Abstract
Cytosine-phosphate-guanine (CpG) preconditioning reprograms the genomic response to stroke to protect the brain against ischemic injury. The mechanisms underlying genomic reprogramming are incompletely understood. MicroRNAs (miRNAs) regulate gene expression; however, their role in modulating gene responses produced by CpG preconditioning is unknown. We evaluated brain miRNA expression in response to CpG preconditioning before and after stroke using microarray. Importantly, we have data from previous gene microarrays under the same conditions, which allowed integration of miRNA and gene expression data to specifically identify regulated miRNA gene targets. CpG preconditioning did not significantly alter miRNA expression before stroke, indicating that miRNA regulation is not critical for the initiation of preconditioning-induced neuroprotection. However, after stroke, differentially regulated miRNAs between CpG- and saline-treated animals associated with the upregulation of several neuroprotective genes, implicating these miRNAs in genomic reprogramming that increases neuroprotection. Statistical analysis revealed that the miRNA targets were enriched in the gene population regulated in the setting of stroke, implying that miRNAs likely orchestrate this gene expression. These data suggest that miRNAs regulate endogenous responses to stroke and that manipulation of these miRNAs may have the potential to acutely activate novel neuroprotective processes that reduce damage.
Introduction
Stroke causes injury to the brain that can result in severe physical disability, cognitive dysfunction, and death. Thus far there is only one treatment available—administration of tissue plasminogen activator—which is limited to <5% of stroke patients because of its narrow window of efficacy and significant risk of dangerous side effects. 1 Research into mechanisms that may protect the brain against stroke injury is needed to decipher this important process and to identify novel therapeutic targets.
Preconditioning is a phenomenon whereby pretreatment with a low dose of an otherwise harmful stimulus induces tolerance against a subsequent injurious event, such as stroke. Several preconditioning stimuli can protect the brain against ischemic injury, including brief ischemia, volatile anesthetics, and Toll-like receptor ligands. 2 We and others have shown that preconditioning reprograms the genomic response to injury resulting in increased expression of protective genes, including interferon-related genes,3, 4 stress responses such as heat-shock proteins,5, 6 and antiapoptotic genes such as Bcl2.5, 6, 7, 8 Although these studies have focused on gene and protein expression, little is known about how these new responses originate.
Functional microRNAs (miRNAs) are short noncoding nucleotide sequences 22 to 25 nucleotides in length that modulate gene expression by degrading messenger RNA (mRNA) or interfering with protein translation. Each miRNA has many potential mRNA targets; thus, a broad spectrum of gene expression can be affected by the presence or absence of an individual miRNA. It is plausible that miRNAs may regulate the genomic changes found in endogenous neuroprotection induced by preconditioning. As such, miRNAs could serve as superior therapeutic targets, compared with individual genes and proteins, because of their ability to target multiple endogenous genes and proteins at one time to promote protection.
The role of miRNAs in stroke and ischemic preconditioning is a recent area of research focus. Several studies using miRNA microarrays reveal robust regulation of miRNAs after ischemic injury as well as other types of neuronal damage or degeneration. These studies have identified potential miRNAs that may influence stroke outcome. For example, the induction of miR-497 induces neuronal death by suppressing Bcl2 and downregulation of miR-181b promotes protection against ischemic injury
Here we exposed mice to a preconditioning stimulus using the Toll-like receptor 9 agonist cytosine-phosphate-guanine (CpG) oligodeoxynucleotide, which is known to reduce ischemic injury dramatically in both rodent and nonhuman primate models of experimental stroke.13, 14 We performed a large-scale miRNA microarray study using multiple time points before and after induction of middle cerebral artery occlusion (MCAO) in mice to evaluate the influence of CpG preconditioning on miRNA expression in the brain. We integrated the miRNA expression data with mRNA gene expression data that was generated from a previous microarray experiment that used CpG-preconditioned animals under the same time points and conditions. To our knowledge, we are the first to integrate miRNA with gene (mRNA) microarray expression in preconditioning and stroke to understand the influence of miRNAs on stroke pathogenesis and protection. We found that CpG preconditioning does not alter miRNA expression in the brain before stroke, which shows that miRNA regulation is not needed before stroke to implement neuroprotection. After stroke there is a large set of miRNAs regulated within 24 hours regardless of exposure to preconditioning. These shared miRNAs associated with genes involved in processes typically seen in stroke, including inflammation, metabolism, and excitotoxicity. Importantly, a unique set of miRNAs was differentially regulated in preconditioned animals revealing insights into the mechanism of neuroprotection. The unique miRNAs that were suppressed in the CpG-preconditioned mice compared with saline-treated mice are associated with the upregulation of several known or potential neuroprotective genes, indicating that these miRNAs promote new gene regulation that could reduce ischemic injury. These data implicate miRNAs as master regulators of endogenous responses to stroke and suggest that manipulation of these miRNAs could serve as a novel therapeutic approach for acute stroke therapy.
Materials and Methods
Animals
C57Bl/6J mice (male, 8 to 12 weeks) were purchased from Jackson Laboratories (West Sacramento, CA, USA). Mice were caged in age-matched cohorts under specific pathogen-free conditions and housed at Oregon Health & Science University (OHSU) in a facility approved by the Association for Assessment and Accreditation of Laboratory Animal Care International. All mice were maintained on a 12-hour light/dark cycle and given free access to food and water. Animal protocols were approved by the OHSU Institutional Animal Care and Use Committee (OWLAW# A3304-01) and met the guidelines set forth by the National Institutes of Health.
Middle Cerebral Artery Occlusion
Mice were injected subcutaneously with 1.6 mg/kg CpG 1826 (InvivoGen, San Diego, CA, USA) or saline 72 hours before MCAO. All mice were randomized at the time of drug treatment with the surgeon masked to treatment. Mice were anesthetized with isoflurane (1.5% to 2%) and subjected to MCAO using the monofilament suture method described previously. 3 Briefly, a silicone-coated 7-0 monofilament nylon surgical suture (Doccol, Sharon, MA, USA) was threaded through the external carotid artery to the internal carotid artery to block the middle cerebral artery, and maintained intraluminally for 45 minutes. The suture was then removed to restore blood flow. Cerebral blood flow was monitored throughout surgery by laser Doppler flowmetry. Any mouse that did not maintain a cerebral blood flow during occlusion of <25% of baseline was excluded from the study. Body temperature was monitored and maintained at 37°C with a thermostat-controlled heating pad.
Tissue Extraction
Under deep isoflurane anesthesia, the mice were perfused with heparinized (2 U/mL) saline followed by rapid removal of the brain. The olfactory bulbs were removed and the first 4 mm of tissue was collected beginning at the rostral end. The striatum was dissected and removed and the remaining cortex was used for RNA isolation. The S5 1-mm coronal section was taken for triphenyltetrazolium chloride staining to assess infarct. The investigator performing the tissue extraction, triphenyltetrazolium chloride staining, and infarct assessment was masked to the experimental condition.
Microarray
MiRNA microarrays were performed on miRNA isolated from cortical tissue obtained from untreated mice that did not undergo MCAO, CpG, or saline-preconditioned mice at 3, 24, or 72 hours after preconditioning and 3 and 24 hours after MCAO (
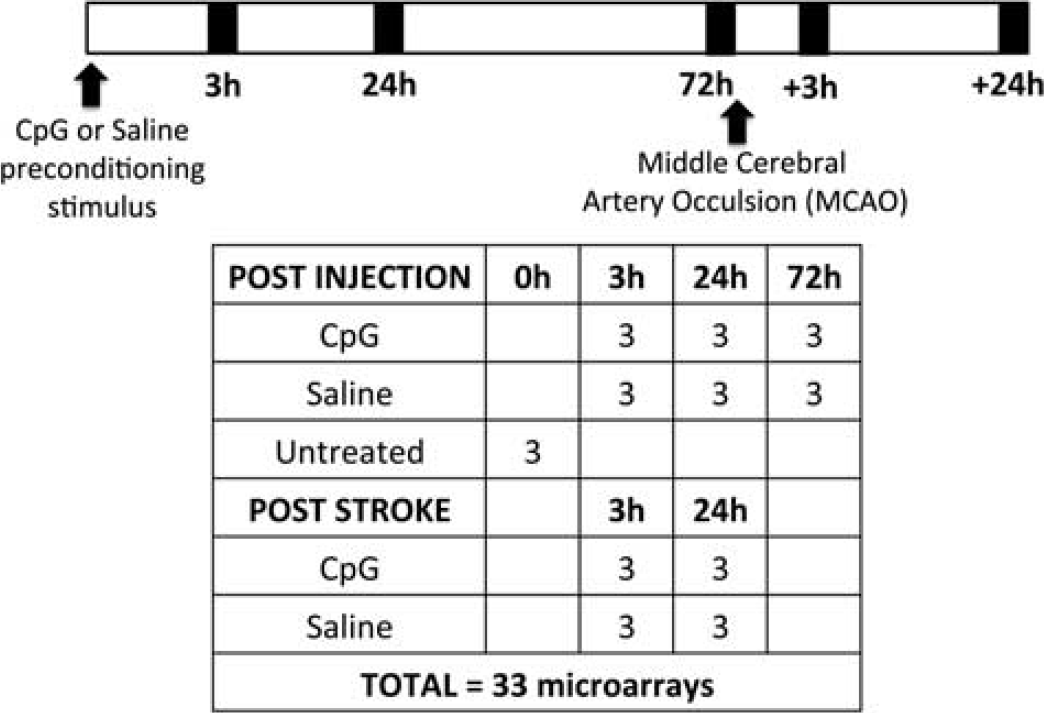
Outline of experimental design. Timeline indicates treatment, middle cerebral artery occlusion (MCAO), and time of total RNA isolation. The table delineates treatment groups and group size (
The gene expression data used in this study was obtained from our previously described microarray experiments wherein the brain cortical response was analyzed in CpG or saline-preconditioned animals at 3, 24, and 72 hours after treatment and 3 and 24 hours after stroke (
Microarray Analysis
Affymetrix CEL files were uploaded into Partek (St Louis, MO, USA) software and normalized using robust multi-array average. Analysis of variance was performed with least-square means
Integration of miRNA and mRNA
The miRWalk ‘Predicted Targets’ web resource (http://www.umm.uni-heidelberg.de/apps/zmf/mirwalk/micrornapredictedtarget.html) was used to acquire miRNA target predictions based on the miRWalk algorithm.
16
There are no database files available for download, thus results from individual queries for each miRNA of interest using default settings were combined to generate a file of miRWalk-based predictions. To convert gene symbols in the predictions to match the probe IDs used in the association networks, each miRNA—gene prediction was expanded to include all probe IDs matching the symbol. Since the acquired targeting predictions all had an associated
Total RNA Isolation, Reverse Transcription, and qtPCR
Total RNA (containing miRNA and mRNA) was isolated from cortical tissue using a mirVana RNA isolation kit (Life Technologies, Grand Island, NY, USA). Reverse transcription of miRNA was performed using TaqMan miRNA reverse transcription kit (Life Technologies) targeted for miR-762, miR-1892, miR-1894-3p, miR-1224, and U6 control. Reverse transcription for mRNA gene expression was performed using Omniscript reverse transcription kit (Qiagen, Valencia, CA, USA). Quantitative PCR (qtPCR) was performed using TaqMan MiRNA expression assays and TaqMan gene expression assays (Life Technologies) for each miRNA or gene of interest on an ABI Prism 7700 (Life Technologies). MiRNA and gene expression results were normalized to U6 and β-actin expression, respectively, and analyzed relative to their saline-treated or -untreated counterparts. We confirmed that U6 and actin expression was not influenced by CpG treatment or ischemic injury (data not shown). The relative quantification of the gene of interest was determined using the comparative cycle threshold method (2−DDCt). Statistics were performed on average delta-delta-ct values from 2 to 3 independent PCR experiments. Samples were considered to be an outlier if they were >2 s.d. outside of the average.
Statistical Analysis
Statistics for miRNA and gene array are described above. Data are represented as mean±s.e.m. The
Results
MiRNA Expression is Regulated Robustly 24 hours after Stroke
MiRNA expression was measured in saline-treated or CpG-preconditioned animals 3, 24, and 72 hours after injection and 3 and 24 hours after stroke (Figure 1). As previously published, CpG-preconditioned mice showed marked neuroprotection with significantly reduced infarct size compared with saline-treated mice (15.2%±3.9% versus 49.6%±16.3%, respectively,
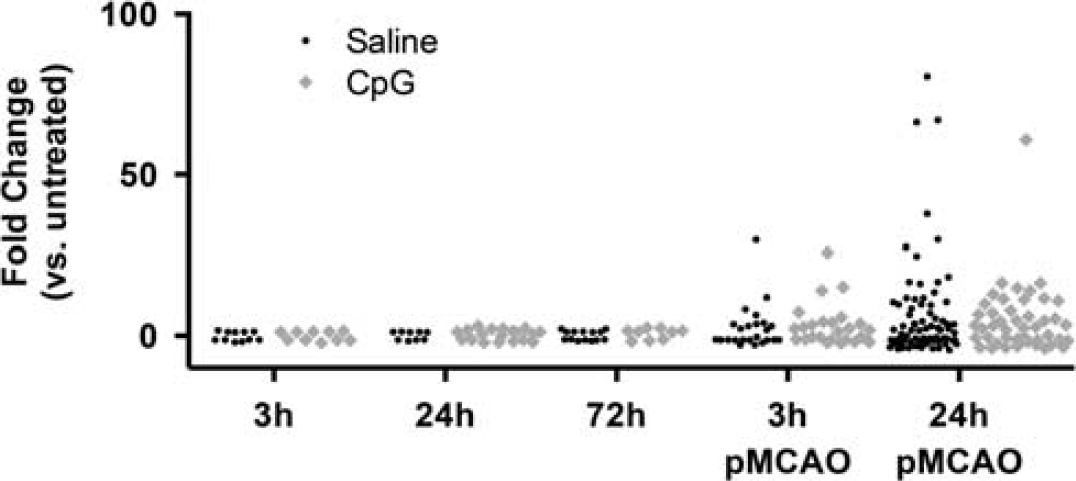
MiRNA (microRNA) expression over time. Significantly regulated miRNAs (
After stroke, an increased number of miRNAs were altered at 3 and 24 hours in both treatment groups. All miRNAs that were regulated at 3 hours after MCAO were also regulated 24 hours after MCAO. The largest number of miRNAs was regulated 24 hours after MCAO and these miRNAs also showed the greatest magnitude of change. Fewer total miRNAs were regulated in the CpG-preconditioned animals (57 miRNAs) compared with saline-treated animals (73 miRNAs). The dramatic reduction in the number of miRNAs regulated in CpG-preconditioned animals versus saline-treated animals indicates that the CpG preconditioning alters the miRNA response to stroke.
Three Distinct miRNA Expression Patterns 24 hours after Stroke
We examined the miRNA expression patterns in CpG-preconditioned and saline-treated animals 24 hours after stroke. We selected all miRNAs that were significantly regulated compared with nonstroked animals in either treatment group. Hierarchical clustering of the miRNAs in the untreated, saline-treated, and CpG-preconditioned animals 24 hours after stroke identified three distinct groups of miRNA expression profiles (Figure 3). The miRNAs that were downregulated 24 hours after stroke in both CpG- and saline-treated animals were assigned to one group referred to as ‘downregulated’ containing 40 miRNA. MiRNAs that were upregulated 24 hours after stroke in both CpG- and saline-treated animals were assigned to another group referred to as ‘upregulated’ containing 11 miRNA. MiRNAs that were upregulated in saline stroke and attenuated in CpG-preconditioned animals after stroke were assigned to a group referred to as ‘differential’ containing 22 miRNAs. The miRNAs were grouped into these categories to analyze the influence of the miRNAs on gene expression after stroke.
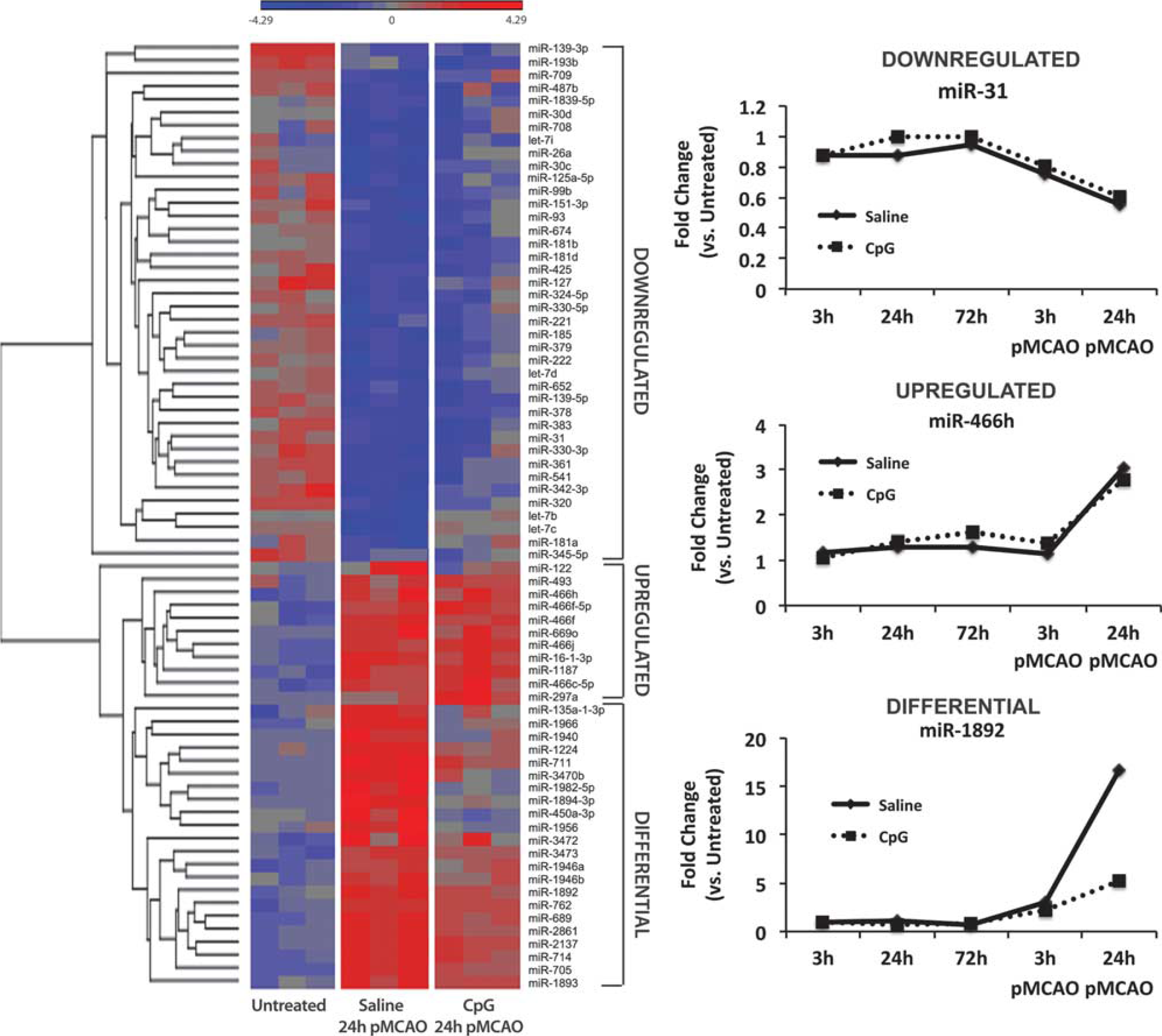
MiRNA expression after stroke. Left: Heatmap of hierarchical clustering showing 73 significantly regulated miRNAs in untreated, saline-treated, and CpG-preconditioned animals 24 hours after stroke. Regulated miRNAs have three distinct expression profiles compared with untreated animals: downregulated, downregulated miRNAs in saline and CpG; upregulated, upregulated miRNAs in saline and CpG; and differential, differentially regulated miRNAs between saline and CpG, where the miRNAs are upregulated in saline-treated animals after stroke and attenuated in CpG-preconditioned animals after stroke. Right: expression of representative miRNA from each group. CpG, cytosine-phosphate-guanine.
MiRNA and mRNA Integration
We have previously performed mRNA gene microarray analyses on mouse cortical tissue under the same conditions and time points. We took advantage of the availability of these data to analyze the influence of miRNA expression on mRNA gene expression in this system. Figure 4 outlines the workflow we used to integrate our miRNA microarray data with our previously obtained mRNA gene microarray data. Genes were selected that were significantly regulated by analysis of variance with false discovery rate
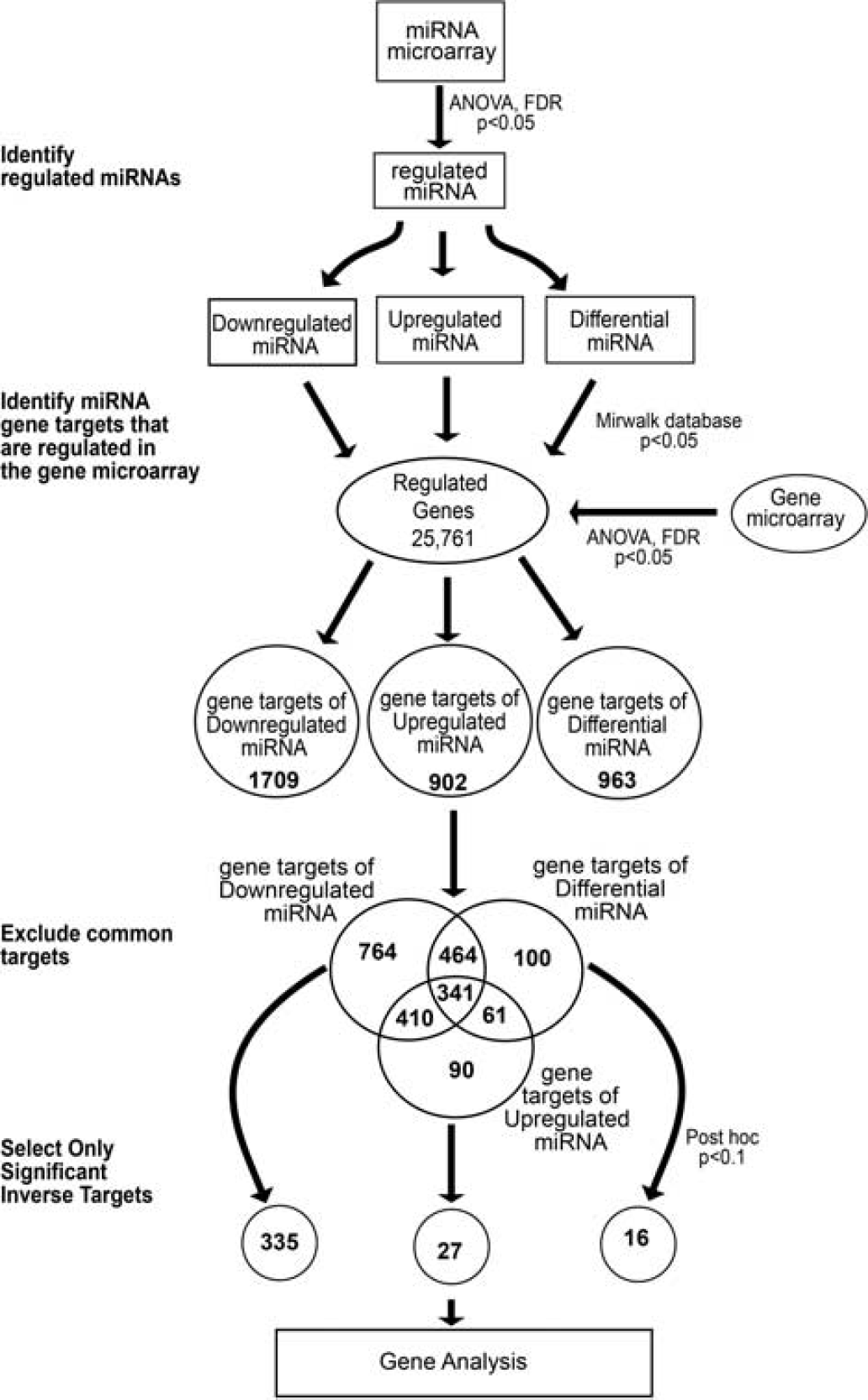
MiRNA-gene integration and analysis. Significantly regulated miRNAs were selected and separated into three groups based on expression pattern. Significantly regulated gene transcripts were selected from previously obtained gene microarray data. miRWalk database was used to identify gene targets of each group of miRNAs; only those genes that were significantly regulated in the gene microarray were included for further analysis. The common gene targets between each miRNA group were excluded to increase specificity. The unique gene targets were separated by regulation and only those that were inversely regulated 24 hours after stroke were selected for further analysis.
Next, we determined the likelihood that the regulated miRNAs were responsible for the gene expression changes observed after stroke. Statistical analysis using Fisher's exact test revealed that there was significant (
Predicted Influence of MiRNA Expression on Stroke
The relationships between the miRNAs and their inverse targets are shown in Figure 5A, whereby the large circles represent miRNAs and small circles represent the gene targets (red, upregulated; green, downregulated). The lines in Figure 5A connect each miRNA to a predicted target. Some miRNAs have a large number of inverse gene targets, including miR-709, miR-181d, miR-1187, and miR-466c-5p; while others have just one inverse target such as miR-127 and miR-16-1-3p; or no inverse targets such as miRNA-487b.
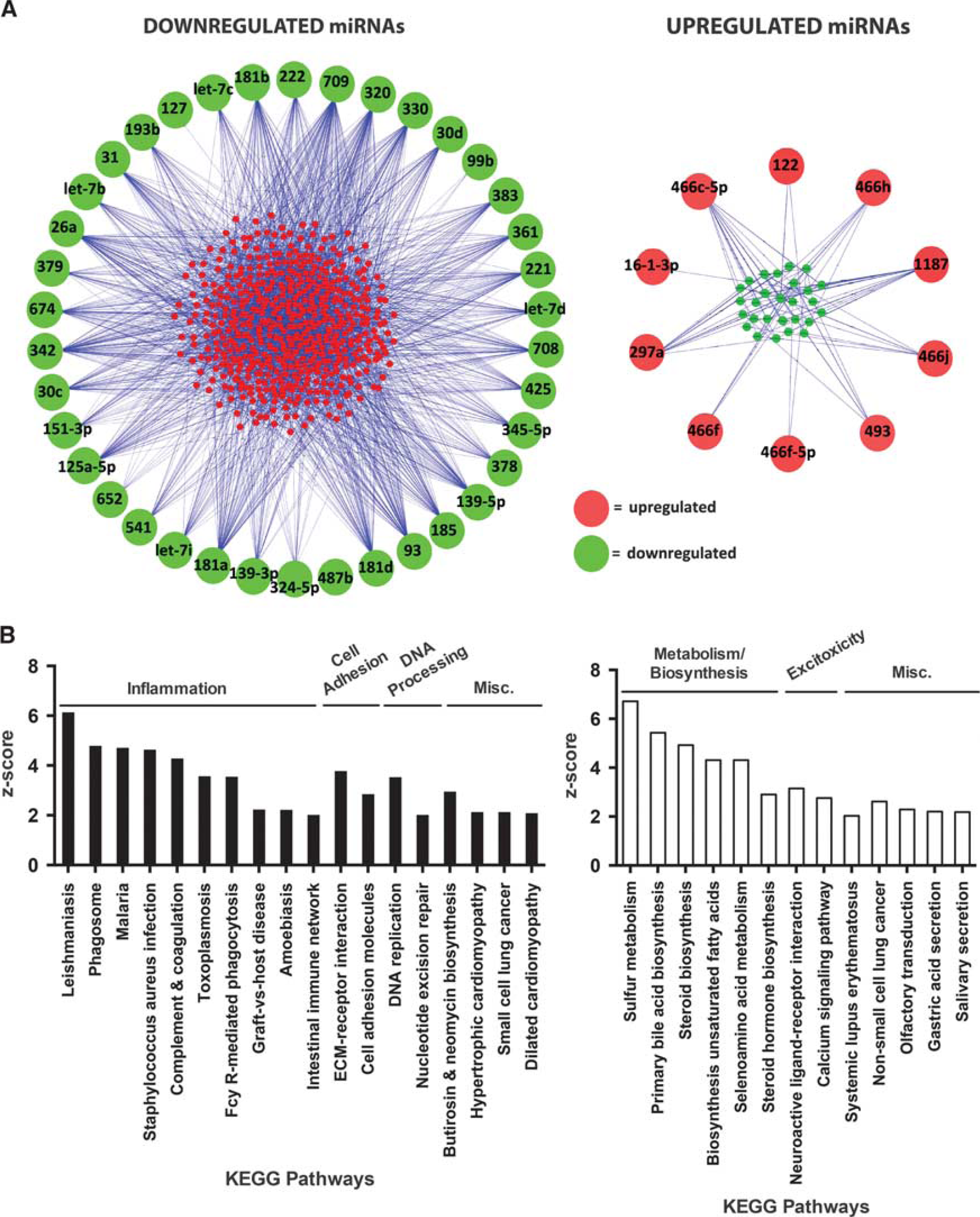
Analysis of gene targets of the shared downregulated and upregulated miRNA groups. (
To determine the potential influence of the shared downregulated miRNAs and upregulated miRNAs after stroke, we analyzed the Kyoto encyclopedia of genes and genomes pathways of their inverse gene targets. Analysis of the increased gene targets of the downregulated miRNA group revealed that pathways associated with inflammatory responses were significantly overrepresented in these genes (Figure 5B, left). Analysis of the decreased gene targets of the upregulated miRNA group revealed overrepresentation of pathways associated with metabolism/biosynthesis and excitotoxicity (Figure 5B, right). These data suggest that the downregulated miRNAs may increase inflammation in response to stroke, while the upregulated miRNAs may suppress metabolism/biosynthesis and excitotoxicity in response to stroke. Increased inflammation is known to exacerbate stroke injury, while suppression of metabolism and excitotoxicity protects against cellular damage in the brain. The shared regulation of these miRNAs between saline-treated and CpG-preconditioned animals after stroke indicates that pathways that are damaging and protective are engaged in response to stroke. In stroke alone, injury may stem from the large number of downregulated miRNAs that influence pathways of injurious inflammation, which outweighs the few upregulated miRNAs that influence protective pathways.
MiRNA Suppression in Neuroprotection
To identify potential differences between neuroprotective and injurious responses, we evaluated the 22 miRNAs in the differential group that had a significantly suppressed expression profile in CpG-preconditioned animals compared with saline-treated animals 24 hours after stroke (Figure 6A). To determine the potential impact of the suppression of these 22 miRNAs on stroke injury, we identified the unique inversely related gene targets following the workflow described in Figure 4. Because this group of miRNAs was downregulated in CpG-preconditioned animals compared with saline-treated animals 24 hours after stroke, we selected inverse gene targets that were upregulated to identify potential differences between the neuroprotective and injurious response. The miRNAs in this group had a total of 16 upregulated gene targets (Figure 6B and Table 1). The relationships between the suppressed miRNAs and their inverse gene targets are shown in Figure 6B indicating that miR-762, miR-1892, miR-1224, and miR-1894-3p all have three or more inversely related targets suggesting that they may highly influence the genomic response to stroke in preconditioned animals. We used qtPCR to validate the expression of these miRNAs. We found a significant decrease in miR-762 and miR-1894-3p expression in CpG-preconditioned animals compared with saline-treated animals (Figure 6C). MiR-1892 showed a trend for suppressed expression in CpG-preconditioned animals compared with saline and miR-1224 showed no difference between treatment groups (Figure 6C). In addition, we found that Nupr1 was targeted by three suppressed miRNAs, including miR-762 and miR-1892, making it the most targeted gene by miRNAs in this group and suggesting that it may be an important gene in neuroprotection. Using qtPCR, we validated that Nupr1 was significantly increased in CpG-preconditioned versus saline-treated animals 24 hours after stroke (Figure 6D).
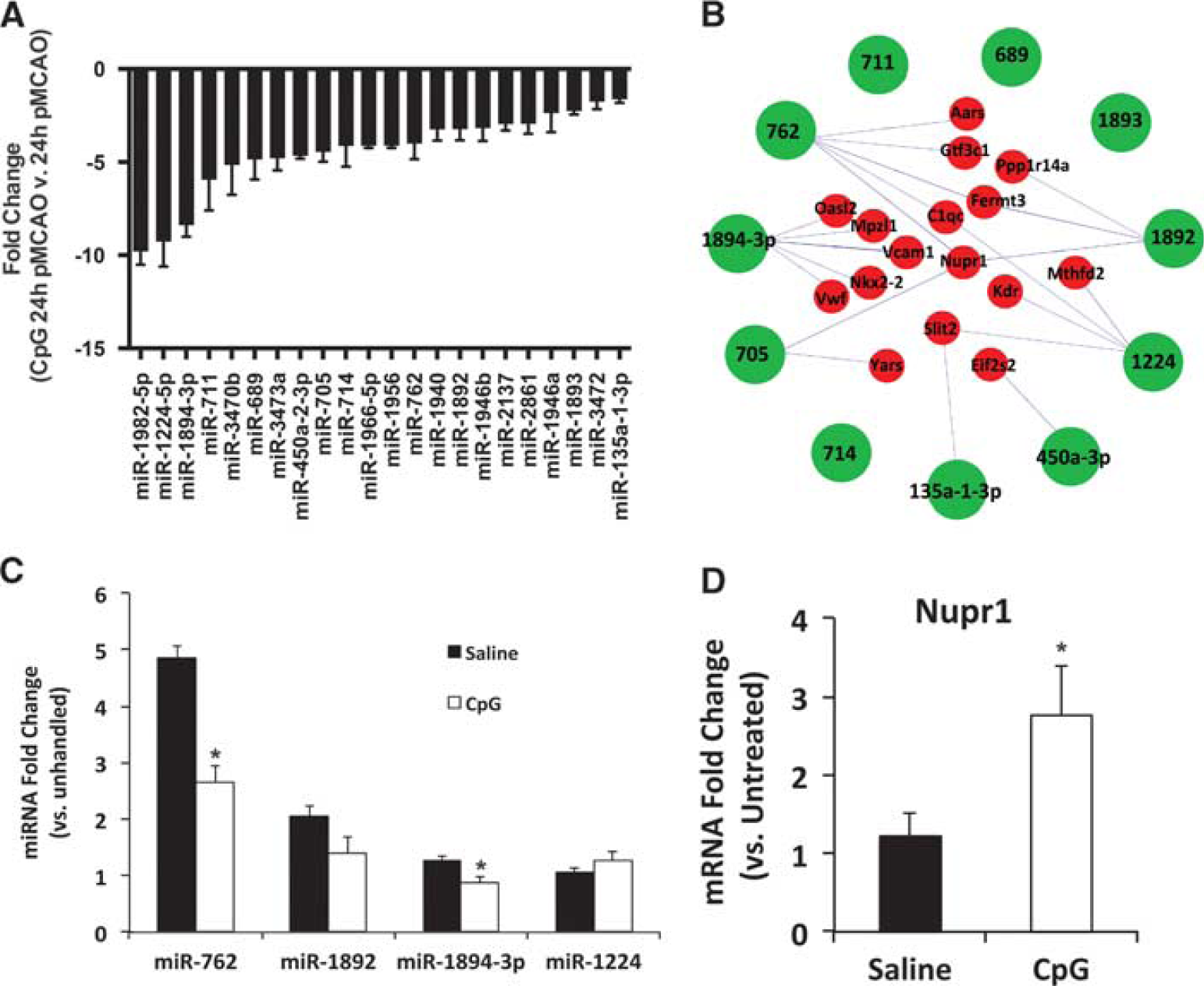
Differentially regulated miRNAs (microRNAs) in CpG-preconditioning—induced neuroprotection. (
Inverse targets of suppressed differential miRNA (CpG versus saline, 24-hour pMCAO)

CpG, cytosine-phosphate guanine; IFN, interferon; pMCAO, permanent middle cerebral artery occlusion; vWF, von Willebrand factor.
Suppressed miRNAs Associated with Reprogrammed Neuroprotective Genomic Response
The functions of each of the 16 upregulated genes targeted by these suppressed miRNAs were evaluated based on the scientific literature. Interestingly, 8 of 16 genes had previously known or potentially protective functions in stroke or ischemia (Table 1). Two genes, Aars and Yars, are both associated with aminoacyl-tRNA biosynthesis and mutations in these genes are known to increase the risk of stroke and neurodegeneration.17, 18 KDR, the vascular endothelial growth factor receptor 2, has several known neuroprotective functions, including induction of angiogenesis and neurogenesis. Slit2 decreases platelet adhesion and vascular reactivity, 19 which may decrease blood coagulation and may prevent detrimental vascular inflammation that causes damage during stroke. Oasl2 has been previously associated with the neuroprotective response induced by several different preconditioning stimuli 3 and Nupr1 is a gene typically associated with cancer that is known to increase cell survival in response to cellular stress.20, 21 Nkx2-2 has neuroprotective potential as it may be involved in restorative processes through adult myelination and oligodendrocyte maturation. 22 Finally, Mthfd2 is part of the folate pathway, and folic acid is known to be neuroprotective against stroke. 23 There were three genes (VWF, 24 VCAM1, and C1qc) that are involved in damaging pathways in stroke and five genes (Gtf3c1, Mpzl1, Fermt3, Ppp1r14a, and Eif2s2) that are associated with general cell functions. Taken together, these results suggest that suppression of these differential miRNAs upregulates key genes in preconditioned animals that may be essential to reduce injury in response to stroke. In nonpreconditioned animals the upregulation of these miRNAs would result in the suppression of these key neuroprotective genes, which is likely detrimental to stroke outcome. Thus, the suppression of these differential miRNAs in CpG preconditioning may reprogram the genomic response to stroke to provide an additional pathway to protection that shifts the endogenous response to stroke to favor a protective outcome.
Discussion
Preconditioning with CpG dramatically reduces injury after stroke. Our laboratory has shown that preconditioning alters gene expression in the brain before stroke and leads to a reprogrammed genomic response to injury that likely contributes to neuroprotection.3, 25 Despite robust gene expression changes in the brain after CpG administration, 3 we failed to find alterations in miRNA expression at several time points before stroke (3, 24, and 72 hours after CpG preconditioning). This indicates that gene changes before stroke may stem from altered transcriptional regulation and not from posttranscriptional miRNA regulation. These data also suggest that substantial alterations in miRNA expression in the brain before stroke are not necessary for the neuroprotection produced by systemic CpG preconditioning. This finding is in contrast to studies involving ischemic preconditioning where several influential miRNAs were identified, including miR-132, miR-182, and miR-429.11, 12 The difference in miRNA responses found between CpG and ischemic preconditioning likely stems from the nature of the stimulus. Ischemic preconditioning is an invasive procedure that directly targets the brain by inducing brief periods of cerebral ischemia. In contrast, CpG is administered systemically, leading to activation of cells in the periphery and effects the brain through blood—brain-barrier transfer or secondary mediators induced by CpG activation of the endothelium. Little is known about miRNA expression in the brain as a result of other systemic preconditioning agents, but these data suggest that systemic preconditioning agents may not require miRNA expression changes in the brain before stroke.
After stroke in both CpG-preconditioned and saline-treated mice, the group of downregulated miRNAs associated with an increase in injurious inflammatory gene expression that may exert damage after stroke. Many of the inflammatory genes that are predicted targets of these miRNAs are related to nuclear factor kappa B activation including Rela, Tifa, Ripk2, and NFκbiz (Supplementary Table I). Nuclear factor kappa B has a well-documented detrimental role in stroke and research shows that suppression of nuclear factor kappa B activation reduces ischemic injury.26, 27 It is somewhat surprising that the CpG-preconditioned animals had the same set of miRNAs downregulated as saline-treated animals that corresponded to increased inflammation after stroke, indicating the presence of detrimental mediators in the neuroprotected state. Such results have been reported showing some aspects of preconditioning that do not suppress inflammation. 28 In particular, preconditioning with the Toll-like receptor 4 ligand lipopolysaccharide did not reduce inflammatory gene expression in the neuroprotected state compared with stroke alone. 28 Thus, preconditioning with the Toll-like receptor ligands lipopolysaccharide and CpG does not appear to suppress expression of genes associated with inflammation.
The upregulated miRNA group of miRNAs in CpG-preconditioned and saline-treated mice is associated with the downregulation of metabolism/biosynthesis and excitotoxicity. It has been shown that at times of reduced oxygen availability, such as during hibernation, suppressed metabolic functions can be neuroprotective by reducing the energy demands of the cell.29, 30, 31, 32 Interestingly, previous studies in our laboratory using brief ischemia to induce neuroprotection showed that the genomic signature of the neuroprotective state included metabolic suppression.
32
Excitotoxicity contributes to neuronal death during stroke and suppression of excitotoxicity has been a focus of therapeutic strategies. For example, synaptotagmin 1, which is a downregulated gene target of the upregulated miRNAs (Supplementary Table II), is involved in neurotransmitter signaling and
Importantly, we identified several miRNAs that were differentially expressed in the preconditioning-induced neuroprotectived state compared with stroke alone. The suppression of the 22 differential miRNAs correlated with upregulation of 16 predicted gene targets. Interestingly, many of these genes have known or potential neuroprotective roles in stroke, indicating that suppression of these miRNAs may initiate new gene expression that reduces ischemic injury (Figure 6). As discussed above, 8 of the 16 gene targets have known or potential neuroprotective functions. In particular, the functions of KDR, Slit2, and Nupr1 suggest they are highly promising neuroprotective gene targets (Table 1). KDR codes for VEGFR2, which modulates vascular endothelial growth factor's well-established neuroprotective activites.
34
It has been shown that vascular endothelial growth factor treatment is neuroprotective
These data provide a global view of the influence of miRNA expression on the genomic response to stroke. In addition, we have identified several miRNAs and genes that may influence stroke outcome. However, the relationships between the identified miRNAs and their gene targets require additional validation to define the influence of the miRNAs on gene expression and stroke outcome. MiRNAs have highly complex regulation including processes downstream of biogenesis that influence how miRNAs function and regulate gene expression. 39 Analysis of the individual roles of these miRNAs in regulation of the identified target genes and proteins and their influence on stroke outcome is the focus of future work in our laboratory.
The data presented here provide strong evidence that miRNAs may serve as critical upstream master regulators of the endogenous responses to ischemia that both induce injury and provide protection. However, in the absence of preconditioning, stroke injury is accompanied by the upregulation of protective miRNAs that appear to be unable to overcome the damaging miRNAs that increase inflammation. CpG preconditioning alters the miRNA response to stroke by suppressing a subset of miRNAs. This suppression of miRNA associates with the induction of several additional neuroprotective genes in the setting of ischemia, implicating these miRNAs in genomic reprogramming induced by CpG preconditioning. The suppression of these miRNAs in response to stroke provides new pathways that function to reduce ischemic injury and ultimately favor a neuroprotective outcome. Thus, these miRNAs may be key targets of future stroke therapies that have a wide window for intervention to promote cell survival in the setting of ischemia.
Footnotes
ACKNOWLEDGMENTS
This work was supported by funds from NINDS NS062381 and Collins Medical Trust. Portions of this work were undertaken at PNNL, which is operated by Battelle Memorial Institute under contract DE-AC05-76RLO-1830.
The authors declare no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
