Abstract
A disturbance in redox balance has been implicated in the pathogenesis of a number of diseases. This study sought to examine the feasibility of imaging brain redox status using a 11C-labeled dihydroquinoline derivative ([11C]DHQ1) for positron emission tomography (PET). The lipophilic PET tracer [11C]DHQ1 was rapidly oxidized to its hydrophilic form in mouse brain homogenate. The redox modulators diphenyleneiodonium and apocynin significantly reduced the initial velocity of [11C]DHQ1 oxidation, and apocynin also caused concentration-dependent inhibition of the initial velocity. Moreover, [11C]DHQ1 readily entered the brain by diffusion after administration and underwent oxidation into the hydrophilic cationic form, which then slowly decreased. By contrast, apocynin treatment inhibited the in vivo oxidation of [11C]DHQ1 to the hydrophilic cationic form, leading to a rapid decrease of radioactivity in the brain. Thus, the difference in the [11C]DHQ1 kinetics reflects the alteration in redox status caused by apocynin. In conclusion, [11C]DHQ1 is a potential PET tracer for imaging of redox status in the living brain.
INTRODUCTION
Reactive oxygen species (ROS) serve as regulatory mediators in signaling processes at moderate concentrations, whereas high concentrations of ROS cause the disruption of redox homeostasis, resulting in nonspecific damage to proteins, lipids, and nucleic acids. 1 A disturbance in the prooxidant/antioxidant balance in favor of the former is called oxidative stress, 2 and has been implicated in the pathogenesis of several brain diseases such as Alzheimer's disease and Parkinson's disease. 3 In vivo imaging of brain redox status would be useful for elucidating pathologic conditions and aiding in diagnoses of brain diseases. Although chemiluminescence and fluorescence probes can measure ROS production in cells or tissues quantitatively,4,5 tissue absorption and scattering of photons prevent their translation to in vivo use. This is less of a problem with positron emission tomography (PET). However, few PET tracers have been reported for imaging brain redox status. In mouse heart, a 18F-labeled analog of hydroethidine, which is a fluorescent probe commonly used for detecting superoxide anion radicals, has recently been synthesized and shown promise as a PET tracer for imaging ROS. 6 [3H]Hydromethidine, an analog of hydroethidine, has also been synthesized and evaluated as a radiotracer to measure brain ROS production. 7 [3H]Hydromethidine showed the desired characteristic of a radical trapping tracer in mouse brain; however, the radiolabeling of hydromethidine with 11C has not been reported to date.
Reduced nicotinamide adenine dinucleotide (NADH) and reduced nicotinamide adenine dinucleotide phosphate (NADPH) are coenzymes involved in redox reactions. 8 1,4-Dihydropyridine and 1,4-dihydroquinoline are the analogs of NADH/NADPH and have been used as redox carriers in chemical delivery systems for delivery of therapeutic drugs to the brain.9–11 Similar to the oxidation of NADH or NADPH, 1,4-dihydropyridine and 1,4-dihydroquinoline derivatives are oxidized to the corresponding quaternary ammonium ions by biological redox systems. In contrast to previous tracers for detecting ROS, we hypothesized that such derivatives labeled with a PET nuclide could provide information on brain redox status by the mechanism shown in Figure 1A. A lipophilic PET tracer (in a reduced form) enters the brain across the blood-brain barrier (BBB) after intravenous injection. A portion of the tracer is oxidized to a hydrophilic metabolite (an oxidized form) depending on brain redox status, while the rest diffuses back into the blood. The metabolite is trapped or eliminated slowly because of its hydrophilicity. Hence, the brain radioactivity increases when oxidative stress occurs or oxidases activity increases. By contrast, enhancement of antioxidant defense systems or inhibition of oxidases involved in oxidative stress leads to the decrease in brain radioactivity. In this study, we synthesized 1-[11C]methyl-1,4-dihydroquinoline-3-carboxamide ([11C]DHQ1; Figure 1B) as a model compound of NADH/NADPH and examined the feasibility of imaging redox status in the brain using [11C]DHQ1.
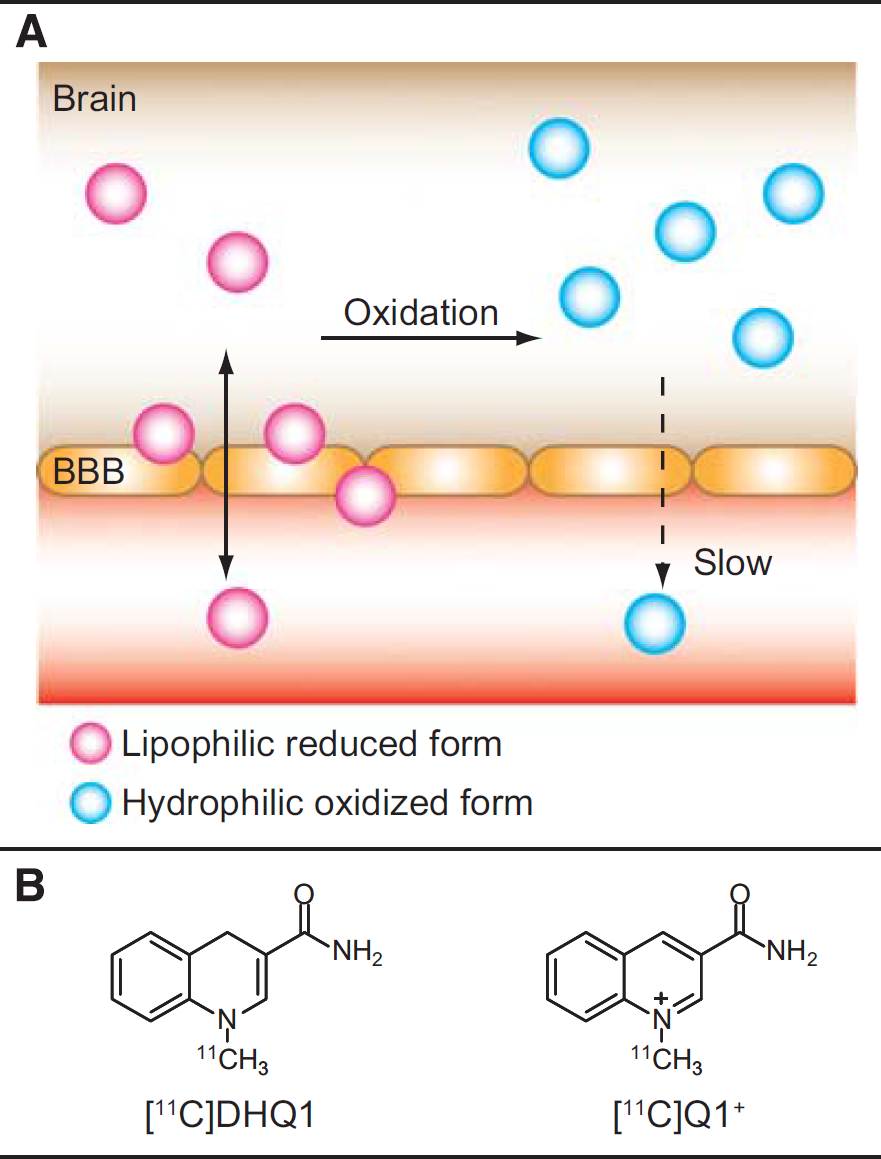
(
MATERIALS AND METHODS
General
All experiments were approved by the committee of National Institute of Radiological Sciences (Chiba, Japan). All commercially available reagents and solvents were used without further purification. Proton nuclear magnetic resonance (1H-NMR) spectroscopy was performed using a JEOL JNM AL-300 (300 MHz) spectrometer (JEOL, Tokyo, Japan) with chemical shifts reported in units of parts per million (p.p.m.). The purification of 1-methyl-1,4-dihydroquinoline-3-carboxamide and the analysis of radiochemical purities of 11C-labeled compounds were performed on a high performance liquid chromatography (HPLC) system consisting of a JASCO PU-2089 plus pump (JASCO, Tokyo, Japan), a multiwavelength detector (MD-2015 plus, JASCO), and a NaI(Tl) scintillation detector with an ACE Mate Amplifier and bias supply (925-SCINT, ORTEC, Oak Ridge, TN, USA) for radioactivity detection.
Apocynin was purchased from Tokyo Chemical Industry (Tokyo, Japan), and diphenyleneiodonium chloride (DPI) was purchased from Toronto Research Chemicals (Toronto, ON, Canada). β-Nicotinamide adenine dinucleotide phosphate, reduced form, tetrasodium salt (NADPH) was purchased from Oriental Yeast (Tokyo, Japan).
The mice were treated and handled according to the ‘Recommendations for Handling of Laboratory Animals for Biomedical Research’ compiled by the Committee on Safety and Ethical Handling Regulations for Laboratory Animal Experiments at the National Institute of Radiological Sciences. Experiments were performed according to the ARRIVE guidelines on animal research. Male C57BL/6J mice were purchased from Charles River Laboratories Japan (Yokohama, Japan). All the mice were kept under conventional laboratory conditions of temperature (23 °), humidity (50%), and light (12:12 h light/dark cycle), and allowed free access to food and water at our institute. Mice weighing 21 to 29 g (aged 8 to 13 weeks) were used throughout this study. The allocation of mice to the two study groups was performed by picking them at random from cages. Masking was not always done, and observer bias may have occurred.
Synthesis
Quinoline-3-carboxamide. This compound was synthesized by the hydrolysis of quinoline-3-carbonitrile in the same manner as described previously. 9 1H NMR (DMSO-d6, 300 MHz): δ 7.69 (1H, ddd, J =1.5, 7.0, 8.4 Hz), 7.69 (1H, br), 7.87 (1H, ddd, J =1.5, 7.0, 8.4 Hz), 8.08 (1H, dd, J =1.5, 8.4 Hz), 8.08 (1H, dd, J =1.5, 8.4 Hz), 8.31 (1H, br), 8.85 (1H, d, J = 2.2 Hz), 9.30 (1H, d, J =2.2 Hz); yield 35%.
3-Carbamoyl-1-methylquinolinium iodide (Q1+). This compound was synthesized by the reaction of quinoline-3-carboxamide with CH3I in the same manner as described previously. 9 1H NMR (DMSO-d6, 300MHz): δ 4.70 (3H, s), 8.13 (2H, m), 8.38 (1H, ddd, J =1.5, 7.0, 7.3 Hz), 8.56 (3H, m), 9.69 (1H, s), 9.91 (1H, s); yield 80%.
1-Methyl-1,4-dihydroquinoline-3-carboxamide (DHQ1). This compound was prepared following a modified literature procedure. 9 To a solution of 67 mg of Q1+ in 20 mL of phosphate-buffered saline (PBS) pH 7.4, 237 mg of NADPH was added, and the mixture was stirred under argon atmosphere at room temperature for 1 hour. Ethyl acetate (40 mL) was then added to the mixture resulting in the formation of a biphasic mixture, and the organic phase was separated from the aqueous phase. The aqueous phase was extracted with ethyl acetate (20 mL), and the organic phase and extractions were combined and dried over anhydrous Na2SO4. The solvent was removed in vacuo to give a crude product, which was purified by the HPLC system with a COSMOSIL 5C18-MS-II column (20 ID × 250 mm; Nacalai Tesque, Kyoto, Japan) and COSMOSIL 5C18-MS-II guard column (20 ID × 20 mm). The column was eluted with a mobile phase of H2O/CH3CN (60:40; v/v) at a flow rate of 6.0 mL/minute, and the peak from 15 to 16.5 minutes was collected. The solvent was then removed in vacuo to give DHQ1 (70%). 1H NMR (DMSO-d6, 300 MHz): δ 3.14 (3H, s), 3.61 (2H, s), 6.65 (2H, br), 6.79 (1H, d, J = 8.1 Hz), 6.88 (1H, dd, J = 7.0, 7.3 Hz), 7.00 (1H, d, J =7.0 Hz), 7.12 (1H, dd, J =7.3, 8.1 Hz), 7.14 (1H, s).
1-[11C]Methyl-1,4-dihydroquinoline-3-carboxamide ([11C]DHQ1). Carbon-11 was generated by the 14N(p, α) 11 C nuclear reaction using a CYPRIS HM-18 cyclotron (Sumitomo Heavy Industries, Tokyo, Japan). Preparation of [11C] CH3I and subsequent 11C-methylations were carried out automatically by using a synthetic apparatus for 11C-labeled compounds developed by Suzuki et al. 12 HPLC was performed using a JASCO HPLC system for radioactive runs, and effluent radioactivity from the HPLC was determined using a NaI (Tl) scintillation detector system. [11C]CH3I was trapped in 150 μL of anhydrous dimethylformamide (DMF) containing quinoline-3-carboxamide (1 mg) at − 10 °. The reaction vessel was heated at 160 ° for 5 minutes under a pressure of 0.167 MPa, and then DMF was removed in a stream of nitrogen at 200 °. After the reaction vessel was cooled to room temperature, 6.5 mg of NADPH dissolved in a solution of PBS (170 μL) and CH3CN (10 μL) was added to the residue, and the resulting mixture was kept at room temperature for 5 minutes. After an addition of 0.6 mL of H2O/CH3CN (7:1; v/v), the radioactive mixture was transferred into a COSMOSIL 5C18-MS-II column (10 ID × 250 mm; Nacalai Tesque) and eluted with H2O/CH3CN/25% ascorbic acid injection (350:150:1, v/v/v) at a flow rate of 4.5 mL/minute. The radioactive fraction corresponding to the desire product (retention time: 9 minutes) was collected in a rotary evaporator flask containing 5 μL of 25% ascorbic acid injection, evaporated in vacuo, and dissolved in 2.5 mL of PBS (pH 7.4). The radioactivity of 110 to 240 MBq of [11C]DHQ1 was obtained in a total synthesis time of 45 minutes after a 15-minute proton bombardment at a beam current of 20μA. The radiochemical yield was 1.6% to 3.7% (decay corrected to the end of bombardment). The identification and radiochemical purity of [11C]DHQ1 obtained after the formulation were determined by HPLC using an authentic sample of DHQ1. HPLC was performed on a Luna C18(2) column (3 μm, 4.6 ID × 50 mm; Phenomenex, Torrance, CA, USA) with a mobile phase of 50 mmol/L HCOONH4 (pH 6.5)/CH3CN (CH3CN content from 20% to 40% over 10 minutes) at a flow rate of 1.0 mL/minute. The retention time of the authentic sample was 5.0 minutes. The radiochemical purity determined by HPLC was greater than 96%. The specific activity of [11C] DHQ1 was determined by comparison of ultraviolet absorbance (342 nm) with that of known standards to be 120 to 360 GBq/μmol at the end of the synthesis.
3-Carbamoyl-1-[11C]methylquinolinium iodide ([11C]Q1+). Quinoline-3-carboxamide was methylated with [11C]methyl iodide as described above and purified as follows. After DMF was removed and the reaction vessel was cooled to room temperature, 0.6 mL of 10 mmol/L HCOONH4 (pH 6.5)/CH3CN (95:5, v/v) was added to the residue. The resulting radioactive mixture was transferred into a CAPCELL PAK C18 AQ column (10 ID × 250 mm; Shiseido, Tokyo, Japan) and eluted with 10 mmol/L HCOONH4 (pH 6.5)/CH3CN (95:5, v/v) at a flow rate of 4.5 mL/minute. The radioactive fraction corresponding to the desire product (retention time: 7.5 minutes) was collected in a rotary evaporator flask, evaporated in vacuo, and dissolved in 2.5 mL of PBS (pH 7.4). The radioactivity of 210 MBq of [11C]Q1+ was obtained in a total synthesis time of 40 minutes after a 15-minute proton bombardment at a beam current of 20 μA. The radiochemical yield was 3.2% (decay corrected to the end of bombardment). The identification and radiochemical purity of [11C]Q1+ obtained after the formulation were determined by HPLC using an authentic sample of Q1+. HPLC was performed on a multimode Scherzo SS-C18 column (4.6 I. D.×150 mm; Imtakt, Kyoto, Japan) with a mobile phase of 25 mmol/L HCOONH4 (pH 6.5)/CH3CN (65:35, v/v) at a flow rate of 0.9 mL/minute. The retention time of the authentic sample was 7.4 minutes. The radiochemical purity determined by HPLC was 94%. The specific activity of [11C]Q1+ was determined by comparison of ultraviolet absorbance (320 nm) with that of known standards to be 78 to 340 GBq/μmol at the end of the synthesis.
Measurement of Initial Velocity in Mouse Brain Homogenates
Immediately after [11C]DHQ1 synthesis, the concentration of total DHQ1 containing [11C]DHQ1 in [11C]DHQ1 solution was determined by the HPLC systems using the Luna C18(2) column as described above. The solution of [11C]DHQ1 was then diluted with PBS/25% ascorbic acid injection (49:1, v/v) so that the initial concentration of total DHQ1 in brain homogenate was 25 nmol/L.
A total of 12 mice were used for this experiment (n = 4 for DPI effect, n = 4 for apocynin effect, n = 4 for dependence of homogenate concentrations). Mice were decapitated, their brains were removed, and the cerebral cortices were dissected. The cerebral cortex was weighed and homogenized in PBS (pH 7.4) under ice cooling, and the homogenate was placed in tubes. Apocynin and DPI were dissolved in dimethylsulfoxide (DMSO). Three microliters of the concentrated inhibitor solution or DMSO alone was added to 297 μL of the homogenate and kept on ice until preincubation (for approximately 30 to 60 minutes). The homogenate containing the inhibitor solution or PBS was preincubated at 37 ° for 30 minutes. The solution of [11C]DHQ1 (20 μL) was then added to the homogenate or PBS and vortexed for about 1 to 2 seconds to initiate the reaction. At given time points after [11C]DHQ1 addition (typically 2,4,6, and 8 minutes), 40 μL of the homogenate was immediately added into 80 11L of ice-cold EtOH, vortexed for about 1 to 2 seconds, and then kept on ice until centrifugation. After the centrifugation (1 minute, 15,000 g, 4°), the supernatant was analyzed using the HPLC system equipped with a sensitive positron detector (Ohyo Koken Kogyo, Tokyo, Japan), and a Chromolith performance column, RP18-e (100×4.6 ID mm; Merck Millipore, Billerica, MA, USA), and guard column system for Chromolith performance RP18-e (5×4.6 ID mm; Merck Millipore). The column was eluted with a mobile phase of 50 mmol/L HCOONH4 (pH 6.5)/CH3CN (80:20, v/v) at a flow rate of 3.0 mL/minute. The retention times of [11C]Q1+ and [11C]DHQ1 were 0.7 and 2.3 minutes, respectively. The fraction of [11C] DHQ1 was calculated following the correction of the 1radiochromatograms for decay, and the concentration of DHQ1 at each time point was calculated on the basis of the fraction of [11C]DHQ1 and the initial concentration of DHQ1 (25 nmol/L) in the homogenate. Initial velocity (nmol/L per minute) was determined from the slope of the linear range of the DHQ1 concentration against time curve, and divided by homogenate concentration (g/mL) to yield initial velocity per unit homogenate concentration. The final concentrations were as follows: brain homogenate, 0.5% (w/v); DMSO, 0.94% (v/v); DPI, 0.1 mmol/L; apocynin, 0.01 to 10 mmol/L. For evaluation of the relationship between initial velocities and homogenate concentrations, the reaction was stopped at 2 and 4 minutes for 1% homogenate (w/v); at 2, 4, 6, 8,12, and 16 minutes for 0.25% (w/v); 2, 4, 6, 8, 16, and 32 minutes for 0.125% (w/v).
Before starting the assay, 20 μL of [11C]DHQ1 solution was added to 300 μL of PBS, and 40 μL of the solution was then added into 80 μL of ice-cold EtOH. The mixture (10 μL) was analyzed by the HPLC systems, and the fraction of [11C]DHQ1 was taken here to be that at 0 minute.
When the concentration of DHQ1 in the [11C]DHQ1 solution was low, the amount of [11C]DHQ1 solution that should be added to the homogenate was increased (~30 μL) to make 25 nmol/L of DHQ1.
Positron Emission Tomography Scanning Procedures
A total of 14 mice were used for PET scanning. Body temperature was monitored using a rectal thermometer throughout PET scanning and was maintained within the normal range using a heat lamp. Scans were performed with an Inveon Dedicated PET system (Siemens Medical Solutions, Knoxville, TN, USA), which has a transaxial field of view of 10 cm and an axial field of view of 12.7 cm. The tracers [11C]DHQ1 (10 to 11 MBq; < 0.061 nmol) or [11C]Q1 + (11 to 12MBq; < 0.14nmol) was administered as a 0.2 mL intravenous bolus injection to each mouse, which was maintained under isoflurane anesthesia during the scanning periods (n = 3 for each tracer). Data were acquired by the animal tomograph for 60 minutes after injection in 17 frames divided as follows: 4×1-, 2×5-, 2 × 5-, and 3 × 10-minute frames. The data were corrected for random coincidences, but not for scatter or attenuation, and reconstructed using Fourier re-binning and filtered back projection with a Hanning filter cutoff at the Nyquist frequency into images with a 128×128 matrix size and three times zoom, resulting in a voxel size of 0.26 × 0.26 × 0.8 mm3. For the reconstructed spatial resolution, this PET system can resolve the 1.75-mm rods of a Derenzo phantom. After image reconstruction, regions of interest were manually placed on a transverse view of summed PET images and transferred to all of the frames of images to generate time-radioactivity concentration (Bq/mL) curves for the cerebrum, using the ASIPro VM software (CTI Concorde Microsystems, Knoxville, TN, USA). The area under the curve (AUC) was calculated by the trapezoidal rule.
Apocynin was dissolved in DMSO and then diluted with the same volume of PBS. Mice (n = 4 for each group) received an intraperitoneal injection of the apocynin solution (200 mg/kg body weight, 0.2 mL/mouse) or vehicle 15 minutes before the intravenous administration of [11C]DHQ1. The tracer [11C]DHQ1 was intravenously administered to each mouse, and data were acquired by animal tomographs for 60 minutes after injection. Time-radioactivity concentration (Bq/mL) curves for the cerebrum were generated as described above.
To shorten the anesthesia duration for the mice, attenuation correction was not performed. This will result in an underestimation of uptake values. However, because the size of mice is very small, the magnitude of attenuation is less in mouse PET than in large animal PET. In addition, data were measured under the same bias conditions. It would not be so critical to compare data between the groups without attenuation correction.
A dose of [11C]Q1+ (0.14 nmol) is more than that of [11C]DHQ1 (0.061 nmol). However, the brain kinetics of 0.14 nmol of [11C]Q1+ was quite similar to those of 0.034 nmol [11C]Q1+ (data not shown). At a dose of 0.14nmol, the carrier would not affect the brain kinetics of [11C]Q1+.
Time Course of Radioactivity in Blood
A total of eight mice (n = 4 per group) received an intraperitoneal injection of apocynin in 50% DMSO in PBS (200 mg/kg body weight) or vehicle 15 minutes before the intravenous administration of [11C]DHQ1. Blood samples were collected at 5, 15, 30, and 60 minutes after injection of [11C]DHQ1 by pricking the lateral tail vein using a sterile needle under isoflurane anesthesia. The samples were weighed, and the radioactivity was measured with an automatic gamma counter (1480 Wizard 3” PerkinElmer, Waltham, MA, USA). The radioactivity in blood was expressed as the percentage of injected dose per gram tissue (% ID/g tissue). One blood sample of a control mouse (at 15 minutes) could not be collected.
Analysis of Chemical Form in the Brain
A total of eight mice (n = 4 per group, n = 2 per time point) received an intraperitoneal injection of apocynin in 50% DMSO in PBS (200 mg/kg body weight) or vehicle 15 minutes before the intravenous administration of [11C]DHQ1. Mice were decapitated 1 and 15 minutes after intravenous injection of [11C]DHQ1 (11 MBq in 0.2 mL PBS, < 0.06 nmol). The head was quickly immersed in liquid nitrogen, and the frozen brain was removed and weighed. The brain was immediately homogenized in 1 mL of MeOH under ice cooling and diluted with 20 mmol/L HCOONH4 (pH 6.5)/CH3CN (1:1, v/v) to make 5% w/v homogenate sample, which was then centrifuged at 15,000 g for 5 minutes at 4°. The recovery of radioactivity into the supernatant was >86%. An aliquot of the supernatant (350μL) to which a small amount of unlabeled Q1+ was added was analyzed using the HPLC systems equipped with the sensitive positron detector and a multimode Scherzo SS-C18 column (10 ID × 50 mm) and guard column system for Scherzo SS-C18 (8 ID × 10 mm). The column was eluted with a mobile phase of 20 mmol/L HCOONH4 (pH 6.5)/CH3CN (CH3CN content 60% for the first 1.5 minutes, and from 60% to 40% over 3.5 minutes) at a flow rate of 3.0 mL/minute. The recovery of radioactivity injected into the HPLC system was >95%. The retention times of [11C]DHQ1 and [11C]Q1 + were 1.5 and 4.0 minutes, respectively. The ratio of the components of the radioactive compounds was calculated after correcting the radiochromatogram for baseline noise and decay.
Statistical Analysis
Data distribution was assumed to be normal, but this was not formally tested. Statistical analyses were performed using Microsoft Excel 2003 (Microsoft, Redmond, WA, USA) with the add-in software Statcel 3 (OMS publishing, Saitama, Japan). Sample sizes were estimated using G*Power (G*Power 3.2.1, Franz Faul, University of Kiel, Kiel, Germany) and data from our pilot studies (a two-tailed, α = 0.05, 1 - β = 0.8). Differences between the two groups were analyzed by two-tailed unpaired Student's t-test, and the level of significance was set at 0.05. In the concentration dependence experiment, statistical significance was examined by one-tailed Williams' test, and P-values of < 0.025 were considered significant.
RESULTS
Stability of [11C]DHQ1 in Phosphate-Buffered Saline and Mouse Brain Homogenate
The time courses of the fraction of [11C]DHQ1 in PBS and mouse brain homogenate are shown in Figure 2A. The PET tracer [11C] DHQ1 was stable in PBS at 37 °, whereas it was rapidly converted to its hydrophilic oxidized form in mouse brain homogenate, and the disappearance was linear (R2>0.99) with time until less than 50% of [11C]DHQ1 was oxidized.
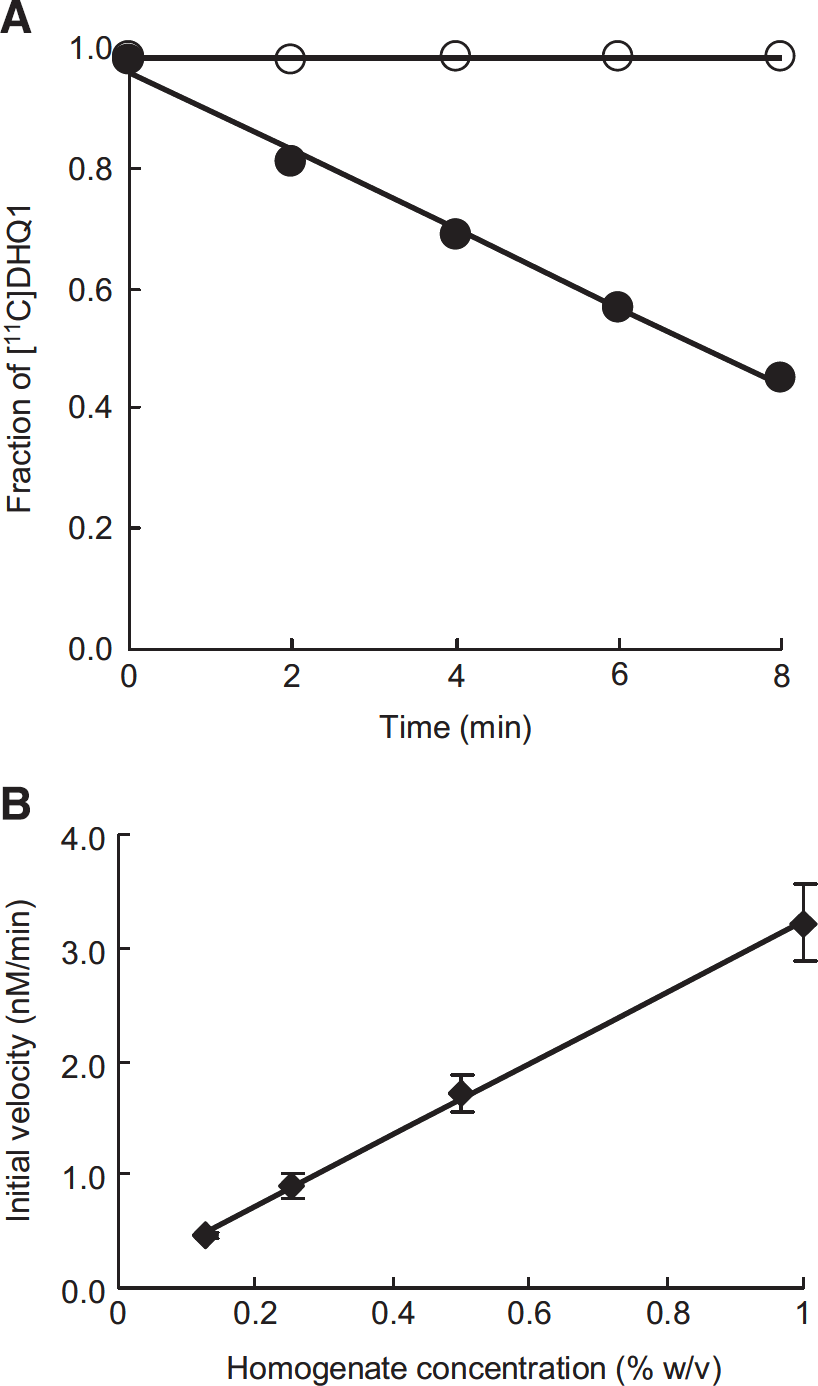
(
Figure 2B shows the effect of the homogenate concentrations on the initial velocity, which was determined from the slope of the linear range of the DHQ1 concentration against time curve. The initial velocity was linearly (R2>0.99) dependent on the homogenate concentrations over this range (0.125 to 1.0% w/v).
Effect of Diphenyleneiodonium Chloride and Apocynin on Oxidation of [11C]DHQ1 in Mouse Brain Homogenate
To confirm whether [11C]DHQ1 can respond to the alteration of redox status, we used DPI and apocynin to modulate redox status and examined their effect on the oxidation of [11C]DHQ1. Diphenyleneiodonium chloride at a concentration of 0.1 mmol/L caused approximately 50% reduction of the initial velocity: the initial velocities (nmol/L per minute per unit homogenate concentration) for control and DPI were 370±39 and 180±21 (mean±s.e.), respectively. As shown in Figure 3, apocynin also reduced the initial velocity in a concentration-dependent manner.
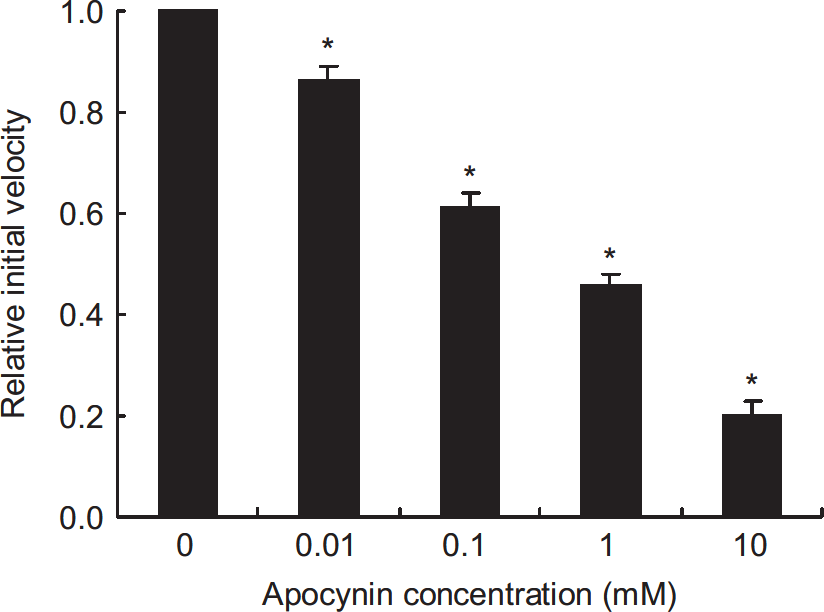
Effect of apocynin on initial velocity in mouse brain homogenate. Data are given as mean ± s.e. (n = 4, each concentration) and were analyzed using Williams' test. *P < 0.025, as compared with the control group. Apocynin (0.01 to 10 mmol/L) caused a concentration-dependent reduction of the initial velocity.
Blood-Brain Barrier Permeability of [11C]DHQ1 and [11C]Q1+
For imaging of redox status in the brain, the reduced form [11C] DHQ1 in blood must enter the brain across the BBB, whereas the oxidized form [11C]Q1+ in blood should not. The PET images and time-activity curves in the brain after injection of [11C]DHQ1 or [11C]Q1+ into mice are shown in Figures 4A and 4B. The radioactivity in the cerebrum after [11C]Q1 + injection was significantly lower than in the case of [11C]DHQ1 for 60 minutes: the AUC0–60 (% ID/mLxminute) of [11C]Q1+ and [11C]DHQ1 were 25 ±0.9 and 120±3 (mean±s.e.), respectively (Student's t-test, P=0.000008).
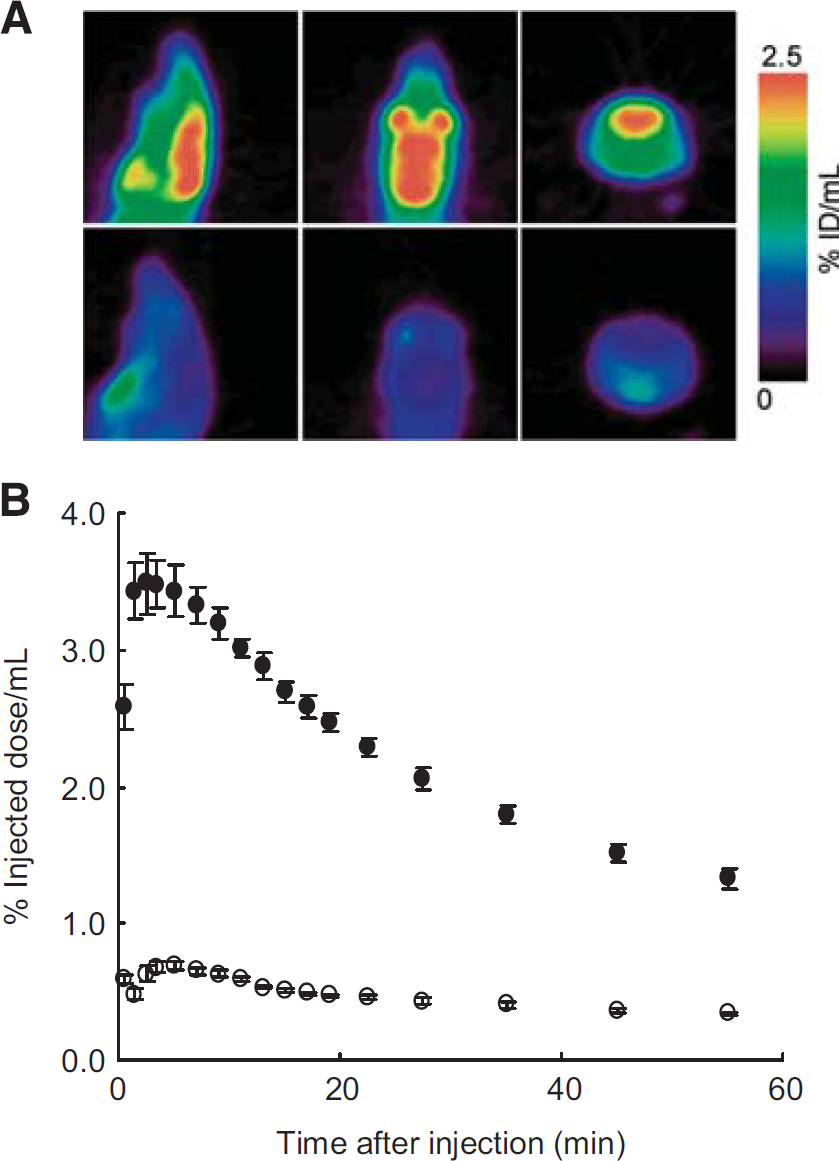
(
Effect of Apocynin on Brain Kinetics of [11C]DHQ1
Figures 5A and 5B show PET images and the time-activity curves in the brain after injection of [11C]DHQ1 into mice pretreated with apocynin or vehicle. Radioactivity in the cerebrum reached a maximum level after injection of [11C]DHQ1 and then slowly decreased, whereas pretreatment with apocynin caused a rapid decrease in brain radioactivity compared with control mice. In the early phase, the maximum uptake (% ID/mL) was similar between the two groups: 4.1 ±0.3 (mean±s.e.) for apocynin-treated mice and 3.8±0.1 for vehicle-treated mice. The AUC4–25 (% ID/mLx minute) of [11C]DHQ1 was 43±1 for the apocynin group and 66±3 for the control group (mean±s.e.), and significant difference in the AUC4–25 was evident between the two groups of mice (Student's t-test, P = 0.0002).
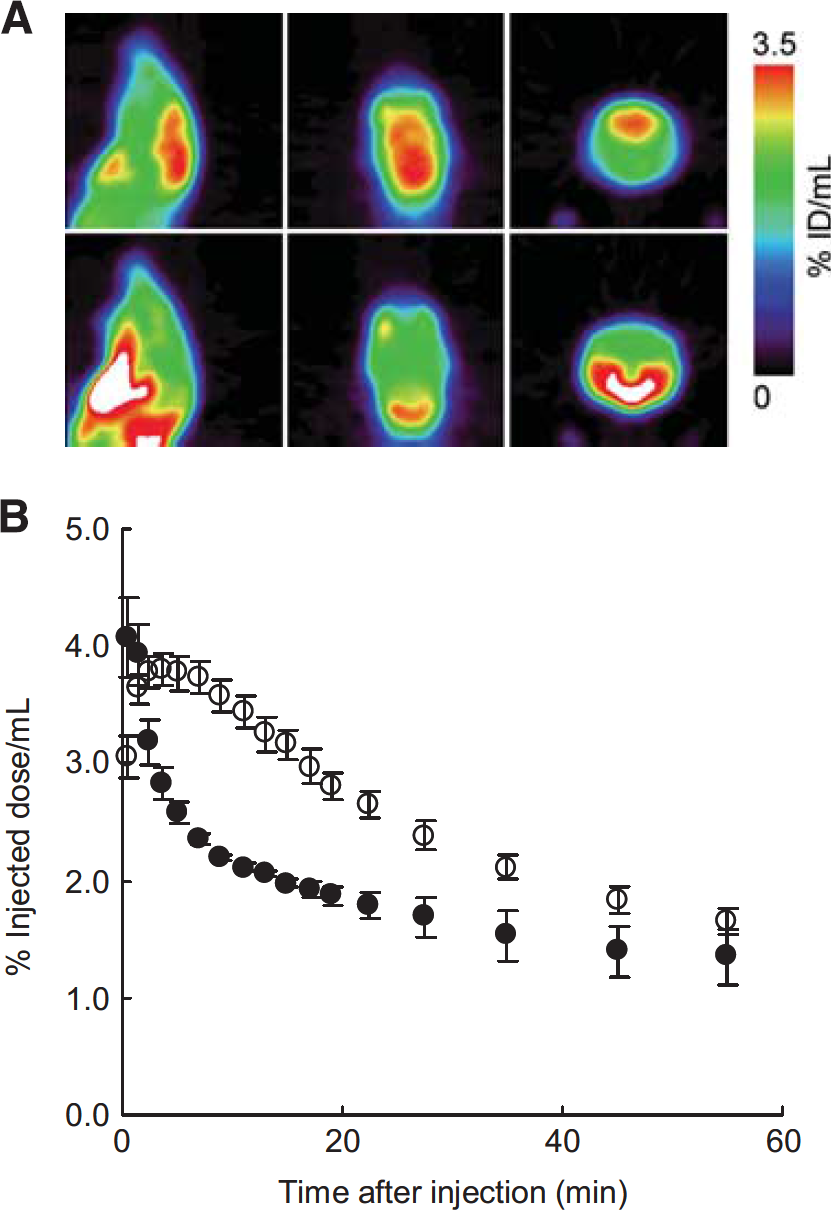
(
Figure 6 shows the radioactivity levels in blood after the intravenous administration of [11C]DHQ1 into mice pretreated with apocynin or vehicle. The radioactivity levels slowly decreased with time, and there were no significant differences in the blood radioactivity at each time point between the two groups of mice.
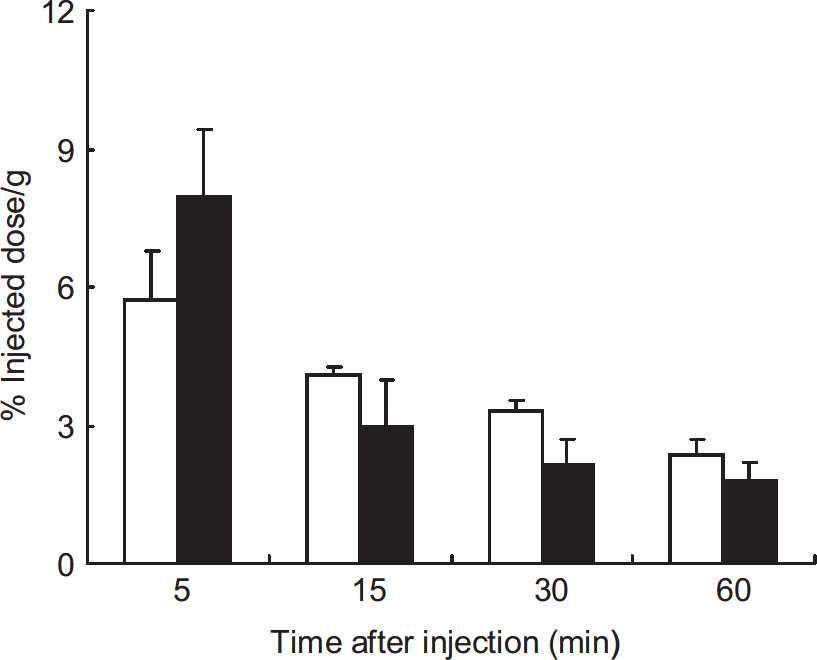
Time course of radioactivity in blood after intravenous injection of [11C]DHQ1 into mice pretreated with vehicle (white bars) or 200 mg/kg of apocynin (black bars). Data are expressed as mean percentage injected dose per gram tissue ±s.e. (n = 3 to 4 per time point) and were analyzed using Student's t-test. There were no significant differences in the blood radioactivity at each time point between the two groups of mice. [11C]DHQ1, 1-[11C]methyl-1,4-dihydroquinoline-3-carboxamide.
The chemical form of radioactive compounds in the brain 1 and 15 minutes after intravenous administration of [11C]DHQ1 were also analyzed. As shown in Figure 7, the reduced form [11C]DHQ1 disappeared from the brain tissue of vehicle-pretreated mice, and the main radioactive compound in the brain 1 and 15 minutes after injection was the oxidized form [11C]Q1+ (>89% of radioactivity). However, 65% (mean, n=2) of the radioactivity in the brain of apocynin-pretreated mice 1 minute after injection was associated with [11C]DHQ1, and 35% of that was present as [11C] Q1+. At 15 minutes after injection, the fractions of [11C]DHQ1 and [11C]Q1+ were 36% and 64%, respectively.
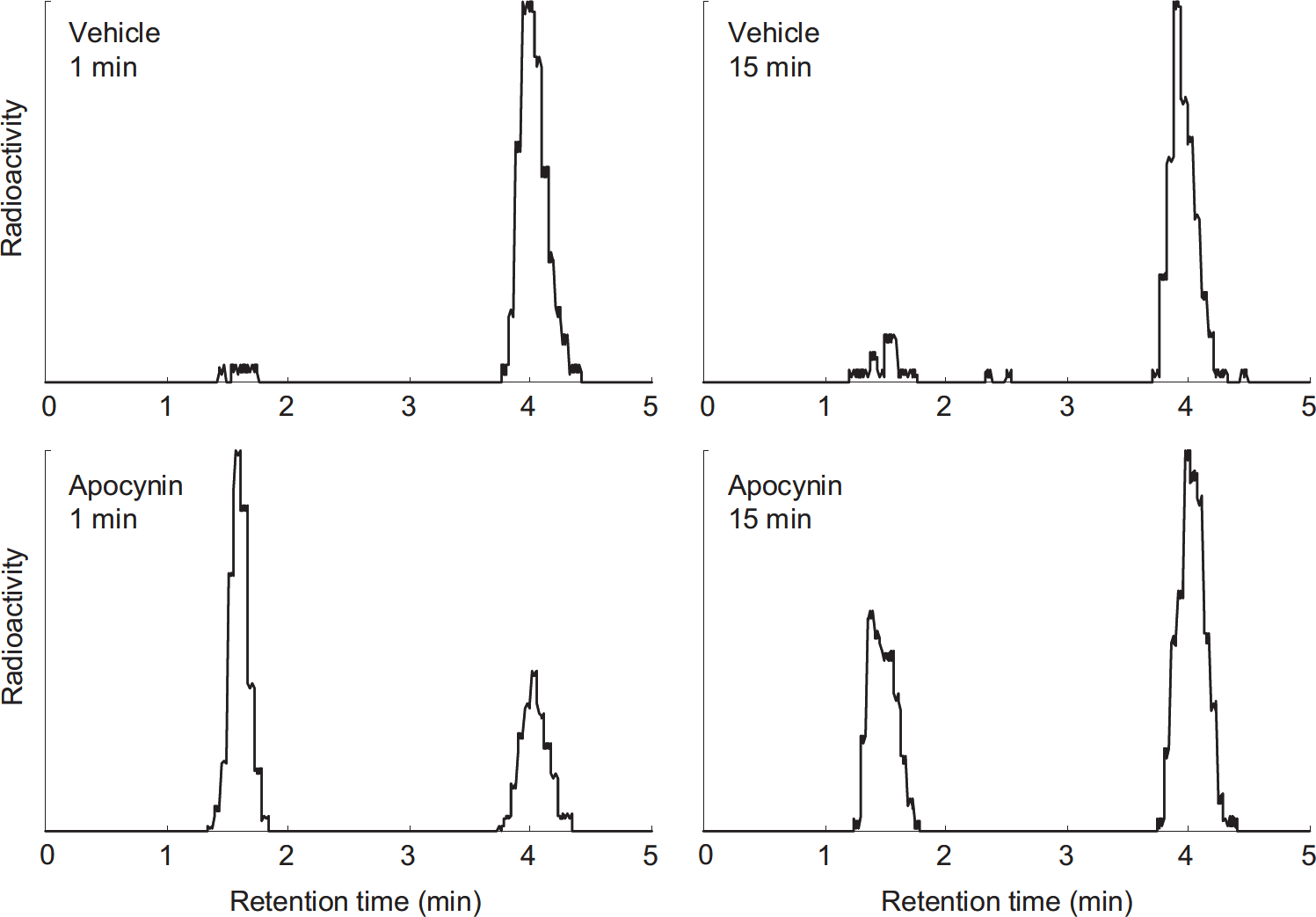
Typical chromatograms of radioactive compounds in the brain at 1 minute (left) or 15 minutes (right) after intravenous administration of [11C]DHQ1 into mice pretreated with vehicle (upper) or 200 mg/kg of apocynin (lower). The chromatograms were corrected for baseline noise and decay. The vertical axes are shown as relative values. The retention times of [11C]DHQ1 and [11C]Q1+ are 1.5 and 4.0 minutes, respectively.
DISCUSSION
For imaging of redox status in the brain by the metabolic trapping (Figure 1A), a tracer should be oxidized to its hydrophilic metabolite to some extent in the brain tissue, while also remaining stable in an aqueous solution for injection. In the NADH/NADPH molecule, 1,4-dihydronicotinamide moiety contributes to redox reactions, and 1,4-dihydronicotinamide may hence be used as a NADH/NADPH model compound. However, a limitation of 3-substituted-1,4-dihydropyridine carriers includes the susceptibility to hydration in acidic media. 9 Once hydration occurs, 1,4-dihydropyridine carriers are no longer oxidized. By contrast, 1,4-dihydroquinoline carriers are resistant to hydration. 9 In addition, unlabeled DHQ1 has been reported to be much more stable against chemical oxidation than the corresponding 1,4-dihydropyridine, 1-methyl-1,4-dihydronicotinamide.9,13 In this study, we synthesized [11C]DHQ1 as a model compound of NADH/NADPH and examined [11C]DHQ1 for its potential in the imaging of brain redox status.
Although unlabeled DHQ1 was relatively stable against chemical oxidation,9,13 the stability of [11C]DHQ1 against oxidation was not so high, and the oxidized product was observed after HPLC purification. The addition of ascorbate to the flask used for collecting [11C]DHQ1 was not enough to prevent the oxidation of [11C]DHQ1 in the preparation processes, and the radiochemical purity was less than 90% immediately after the preparation.
However, further addition of ascorbate to the HPLC eluent provided [11C]DHQ1 with high radiochemical purity (>95%), which remained stable in PBS for more than 90 minutes at room temperature. The [11C]DHQ1 solution used in this study contained ascorbate derived from the synthesis and preparation processes, and hence ROS would not critically affect the oxidation of [11C]DHQ1 in the mouse brain homogenate even if ROS were generated in the homogenate.
The PET tracer [11C]DHQ1 was stable in PBS at 37° (Figure 2), and the oxidized form was hardly observed even 90 minutes after incubation (data not shown), similar to room temperature. By contrast, [11C]DHQ1 disappeared linearly with time in mouse brain homogenate, and there was a linear relationship between the initial velocity and homogenate concentration. These results suggested that [11C]DHQ1 was oxidized by some enzymes to [11C]Q1+ in mouse brain homogenate. This tracer was also expected to be oxidized to some extent in mouse brain in vivo. In addition, we examined whether [11C]DHQ1 can respond to the alteration of redox status modulated by DPI and apocynin. These compounds inhibit enzymes that contribute to cellular redox balance and have been commonly used to inhibit ROS-producing NADPH oxidases. 14 Although apocynin and DPI have been shown to reduce ROS production or protect oxidative stress,15–20 reports have also shown that they can induce oxidative stress in glial cells.21,22 In this study, the initial velocity of [11C]DHQ1 was reduced to 50% at 0.1 mmol/L of DPI, although the effect of DPI concentration on the initial velocity was not examined because of the limited solubility at higher concentrations. Apocynin also caused the reduction of the initial velocity in a concentration-dependent manner (Figure 3). Thus, [11C]DHQ1 responded to the changes of redox status in mouse brain homogenate. A previous study has shown that unlabeled DHQ1 was stable in rat brain homogenate without any sign of oxidation within the time of experiment (6 hours), 9 whereas our study showed that [11C]DHQ1 was rapidly oxidized in mouse brain homogenate (Figure 2). This may be due to species differences in the activity of enzymes involved in the oxidation of DHQ1 between mouse and rat.
We examined the BBB permeability of [11C]DHQ1 and the oxidized form [11C]Q1+ by PET (Figures 4A and 4B). As expected, the brain radioactivity of the cationic compound [11C]Q1+ was much lower than that of [11C]DHQ1 for 60 minutes, demonstrating that [11C]Q1+ generated in peripheral tissues cannot enter the brain from blood. After intravenous injection, [11C]DHQ1 was distributed to the brain. The radioactivity in the brain of vehicle-treated mice was not constant but decreased after reaching a maximum level (Figure 5B). This appears to be because of either the elimination of the polar compound [11C]Q1+ or the reduction of [11C]Q1+ to [11C]DHQ1 that can diffuse back into the blood. However, the latter is unlikely because [11C]DHQ1 was hardly observed in the brain of vehicle-treated mice. In peripheral tissues, there seems to be some system that can reduce [11C]Q1+ back to [11C]DHQ1 (Supplementary Figure S1). In general, hydrophilic substances cannot cross the BBB, but [11C]Q1+ was moved from the brain into the blood in spite of the hydrophilicity. In the mouse brain, the elimination of [11C]Q1+ might thus be mediated by transporters such as P-glycoprotein or organic cation transporters. Given the analysis of chemical form (Figure 7), the rates of decline of radioactivity in the brain of vehicle- and apocynin-treated mice from peak to 15 minutes can be roughly considered the clearance rate of [11C]Q1+ and [11C]DHQ1, respectively. The rate of [11C]Q1+ was slower than that of [11C]DHQ1, and [11C]DHQ1 may, therefore, be used as the redox tracer on the basis of our model, although further chemical modifications, such that the oxidized form is trapped in the brain, will be needed.
We investigated whether [11C]DHQ1 can respond to the brain redox status altered by apocynin. Diphenyleneiodonium chloride was not used in the in vivo experiments as this inhibitor would not readily enter the brain across the BBB because of the cationic compound. The apocynin treatment resulted in the rapid clearance of radioactivity from brain compared with control mice (Figure 5). Although there were no significant differences in the blood concentration at any time point between the two groups of mice (Figure 6), the shape of the blood concentration in the apocynin group may not be the same as that in the control group, or the venous concentration may not be the same as the arterial concentration. These might affect the brain AUC (uptake) in addition to the oxidation rate of [11C]DHQ1 in the brain. However, the fraction of unchanged [11C]DHQ1 in the blood of vehicle-treated mice was much less than that of apocynin-treated mice at each time point (1, 5, and 15 minutes) as shown in Supplementary Figure S1. Despite this fact, the brain AUC for the control group was higher than that of the apocynin group. The brain AUC would thus reflect the difference in the oxidation rate of [11C]DHQ1 in the brain, rather than in the blood concentration between the two groups. Actually, [11C]DHQ1 mostly disappeared from the brain tissue of the control group after injection and remained largely intact in the brain of the apocynin group (Figure 7). Therefore, the rapid clearance of radioactivity for the apocynin group would be due to the inhibition of the oxidation of [11C]DHQ1 to the hydrophilic [11C]Q1+ in the brain followed by the rapid diffusion of the lipophilic [11C]DHQ1 out of the brain.
Similar to the oxidation of NADH/NADPH, we can expect the model tracer [11C]DHQ1 to be oxidized by biologic redox systems. The results of this study showed that the oxidation of [11C]DHQ1 would proceed enzymatically in the mouse brain. Under physiologic conditions, it is unlikely that [11C]DHQ1 will react with ROS in the brain because numerous antioxidant systems remove deleterious ROS, although we cannot exclude the possibility that [11C]DHQ1 reacts with ROS under pathologic conditions that produce excessive ROS. Hence, the imaging of redox status by this tracer may reflect the activity of some enzyme or several enzymes participating in redox balance, rather than the amount of ROS itself. Such enzymes include NADPH oxidases because the oxidation velocity and kinetics of [11C]DHQ1 were changed by the putative inhibitors for NADPH oxidases, DPI and apocynin.
In conclusion, this study has shown that [11C]DHQ1 is a potential PET tracer for imaging of redox status in the living brain. Oxidative stress has been recognized as a common underlying mechanism for many diseases such as neurodegenerative disorders, cardiovascular diseases, and cancer. In addition, reductive stress, characterized by an abnormal increase of reducing equivalents, has shown to have deleterious effects in mouse heart.23,24 In the brain, it has been suggested that scavenging ROS prevents hippocampal processes such as long-term potentiation and memory.25,26 It is important to noninvasively evaluate not only oxidative stress, but also an excessively reduced environment for the elucidation of various brain diseases. Brain redox status cannot be accurately evaluated by one method alone at present. However, a combination of [11C]DHQ1 and either biomarkers or other methods could allow for more accurate assessment of redox status, and be helpful for studying the relationship between the development or pathogenesis of brain diseases and oxidative/reductive stress.
Footnotes
TO designed the experiments, conducted the syntheses and in vitro and in vivo experiments, analyzed the data, and wrote the manuscript. MO, TK, and HW contributed to the PET studies and revising the manuscript. M-RZ supervised the study and contributed to revising the manuscript. All the authors gave final approval of the version to be published.
The authors declare no conflict of interest.
ACKNOWLEDGMENTS
The authors thank Mr M Ogawa, Mr N Nengaki, and Mr Y Kurihara (SHI Accelerator Service) for their technical assistance with radiosynthesis. The authors are also grateful to the technical team of the Cyclotron Section and Radiopharmaceuticals Section of the National Institute of Radiological Sciences for their support during cyclotron operation and the production of radioisotopes.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
