response. The decrease in the mean motor UPDRS motor score (31.1 at baseline vs. 21.1 at scan2) indicates that the dose used was adequate. However, the possibility remains that high doses of levodopa may have some effect, although this is unlikely because even high-dose L-dopa (50 mg/kg intravenously) did not affect dopamine transporter binding in nonhuman primates (Laruelle et al., 1993).
One particular consequence of the unchanged [18F]CFT uptake on levodopa treatment is that the patients need not cease taking levodopa long before the scan. In the current study levodopa treatment was stopped six hours before the PET scan. The low variation from scan-to-scan in healthy controls and PD patients enables detection of even small effects of various drugs on dopamine transporter function.
In a SPECT study with [123I]β-CIT, uptake values showed a 12.8% test–retest variability of striatal V3” in healthy subjects (Seibyl et al., 1997). The corresponding figure for [18F]CFT PET found in the current study was considerably lower (varying from 1.1% to 4.0% in controls). There was a slight difference in the method for calculating the variability. In the [123I]β-CIT SPECT study (Seibyl et al., 1997), percent striatal uptake within subjects was measured as the absolute value of the difference of the test and retest measure divided by the mean of test and retest and expressed as a percentage. In contrast, in the current study, percent change was calculated as the difference between scan1 and scan2 measurements divided by the scan1 measurement. This small methodological difference can not explain the variation between these two studies because in the current controls the [18F]CFT uptake ratios were almost the same at the two PET scans. Thus, using either the mean of the scan1 and scan2 values or the scan1 value as a divisor, the result would essentially be the same. The better sensitivity and resolution of PET compared with SPECT, and also the use of anatomic reference (MRI) in the current study are factors contributing to this difference in the test–retest variability.
In previous studies of progression in PD, the annual rate of decline in dopamine transporter ligand uptake has been much greater than the test–retest variability found in the current study. In a [18F]CFT PET study, the annual rate of decline of the baseline mean was 13.1% in the putamen and 12.5% in the caudate nucleus (Nurmi et al., 2000). This is in agreement with the [123I]β-CIT SPECT study in which the annual rate of decline of tracer uptake was 11.8% in the total striatum (Marek et al., 1998). Thus, dopamine transporter ligands are sensitive both to detecting dopaminergic hypofunction in PD and to monitoring its progression.
The current findings indicate good reproducibility of measuring dopamine transporter function in normal controls and patients with PD. Moreover, levodopa treatment does not have a significant effect on dopamine transporter ligand binding in PD. Therefore, dopamine transporter ligands are feasible in studies of disease progression of PD.
Footnotes
Acknowledgements
The authors gratefully acknowledge the assistance of the staff of the Turku PET Centre.
