Abstract
Hypertension is linked with an increased risk of white matter hyperintensities; however, recent findings have questioned this association. We examined whether hypertension and additional cerebrovascular risk factors impacted on white matter integrity in an inducible hypertensive rat. No white matter hyperintensities were observed on magnetic resonance imaging either alone or in conjunction with ageing and high-fat diet. Aged hypertensive rats that were fed a high-fat diet had moderately reduced fractional anisotropy in the corpus callosum with no overt pathological features. Herein we show that moderate hypertension alone or with additional risk factors has minimal impact on white matter integrity in this model.
Introduction
Hypertension has been associated with an increased risk of white matter (WM) hyperintensities (WMHs) 1 and is the second most important factor after ageing for the development of cerebrovascular disease, a known contributor to vascular dementia. Clinically, WM lesions (WMLs) are characterized on MRI as hyperintense regions on fluid-attenuated inversion recovery or T2-weighted magnetic resonance imaging (MRI), which are associated with disruption of the normal cellular architecture of myelinated fibers. However, increased tissue water mobility without altered fractional anisotropy (FA) is an early imaging feature, as shown by diffusion tensor (DT-) and magnetisation transfer (MT-) MRI that have been utilized to explore WM alterations in hypertension.2, 3 Hypertension is associated with decreased FA, increased mean diffusivity (MD), and reduced magnetization transfer ratio (MTR) in both WMLs and normal-appearing WM,2, 4 commonly including periventricular WM and the corpus callosum (CC). 3 However, hypertension may not be the primary precipitant of WMHs with other nonvascular factors involved, 5 which is further supported by the failure of antihypertensive treatment to prevent the progression of WMLs. 6
Animal models have been used to explore the impact of hypertension on the central nervous system, (see Bailey
Given the discrepancies in the influence of hypertension between the two commonly utilized models, and the difficulty in exploring isolated hypertension in humans because of a multitude of comorbid risk factors including diet, diabetes, and genetic susceptibility,1, 11 we aimed to explore the alterations in WM integrity using MRI and immunohistochemistry in an inducible hypertensive transgenic rat, in which the onset and duration of hypertension are controllable. 12 We have previously demonstrated that MRI is sensitive to subtle WM alterations in a mouse model of modest cerebral hypoperfusion, 13 and as such we hypothesize that sustained hypertension would lead to microstructural alterations in central nervous system WM and further that ageing or additional cerebrovascular risk factors, such as high-fat intake, would exacerbate WM disruption.
Materials and Methods
Animals
The Cyp1a1Ren2 transgenic rats on a Fisher-F44 background, expressing the mouse
Magnetic Resonance Imaging
Rats were undefined and placed in an MRI holder (Rapid Biomedical, GmbH, Würzburg, Germany). Rectal temperature and respiration were controlled to ensure normal physiological parameters. T2, DT-, and MT-MRI data were collected using an Agilent 7T preclinical scanner (Agilent Technologies, Yarnton, UK); with a 72-mm volume coil and a two-channel phased-array rat brain coil (Rapid Biomedical). Diffusion tensor imaging was conducted using an axial fast spin—echo sequence and FA and MD were computed, MT-MRI was conducted using two spin—echo sequences (Supplementary Data). Mean diffusivity, FA, and MTR were measured in the CC, internal capsule, and fimbria.
Immunohistochemistry
After MRI, rats were perfused under deep isoflurane anesthesia with heparinized saline followed by 4% parafomaldehyde, their brains removed and postfixed before paraffin embedding. Appropriate 6 μm sections were cut corresponding to the regions assessed for MRI (0.86 to −0.1 mm and −1.34 to −2.18 mm bregma) as these areas have previously demonstrated vulnerability in spontaneously hypertensive rat and SHRSP strains. Immunostaining was conducted using standard methods after pretreatment to remove paraffin and for amyloid precursor protein antigen retrieval with microwaving in 10 mM citrate buffer (pH 6). Antibodies were selected to visualize the different cellular components of WM fiber bundles. Myelin integrity was assessed using anti-myelin basic protein (MBP) (1:5,000; Millipore, Watford, UK). Axonal damage was assessed using anti-amyloid precursor protein (1:1,000; Millipore). Sections were incubated with primary antibody overnight at 4°C after blocking then biotinylated secondary antibodies (1:100) and a solution of streptavidinbiotin—peroxidase complex. Peroxidase activity was localized using 3,3′diaminobenzadine tetrahydrochloride (Vector, Peterborough, UK). The percentage area stained of MBP was assessed in the CC above the lateral ventricle using Image J (v1.43, NIH, Bethesda, MD, USA). Bilateral images were taken at x200 magnification and the CC manually delineated by an observer blinded to the intervention. Four predefined regions-of-interest were then randomly placed over the images and the percentage area of immunopositive MBP calculated as a measure of myelin density. To assess axonal integrity the CC in all animals were carefully screened for the presence of intense amyloid precursor protein immunoreactivity in swollen or bulbous axons.
Statistical Analysis
Blood pressure data are presented as the average 2-weekly values across the different groups. Statistical comparisons were conducted using
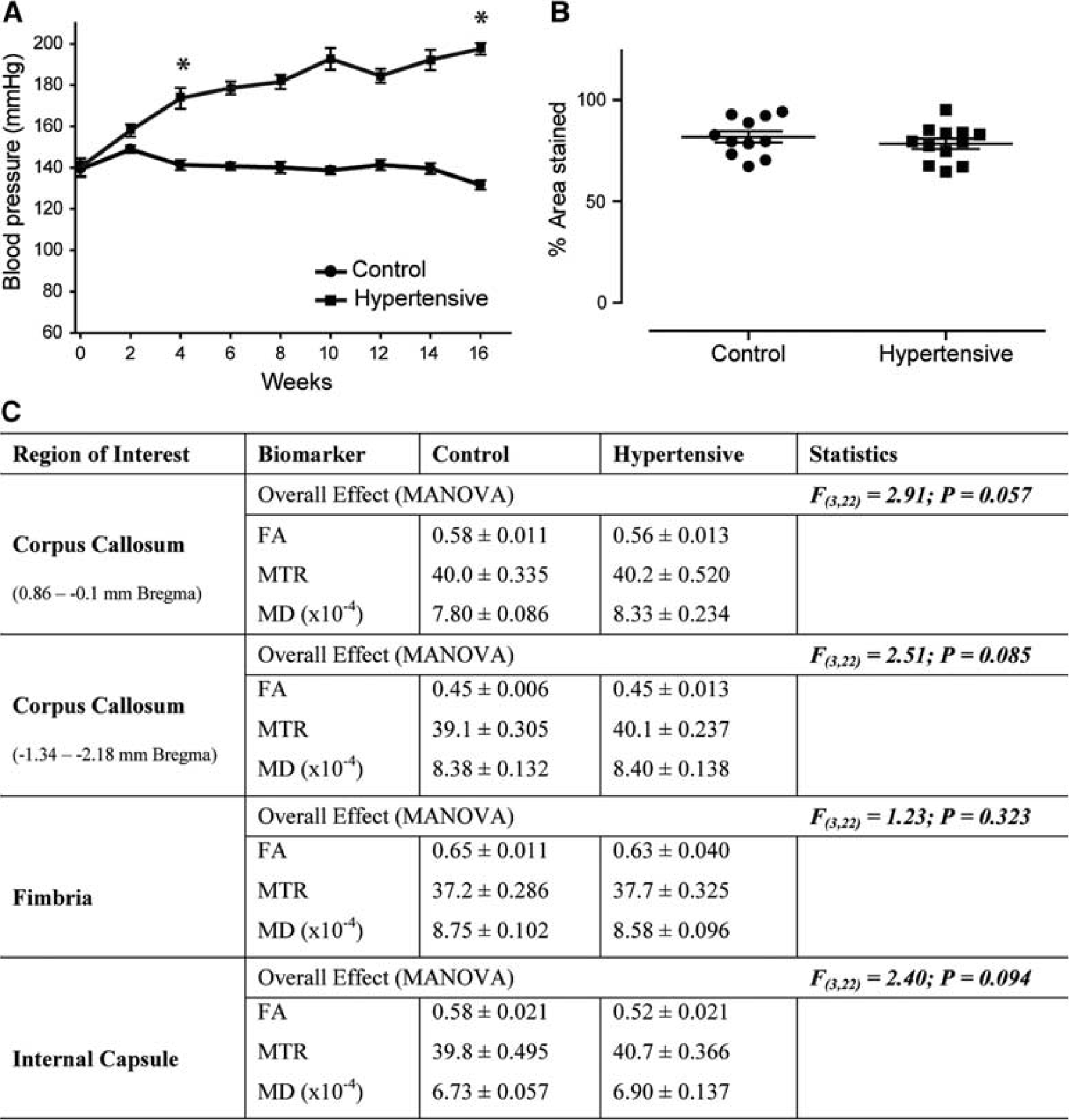
Physiological and magnetic resonance imaging findings in young rats. Addition of 0.15% I3C in normal rat chow results in a gradual increase in blood pressure in Cyp1a1Ren2 rats that is significantly different from control littermates from weeks 4 to 5 reaching >160 mm Hg systolic and remaining significantly increased for the duration of the study (
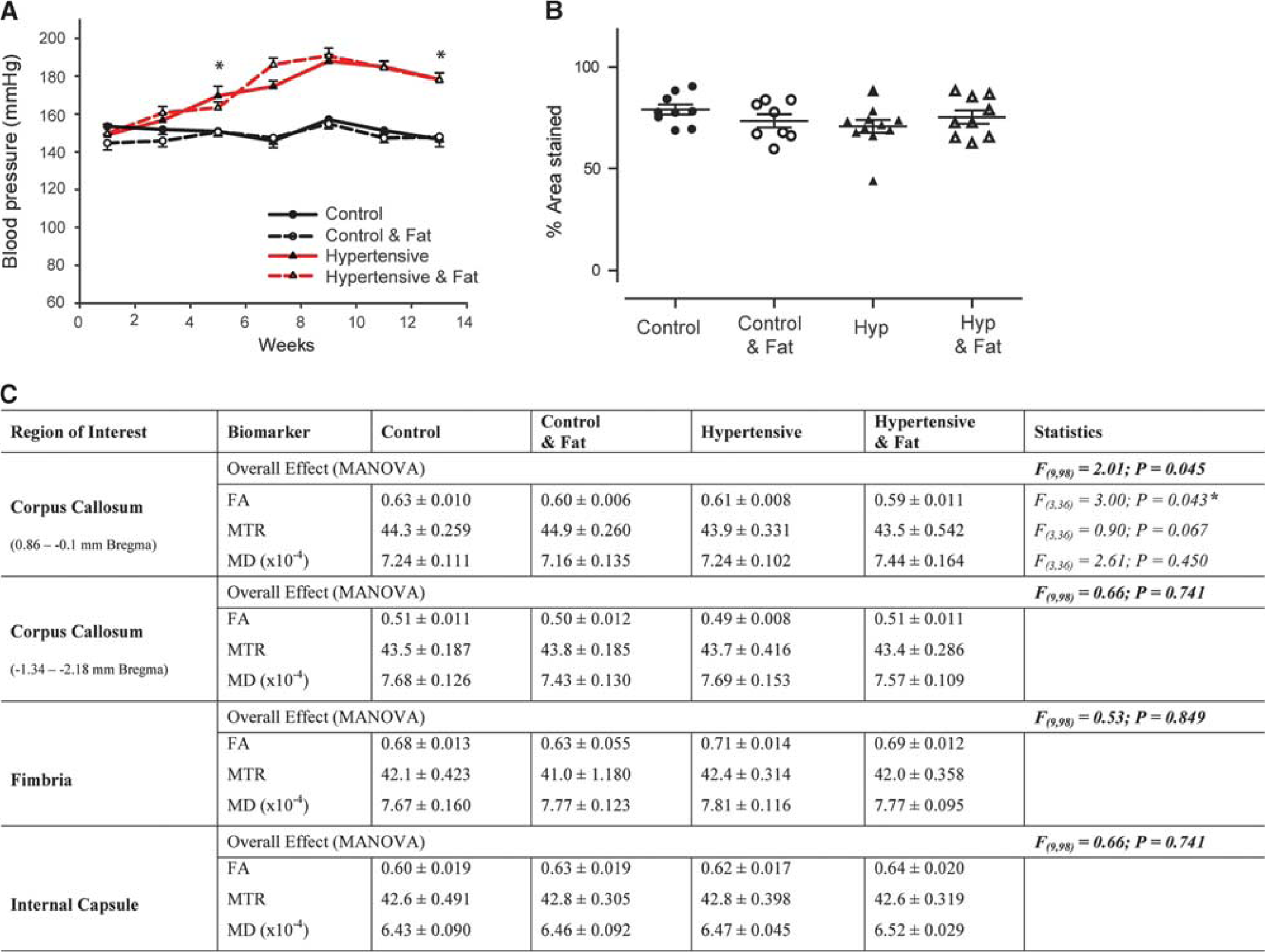
Physiological and magnetic resonance imaging findings in aged rats. Addition of 0.15% I3C to the diet of aged Cyp1a1Ren2 rats results in a gradual increase in blood pressure that is significantly different from control littermates from weeks 4 to 5, reaching >160 mm Hg systolic and remaining significantly increased for the duration of the study (
Results
Impact of Hypertension in Young Rats
Addition of 0.15% I3C to young Cyp1a1Ren2 transgenic rats resulted in a gradual increase in BP which was significantly different from control littermates from week 4 to 5 onward (t(18)=5.80,
There were no overt WMH's on T2-weighted MRI (Supplementary Figure 2) in young rats, and overall analysis failed to detect any significant microstructural alterations in all regions studied (Figure 1C). In agreement with the absence of WM alterations on MRI there was no overt axonal pathology in the CC and no significant change in myelin density (t(21)=0.88,
Impact of Hypertension in Aged Rats
Addition of 0.15% I3C to aged Cyp1a1Ren2 transgenic rats (groups 3 to 4) resulted in a gradual increase in BP that was significantly different from control littermates (groups 1 to 2) from weeks 4 to 5 onward (t(18)=3.02,
There were no overt WMLs on T2-weighted MRI in aged rats (Supplementary Figure 2), although overall analysis identified significant microstructural alterations between the four groups, in the CC (F(9,98)=2.01;
Discussion
White matter lesions are commonly seen on MRI in older subjects and their presence is associated with age and cerebrovascular risk factors, including hypertension. 1 Herein we demonstrate that hypertension alone or in conjunction with further cerebrovascular risk factors, including ageing and high-fat intake does not result in gross WMLs as evidenced by the lack of WMHs on T2-weighted MRI. Recently alternate approaches including DT- and MT-MRI have demonstrated sensitivity for subtle WM disruption. 13 In agreement we detected a significant subtle decrease in FA in the CC dorsal to the lateral ventricle; however, this was only evident in aged hypertensive rats fed a high-fat diet, suggestive of a summation effect. Despite the significant increase in BP there was no significant correlation between these indices and the observed alterations in FA. Our data are in agreement with others who recently identified no overt disruption in the WM of SHRSP rats, who survived stroke free up to ten months 10 or required the addition of chronic hypoperfusion to elicit WMLs. 14 Taken together the data suggest that hypertension alone for the duration tested here is insufficient to drive WM alterations and we further show that in aged rats (∼21months) the combination of ageing and late onset hypertension does not impact on WM integrity in this model. As myelin damage may be a relatively late feature of WM damage secondary to vascular risk factors (as suggested by pathologic study), it may be that we did not expose the rats to hypertension or high-fat for a sufficient length of time.
Although the use of a relatively novel model is beneficial to allow controllable hypertensive induction, it is a limitation of the study that the model has not been widely used to study central nervous system pathologic assessment after hypertension. To this end we have previously explored cerebrovascular alterations in young Cyp1a1Ren2 transgenic rats with varying durations of hypertension
15
and identified thalamic cerebrovascular alterations. Thus the absence of overt WM alterations in hypertensive animals may indicate a regional vulnerability within thalamic regions. Alternatively, damage to myelin may occur late as suggested by human studies where post-mortem findings focus on loss of myelin, but much earlier changes detectable
In conclusion both young and aged hypertensive rats show no overt WM disruption raising further questions regarding the impact of hypertension on the development of WMHs. White matter structural integrity was only modestly altered in the combined presence of hypertension, ageing, and high-fat diet suggesting a cumulative effect of multiple risk factors.
