Abstract
Cerebral parenchymal arterioles (PAs) have a critical role in assuring appropriate blood flow and perfusion pressure within the brain. They are unique in contrast to upstream pial arteries, as defined by their critical roles in neurovascular coupling, distinct sensitivities to chemical stimulants, and enhanced myogenic tone development. The objective of the present study was to reveal some of the unique mechanisms of myogenic tone regulation in the cerebral microcirculation. Here, we report that
INTRODUCTION
While the regulation of vascular tone in cerebral pial arteries has been intensely investigated, mechanistic studies of the intracerebral vessels branching from the surface arteries, i.e., the cerebral parenchymal arterioles (PAs), are uncommon, despite the fact that the PAs have unique functional roles and properties. Parenchymal arterioles contribute to as much as 40% of cerebrovascular resistance and have a key role in regulating local blood flow within the brain. 1 By maintaining appropriate local perfusion pressure, PAs serve to protect downstream capillaries from the potential damaging effects of high intravascular pressure. 1 When these small arterioles branch from pial arteries to perfuse the cerebral cortex, they enter a unique environment where surrounding astrocytes and neurons modulate vascular diameter and blood flow through a process called neurovascular coupling.2,3 In addition, compared with pial arteries, the distinct features of PAs involve different responses to vasoconstrictors,4,5 altered ion channel function, 6 and enhanced myogenic responsiveness. 2 It is therefore likely that some of the fundamental contractile mechanisms in PA smooth muscle are unique as well. Since PAs are major sites of cerebral small vessel diseases, 7 a better understanding of vascular tone regulation in PAs may reveal potential therapeutic targets for treatment of cerebral microvascular diseases.
As one of the essential processes that modulate vascular tone, myogenic reactivity represents the ability of small arteries and arterioles to constrict and reduce their diameter in response to increased intravascular pressure. Myogenic regulation ensures that blood flow remains relatively constant despite moment-to-moment fluctuations in arterial pressure. In the brain, myogenic tone is an essential contributor to cerebral autoregulation. Mechanisms of myogenic tone development have been extensively studied and it is well established that vascular smooth muscle cells respond to increased intraluminal pressure via membrane depolarization, 8 calcium entry through voltage-dependent calcium channels and subsequent vasoconstriction. 9 However, the signaling pathways responsible for pressure-induced membrane depolarization have not been fully elucidated. Prior research in our laboratory showed a critical role of transient receptor potential (TRP) channels, specifically TRPM4, in this mechanism involving monovalent cation influx (mainly Na+) and membrane depolarization in the smooth muscle cells of cerebral pial arteries. 10 Further studies of this channel revealed that it is regulated by protein kinase C (PKC) and Ca2+ release from the sarcoplasmic reticulum.11,12 However, neither the significance nor the regulation of TRPM4 channels has been investigated in the PAs.
Another recent finding from our laboratory indicates the essential contribution of G protein-coupled receptors (GPCRs), specifically P2Y4 and P2Y6 purinergic receptors, to myogenic responsiveness of PAs via mechanosensitive receptor activation, which triggers vascular smooth muscle contraction, 5 similar to a proposed role for angiotensin II receptors in pial arteries. 13 Following from this novel finding, a major goal of the current study was to explore the possible interactions between TRPM4 channels and P2Y receptors. We hypothesized that P2Y4 and P2Y6 receptors regulate TRPM4 channel activity to mediate pressure-induced depolarization and myogenic vasoconstriction in cerebral PAs. The results of the present study support this hypothesis by showing that downregulation of TRPM4 channel expression or pharmacologically blocking TRPM4 channels substantially attenuates pressure- and P2Y receptor ligand-induced vascular and cellular responses, indicating a key role of TRPM4 channels in the P2Y receptor-mediated myogenic regulation of cerebral PAs.
MATERIALS AND METHODS
Animals
All animal procedures were approved by the Institutional Animal Care and Use Committee at the University of Vermont and performed in accordance with the National Institutes of Health Policy on the care and use of laboratory animals. Male, Sprague-Dawley rats (15 to 20 weeks old; Charles River Laboratories, Saint Constant, QC, Canada) were used for all experiments.
Suppression of TRPM4 Channels In Vivo
Antisense and sense oligodeoxynucleotides (ODNs) were designed based on the published rat TRPM4 sequence (RGD: 620244). Antisense ODN sequences were TRPM4 AS-1, 5′-GTGTGCACCGCGGTACCCCG-3′; TRPM4 AS-2, 5′-GCCCTCATGAATCCCAGTAA-3′. Control animals were treated with sense ODNs: TRPM4 S-1, 5′-CGGGGTACCGCGGTGCACAC-3′; TRPM4 S-2, 5′-TTACTGGGATTCATGAGGGC-3′. The last four bases on the 5′ and 3′ ends were phosphorothioated to limit ODN degradation. All ODNs were synthesized by Integrated DNA Technologies Inc. (San Diego, CA, USA). Oligodeoxynucleotides were dissolved in bicarbonate-free artificial cerebral spinal fluid (aCSF) solution (in mmol/L): 130 NaCl, 3 KCl, 2 CaCl2 · 2H2O, 1 MgCl2, 3 Na2HPO4-12H2O, 0.45 NaH2PO4H2O. Rats were initially anesthetized by isoflurane (5%) in an induction chamber and then maintained on isoflurane (2% to 3%) inhaled via a nose cone. A small (0.5 to 1.5 cm) longitudinal, midline incision was centered over the foramen magnum, and the neck muscle was dissected until the dura was visualized. Oligodeoxynucleotides (100
Tissue Preparation
Rats were euthanized by an overdose of sodium pentobarbital (120 mg/kg, intraperitoneal injection) followed by exsanguination. The brain was rapidly removed and placed in cold (4°C) 3-(N-morpholino)propanesulfonic acid-buffered physiological salt solution (in mmol/L): 3 3-(N-morpholino)-propanesulfonic acid, 145 NaCl, 5 KCl, 1 MgSO4, 2.5 CaCl2, 1 KH2PO4, 2 sodium pyruvate, 5 glucose, 1% albumin from bovine serum, pH 7.4. Middle cerebral arteries and attached PAs were isolated from the brain as previously described 14 and transferred to a small volume of 3-(N-morpholino)propanesulfonic acid-buffered physiological salt solution for further experimental use.
RT-PCR for TRPM4 Channel Message
Total RNA was prepared from isolated cerebral pial and parenchymal arteries using RNA STAT-60 total RNA isolation reagent (Tel-test Inc., Friendswood, TX, USA). First-strand cDNA for pial and parenchymal arteries was prepared from 1,320 ng total RNA and 620 ng total RNA, respectively, using the Qiagen (Valencia, CA, USA) Sensiscript Reverse Transcriptase kit. Semiquantitative PCR was performed using HotStarTaq DNA Polymerase (Qiagen). Amplification of the same templates for glyceraldehyde-3-phosphate dehydrogenase (GAPDH) served as an internal control, and TRPC6 channel transcript expression served to indicate the specificity of TRPM4 antisense ODNs. PCRs were hot started at 94°C for 15 minutes and exposed to 38 cycles of 94°C for 60 seconds, 60°C for 60 seconds, and 72°C for 60 seconds, followed by final extension at 72°C for 10 minutes. All reaction products were resolved on 1.8% agarose gels. Primer sequences are listed in Table 1.
Primer information for PCR studies examining TRPM4 channel transcript expression

GAPDH, glyceraldehyde-3-phosphate dehydrogenase; TRP, transient receptor potential.
Pressure Myography
Parenchymal arteriolar segments were cannulated on glass pipettes in an arteriograph chamber (Living Systems, Burlington, VT, USA) containing a bicarbonate-buffered (aCSF of the following composition (in mmol/L): 136 NaCl, 3 KCl, 15 NaHCO3, 1.25 NaH2PO4, 1 MgCl2, 4 glucose, 2 CaCl2 (temperature 37°C and pH 7.3). For most experiments, to eliminate endothelial responses to P2Y receptor agonists, arterioles were stripped of endothelium by passing air bubbles through the arteriolar lumen. Arterioles were pressurized at 5 mm Hg (with no flow), and superfused with warmed (37°C), gassed (20% O2/5% CO2/balance N2) aCSF. As an initial check of tissue viability, arterioles were reversibly constricted with the thromboxane receptor activator U46619 (100 nmol/L) (Enzo Life Sciences Inc., Farmingdale, NY, USA). Vessels were rejected for study if constrictions to U46619 were less than a 60% decrease in diameter. In experiments where myogenic constriction was not desired, e.g., agonist-induced vasoconstriction, intraluminal pressure was controlled at 3 to 5 mm Hg. In experiments where steady myogenic tone was desired, PAs were pressurized to 40 mm Hg. After steady myogenic tone developed, PAs were exposed to IKCa and SKCa channel activator NS 309 (1
Membrane Potential
Arterioles were cannulated as described above and pressurized to 5 mm Hg. The endothelium was removed from all vessels to eliminate any contribution of endothelial components to membrane potential. Nimodipine (300 nmol/L) (Calbiochem, San Diego, CA, USA) was included in aCSF before membrane potential measurements to prevent excessive vessel movement. Smooth muscle membrane potential was measured by repeated insertion of a sharp glass microelectrode (~200 MΩ resistance when filled with 0.5 mol/L KCl) into the vessel wall. The criteria for successful impalement were (1) an abrupt negative potential deflection upon entry, (2) a stable membrane potential for at least 30 seconds, and (3) an abrupt positive potential deflection to 0 mV upon electrode withdrawal. Measurements were made with an electrometer (World Precision Instruments, Sarasota, FL, USA) and recorded via computer with the AxoScope (Molecular Devices, Sunnyvale, CA, USA) and Dataq software (Dataq Instruments, Akron, OH, USA). At least three repeated impalements were made and the average membrane potential was calculated under each pharmacological treatment for every PA.
Single Cell Isolation
Arterioles were placed in a cell isolation solution containing (in mmol/L): 55 NaCl, 80 Na Glutamate, 5.6 KCl, 2 MgCl2, 10 HEPES, 10 Glucose (pH 7.3). Arteriolar segments were initially incubated in 0.5 mg/ml papain (Worthington Biochemical Corp., Lakewood, NJ, USA) and 1.0 mg/ml dithioerythritol for 12 minutes at 37°C and then in 1.0 mg/ml collagenase type F for 10 minutes at 37°C. The digested segments were washed three times in ice-cold cell isolation solution and incubated on ice for 30 minutes. Digested arterioles were triturated to liberate smooth muscle cells and stored in ice-cold cell isolation solution for use. Smooth muscle cells were studied within 5 hours after isolation.
Patch Clamp Recordings
Isolated smooth muscle cells were placed into a recording chamber and allowed to adhere to glass coverslips for 20 minutes at room temperature. Whole-cell currents were recorded with an AxoPatch 200B amplifier (Axon Instruments, Molecular Devices, Sunnyvale, CA, USA) equipped with an Axon CV 2032wBU headstage (Molecular Devices). Recording electrodes (3 to 5 MΩ) were pulled from borosilicate glass (Sutter Instrument, Navato, CA, USA). Gigaohm seals were obtained in bath solution of the following composition (in mmol/L): 134 NaCl, 6 KCl, 1 MgCl2, 2 CaCl2, 10 HEPES and 10 glucose, pH 7.4. The pipette solution contained (in mmol/L): 85K-aspartate, 1 MgCl2, 30 KCl, 10 NaCl, 10 HEPES, 5
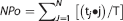
where
Chemical and Reagents
Buffer reagents, dissociation enzymes, nystatin, UDP and 9-phenanthrol were purchased from Sigma-Aldrich (St Louis, MO, USA). UTPyS was purchased from Tocris (Minneapolis, MN, USA). Nimodipine was dissolved in DMSO to a final solvent concentration of 0.1%. All other compounds were dissolved in the appropriate salt solution.
Statistical Analysis
Values are expressed as mean ± s.e.m., and
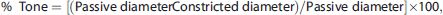
where ‘Passive diameter’ is the diameter in Ca2+-free aCSF.
For P2Y receptor agonist responses, constrictions were expressed as percent of the maximum contraction induced by the thromboxane receptor activator U46619 (100 nmol/L). Dilation to NS309 (1
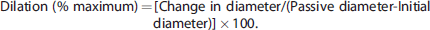
Student's
RESULTS
Inhibition of TRPM4 Channels Reduces Pressure-Induced Vasoconstriction of Parenchymal Arterioles
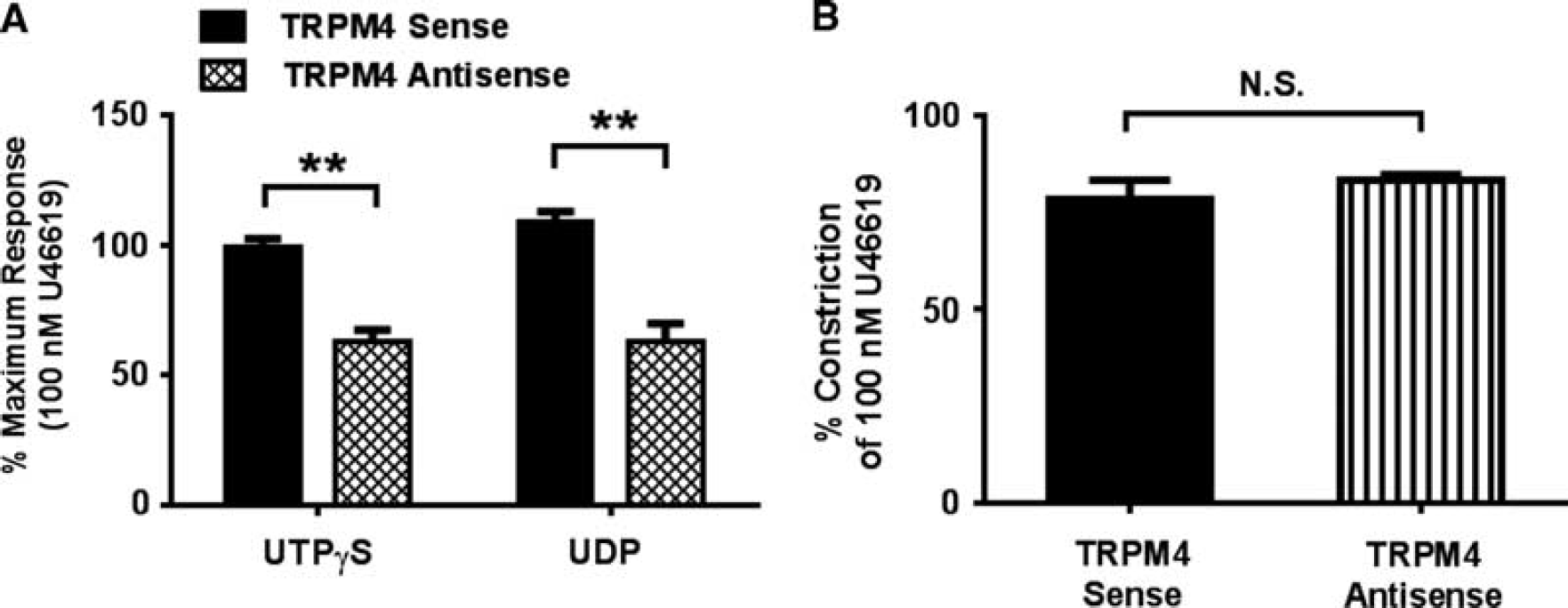
Previous studies have shown that TRPM4 channels are involved in pressure-induced (myogenic) membrane depolarization and constriction of pial arteries. However, the roles of these channels in PAs have not been investigated. Therefore, we first determined the presence of TRPM4 channel message in parenchymal arteriolar extracts. In agreement with previous studies examining the localization of TRPM4 channels in cerebral pial arteries, TRPM4 message was detected in PAs (Figure 1A). Next, to explore the functional significance of TRPM4 channels, we tested the effects of suppressing TRPM4 expression on myogenic responses of PAs. Exposure of PAs to TRPM4 antisense ODNs
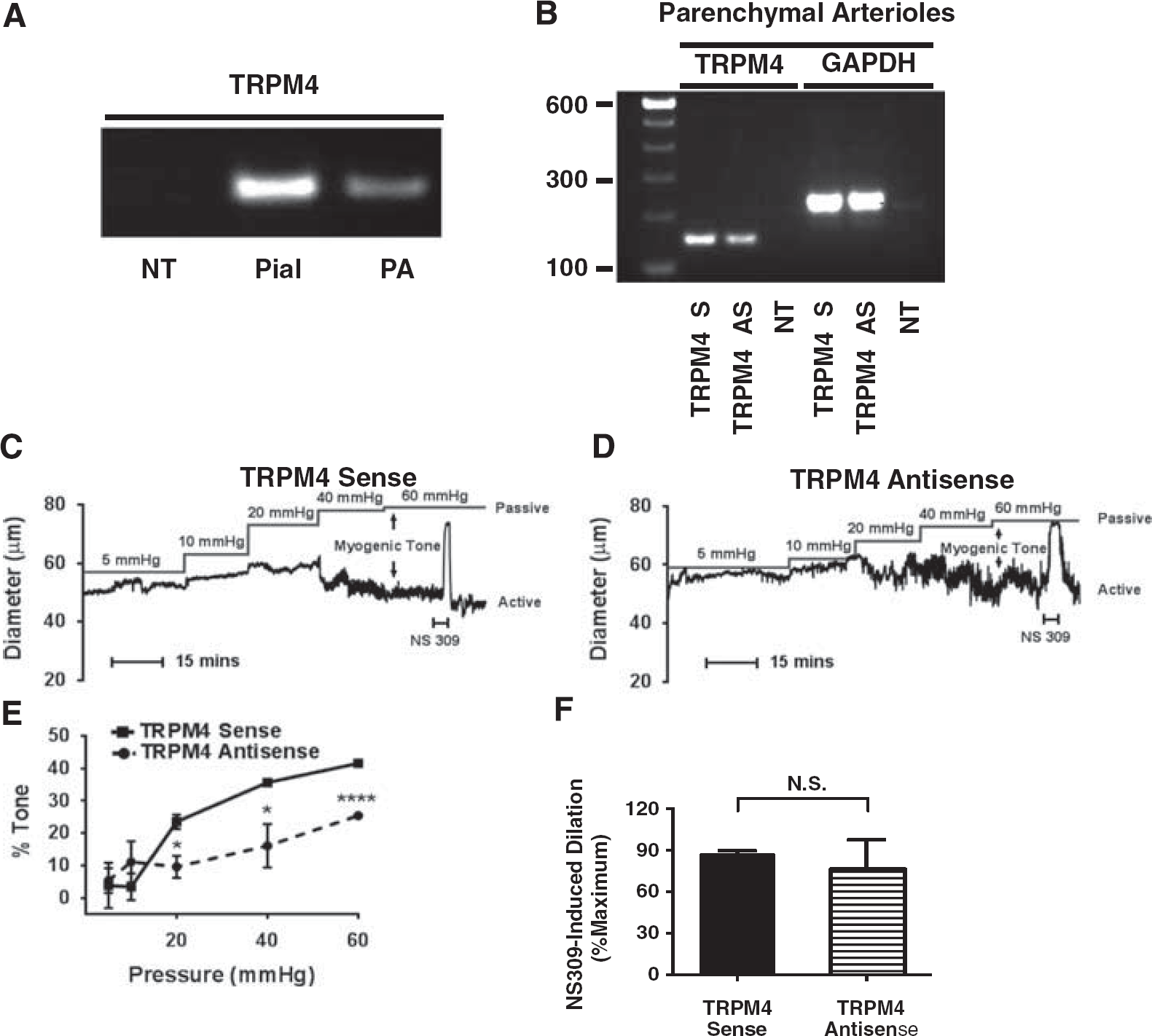
Effects of
Functional assessment of contractility revealed that PAs exposed
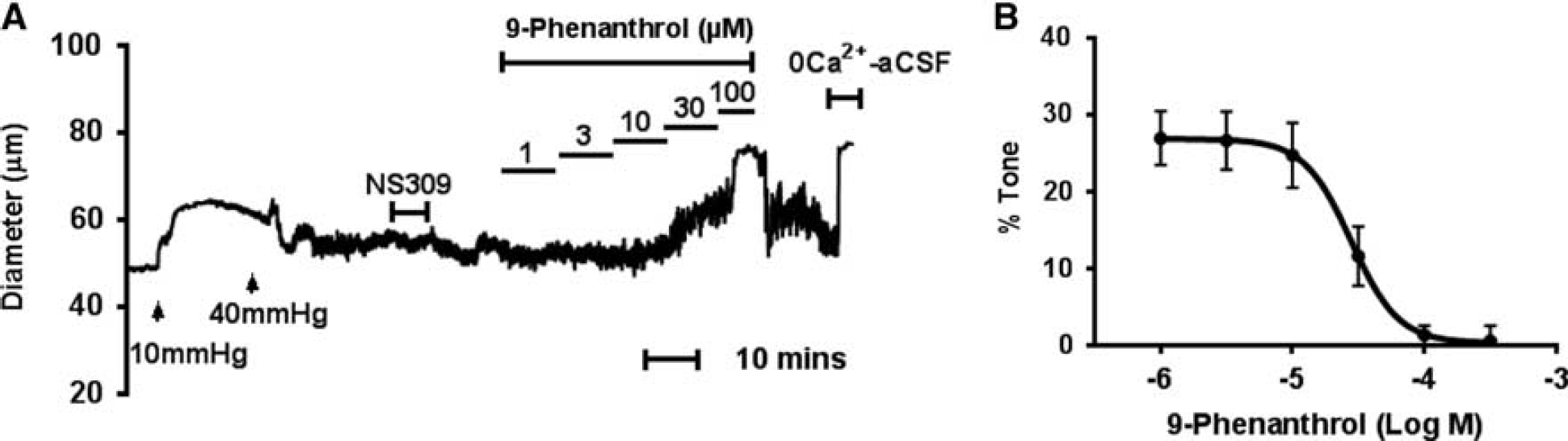
Effects of 9-phenanthrol on myogenic tone of endothelium-denuded parenchymal arterioles (PAs). (
Inhibition of TRPM4 Channels Suppresses P2Y Purinergic Receptor Ligand-Evoked Vasoconstriction and Membrane Depolarization of Parenchymal Arterioles
Interestingly, previous studies from our laboratory have shown the significant contributions of P2Y4 and P2Y6 purinergic receptors to coupling of membrane stretch and vasoconstriction in PAs, which is primarily mediated by calcium entry through voltage-gated calcium (Cav) channels. 5 Since TRPM4 channels are also implicated in the myogenic responses of PAs, presumably through depolarization and activation of Cav channels, it is logical to hypothesize that TRPM4 channels are involved in the mechanosensitive P2Y receptor-mediated myogenic vasoconstriction of PAs.
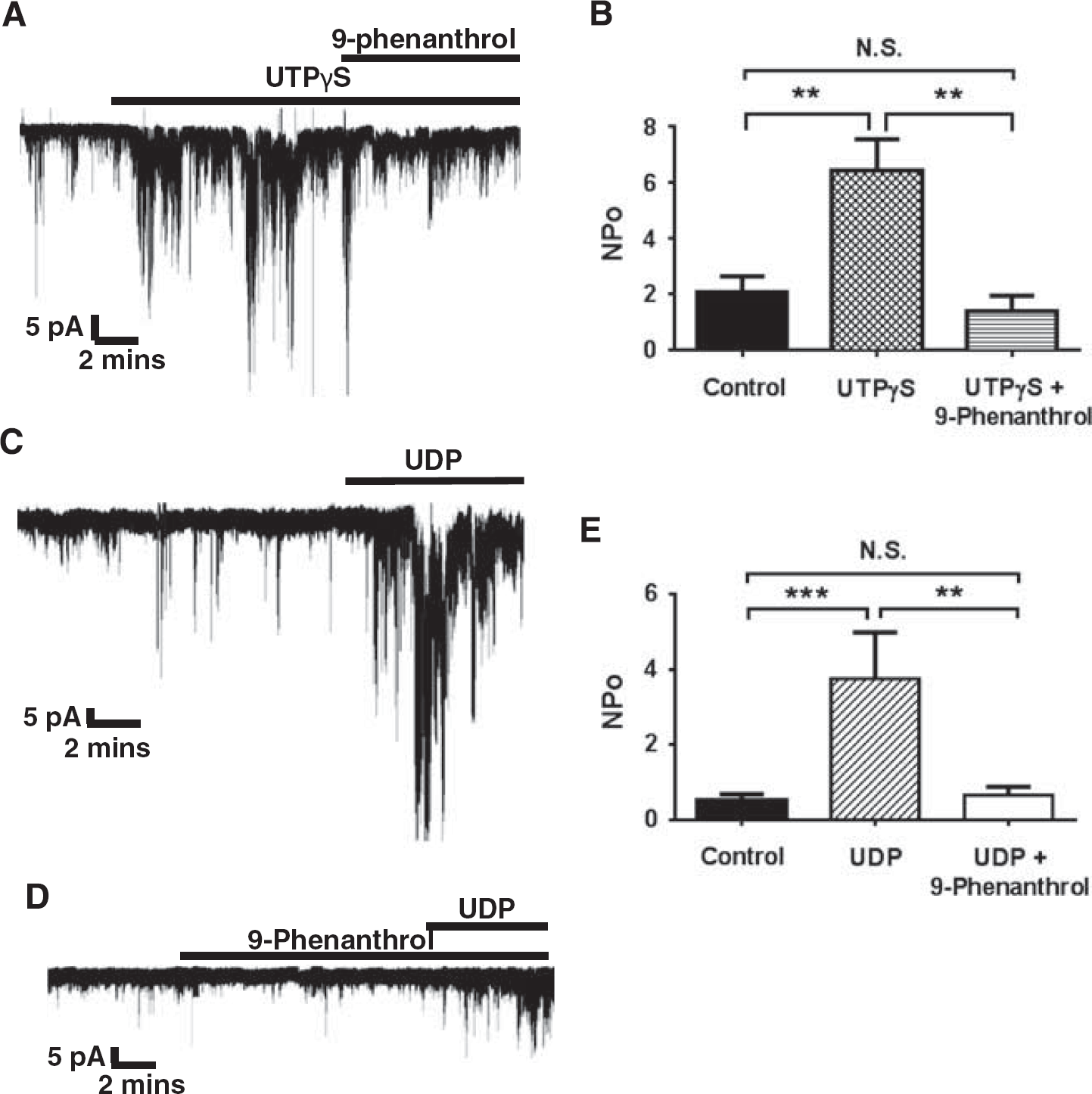
To establish a possible link between P2Y receptors and TRPM4 channels, we first examined the effects of suppressing TRPM4 expression on the contractile responses to P2Y4 and P2Y6 receptor ligands in endothelium-denuded PAs. We found that the selective P2Y4 receptor agonist UTP
Effects of
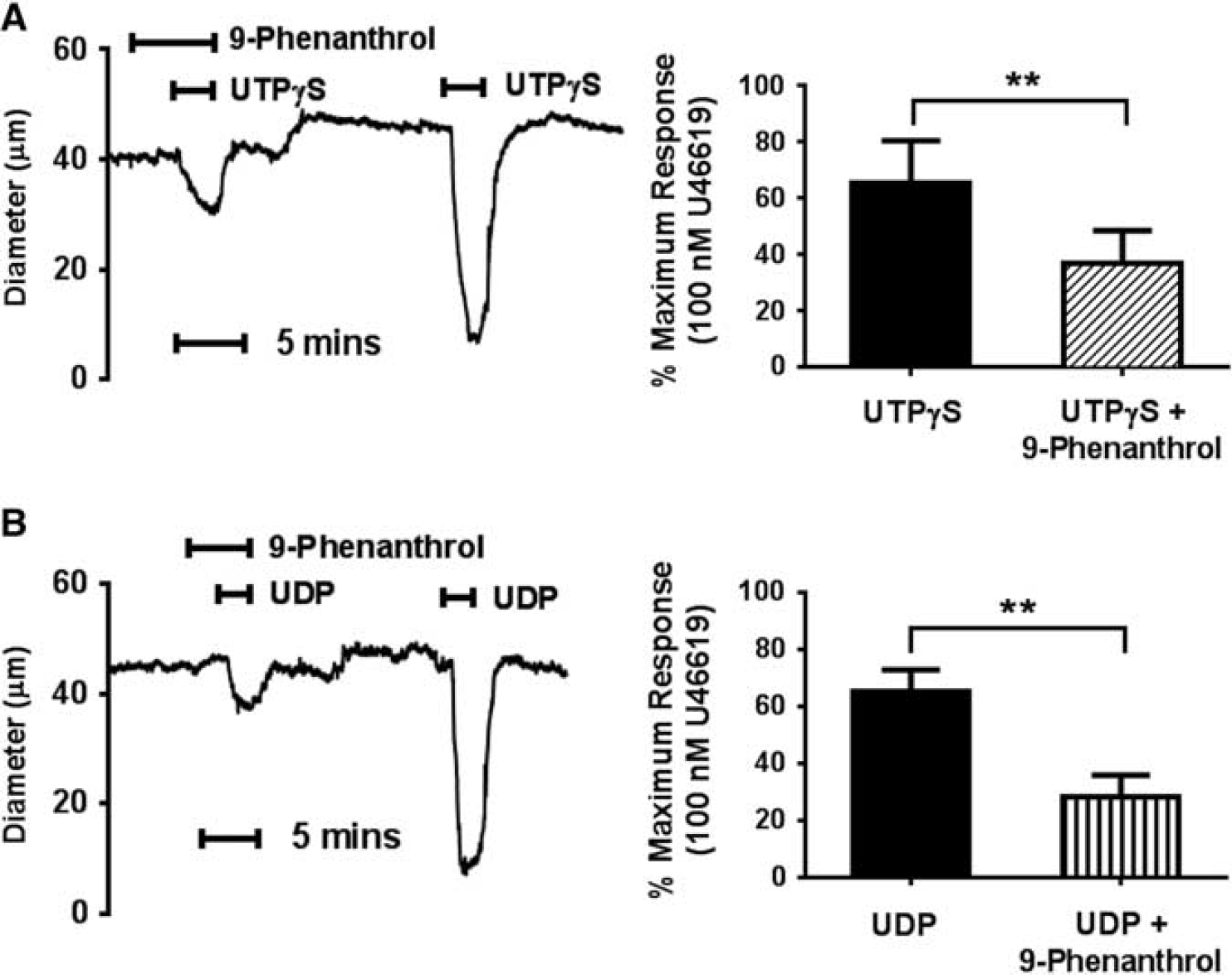
9-Phenanthrol (10
The proposed link between P2Y receptor activation, TRPM4 channels, and constriction of PAs implies that activation of P2Y receptors should induce a depolarization of PA myocytes that is inhibited by TRPM4 channel block. Thus, in the next series of experiments, we measured smooth muscle membrane potential using intracellular microelectrodes before and after the administration of P2Y receptor ligands, in the absence and presence of 9-phenanthrol. At 5 mm Hg, PA smooth muscle cells depolarized from −43 ± 1 mV to −25 ± 1 mV in response to UTP
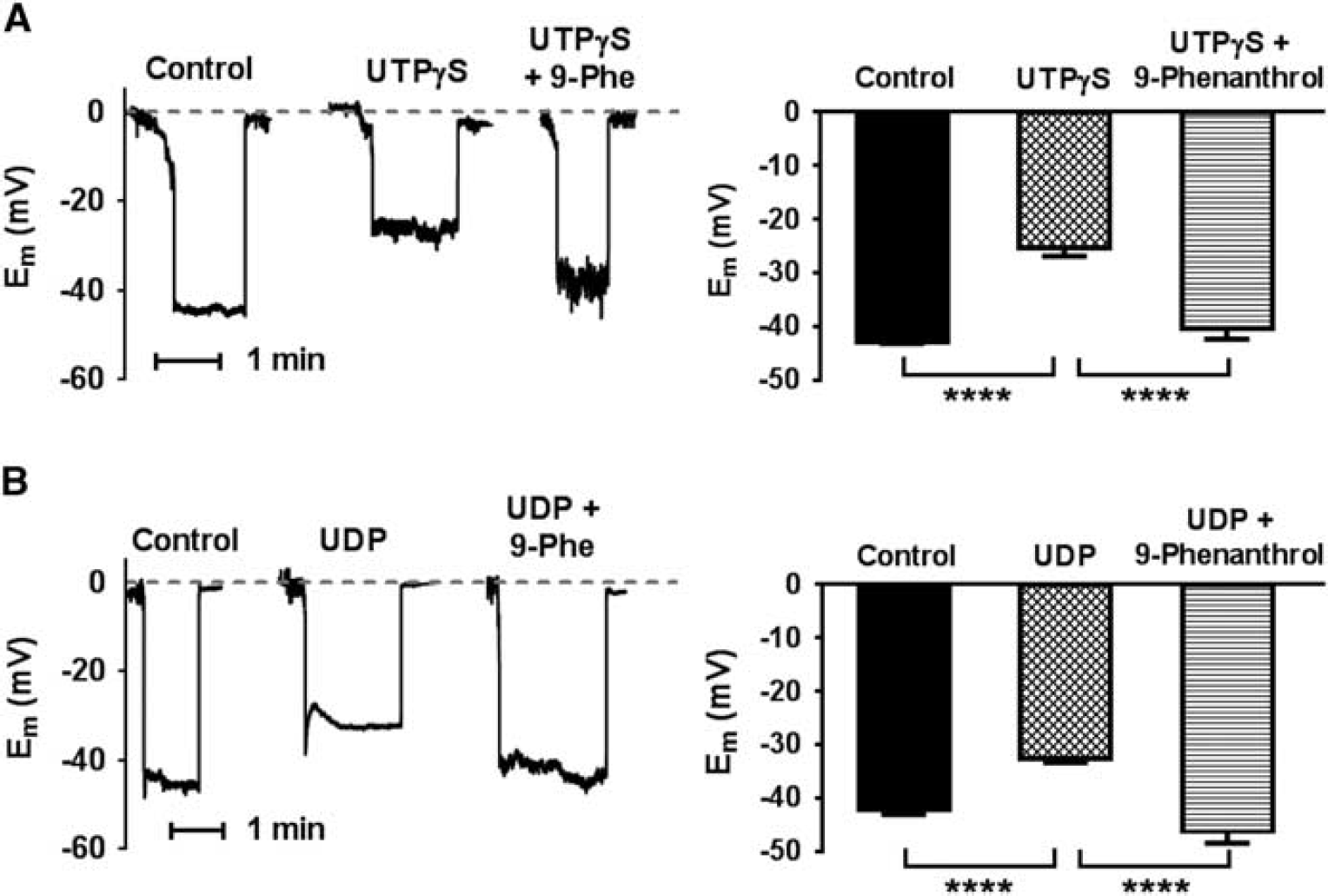
Effects of P2Y receptor ligands and 9-phenanthrol on parenchymal arteriolar smooth muscle membrane potential (Em). (
Activation of P2Y Receptors Potentiates TRPM4 Channel Activity in Parenchymal Arteriolar Smooth Muscle Cells
In a final series of experiments, direct coupling between P2Y receptors and TRPM4 channels was assessed using the nystatin-perforated whole-cell patch clamp technique. The P2Y4 receptor-specific ligand UTP
Effects of P2Y receptor ligands and 9-phenanthrol on TRPM4 channel activity in isolated parenchymal arteriolar smooth muscle cells. (
DISCUSSION
Here, we investigated the expression and functional roles of smooth muscle TRPM4 channels in cerebral intraparenchymal arterioles. The major findings of the present study are (1) TRPM4 channels are expressed in rat PAs; (2) administration of TRPM4 antisense ODNs
TRPM4 Channel Presence and Function in Parenchymal Arteriole
Myogenic tone development is a process by which arterial pressure regulates vascular diameter to maintain constant blood flow. The mechanisms underlying the myogenic response are not entirely clear. However, the primary roles of membrane potential and voltage-dependent Ca2+ (Cav) channels have been established. It is clear that increased intraluminal pressure is sensed by vascular smooth muscle cells, which subsequently depolarizes the cell membrane potential and increases the open probability of Cav channels, resulting in Ca2+ influx into smooth muscle cells and vasoconstriction. 9 Nevertheless, how a mechanical signal (i.e., membrane stretch) is converted into a cellular response (i.e., membrane depolarization) remains obscure. Previous studies from our laboratory focused on the roles of TRP channels and showed that both TRPM4 and TRPC6 channels are activated by increased intravascular pressure and mediate cation influx, which leads to myogenic membrane depolarization and constriction of cerebral pial arteries.10,15 Subsequently, other depolarizing contributors implicated in this mechanism have been reported in cerebral pial arteries, including TRPP2 channels, 16 epithelial Na+ channels (ENaC), 17 and TMEM-16A chloride channels, 18 which further emphasize the significance of membrane potential regulation in myogenic tone development.
Interestingly, compared with pial arteries, downstream PAs are considerably more sensitive to mechanical forces. Specifically, we found that PA smooth muscle is more depolarized and more constricted than pial arteries over the same range of intraluminal pressures.2,14 This could be due in part to reduced hyperpolarizing influence provided by large-conductance Ca2+-activated K+ (BKCa) channels in Pas.
6
However, a more depolarized membrane potential may also be attributed to enhanced depolarizing stimuli. The present study provides the first evidence that TRPM4 channels have this central role in myogenic tone development of PAs. In agreement with previous studies testing the presence of TRPM4 in cerebral pial arteries, transcripts of this channel were detected in PAs. Importantly, we found that knocking down TRPM4 channels through
As an alternative tool to confirm the functional significance of TRPM4 channels, the selective TRPM4 channel blocker 9-phenanthrol
20
elicited concentration-dependent inhibition of myogenic tone in endothelium-denuded arterioles, supporting an essential role of smooth muscle TRPM4 channels in the coupling of mechanical stimulation and vasoconstriction in PAs. The specificity of 9-phenanthrol has been tested by various groups. 9-Phenanthrol does not inhibit TRPM5 channels.
21
At a concentration of 30
P2Y4 and P2Y6 Receptors Are Coupled to TRPM4 Channels to Mediate Myogenic Constriction in Parenchymal Arterioles
Purinergic signaling has an important role in control of vascular contractility in a variety of blood vessels. 23 Although different vessels in various species express distinct purinergic regulatory mechanisms, it is clear that vascular tone is under the control of both the dilator effects of endothelial purinergic receptors and contractile responses of smooth muscle-associated purinergic signaling. In rat cerebral arteries, P2Y1 and P2Y2 receptors are located on endothelial cells and mediate endothelium-dependent vasodilation, 24 whereas P2Y2, P2Y4, and P2Y6 receptors are abundantly expressed in smooth muscle and mediate vasoconstriction in response to purine and pyrimidine stimulation.5,25
Several earlier studies have explored the possible contributions of pyrimidine receptors in myogenic regulation. For instance, P2Y6 receptors likely have an important role in mouse mesenteric arteries, 26 as the P2Y6 antagonist (MRS2567) inhibits myogenic vasoconstriction of these blood vessels. 27 Moreover, recent findings from our laboratory revealed the central involvement of P2Y4 and P2Y6 receptors in myogenic tone development of cerebral PAs. 5 Interestingly, rather than endogenous release of nucleotides from local sources as was reported for systemic arteries, 26 pressure-induced activation of these receptors in the PAs appeared to be caused by direct mechanoactivation, since neither potentiation nor suppression of ectonucleotidase activity has any effect on the myogenic behavior of PAs. 5 This observation is in fact consistent with a previous study on rat cerebral pial arteries that myogenic tone is mediated by mechanosensitive GPCRs. 13 In that study, an inverse agonist of angiotensin II (Ang II) receptor attenuated myogenic responses by 50%. Further, because pretreatment of isolated pial arteries with an angiotensin I converting enzyme inhibitor had no effect on myogenic tone development, it was proposed that pressure-evoked vasoconstriction is facilitated by direct mechanical activation of Ang II receptors. 13
The mechanisms of direct mechanoactivation of GPCRs (Ang II receptors, P2Y receptors, etc.) are not clear. Presently, a few possibilities have been proposed as to how GPCRs perceive mechanical force and initiate a conformational alteration to an active state. First, a ‘tethered’ model has been proposed suggesting that intracellular and extracellular anchorage to other proteins acts as a molecular spring to activate GPCRs upon membrane stretch. 28 Second, a so-called ‘membrane’ model proposes that the altered lateral pressure profile at the protein/phospholipid bilayer boundary can directly force a conformational change of the GPCRs. 28 Additional mechanisms including the interactions between receptors and cytoskeletal elements to facilitate stimulation of GPCRs have also been presented. 29 Clearly, identification of the specific mechanisms involved in GPCR regulation of myogenic tone will require deeper and more detailed biochemical and biophysical investigations.
A major finding of the present study is that TRPM4 channels are regulated by P2Y4 and P2Y6 receptors, providing the first direct evidence that TRPM4 channels are associated with mechanosensitive GPCRs to mediate pressure-evoked responses of the cerebral PAs. Knocking down TRPM4 channels or blocking them with 9-phenanthrol significantly inhibited the constrictor responses to both UTP
Regulation of TRPM4 Channels upon Myogenic Stimulation
Previous studies in cerebral arteries have shown that TRPM4 channel activity is regulated by PKC, whereby PKC increases the sensitivity of TRPM4 to intracellular Ca2+. 11 Moreover, Ca2+ release from the sarcoplasmic reticulum through inositol trisphosphate (IP3) receptors is required for sustained TRPM4 activity. 12 Both findings suggest that TRPM4 channels may be physically associated with GPCR signaling partners in smooth muscle cells. Furthermore, there is abundant evidence in support of a role of GPCR downstream signaling mechanisms in myogenic tone development. For instance, phospholipase C has a key role in the genesis of myogenic responses via membrane depolarization and increased calcium influx. 31 Contribution of diacylglycerol in pressure-evoked vasoconstriction has also been reported in cerebral arteries. 32 Additionally, PKC exerts substantial influence on various ion channels in smooth muscle of different vascular beds, including Cav, 33 Kv, 34 and TRPM4 11 channels, which are clearly involved in myogenic regulation. Moreover, IP3 receptors and intracellular levels of IP3 have been shown to correlate with mechanical stimulation of arteries or arterioles. 35 Since P2Y4 and P2Y6 are associated with Gq/11 protein signaling, phospholipase C-diacylglycerol-PKC and IP3 are logical candidate pathways that couple mechanostimulation of purinergic receptors to activation of TRPM4 channels in PA smooth muscle.
In addition to Gq/11 protein signaling, P2Y receptors are also coupled to the Rho-associated protein kinase (ROCK) pathway. 36 Interestingly, correlation between ROCK signaling and myogenic responses has been investigated in various vascular beds, including rat cerebral arteries. 37 Although Ca2+ sensitization through inhibiting myosin light chain phosphatase is generally accepted as a major mechanism of ROCK-associated myogenic vasoconstriction, 37 it has also been shown that ROCK can regulate the activity of various ion channels, including Cav and Kv channels38,39 and can modulate cerebral artery membrane potential. 39 These findings provide further evidence that ROCK signaling may contribute to the depolarizing mechanisms in smooth muscle, suggesting a possible role of ROCK in myogenic activation of TRPM4 channels. Understanding the relative contributions of these pathways may reveal therapeutic targets that can be manipulated in clinical settings.
CONCLUSION
The current results show that TRPM4 channels contribute substantially to P2Y4 and P2Y6 receptor-mediated myogenic tone development of cerebral intraparenchymal arterioles. A more thorough understanding of the vasomotor mechanisms within the brain may contribute to developing novel medical approaches and revealing new therapeutic targets that could help patients with cerebral microvascular diseases.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
