Abstract
Cerebral microangiopathy (CMA) has been associated with executive dysfunction and fronto-parietal neural network disruption. Advances in magnetic resonance imaging allow more detailed analyses of gray (e.g., voxel-based morphometry—VBM) and white matter (e.g., diffusion tensor imaging—DTI) than traditional visual rating scales. The current study investigated patients with early CMA and healthy control subjects with all three approaches. Neuropsychological assessment focused on executive functions, the cognitive domain most discussed in CMA. The DTI and age-related white matter changes rating scales revealed convergent results showing widespread white matter changes in early CMA. Correlations were found in frontal and parietal areas exclusively with speeded, but not with speed-corrected executive measures. The VBM analyses showed reduced gray matter in frontal areas. All three approaches confirmed the hypothesized fronto-parietal network disruption in early CMA. Innovative methods (DTI) converged with results from conventional methods (visual rating) while allowing greater spatial and tissue accuracy. They are thus valid additions to the analysis of neural correlates of cognitive dysfunction. We found a clear distinction between speeded and nonspeeded executive measures in relationship to imaging parameters. Cognitive slowing is related to disease severity in early CMA and therefore important for early diagnostics.
Keywords
Introduction
Cerebral microangiopathy (CMA) describes a status of impaired blood circulation in the arterioles of the brain causing vascular dementia, the second most common cause of dementia after Alzheimer's disease. Causal factors discussed are arteriolosclerotic alterations such as fibrosis, wall thickening, and reduced vasoreactivity of the small vessels (Román et al, 2002; Schroeter et al, 2004, 2005; Brundel et al, 2012). The impaired microcirculation results in lacunar infarcts if complete occlusion of arterioles occurs and in cell loss mainly in deep white matter if incomplete. The resulting clinical condition is subcortical ischemic vascular dementia or its prestage vascular mild cognitive impairment (Erkinjuntti and Gauthier, 2009). It is associated with the disruption of fronto-parietal brain circuits (Cummings, 1995). Neuropsychologically, it has been described as a predominant dysexecutive syndrome (Román et al, 2002; Schroeter et al, 2007a). However, individual differences in cognitive performance are high and the relationship to various brain imaging markers is controversial (Patel and Markus, 2011).
Vascular white matter lesions might be assessed with neuroradiological rating scales such as the age-related white matter changes scale (ARWMC; Wahlund et al, 2001). It allows global and regional classification of white matter lesion severity by visual rating of magnetic resonance images. Subcortical, especially periventricular hyperintensities on visual rating have been associated with executive and speed impairment in several types of mild cognitive impairment (Debette et al, 2007) and normal aging (Prins et al, 2005). Ratings of lacunes have been associated with cognitive deficits in normal aging (Carey et al, 2008). However, other authors failed to show correlations between visual ratings and neuropsychological data (O’Sullivan et al, 2004), wherefore it is still under debate whether visual ratings lack validity in explaining and predicting cognitive performance or whether there is in fact no association.
During the last years, various new imaging parameters have been introduced. As CMA affects white matter structures, the most promising magnetic resonance imaging (MRI) marker in describing and predicting effects of the disease has been diffusion-weighted imaging (Nitkunan et al, 2008). Diffusion-weighted imaging measures the diffusion of water molecules in the brain. A diffusion tensor can be computed from the diffusion-weighted imaging data (diffusion tensor imaging, DTI) and its orthogonal components describe diffusion within the three-dimensional space. In combination, they indicate white matter tract destruction. Axial diffusivity measures water diffusion along the fiber tract and has been associated with axonal loss (Song et al, 2003), whereas radial diffusivity measures diffusion perpendicular to the fiber tract and has been associated with myelin degeneration (Alexander et al, 2007). Mean diffusivity describes the overall diffusion independent of its direction and is a measure of ultrastructural damage, whereas fractional anisotropy is a measure of the directionality of diffusion and associated with changes in white matter fiber parallelism, density, axonal diameter, and myelination (Kochunov et al, 2007). Recently, tract-based spatial statistics (TBSS; Smith et al, 2006) has been introduced as a refinement of voxelwise group analysis of DTI data overcoming alignment problems inherent to approaches inspired by voxel-based morphometry (VBM; Ashburner and Friston, 2000).
In several VBM style DTI correlation analyses, associations were found between reduced fractional anisotropy, increased mean diffusivity and neuropsychological impairment, most prominently executive deficits. This was found in middle-aged patients with a genetic variant of CMA with mild to severe white matter lesions and a wide range of cognitive functioning (O’Sullivan et al, 2005) as well as in elderly patients with more severe white matter lesions and mild to severe cognitive impairment (Kim et al, 2011). In a group study, a consistent pattern of increased mean diffusivity and reduced fractional anisotropy in CMA patients as compared with healthy control subjects was found pointing to white matter tract disruption in CMA (Nitkunan et al, 2008). Only two studies used the refined TBSS approach in CMA patients. A large cohort study showed a relationship between gait disturbance and increased mean diffusivity and reduced fractional anisotropy in a group of nondemented elderly with mild to severe white matter or lacunar lesions (de Laat et al, 2011). A second study in severe leukoaraiosis (Fazekas scale grade 3) with a wide range of cognitive functioning found an association between global cognitive functioning and reduced fractional anisotropy (Otsuka et al, 2012). However, no study has investigated axonal and radial diffusivity in CMA patients so far, which allows deeper insights into the nature of white matter alterations (Alexander et al, 2007). Moreover, no study has used TBSS data in combination with executive functions in CMA patients until now.
Voxel-based morphometry (Ashburner and Friston, 2000) is well suited to study gray matter changes complimentary to TBSS analyses focused on white matter tracts. The VBM studies showed widespread atrophy in severely impaired vascular dementia patients (Li et al, 2011) and an association between white matter lesions and atrophy in healthy elderly subjects (Raji et al, 2012) mainly in the frontal lobes.
Recent studies investigated neuropsychological impairment and related neural correlates in vascular dementia due to CMA. Our study aimed at examining these factors in its prestage—vascular mild cognitive impairment (Erkinjuntti and Gauthier, 2009). As discussed above, CMA is characterized by executive dysfunctions, although the heterogeneity of the concept makes results viable to discrepancies in interpretation depending on the operationalization chosen for measuring executive function (Stuss and Alexander, 2007). Moreover, most executive tasks include a strong speed component that might be particularly impaired in CMA (Peters et al, 2005), whereas others find associations exclusively with the executive component of the tasks (O’Sullivan et al, 2005). In the current study, we thus examined in detail the relationship between speed and other subcomponents of executive functions and their relationship to several brain imaging markers in early CMA. We used a focused neuropsychological test battery and a wide range of imaging markers combining diffusion-weighted imaging (TBSS), visual ratings of structural MRI (ARWMC), and voxelwise analysis of structural MRI (VBM). Based on the current literature, we hypothesized associations between executive deficits and mainly white matter imaging parameters particularly in fronto-parietal brain areas.
Materials and methods
Subjects
Table 1 describes characteristics of the involved samples. Twelve patients with early CMA were recruited among former patients of the Clinic for Cognitive Neurology of the University Hospital Leipzig who had initially presented with cognitive complaints. Twenty-five healthy control subjects matched for age, intelligence, education, and gender were included from the volunteer database of the Max Planck Institute for Human Cognitive and Brain Sciences, Leipzig. Four of the healthy control subjects were subsequently excluded due to microangiopathic alterations on structural MRI images (ARWMC total score >2) and one patient due to an additional cerebellar infarct. Patients had been diagnosed with CMA after thorough clinical examination and structural MRI and all had a total ARWMC score of >2. Further exclusion criteria for both patients and control subjects were a history of psychiatric or neurologic disorders including stroke, craniocerebral injury or neurodegenerative disease and dementia. The research protocol was approved by the ethics committee of the University of Leipzig and was in accordance with the latest version of the Declaration of Helsinki. All participants were native German speakers and gave informed written consent. All had normal or corrected-to-normal vision.
Clinical characteristics of the sample
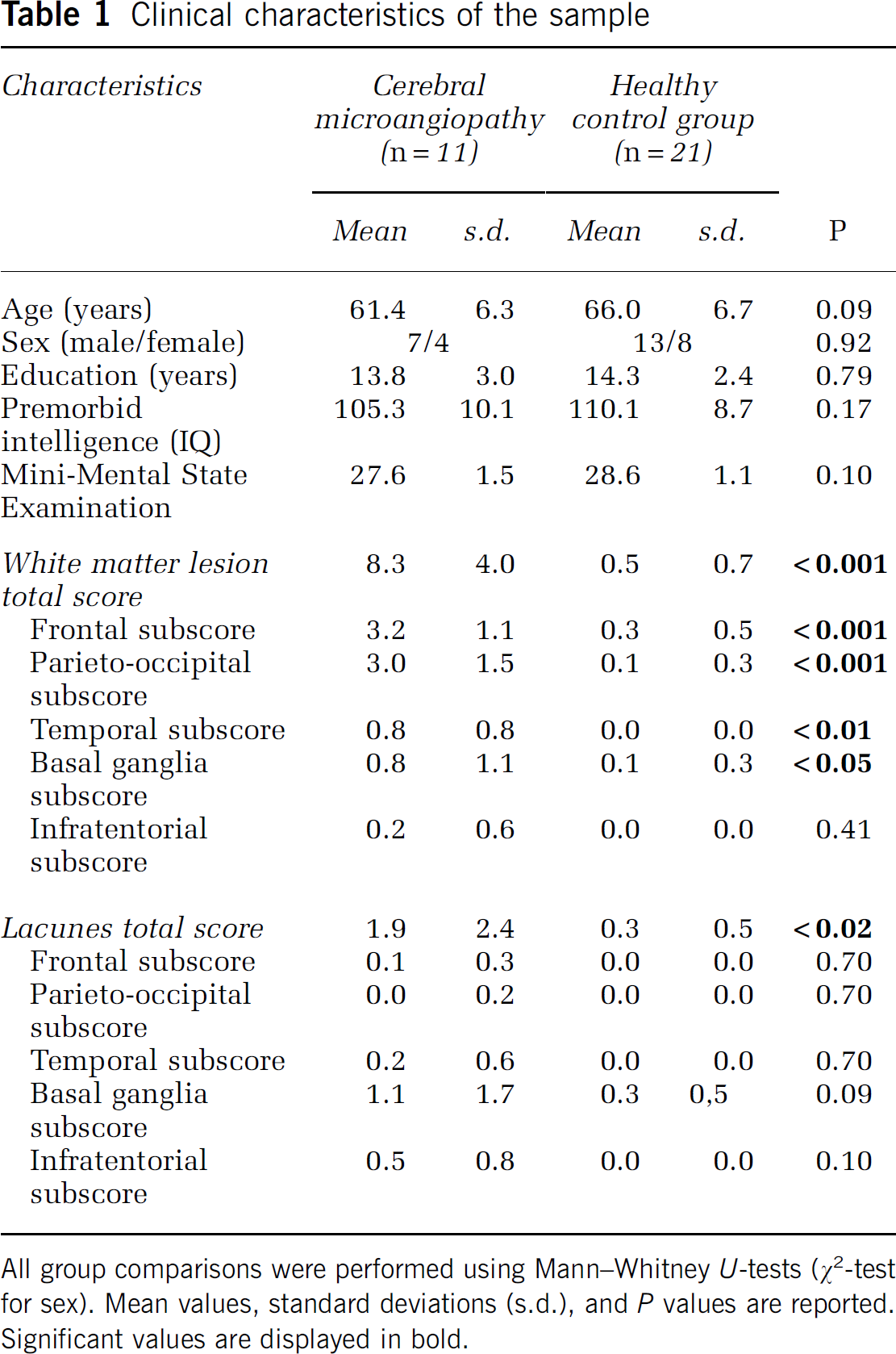
All group comparisons were performed using Mann–Whitney
Neuropsychological Testing
Neuropsychological testing was performed by experienced staff. First, the Consortium to Establish a Registry for Alzheimer's disease (CERAD) (Morris et al, 1988) test battery was applied: it includes global functioning by means of the Mini-Mental State Examination. It moreover includes executive function tests such as the Trail-Making-Test parts A and B measuring processing speed and cognitive flexibility as well as a phonemic and semantic fluency task requiring speeded word production. Executive functions were further tested by a computerized version of the Stroop task (Zysset et al, 2001), testing for speed and inhibition of highly automated processing. Further subtests of the CERAD were included as control variables: Memory abilities were tested by means of the word list immediate and delayed recall, recognition and figure recall, visuoconstructive abilities were tested by figure copying and word finding by the Boston naming test (Morris et al, 1988). Furthermore, premorbid verbal intelligence was tested by the Wortschatztest (Schmidt and Metzler, 1992). Overall testing time was 90 minutes.
Magnetic Resonance Imaging Data Acquisition
Magnetic resonance imaging was performed on a 3-Tesla TIM Trio scanner (Siemens, Erlangen, Germany) equipped with a 32-channel head array coil. We included several sequences to assess changes in gray matter density (T1-weighted images), vascular white matter lesions (T2-weighted and fast fluid-attenuated inversion recovery—FLAIR images), and alterations in white matter parameters (diffusion-weighted images).
T1-weighted images were acquired using a three-dimensional magnetization-prepared rapid gradient-echo sequence (sagittal orientation) with selective water excitation and linear phase encoding. The following imaging parameters were used: inversion time 650 ms, repetition time (TR) 1.3 seconds, TR of the gradient-echo kernel 9.1 ms, echo time 3.46 ms, flip angle 10°, field of view 256 × 240 mm2, 2 averages. To avoid aliasing, oversampling was performed in read direction (head to foot). Reconstructed images were calculated using zero filling with 1 × 1 × 1 mm3 voxel size. T2-weighted contrasts were calculated from a fast low angle shot, FLASH sequence (Frahm et al, 1986) sequence with the following parameters: TR 700 ms, echo time 15 ms, flip angle 25°, field of view 220 × 193 mm2, 0.9 × 0.9 × 4 mm3 voxel size, 1 average. A fast fluid-attenuated inversion recovery, FLAIR sequence (Hajnal et al, 1992) was acquired with the following parameters: inversion time 2.5 seconds, TR 10 seconds, echo time 86 ms, flip angle 180°, field of view 220 × 165 mm2, 0.9 × 0.9 × 4 mm3 voxel size, 1 average.
Diffusion-weighted images were acquired with a twice-refocused spin echo echo-planar-imaging sequence (Reese et al, 2003) using the following parameters: TR 12 seconds, echo time 94 ms, image matrix 128 × 128, field of view 220 × 220 mm2: 88 axial slices (no gap), 1.7 × 1.7 × 1.7 mm3 voxel size. Additionally, fat saturation was employed together with 6/8 partial Fourier imaging and generalized autocalibrating partially parallel acquisitions (Griswold et al, 2002; acceleration factor 2). Diffusion weighting was isotropically distributed along 60 diffusion-encoding gradient directions with a gradient pulse of
Data Analysis
T1-weighted images were preprocessed using standard parameters of the VBM8 toolbox (http://dbm.neuro.uni-jena.de/vbm.html). The VBM8 toolbox is designed for standard preprocessing of structural MRI data for later use in VBM analyses. The toolbox is based on the Statistical Parametric Mapping software version 8 (SPM8, Wellcome Trust Centre for Neuroimaging, University College London, London, UK; http://www.fil.ion.ucl.ac.uk/spm), a standard software for analyzing MRI data. SPM8 was run on Matlab version 7.11 (Mathworks Inc., Natick, MA, USA; www.mathworks.com/matlabcentral). Images were bias-field corrected, segmented and registered to standard Montreal Neurological Institute (MNI) space using rigid-body transformation and the unified segmentation approach (Ashburner and Friston, 2005). Subsequently, images were smoothed with a Gaussian kernel of 8 mm3 full width at half maximum. Resulting gray matter images were entered into a two-sample
T2-weighted images and FLAIR images were rated independently by two experienced clinicians blind to the clinical data according to the ARWMC scale (Wahlund et al, 2001). The two ratings were then averaged for further analyses. White matter lesions were defined as hyperintensities on both T2-weighted and FLAIR images of >5 mm diameter. Total score (range 0 to 30) as well as subscores for frontal, parieto-occipital, temporal, basal ganglia (including striatum, globus pallidus, thalamus, insula and internal and external capsules), and infratentorial regions (range 0 to 6) were calculated for white matter lesions. Lacunes were defined as hypointense signal alterations on both T2-weighted and FLAIR images >2 mm in minimal diameter and rated within the same regions. Interrater agreement was assessed by intraclass correlation coefficient
The DTI data were processed as follows: the seven images without diffusion weighting distributed in the whole sequence were used to estimate rigid motion correction parameters. Motion correction parameters were interpolated for all 67 volumes and combined with a global registration to the T1-weighted images. Diffusion-weighted images were skull stripped using the T1-weighted images and finally coregistered into MNI space. The gradient direction for each volume was corrected using the rotation parameters. The registered images were interpolated to the new reference frame with an isotropic voxel resolution of 1 mm3 and the three corresponding acquisitions and gradient directions were averaged. Finally, for each voxel, a diffusion tensor was fitted to the data and used to compute the parameters’ mean, axial and radial diffusivity and fractional anisotropy. For voxelwise statistical analysis of the diffusion data TBSS (Smith et al, 2006; provided by Functional magnetic resonance imaging of the brain Software Library, FSL) was used.
The next steps were performed analogously for all four parameters and will be explained only once for the fractional anisotropy. Fractional anisotropy maps of all participants were computed and subsequently skeletonized. The individual fractional anisotropy parameters were then back projected on the standard FSL fractional anisotropy template skeleton. Voxelwise cross-subject statistical analysis based on randomization tests (Nichols and Holmes, 2002) with 100,000 permutations was performed on the resulting data. Age and sex were included as covariates for all analyses, and the respective neuropsychological variable was added for the correlation analyses. The significance threshold was set to
Results
Clinical and Neuropsychological Characteristics
Samples were matched for age, sex, education, and premorbid intelligence. Overall cognitive functioning as assessed with the Mini-Mental State Examination was not different between patients and control subjects (Table 1). As expected, the patient group showed more white matter lesions than the healthy control group on the ARWMC rating scale except in infratentorial regions, and more overall lacunes. Regional subscales of the lacunar rating did not reach significance. Interrater reliability of the ARWMC ratings as assessed by intraclass correlation coefficient
Cognitive performance in patients with cerebral microangiopathy and healthy control subjects
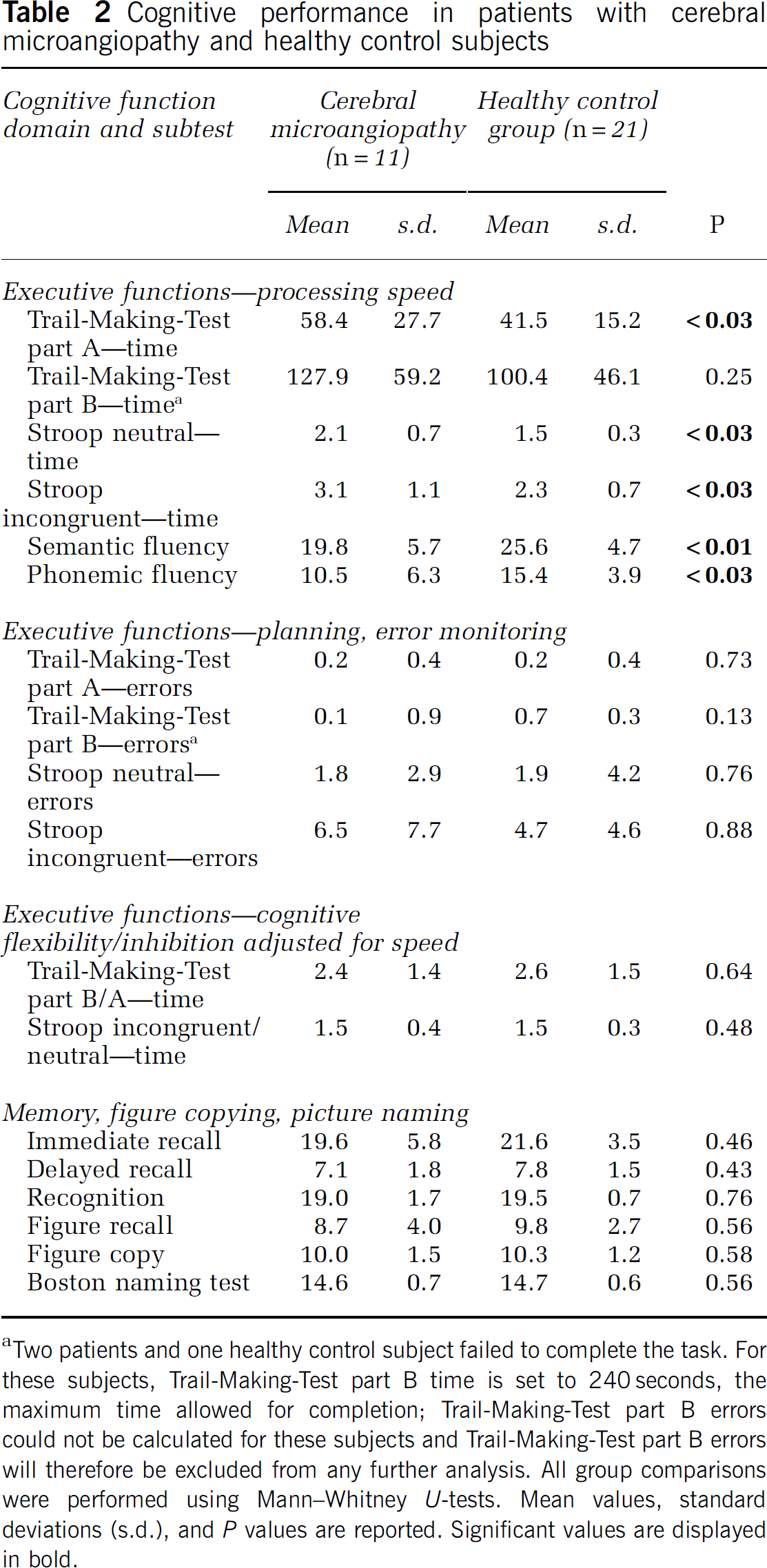
Two patients and one healthy control subject failed to complete the task. For these subjects, Trail-Making-Test part B time is set to 240 seconds, the maximum time allowed for completion; Trail-Making-Test part B errors could not be calculated for these subjects and Trail-Making-Test part B errors will therefore be excluded from any further analysis. All group comparisons were performed using Mann–Whitney
Imaging Data—White Matter Parameters
The group comparison of DTI-derived white matter parameters yielded significant alterations in early CMA. Patients showed widespread increases in mean, axial and radial diffusivity, but reduced fractional anisotropy in comparison with the healthy control group (Figure 1). Further exploratory analysis using SPM8-based parametric methods revealed that effect sizes at peak coordinates of clusters showing significant group differences were equally large for axial (range 2.1 to 4.2) and radial (range 1.9 to 3.9) diffusivity, slightly smaller for mean diffusivity (range 0.9 to 2.5), and markedly smaller for fractional anisotropy (range 0.1 to 0.2).
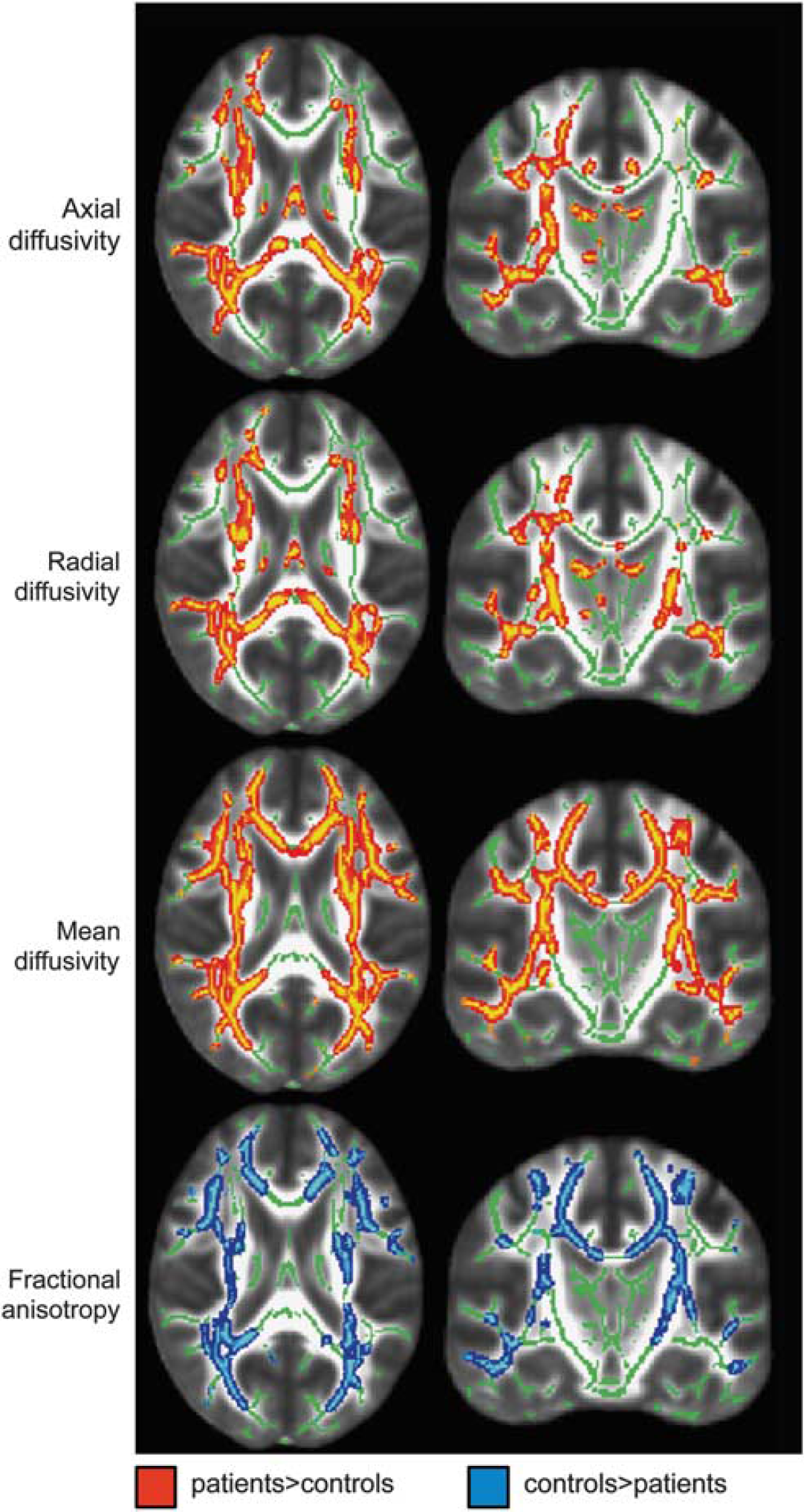
Transversal (
The following analyses explored the association between white matter changes and executive performance in the whole sample. Significant correlations were found between the total score as well as the frontal and parieto-occipital subscales of the ARWMC scale with five of the six processing speed tasks (Table 3). No correlations were found for further subscales of the white matter lesion rating and any other test measure. With respect to the lacunar rating, only the total score correlated with Trail-Making-Test part A errors (
Spearman correlation coefficients between age-related white matter changes rating scale and executive functions
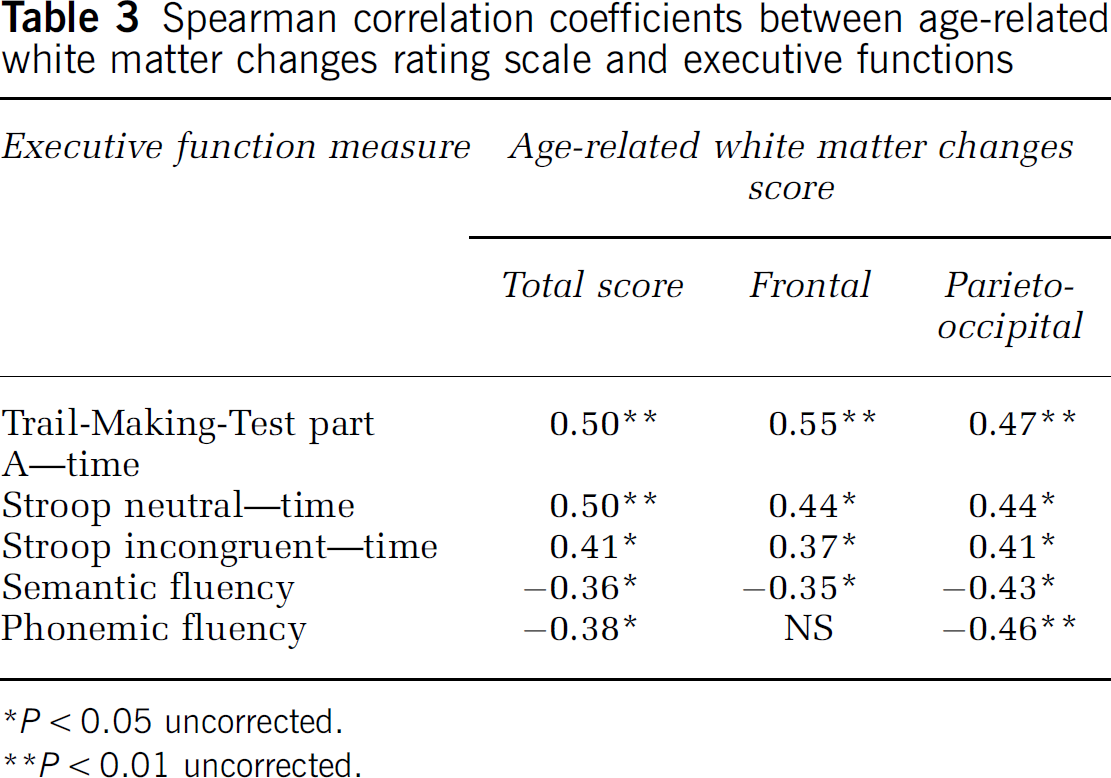
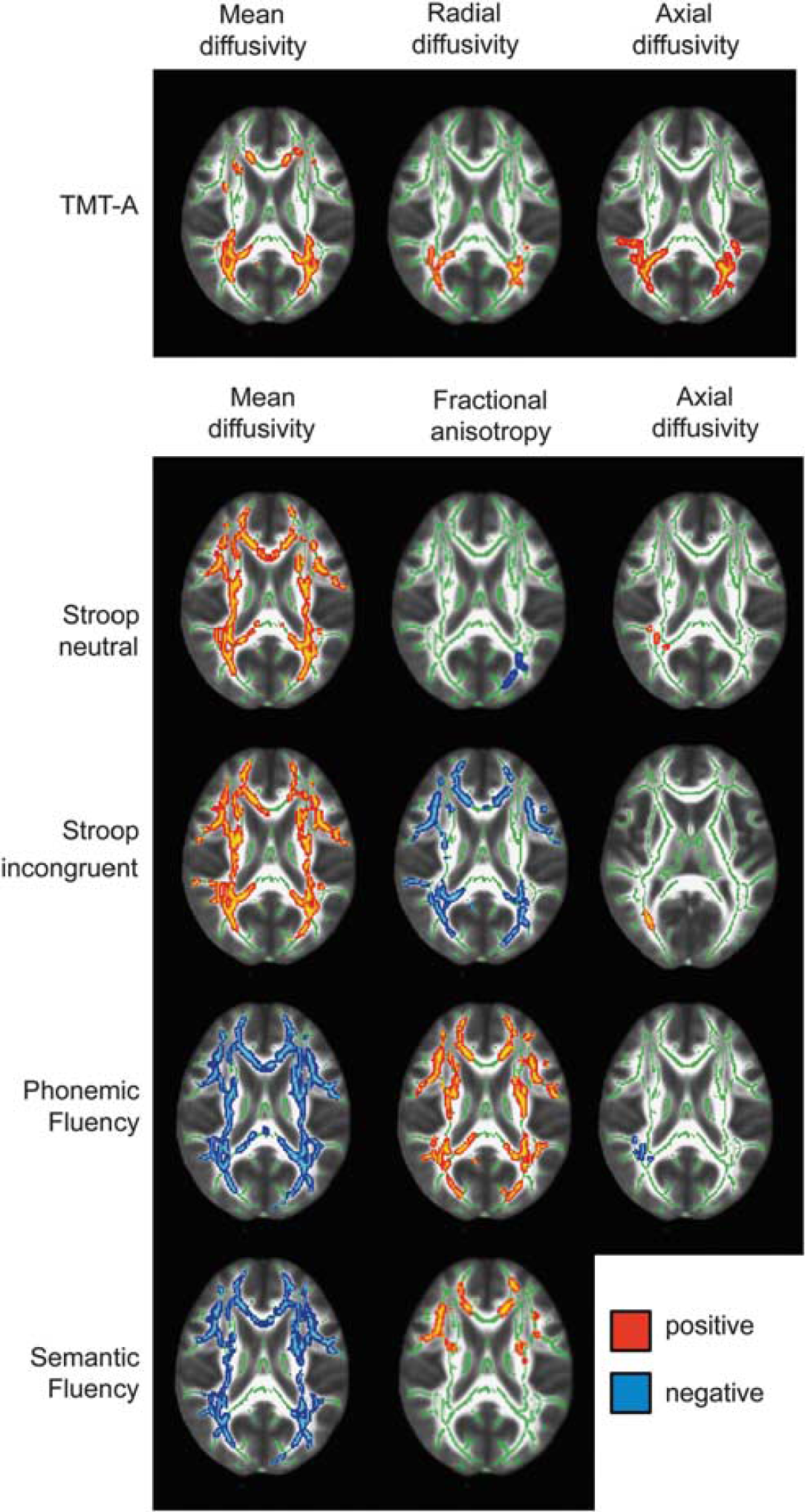
Transversal brain slices (
Imaging Data—Gray Matter Parameters
In contrast to white matter parameters, the group comparison of gray matter parameters revealed only small group differences in early CMA. Patients showed reduced gray matter density in left (peak at
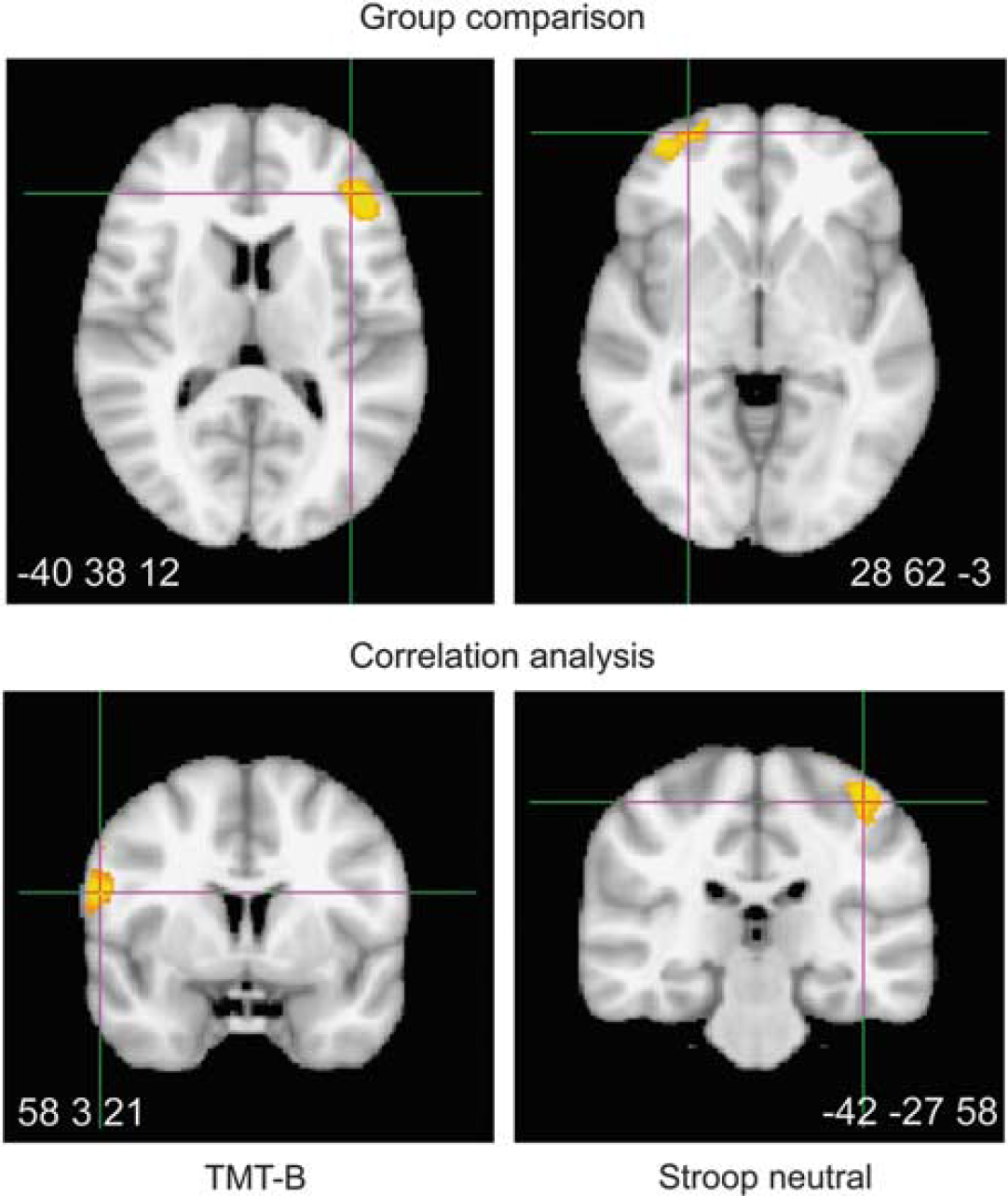
Group comparison and significant correlations with executive functions in voxel-based morphometry analysis shown at Montreal Neurological Institute coordinates of peak activation: Patients showed reduced gray matter density in two frontal regions (voxelwise
Additionally, to explore nonvascular influences on the present results, total gray matter atrophy was compared between groups by analyzing total gray matter volume and gray matter volume relative to total intracranial volume as calculated from the VBM8 toolbox. No significant group differences were found for either gray matter volume (patients mean=606.8, s.d.=46.9; control subjects mean=612.5, s.d.=46.9;
Discussion
Our study aimed at specifying executive dysfunctions and relating these deficits to neural correlates in early CMA—vascular mild cognitive impairment. Regarding imaging data, patients with early CMA showed widespread alterations in white matter parameters, in particular increases in mean diffusivity, axial and radial diffusivity as well as a reduction in fractional anisotropy. Taken together, this implies widespread disruption of white matter tracts. Exploratory parametric analyses revealed that large and comparable effect sizes are present for mean diffusivity, axial and radial diffusivity whereas the effect of fractional anisotropy reduction is smaller. This result is plausible keeping in mind the relationship between the DTI parameters. As both axial and radial diffusivity are increased to a similar extent, the overall directionality of diffusion cannot change much. Pathophysiologically, though necessarily interpretative, our results indicate that both myelin and axonal destruction within white matter tracts occur in early CMA caused by microangiopathic pathology. From the results of the TBSS group comparison alone, we can however not draw any conclusions concerning regionally specific hypotheses as alterations were widespread throughout.
Neuropsychologically, we found evidence in favor of a specific involvement of the speed component of executive tasks, in line with other findings (Peters et al, 2005; Jokinen et al, 2009). Interestingly, these also correlated most strongly with total and regionally specific ARWMC and diffusion parameters. More specifically, timed tasks correlated with white matter lesion ratings especially in frontal and parieto-occipital regions. This is in concordance with the longstanding hypothesis that fronto-parietal networks are primarily disrupted in CMA (Cummings, 1995).
Most interesting results were obtained for white matter diffusion-weighted parameters. The more unspecific parameters mean diffusivity and fractional anisotropy correlated with nearly all speeded tasks but none of the speed-corrected executive scores, which is opposing O’Sullivan et al (2005). They found correlations between mean diffusivity and speed-corrected executive measures only. They investigated a group of middle-aged patients (mean age 46 years) with a genetic variant of CMA. Differences in disease etiology, age, and analysis details—VBM style versus TBSS approach—may account for the discrepancies. Axial and radial diffusivity represent more specific parameters indicating degeneration in axonal and myelin structures (Alexander et al, 2007; Song et al, 2003). Here, we revealed regionally more specific results. Whereas axial diffusivity correlated closely with various speeded measures in parietal regions, radial diffusivity showed such a correlation for the Trail-Making-Test part A only. Furthermore, axial and radial diffusivity again did not correlate with speed-corrected executive measures. Interestingly, the results of the ARWMC rating and TBSS were highly concordant, opposing results that DTI data show stronger correlations with neuropsychological data than white matter lesion ratings (Patel and Markus, 2011).
Taken together the group comparison and the correlation analyses, we can thus conclude that already in early CMA white matter is widely disrupted and that the disruption of putatively axonal structures particularly in parietal areas causes cognitive slowing. On the contrary, more purified measures of executive functions by name inhibition, cognitive flexibility, error monitoring, and planning were not associated with white matter changes.
This is an important finding especially considering the vast literature claiming specific executive dysfunction in vascular type dementia (Román et al, 2002; O’Sullivan et al, 2004; Debette et al, 2007; Carey et al, 2008) from studies that largely included speeded executive tasks. Clearly, the concept of executive functions includes speed as one of its components (Stuss and Alexander, 2007). However, the speed component is overrepresented in executive function results and interpretations. The current results however imply distinct relationships between speeded and nonspeeded executive components and thus favor approaches that separate the broad concept of executive functions into its subcomponents (O’Sullivan et al, 2005; Peters et al, 2005; Prins et al, 2005). Considering the small sample size, sensitivity may have been too low to detect correlations with speed-corrected measures. Therefore, the proposed distinction between speeded and nonspeeded executive subcomponents in the context of CMA should be confirmed in a larger sample. Furthermore, we investigated patients in an early disease stage only and the subcomponents of executive functions may be distinctively affected during the course of the disease so that their relationship changes with disease progression. An isolated impairment of cognitive speed may thus be characteristic of early disease stage only (Peters et al, 2005; Prins et al, 2005). Nonetheless, a separate analysis of executive subcomponents seems to be a reasonable approach to increase the understanding of executive dysfunctions in clinical studies and we thus recommend doing so in future studies.
Gray matter was reduced exclusively in frontal areas, again in line with the fronto-parietal circuit hypothesis and previous work (Cummings, 1995; Li et al, 2011; Raji et al, 2012). Correlations were found between two timed tasks and premotor and somatosensory areas, implying the involvement of basic perceptive and motor functions in the observed slowing. In sum, we found consistent correlations with speed in both white and gray matter with an equally consistent stress on frontal and parietal areas.
Shortcomings of the present study are the small sample size and the cross-sectional design. We examined a homogenous group of patients with comparable disease status and carefully excluded comorbidities such as stroke or psychiatric illness resulting in a small but well-defined sample.
A close association between small vessel disease and Alzheimer's disease has been discussed (Iadecola, 2004). Longitudinal data on cognitive decline and information on the conversion to different types of dementia would therefore be especially interesting in a cohort of mildly affected patients with CMA as tested here. This was however beyond the scope of the current study as our sample would be too small to reliably analyze conversion data. We have however several indicators against substantial nonvascular influences on the current results. First, patients did not differ from control subjects on memory, visuoconstructive tasks or word finding, which is the main clinical finding in Alzheimer's disease (Dubois et al, 2007; McKhann et al, 2011). Second, absolute and relative gray matter volumes did not differ between groups. Third, whole-brain VBM analysis revealed group differences exclusively in frontal areas and visual ratings likewise found predominantly frontal and parieto-occipital involvement in the studied patient group and none of the results points to any hippocampal involvement well known from Alzheimer's disease (Schroeter et al, 2009). Therefore, it seems unlikely that Alzheimer's disease-related brain changes account for the reported group differences. Another differential diagnosis—frontotemporal dementia as the second most common dementia syndrome in subjects under 65 years—is unlikely confounding present results either as patients did not show personality and behavioral changes (Neary et al, 2005; Schroeter et al, 2007b).
A further limitation of our study is the use of a relatively brief test battery of executive tasks rather than a comprehensive test battery including more basic reaction time tasks. However, we were able to include a series of tasks traditionally used for studying executive functions in diseases so that our results are well comparable to previous work. We however acknowledge the benefit of including more basic reaction time tasks as well as more speed-independent executive tasks in future studies, especially given the current results that show a clear distinction between speeded and nonspeeded task components.
In conclusion, our study shows that early CMA disrupts white matter tracts throughout the brain. Axonal degeneration particularly in parietal areas is associated with cognitive slowing. On the contrary, we could not show any impairment in speed-corrected measures of executive functions in early CMA and no association between speed-corrected executive measures and white matter.
