Abstract
The human brain changes structurally and functionally during adolescence, with associated alterations in cerebral perfusion. We performed dynamic arterial spin labeling (ASL) magnetic resonance imaging in healthy subjects between 8 and 32 years of age, to investigate changes in cerebral hemodynamics during normal development. In addition, an inversion recovery sequence allowed quantification of changes in longitudinal relaxation time (
Keywords
INTRODUCTION
The transition from childhood, through adolescence and into adulthood, marks a period of structural and functional change in the normal development of the human brain. The cerebral cortex undergoes its most dynamic structural change in adolescence, 1 and previous studies have indicated that this is accompanied by a further rapid change in cerebral perfusion (subsequent to the dynamic changes that occur in the first year of life), which occurs between the ages of 10 to 20 years. 2
The nonionizing nature of magnetic resonance imaging (MRI), coupled with its superior spatial resolution, makes it an attractive technique for studying the developing physiology of the brain. Although metabolism is not measured directly with MRI, cerebral perfusion can be quantified, which under physiologic steady-state conditions is coupled to the level of cerebral oxygen and glucose consumption, 3 and as such mirrors the brain's metabolic demand and neuronal function.
Traditionally, MRI-based perfusion techniques have involved serial imaging after the injection of a paramagnetic contrast agent, to dynamically track the passage of a labeled bolus through the vasculature. However, the injection of an exogenous contrast agent limits the applicability of these techniques in longitudinal studies, and precludes their use in patients with renal failure, due to the associated risk of nephrogenic systemic fibrosis. 4 Its application in children has also been limited due to reduced patient comfort, and technical difficulties in administering the intravenous contrast agent.
Arterial spin labeling (ASL) is an emerging alternative MRI technique for fully noninvasive quantification of cerebral blood flow (CBF).5,6 Although the signal-to-noise ratio (SNR) is inherently lower than measurements acquired using injected paramagnetic contrast agents, ASL has the advantage that it uses an endogenous tracer, by magnetically labeling water in the arterial blood supply. As such, it is a completely noninvasive and nonionizing technique, allowing for safe repeated measurements and increased patient comfort. Furthermore, ASL provides measurements of CBF in fully quantitative physiological units (such as mL blood/100 g tissue per minute), and as such is well suited for use in longitudinal or multicenter studies. These combined benefits have seen ASL move from the field of research into routine clinical practice in recent years.7,8
Arterial Spin Labeling Perfusion Quantification
Perfusion quantification using ASL is performed by inverting the longitudinal magnetization of the arterial blood flowing into the tissue, waiting for a given ‘inflow time’ (TI), then acquiring an image (known as the
A source of potential systematic error when estimating CBF using ASL is the delay between the application of the label and the arrival of the labeled bolus of blood at the imaging voxel (known as the bolus arrival time, BAT). Spatial variations in BAT will result in a variable amount of tagged blood having been delivered at the time of the acquisition (TI), and failure to account for this can lead to significant errors in CBF quantification.
A second source of error is the ‘through-flow’ effect, which occurs when the acquisition is made at an early TI, leading to an overestimation in CBF, as the d
A simplified kinetic model was introduced by Buxton
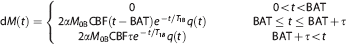
where
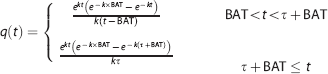
and

Here, α = inversion efficiency,
If measurements are made at multiple TIs, then the d
A further point to note is that, for accurate quantification of CBF,
Due to the rapid alterations in CBF that occur during the transition from late childhood into adulthood,
2
it is important that age-related changes in both the dynamic ASL signal and the
MATERIALS AND METHODS
Subjects
Approval to conduct this study was obtained from the Research Ethics Committee at Great Ormond Street Hospital. All healthy subjects provided written informed consent, in accordance with our institutional ethical review board. A total of 30 healthy subjects (7 males and 9 females below 18 years, 7 males and 7 females above 18 years) were studied, aged between 8 and 32 years (mean 18.5 years). In addition, ASL was performed in conjunction with the standard clinical imaging protocols in use in our institution in two patients. The first was a 17–year-old Sturge–Weber patient with a right pial angioma, who was experiencing symptomatic focal epilepsy, migraine headache, learning difficulties, and intracranial hypertension. The second was a 12–year-old patient with homozygous SCA with no evidence of neurologic abnormality seen on T2-weighted MRI. Blood taken in this patient 6 weeks before MRI revealed a hemoglobin level of 8.6g/dL (normal range: 11.5 to 15.5 g/dL) and hematocrit of 23% (normal range: 35% to 45%). Peripheral blood oxygen saturation was taken as 96% on the day of MRI.
Magnetic Resonance Imaging
All experiments were performed on a 1.5–T Siemens Magnetom Avanto scanner (Siemens, Erlangen, Germany), equipped with 40 mT/m gradients and a 12 channel head receive coil. Arterial spin labeling data were acquired using a flow-sensitive alternating inversion recovery pulsed-ASL sequence, with 3D single shot GRASE data acquisition,
21
with the following imaging parameters: repetition time = 3.0 seconds, echo time = 31.6 ms, 8 averages, field of view = 230 mm, matrix size = 64 × 64, 20 contiguous slices with 5 mm thickness. Measurements were made at six TIs, ranging from 0.2 to 2.2 seconds in 0.4-second intervals, with a total scan time of 4.8 minutes. Background suppression of static tissue was used, as described by Günther
Postprocessing
All data analysis was performed using Matlab (MathWorks Inc., Natick, MA, USA), and all model fitting was performed using an iterative Nelder–Mead nonlinear least squares algorithm.
First, the GM in the ASL and T1-mapping images was segmented into major vascular territories, using the flow territory templates established by Tatu
After image segmentation, regional values of
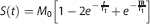
with
Reference Range Regions
Using the method described by Royston, 24 data from all healthy subjects were used to calculate both correlations and reference ranges for normal values of each fitted parameter as a function of age. This technique provides an age-dependent mean value and a ‘normal range’ for a given parameter, accounting for the fact that both the mean and the standard deviation of the raw data may vary with age. Data were log transformed where necessary to ensure normality of the residuals, and a backwards stepwise regression method was used, starting with a second-order polynomial, then reducing the degree to a first-order polynomial (i.e., linear fit), provided a significant worsening of the fit was not observed (details in ref. 24). All reference ranges were calculated with 95% confidence intervals.
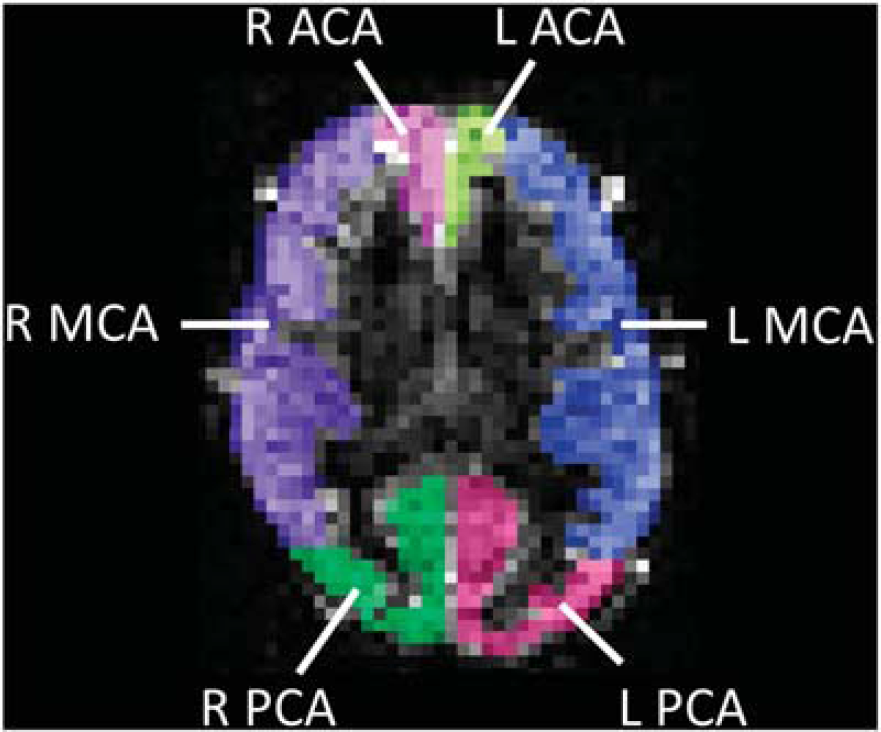
Example of arterial spin labeling (ASL) image segmentation, based on vascular flow territories. An axial ASL difference image (d
Statistical Analysis
All comparisons between correlations were performed using an analysis of covariance (ANCOVA) test, followed by a Tukey-Kramer
RESULTS
In all ROIs, none of the fitted parameters showed a significant difference in their correlation with age as a function hemisphere (
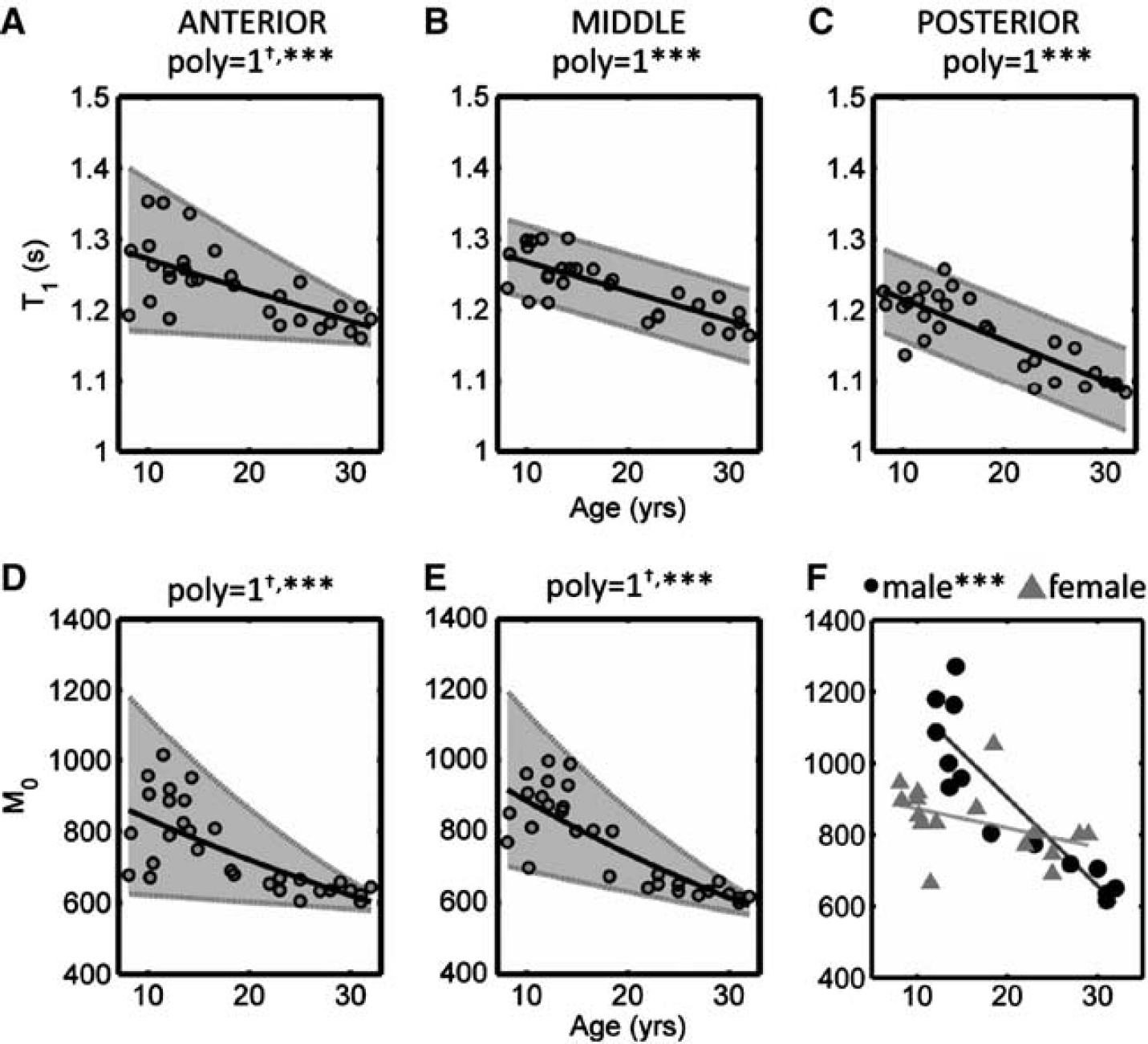
T 1/M 0 Values
The variation in
dM max/TImax Values
The variation in d
Bolus Arrival Time, CBF, and T Values
The variation in BAT, CBF, and T as a function of age is shown in Figure 4. Fitted values of BAT showed no correlation with age in all vascular territories, and the PMM value in the PCA (0.53 seconds) was significantly higher than that found in the ACA (0.40 seconds) and MCA (0.41 seconds) territories. Cerebral blood flow showed a negative correlation with age in all regions, which was particularly strong in the ACA territory (
Variation in fitted values of
The raw values for the mean curves, and upper and lower reference range limits, for all fitted parameters shown above, are given in the Supplementary Material.
Examples of the dynamic ASL signal, calculated using equation (1) and incorporating the mean, age-matched values of BAT, CBF,
Clinical Example in a Sturge–Weber Syndrome Patient
Figure 6 shows example
Clinical Example in a Neurologically Asymptomatic Sickle Cell Anemia Patient
Figure 7 shows example
DISCUSSION
Healthy Subjects
Our CBF measurements in normal GM agree well with a number of previous ASL studies. For instance, in a cohort with a mean age of 11 ± 3 years (mean ± s.d.), Jain
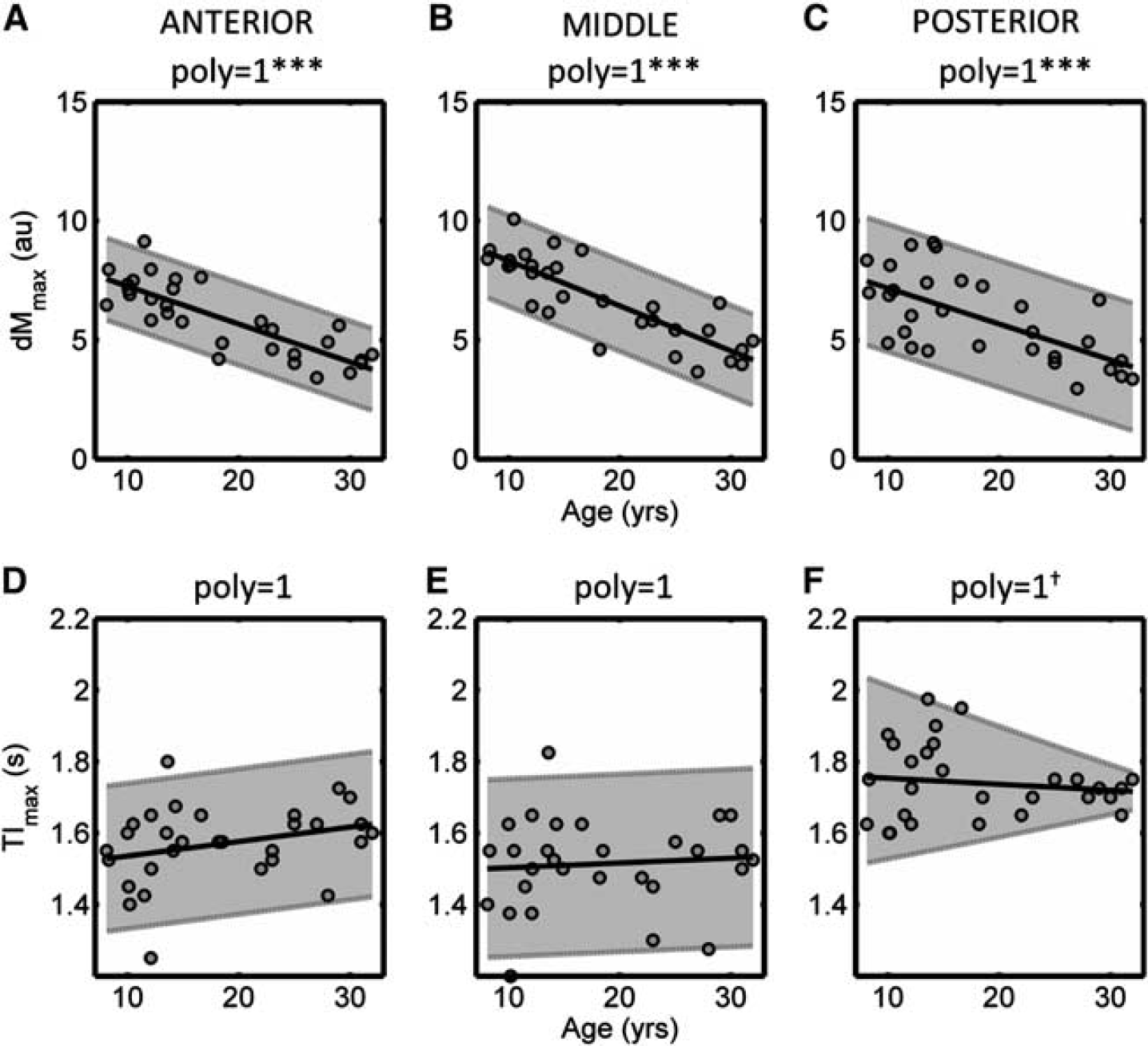
Variation in the magnitude of the peak arterial spin labeling (ASL) signal (d
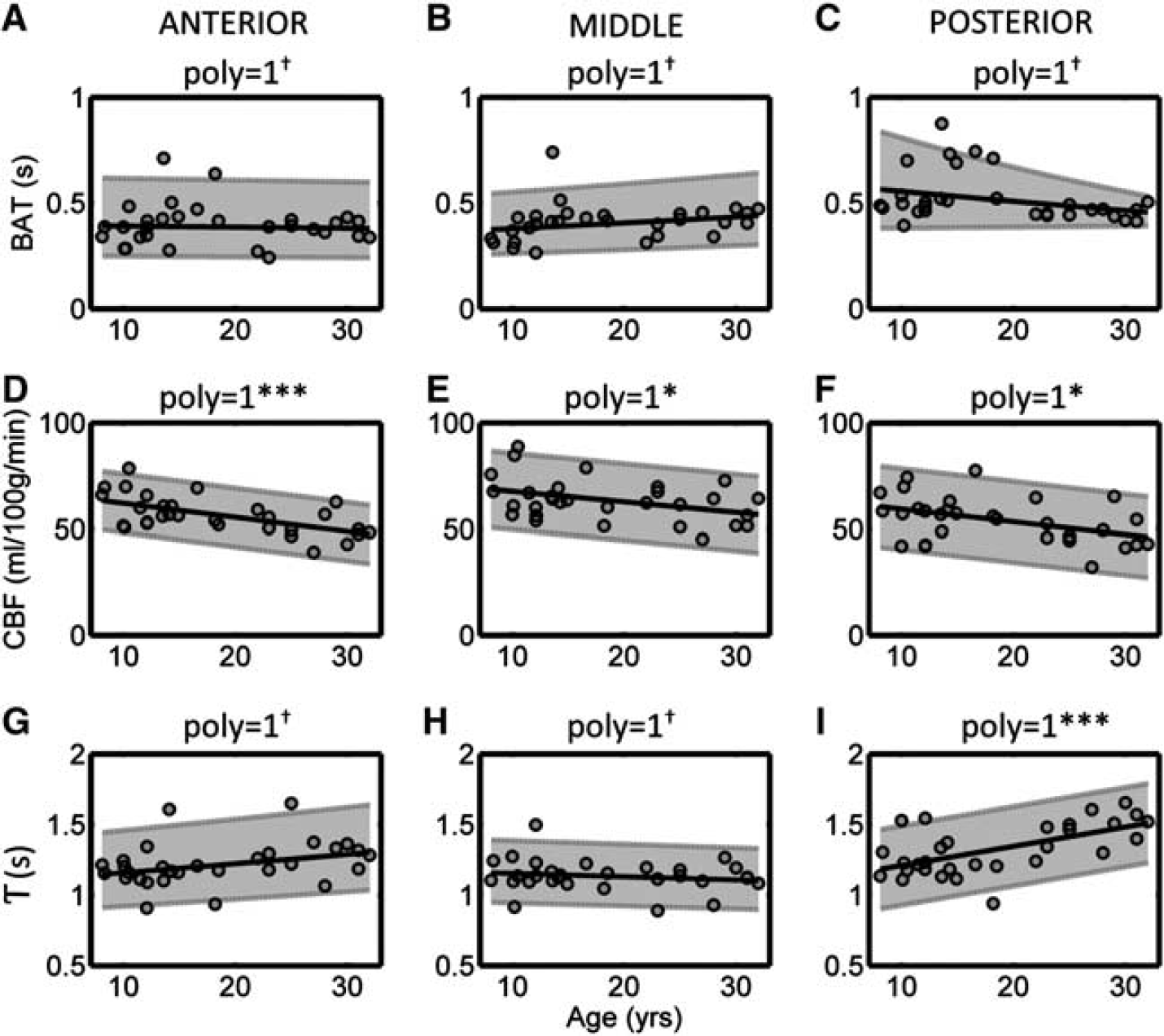
Variation in the fitted values of bolus arrival time (BAT,
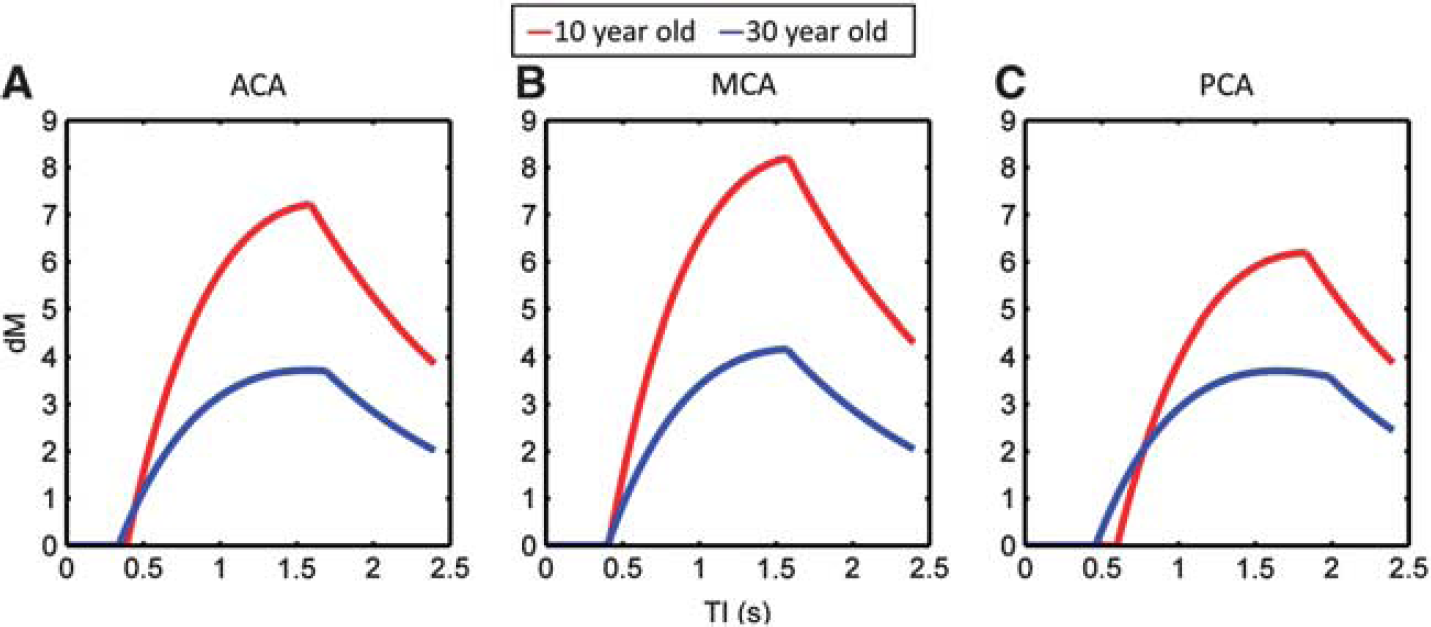
Examples of the typical dynamic arterial spin labeling (ASL) signal (d
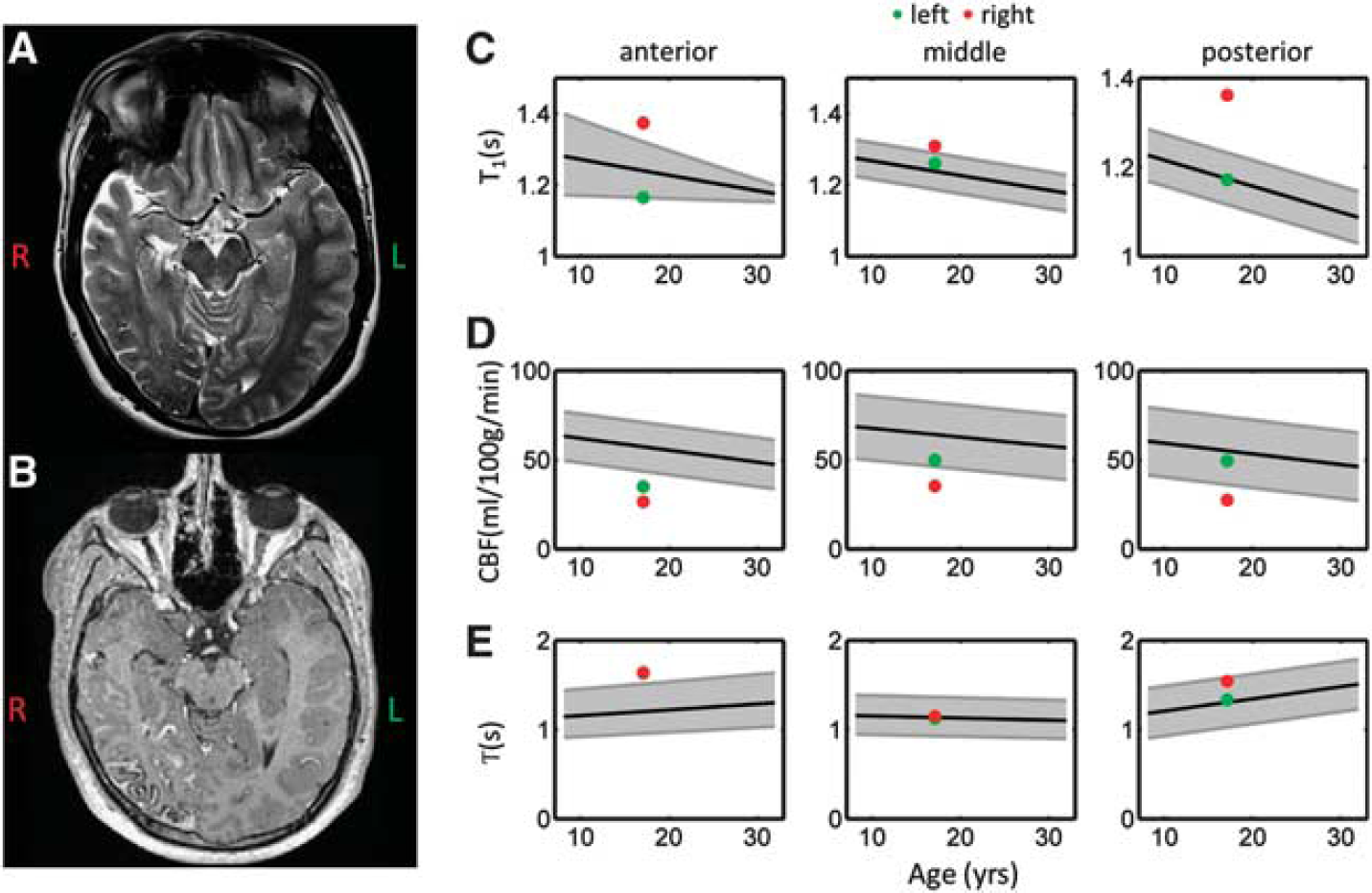
Example standard clinical images and arterial spin labeling (ASL) perfusion parameters acquired in a 17-year-old Sturge–Weber patient. Axial
The strong negative correlation between d
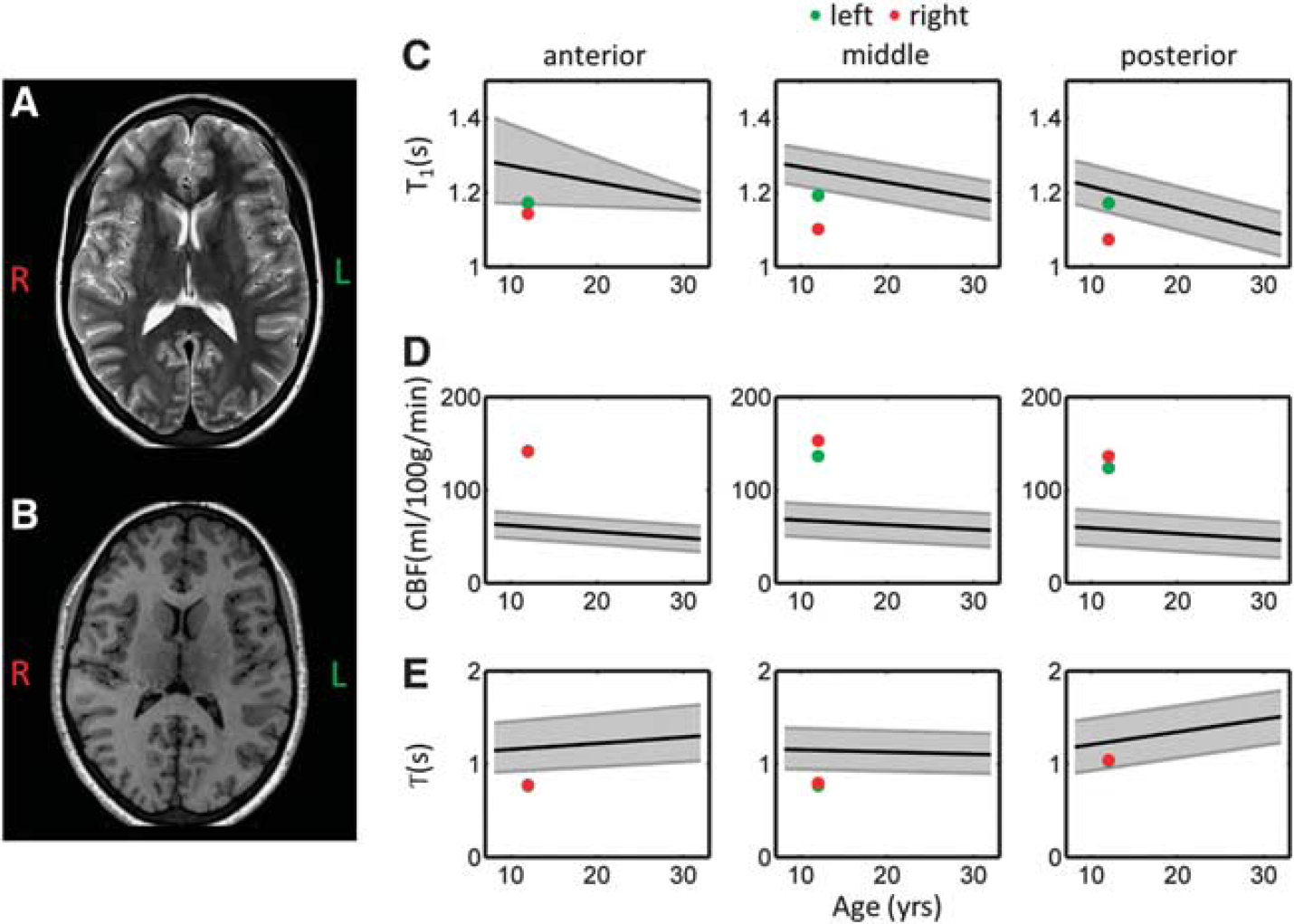
Example standard clinical images and arterial spin labeling (ASL) perfusion parameters acquired in a neurologically asymptomatic 12-year-old sickle cell anemia patient. Axial
Our data confirm that
Previous transcranial Doppler ultrasound studies have shown a significant decline in time-averaged flow velocities (TAV) in ACA, MCA, and PCA, between children aged 10 to 17.5 years, and adults aged 20 to 63 years. 30 Between children aged 1.5 to 9.9 years and children aged 10 to 17.5 years, no change in TAV was observed in ACA and PCA, but a decline was seen in the MCA. In general, TAV was highest in the MCA, followed by the ACA, and lowest in the PCA. However, in this study we observed no change in BAT as a function of age in all vascular territories. As a higher TAV should lead to shorter values of BAT, this would suggest that TAV is constant in all vascular territories between 8 and 32 years of age. However, these results should be interpreted with caution, as the temporal resolution of the ASL measurements made in this study is likely to be too coarse to identify small-scale changes in the underlying BAT. Furthermore, although some voxels within each ROI will include signal from large arteries branching from ACA, MCA and PCA, the majority of voxels will be distal to these feeding arteries, and hence the ASL parameters predominantly reflect blood flow in arterioles and capillaries, which will be less sensitive to changes in the TAV upstream in the feeding artery. However, the fact that that the PMM value of BAT was significantly higher in the PCA vascular territory, compared with both the MCA and ACA territories, does support previous findings that TAV is lowest in the PCA. Furthermore, our finding that CBF was significantly higher in the MCA territory compared with both the ACA and PCA territories agrees with previous evidence that TAV is highest in the MCA.
Around the age of 8 years, all three regions (ACA, MCA, and PCA territories) showed similar values of
Clinical Examples
The aim of this study was to provide reference range values to describe the hemodynamic properties of the ASL signal in healthy children and young adults, which can be used as a tool in future studies to investigate pathologies that lead to abnormal cerebral perfusion. Sturge–Weber syndrome is a good example of one such pathology, characterized by intracranial venous dysplasia, a cutaneous capillary angioma, and ocular abnormalities due to anomalous embryonic development.
34
An abnormal, tortuous pial vasculature is present, associated with venous stasis, leading to perfusion defects, which have been observed both focally in regions of leptomeningeal disease and in more distant GM in ipsilateral and contralateral regions.
35
Clinically, there is commonly epilepsy and contralateral hemiparesis in addition to high intraocular pressure (glaucoma). The Sturge–Weber syndrome patient included in this study had a right pial angioma, with evidence of leptomeningial enhancement in the right PCA territory (identified on
Sickle cell anemia is another pathology in which the reference range tool we have developed would be particularly useful. It is a genetic condition that results in the production of abnormal hemoglobin and subsequent chronic hemolytic anemia. As a compensatory mechanism for low hematocrit, both high cerebral blood velocity and flow have been described.37–39 Children with SCA are at high risk of both clinical and silent cerebral infarction, narrowing of large vessels, and insufficient oxygen/glucose delivery, which may result in deficits in brain function even in patients without any abnormality seen on
CONCLUSION
Overall, our results show that during the transition from childhood into adulthood, significant changes occur in both
Footnotes
The authors declare no conflict of interest.
ACKNOWLEDGMENTS
The authors would like to thank Tina Banks for radiological support, and the subjects scanned in this study for their assistance.
