Abstract
Arterial spin labeling (ASL) is a noninvasive method to measure cerebral blood flow (CBF). Arterial spin labeling is susceptible to artifact generated by head motion; this artifact is propagated through the subtraction procedure required to calculate CBF. We introduce a novel strategy for mitigating this artifact based on weighting tag/control volumes according to a noise estimate. We evaluated this strategy (DVARS weighting) in application to both pulsed ASL (PASL) and pseudo-continuous ASL (pCASL) in a cohort of normal adults (N = 57). Application of DVARS weighting significantly improved test-retest repeatability as assessed by the intra-class correlation coefficient. Before the application of DVARS weighting, mean gray matter intra-class correlation (ICC) between subsequent ASL runs was 0.48 and 0.51 in PASL and pCASL, respectively. With weighting, ICC was significantly improved to 0.63 and 0.58.
INTRODUCTION
Arterial spin labeling (ASL) is a noninvasive functional magnetic resonance imaging (fMRI) technique that measures cerebral blood flow (CBF). Arterial spin labeling acquisition involves tagging arterial blood water, waiting for the blood to perfuse the slice of interest, and measuring the signal. The physical principal underlying ASL is observing the intensity difference between tagged and untagged images to infer CBF. The ASL-based CBF measurements approximate CBF estimates derived from positron emission tomography using radio-labeled water. 1 The favorable properties of ASL (e.g., no ionizing radiation exposure, superior temporal, and spatial resolution) motivate further methodological development. However, ASL, like most fMRI techniques, is sensitive to motion-induced artifact. 2
The problem of motion artifact and strategies to mitigate such artifacts have been extensively discussed in the context of blood oxygen level-dependent fMRI.3,4 Motion also impacts ASL-derived measurements of CBF but discussion of this issue has been limited.5,6 Extant strategies focus on removing signals presumed to be artifactual (e.g., residual motion artifacts and global signal 2 ) or removing frames contaminated by movement (i.e., frame censoring 5 ). However, the efficacy of these strategies has not been quantitatively evaluated.
The procedure used to calculate CBF makes ASL particularly susceptible to head motion artifacts. Arterial spin labeling, like blood oxygen level-dependent fMRI, is subject to artifacts induced by head motion (spin history effects). 7 These artifacts introduce noise, which corrupts the measurement and cannot be corrected by simple realignment. However, the calculation of CBF from ASL data involves subtracting subsequently acquired volumes. Thus, head motion over two frames contribute artifactual signal which exacerbates the confound.
Here, we report a technique that reduces the effect of motion by systematically discounting motion-contaminated frames. We demonstrate that this weighting scheme improves test-retest repeatability of ASL using both pulsed ASL (PASL) and pseudo-continuous ASL (pCASL) in a cohort of healthy individuals. Further, we demonstrate that noise weighting reduces the variance because of artifact in estimated CBF.
MATERIALS AND METHODS
Participants
Fifty-seven participants (34F/27M; mean age = 35 years old, standard deviation = 16 years) were included in this study. Each participant was cognitively normal and had no history of head injury with loss of consciousness greater than 30 minutes, major psychiatric disorders, or contraindications to scanning. The Human Research Protection Office at Washington University in St. Louis approved all the procedures and each participant provided written informed consent. All the procedures conformed to the ethical standards put forth by the Washington University in St. Louis Human Research Protection Office and the Declaration of Helsinki.
Image Acquisition
Participants underwent imaging with a Siemens 3T Trim Trio scanner equipped with a standard 12-channel head coil. A high-resolution three-dimensional, sagittal, MPRAGE (magnetization-prepared rapid gradient echo scan) T1 scan was acquired (TR (time to repeat) = 2,400 milliseconds, TE (time to echo) = 3.16 milliseconds, flip angle = 8°, 256×256 acquisition matrix, inversion time = 1,000 milliseconds, voxel size = 1×1 ×1 mm). A two-dimensional multi-slice oblique axial spin density/T2-weighted fast spin echo scan (TE = 450 milliseconds, TR = 3,200 milliseconds, 256 × 256 acquisition matrix, 1× 1 ×1 mm voxels) was also acquired for image registration. Pulsed ASL was acquired using a common Siemens sequence with PICORE Q2T (TI1/TI of 700/1,800 milliseconds, TR/TE of 2,600/13 milliseconds, voxel size = 3.4 ×3.4×6.0 mm with 1.5 mm slice gap, 64 × 64 acquisition matrix, 15 axial slices, and 90° flip angle). Two PASL runs each containing 63 volumes (duration of 2.7 minutes) were acquired. Pseudo-continuous ASL was also obtained (1.5 seconds labeling time,1.2 seconds post-labeling delay, TR of 3,500 seconds, TE of 9.0 milliseconds, 64× 64 acquisition matrix, 90° flip angle, 22 axial slices with a 1 mm gap, and voxel size of 3.4× 3.4 × 5.0). Two pCASL runs each containing 60 volumes (duration of 3.5 minutes) were acquired. Pulsed ASL and pCASL run pairs were acquired in immediate succession. Pulsed ASL preceded pCASL in all but one subject. There was approximately 5 minutes between PASL and pCASL acquisitions.
Preprocessing—Structural Data
Affine transforms were calculated to align each participant's T1 image to a common atlas space. This affine transform was combined with the transformation relating the T2 image to the T1 scan to enable alignment of the ASL data to a common space.
Preprocessing—Functional Data Alignment
The ASL data were aligned (6 degrees-of-freedom rigid body motion) within and across runs using a standard algorithm minimizing the squared intensity difference between input and reference images. Mean PASL and pCASL images (across each scan) were aligned to the T2 structural scan. The overall scheme achieves atlas transformation by a composition of affine transforms (EPI → T2w → MPRAGE → atlas). 8 The final preprocessing step generated volumetric time-series EPI data in 3 mm3 atlas space, combining head motion correction and atlas transformation in one resampling step.
Using the realignment time-series, we calculated the root sum squared frame displacement for the first and second run for PASL and pCASL separately. The mean and standard deviation frame displacement across subjects was 0.22 (0.2) for the first PASL run, 0.28 (0.2) for the second PASL run, 0.18 (0.2) for the first pCASL run, and 0.21 (0.2) for the second pCASL run. We subjected these frame displacement values to a run by sequence repeated measures analysis of variance and found no effect or run (F1,56 = 1.55,
Cerebral Blood Flow Calculation
Cerebral blood flow was calculated using a previously defined mathematical model.9–11 Variables and constants are defined in Table 1. Cerebral blood flow for PASL was calculated using a single blood-compartment model:
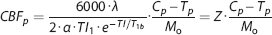
Model constants
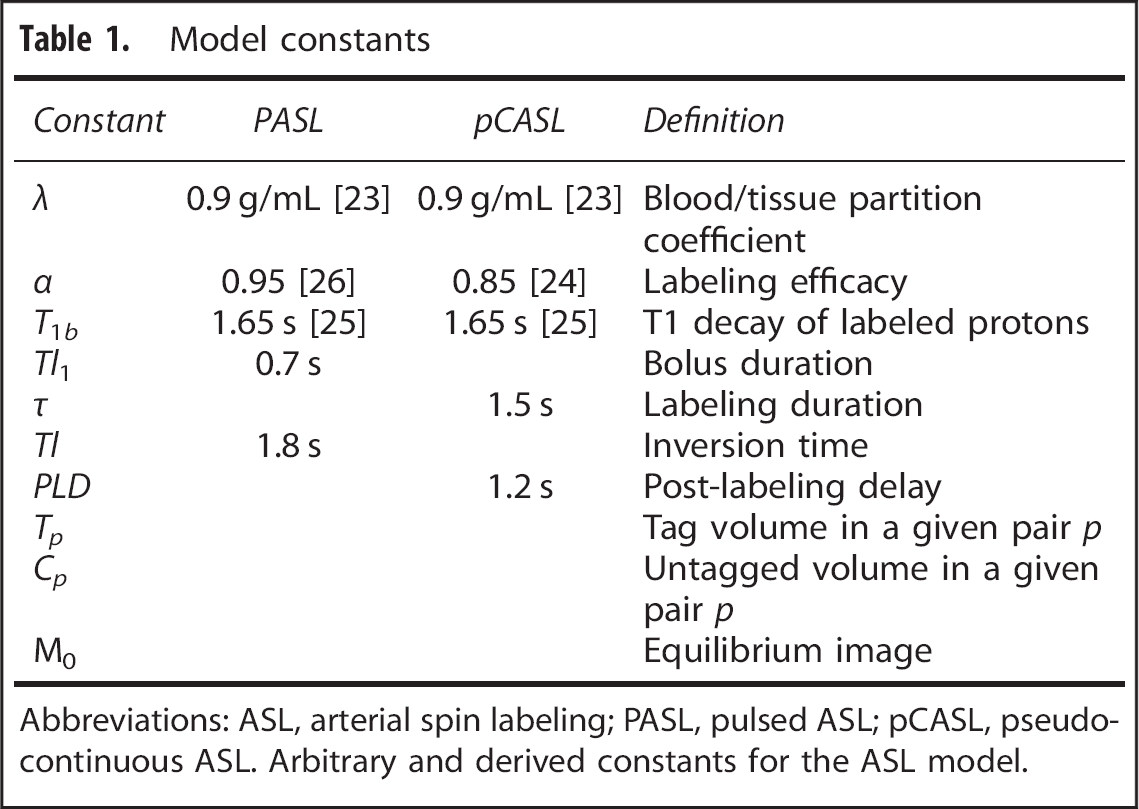
Abbreviations: ASL, arterial spin labeling; PASL, pulsed ASL; pCASL, pseudo-continuous ASL. Arbitrary and derived constants for the ASL model.
Similarly, 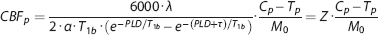
The expression parameterized as Z accounts for the serial acquisition of slices.
The sliced nature of the ASL acquisition used in this study introduces a time dependence on the axial slice number with slices closer to the vertex acquired at a later time than those closer to the base of the brain. Tagged blood decays over time. In participant space, correcting for this decay is a function of position along the axial dimension. However, alignment to atlas space disrupts this relationship. To compensate, an image was generated where each voxel contained the appropriate
Weighted Cerebral Blood Flow Calculation
Given multiple samples of the same quantity plus additive noise, the least squares estimate is achieved by averaging the individual samples weighted inversely in proportion to noise power.
12
Arterial spin labeling data are contaminated by multiple sources of noise, the most important being head motion.
5
In overview, weighting consists of computing a weighted mean of 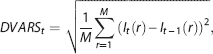
where
As 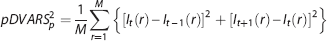
where 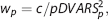
where
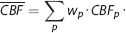
The first tag/control pair was excluded because
Statistics
The principal analytic question assessed here is the repeatability of measured blood flow across immediately successive measurements assuming that the mean CBF measured in both runs is identical. Any difference in calculated
RESULTS
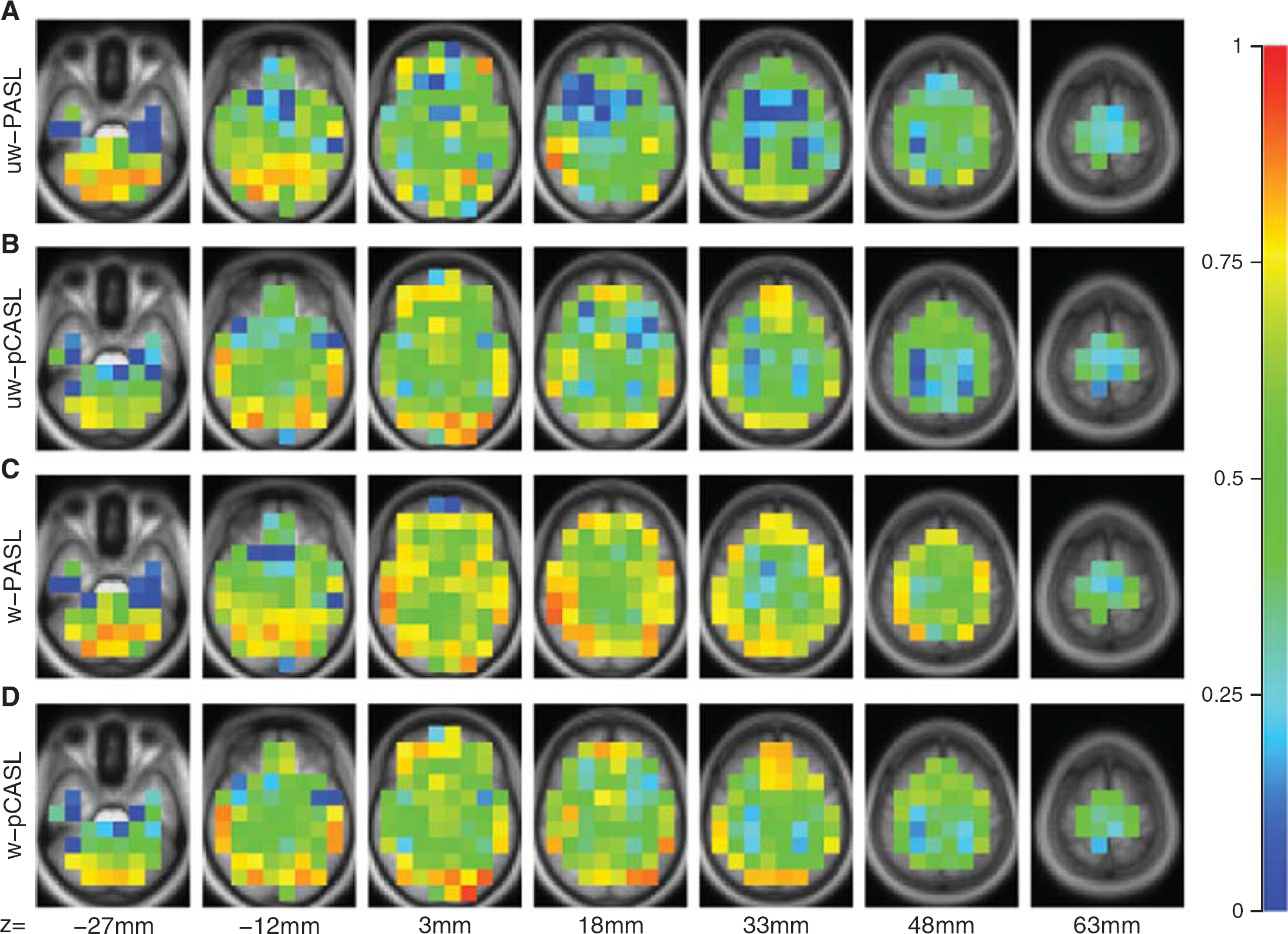
Group average CBF images derived from PASL and pCASL with and without DVARS weighting are shown in Figure 1. Pulsed ASL- and pCASL-derived
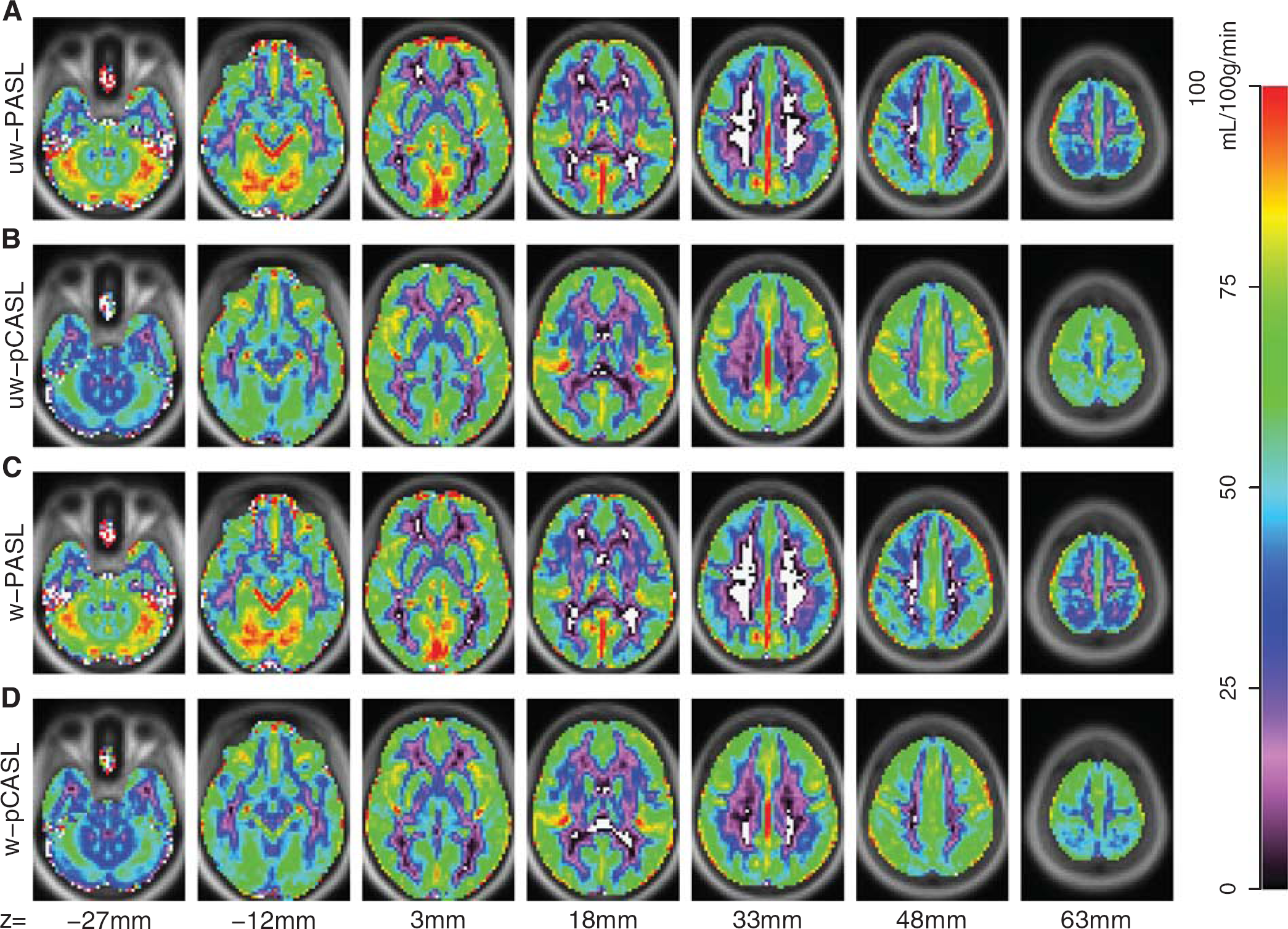
Arterial spin labeling (ASL) derived cerebral blood flow (CBF). Mean CBF from 57 participants is displayed for PASL and pCASL with and without DVARS weighting. (
DVARS-based weighting did not appreciably change mean computed
Weighting comparison
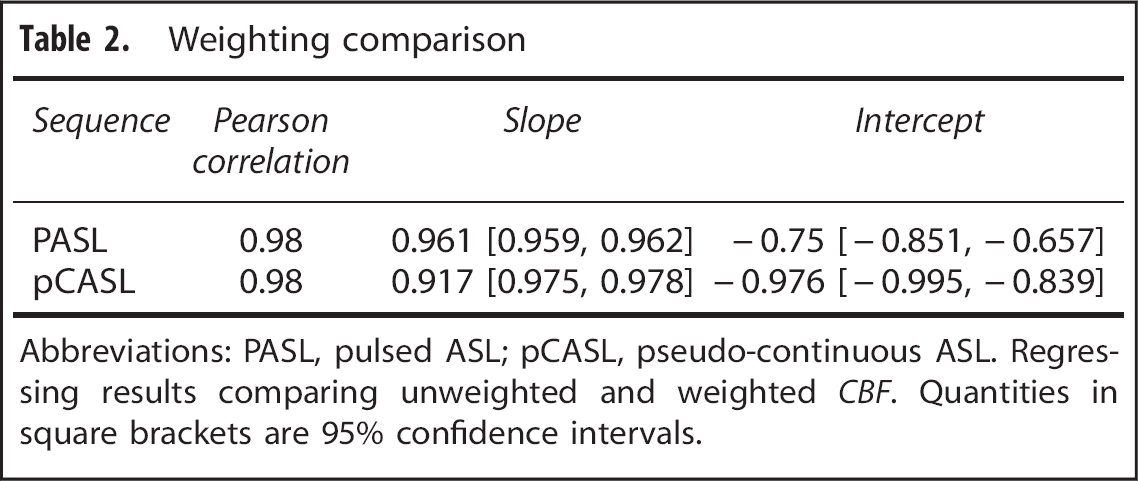
Abbreviations: PASL, pulsed ASL; pCASL, pseudo-continuous ASL. Regressing results comparing unweighted and weighted
Test–retest repeatability of each technique was quantified using
Intra-class correlation coefficient comparison
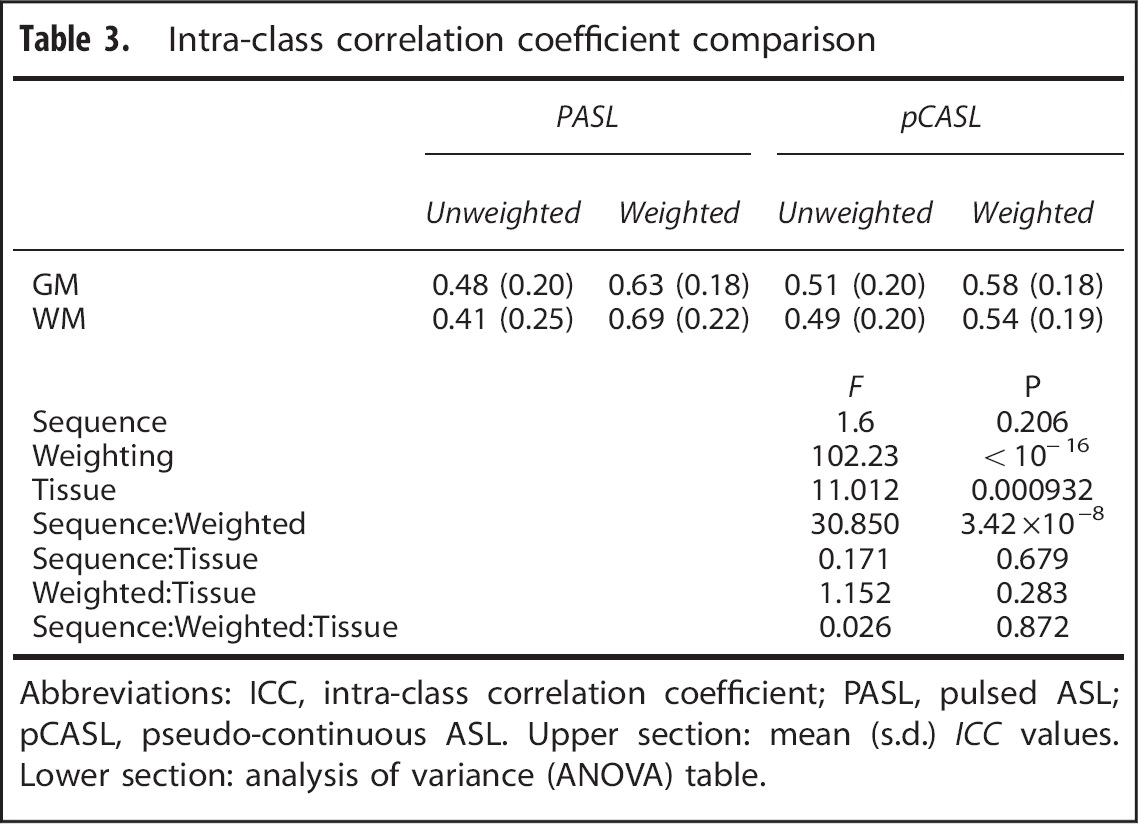
Abbreviations: ICC, intra-class correlation coefficient; PASL, pulsed ASL; pCASL, pseudo-continuous ASL. Upper section: mean (s.d.)
Intra-class correlation coefficient (ICC) calculated between duplicate ASL runs, evaluated in (15 mm3) cubes. Values near 1 indicate perfect repeatability. (
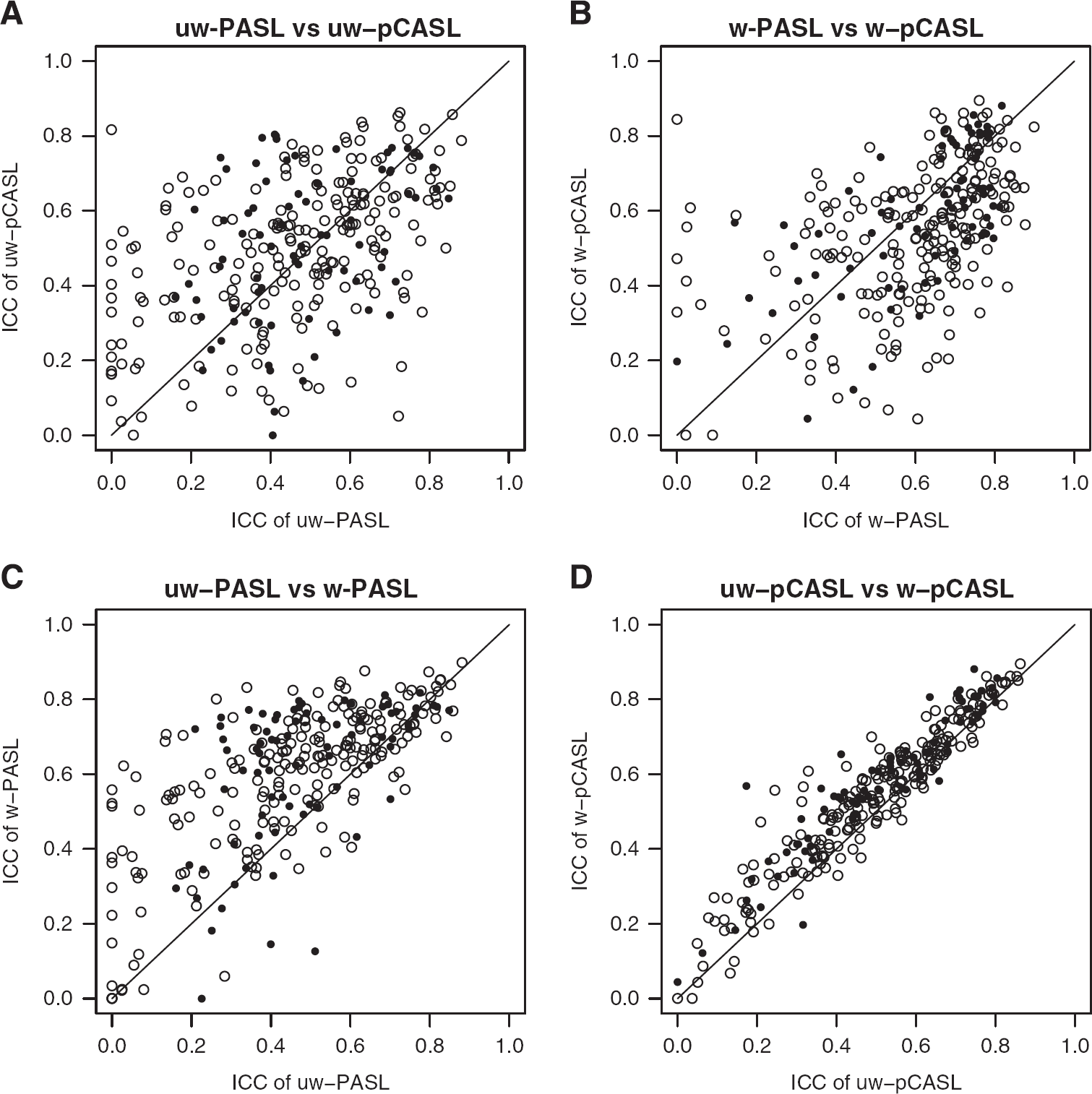
ICC comparisons. Paired ICC comparisons across conditions. (
DISCUSSION
We demonstrate that the DVARS-weighting approach described here is effective at improving the repeatability of ASL by reducing within-participant variance. Evidence derived from resting-state fMRI suggests that this variance is largely a consequence of head motion.4,15 Importantly, DVARS weighting of ASL data does not bias empirically calculated mean
Head Motion in Functional Imaging
It has been recognized for nearly two decades that movement corrupts blood oxygen level-dependent fMRI, the physical basis being the interaction of head motion with echo planar imaging (spin history effects). 7 This artifact manifests as spurious changes in reconstructed image intensity that cannot be corrected by simple realignment. Extant approaches for mitigating the effects of motion in blood oxygen level-dependent fMRI include regression of realignment time-series,2,8 censoring of movement-contaminated frames, 4 independent component analysis, 16 and regression of global signals.17,18 Reported approaches for reducing spurious noise in ASL include correcting for known sources of physiologic noise,19,20 low pass filtering subtracted image pairs, 21 suppression of background signal, 22 regression of nuisance time-series, 2 and removal of motion-contaminated frames. 5 Removal of contaminated frames is conceptually close to DVARS weighting. However, here we extensively characterize the effect of DVARS weighting in terms of ICC in a cohort of 57 normal adults.
SUMMARY AND CONCLUSIONS
We demonstrate that DVARS weighting of ASL data improves ICC, more so with PASL in comparison with pCASL. DVARS weighting does not significantly bias measures of CBF. Moreover, this technique is easily implemented and does not involve arbitrary constants (e.g., filter settings). Our results were obtained by systematically discounting frames with low SNR. Such a SNR-weighting approach improves mean estimates generally. In principle, the inverse SNR weighting is applicable to other ASL implementations (e.g., multiple post-labeling delays/inversion times or three-dimensional acquisitions), although the weighting scheme would have to be appropriately modified.
Footnotes
ABT contributed to the conception of the study, analyzed and interpreted the data, drafted and approved the final version of the manuscript. AZS contributed to the conception of the study, analyzed and interpreted the data, drafted and approved the final version of the manuscript. MRB contributed to the analysis and interpretation of the data, drafted and approved the final version of the manuscript. BMA conceived the study, oversaw data collection and analysis, interpreted the results, drafted and approved the final version of the manuscript.
The authors declare no conflict of interest.
ACKNOWLEDGMENTS
The authors acknowledge Danny J.J. Wang for helpful discussions and Elizabeth Westerhaus for data collection.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
