Abstract
We hypothesized that the glycocalyx, which is important for endothelial integrity, is lost in severe malaria. C57BL/6 mice were infected with
INTRODUCTION
The human malaria parasite,
The healthy endothelium is covered luminally in the glycocalyx; a complex matrix consisting of proteoglycans and glycoproteins. 3 The glycocalyx is involved in mechanosensing, upkeeping endothelial homeostasis and maintaining the barrier function of the endothelium. Moreover it shields endothelial cell adhesion molecules from unwanted leukocyte binding. 4 Several types of pathophysiology including hyperglycemia, ischemia, inflammation and atherosclerosis3,5 involve breakdown of the glycocalyx.
MATERIALS AND METHODS
Mice, Malaria Parasites and Infection
Female, 5-week-old, C57BL/6 mice (Taconic, Ejby, Denmark) were randomly split in three groups: Uninfected, infected intraperitoneal with 10
4
PbA-infected erythrocytes and infected intraperitoneally with 10
4
PcAS-infected erythrocytes. Each group consisted of six to eight mice; the study was repeated and a total of 42 mice were included in the study. Mice were kept in individually ventilated cages with
Killing of Mice and Tissue Processing
The experiment was terminated for all groups when PbA-infected mice developed clear signs of CM. In deep hypnorm/dormicum 8 anesthesia, blood was collected from the orbital sinus into EDTA-coated tubes and stored frozen until further processed. The mice were then transcardially perfused with heparinised saline (15,000 U/L) followed by 5 minutes fixation with 2.5% glutaraldehyde (Electron Microscopy Sciences (EMS), Hatfield, PA, USA) and 0.5% Ruthenium red (Sigma-Aldrich, Brondby, Denmark) in 0.1 mol/L cacodylate buffer (EMS). Brains were stored in the fixative for two hours then transferred to 2.5% glutaraldehyde in 0.1 mol/L cacodylate buffer. With an 18G biopsy punch (Tru-Cut, San Diego, CA, USA), two small, random blocks of cerebral tissue were removed and immersed in osmium tetroxide (1%, AlfaAesar, Ward Hill, MA, USA) for secondary fixation. Tissue was dehydrated according to standard protocols using propylene oxide (EMS) as an intermedium and embedded in Epon (TAAB, Berkshire, UK).
Transmission Electron Microscopy (TEM) and Image Analysis
Ultrathin sections (60 nm) were either stained with Reynold's lead citrate and uranyl acetate as previously described 9 or left unstained. Unstained sections were analyzed for glycocalyx area change. At × 92,000 magnification and at eucentric height, vessels were imaged and with the free-hand selection tool, the luminal surface of the plasma membrane including the stained glycocalyx was selected with open source Fiji software 10 and thresholded. Also a line along the plasma membrane was drawn. The ratio between the two measures provided a surface area/length measure used for the analyses. Stained sections were analyzed for endothelial apoptosis based on heterochromatin aggregation in nuclei. 11 All TEM was performed using a Philips CM100 (Philips, Eindhoven, Holland) with a Morada or an Olympus Veleta digital camera (Olympus, Tokyo, Japan) connected to a workstation with SIS Analysis software (iTEM). All analyses were carried out by CH on masked specimens.
Enzyme Linked Immunosorbent assay (ELISA)
Hyaluronic acid (HA) in plasma was quantified by using a commercially available ELISA (Enzyme Linked Immunosorbent assay) adjusted to 60μL loading volume (Echelon Biosciences, Salt Lake City, UT, USA).
Dot Blots
A total of 2 μL of plasma was dotted on a cationic nitrocellulose membrane (Hybond N+, Amersham, GE Healthcare, Brondby, Denmark). For the analysis of sulfated GAGs, the membrane was immersed in 2.5 mg/mL Alcian blue 8X (Sigma-Aldrich) dissolved in 0.4mol/L Guanidine-HCl, 0.018 mol/L sulfuric acid and 0.25% Triton-X-100 (Sigma-Aldrich). 12 Chondroitin sulfate A (Sigma-Aldrich) was used to generate a standard curve. The membrane was stained for 30 minutes at room temperature and washed for 60 minutes in MiliQ water. For the analyses of HA 2μL of plasma was dotted as described for sulfated GAGs. The membrane was blocked for 1 hour at room temperature with 2% skimmed milk powder (Sigma-Aldrich) in tris-buffered saline. HA was detected by goat anti-HA antibody (5 μg/mL, AbDSerotec, Bio-Rad Laboratories, Hercules, CA, USA). An HRP-conjugated rabbit anti-goat antibody (Dako, Glostrup, Denmark) detected the primary antibody. Supersignal West femto (Pierce, Thermo Fischer Scientific, Waltham, MA, USA) was used as luminiscent substrate. Dot blots were visualized using a Chemidoc XRS in epi-white or chemiluminiscence mode respectively (Bio-Rad Laboratories). The level of staining was quantified by densitometry using Fiji software.
Statistical Analyses
Data following a normal distribution were analyzed with one-way analysis of variance;
RESULTS
Glycocalyx Disruption is Associated with Severe Infection
At day seven post infection PbA-infected mice showed signs of terminal CM including hypothermia, convulsions and motor and balance impairments, whereas no PcAS-infected mice were hypothermic or had cerebral signs (
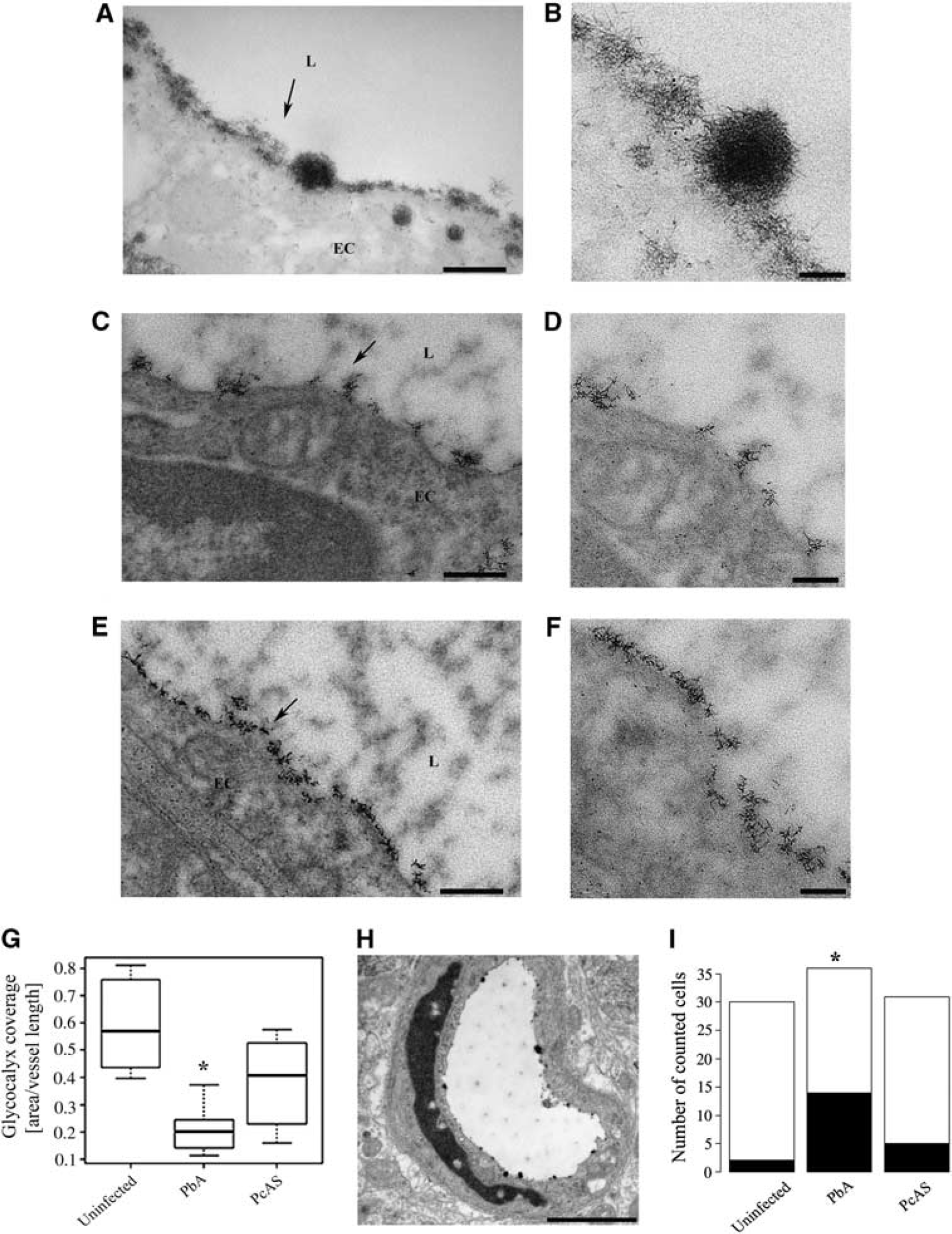
Endothelial glycocalyx is lost and endothelial apoptosis increased in response to experimental CM. (
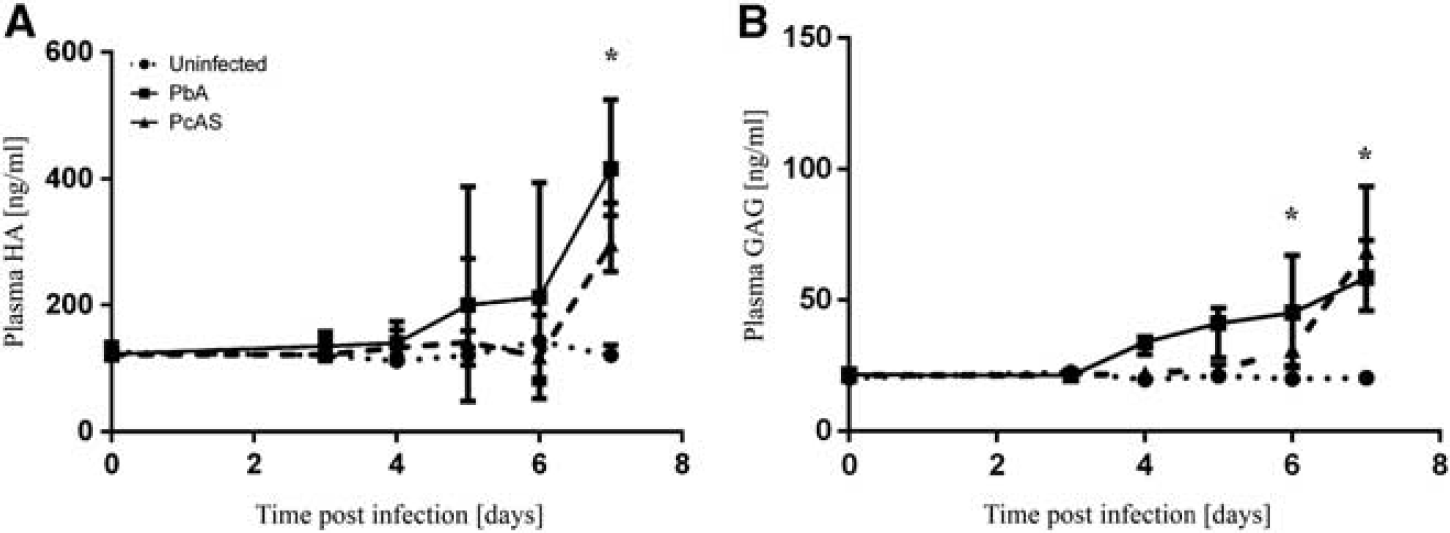
Plasma levels of glycocalyx elements increase owing to infection. (
Glycocalyx Loss is Independent of Endothelial Apoptosis
We investigated whether the loss of endothelial glycocalyx was associated with and thus potentially secondary to endothelial apoptosis. The proportion of apoptotic endothelial cells based on apoptotic chromatin condensation (Figure 1H) was significantly different owing to infection (Figure 1I,
Plasma Markers Confirm the Loss of Endothelial Glycocalyx
At day 7, HA levels in plasma were significantly increased by infection in both PbA- and PcAS-infected mice compared with uninfected controls (Figure 2A,
DISCUSSION
To the best of our knowledge, this is the first demonstration of preservation, visualization and quantification of the endothelial glycocalyx in malaria research. The glycoconjugates on the luminal surface of the endothelial cells have been ignored in previous studies because they are invisible with routine fixation and staining methods for TEM. We used contrasting with ruthenium red and TEM to visualize the glycocalyx in high resolution enabling the identification of individual GAG chains. The condensed tufts of GAG chains characterizing a healthy glycocalyx 3 were either lost completely or seemed to be severely depleted during the terminal phase of CM. The loss of GAGs on the cerebral endothelial cells corresponded with an increased level of sulfated GAGs and HA in the plasma in PbA-infected mice. The size of glycocalyx in uninfected mice was comparable with previous reports. 3
The loss of glycocalyx was not merely secondary to endothelial apoptosis. Instead, it is likely an early marker of endothelial activation and inflammation because its presence shields cellular adhesion molecules that serve as receptors for leukocytes and support sequestration of malaria-infected erythrocytes.1,4 Malaria-infected erythrocytes binding heparan sulfate and chondroitin sulfate, two major constituents of the endothelial glycocalyx, are thus potentially important for establishing an infection. 6
Plasma levels of sulfated GAGs increased markedly during infection with PbA and the levels were significantly different from uninfected mice at day 6 post infection. These data strongly suggest that the endothelial glycocalyx is being shed before the terminal phase of the infection resulting in CM. Hyaluronic acid seemed to increase before the terminal phase as well, but not to the same extent. Interestingly, PcAS-infected mice showed minor changes to the endothelial glycocalyx in the brain, although we detected high plasma levels of both sulfated GAGs and HA, an unsulfated GAG at day 7 post infection. The high plasma levels of HA and sulfated GAGs in PcAS-infected mice was unexpected given that only minor loss of GAG was noticed in TEM analyses. The cationic dye ruthenium red reacts with both sulfated GAG and HA and thus the discrepancy between plasma levels and the quantifications on the cerebral endothelium may also arise from substantial amounts being shed from other organs than the brain. Indeed, PcAS has been shown to adhere to and modulate the endothelium both in the liver and in the brain of CBA mice. 14 However, as uninfected mice were injected with saline rather than with uninfected erythrocytes, we cannot rule out that an unspecific inflammatory response against allogenic erythrocytes may have contributed to glycocalyx disruption. Nevertheless, using the plasma levels of HA and sulfated GAGs as diagnostic markers of focal pathologic examination does not seem appropriate for malarial infections and may limit the possibility of studying glycocalyx changes in human malaria.
Loss of endothelial glycocalyx may be caused by several mechanisms including inflammation and hypoxia3,5 being central in CM pathogenesis.1,8 The loss of glycocalyx promotes leukocyte and platelet interactions with the endothelium which is part of malaria pathogenesis. 1 Also, the glycocalyx has several enzymes and proteins including antithrombin bound to it. Lately, antithrombin has been implicated in severe malaria pathogenesis, as its loss makes the endothelium more vulnerable to thrombin. 15 In murine CM, we have previously assessed the positive effects of adjunctive therapy with erythropoietin, 8 which also exerts its neuroprotective effects via the endothelium. Thus, upkeeping endothelial homeostasis and barrier function is essential for protection against CM. Previous studies using antithrombin protected against ischemia-induced glycocalyx damage 4 and antioxidatory enzymes and scavengers including allopurinol have shown to be protective against reactive oxygen species that mediate glycocalyx breakdown. 3
Type 2 diabetes with significant vascular complications is on the rise in countries, where malaria is also present, and the two diseases could synergize to disrupt the glycocalyx and endothelial function. The disease combination may further reduce nitric oxide bioavailability, increase endothelial apoptosis and inflammatory stress thus possibly aggravating malaria pathology.
In summary the data present loss glycocalyx relating to the severity of murine malaria infections. Future studies will explore the mechanisms behind the loss of endothelial glycocalyx in malaria and its relevance at various steps of disease progression.
Footnotes
The authors declare no conflict of interest.
ACKNOWLEDGMENTS
The superb technical assistance from Hanne Holm (University of Copenhagen) is highly acknowledged. Also the scientific discussions in general and glycocalyx discussions in particular with Professor Henrik Clausen (Centre for Glycomics, University of Copenhagen) are acknowledged. The use of and support for using the Philips CM100 at Core Facility for Integrated Microscopy, Faculty of Health and Medical Sciences, University of Copenhagen, is highly acknowledged. Lasse Maretty (University of Copenhagen) is acknowledged for the use of his stock of PcAS. Jens R. Nyengaard (University of Aarhus) is acknowledged for his idea on how to randomize tissue sampling.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
