Abstract
Cerebral arteriovenous malformations (AVMs) are vascular anomalies that carry a high risk of stroke and death. To test potential AVM therapies, a reverse genetics approach was used to model AVMs in zebrafish. Antisense morpholino oligonucleotides were used to knockdown
INTRODUCTION
Arteriovenous malformations (AVMs) of the brain are vascular anomalies of children and adults that carry a high risk of stroke. 1 Their primary pathologic phenotypic characteristic is a direct communication between arteries and veins without an intervening capillary bed. Treatment of these lesions is limited to surgery, focused external beam radiation, and/or endovascular embolization; no specific FDA (Federal Drug Administration)-approved pharmacologic therapies currently exist. For unruptured cerebral AVMs, a recent randomized trial of these interventional procedures versus conservative management (A Randomized Trial of Unruptured Brain Arteriovenous Malformations, ‘ARUBA’— ClinicalTrials.gov identifier NCT00389181) was undertaken to determine the best management paradigm. In 2013, the National Institute of Neurological Disorders and Stroke halted randomization and enrollment within the ARUBA trial, underscoring the procedural risk associated with treatment of cerebral AVMs with currently available modalities and the need for new treatments.
The process by which AVMs develop is poorly understood, although aberrations in blood vessel formation and segregation during embryonic development and adult life are thought to be responsible. In development, the establishment of a vascular identity (be it arterial or venous) initiates from molecular signals that result in functional, and subsequently structural changes.2,3 There are several hierarchical signaling pathways that promote or inhibit divergent endothelial cell fates, including hedgehog, 4 vascular endothelial growth factor,5,6 notch,7,8 transforming growth factor (TGF)-beta, 9 and the ephrin ligand-receptor pathway, 10 among others. These pathways have a complex interaction and are crucial regulators of vascular assembly, differentiation, and boundary formation.
Despite a robust understanding of vasculogenesis and angiogenesis, the mechanisms behind the formation of discrete AVMs are not well known. These lesions occur either sporadically or much more rarely, in the context of a hereditary syndrome. One such syndrome in humans, hereditary hemorrhagic telangiectasia type-2, is caused by a mutation in the gene encoding activin receptor like kinase 1 (
MATERIALS AND METHODS
The investigation was approved by the Massachusetts General Hospital Institutional Review Board and conforms to the ‘Guide for the Care and Use of Laboratory Animals' published by the US National Institutes of Health (NIH Publication No. 85-23, revised 1996).
Morpholino Injection
A custom,
Zebrafish Maintenance and Drug Exposure
Zebrafish were raised in accordance with standard husbandry protocols. (The age of embryos is indicated by the hours after fertilization and days after fertilization for all experimental data shown.) Morpholino-injected or -uninjected embryos were placed into a 96-well microtiter plate with buffered embryo water (E3). The total volume of the embryos and the E3 was 100
Morphometric Analysis
Embryos at 4 d.p.f. were anesthetized by adding Tricaine (0.02% w/v) to E3 solution. They were then transferred to a petri dish, oriented in a sagittal profile, and examined under a Zeiss Examiner A1 stereomicroscope (Carl Zeiss Microscopy, Thornwood, NY, USA) Images were captured and then analyzed using ImageJ version 1.46 (NIH, Bethesda, MD, USA). A straight line was generated from the center of the lens to the most posterior aspect of the yolk sac. A separate straight line was generated from the posterior aspect of the yolk sac to the tip of the tail. The angle between these lines was automatically calculated, yielding a continuous variable that represented the systemic manifestations of the AVM on whole organism morphology. It is suspected that paucity of flow to the body impairs normal developmental processes, particularly with respect to renal organogenesis, resulting in organism curvature. Sixteen organisms were measured for each group (uninjected, morphant-control, and morphant-treatment).
Fluorescent Histology
JB-4 plastic resin was selected to examine the cranial vasculature given its high level of tissue preservation, fluorescence preservation, and cellular resolution. In all, 4 d.p.f. embryos were embedded, serially sectioned, and visualized using a modification of methods previously described.
14
The embryos were fixed in a 4% paraformaldehyde solution at 4°C overnight. They were then dehydrated in EtOH and infiltrated overnight. Embryos were then transferred to molds where embedding solution was applied. Before solidification, organisms were oriented in the same direction, parallel to the floor. Resin blocks were allowed to harden in the dark at 4°C to preserve fluorescence. A glass microtome was used to section 8
Phosphorylated SMAD1 Enzyme-Linked Immunosorbent Assay
The InstantOne ELISA kit (eBioscience, San Diego, CA, USA) was used to measure levels of phosphorylated SMAD1 (Ser463/465) in whole embryo lysates. This kit is also predicted to detect the analogous phosphorylation sites of SMAD5 and SMAD8. Using a modified protocol, six organisms per well were lysed at 4 dpf with a combination of cell lysis buffer and mechanical homogenization. Lysate and phospho-SMAD1 (Ser463/465) capture and detection antibody reagents were added simultaneously to the InstantOne assay plate. After 1 hour of incubation, wells were washed and a detection solution was applied. Absorbance was measured at 450 nm. Positive control cell lysate and negative control (cell lysis buffer) confirmed antibody efficacy. Twenty-four organisms were measured for each group (uninjected, morphant-control, and morphant-treatment). Experiments were repeated in triplicate.
Statistical Methods
To compare continuous variables, a two-tailed Student's
RESULTS
Morpholino Knockdown of alk1 Results in Cerebral Arteriovenous Malformation Formation in Zebrafish
The genetic basis for AVMs is not well understood, making evaluation of targeted therapies challenging. However, mutations in
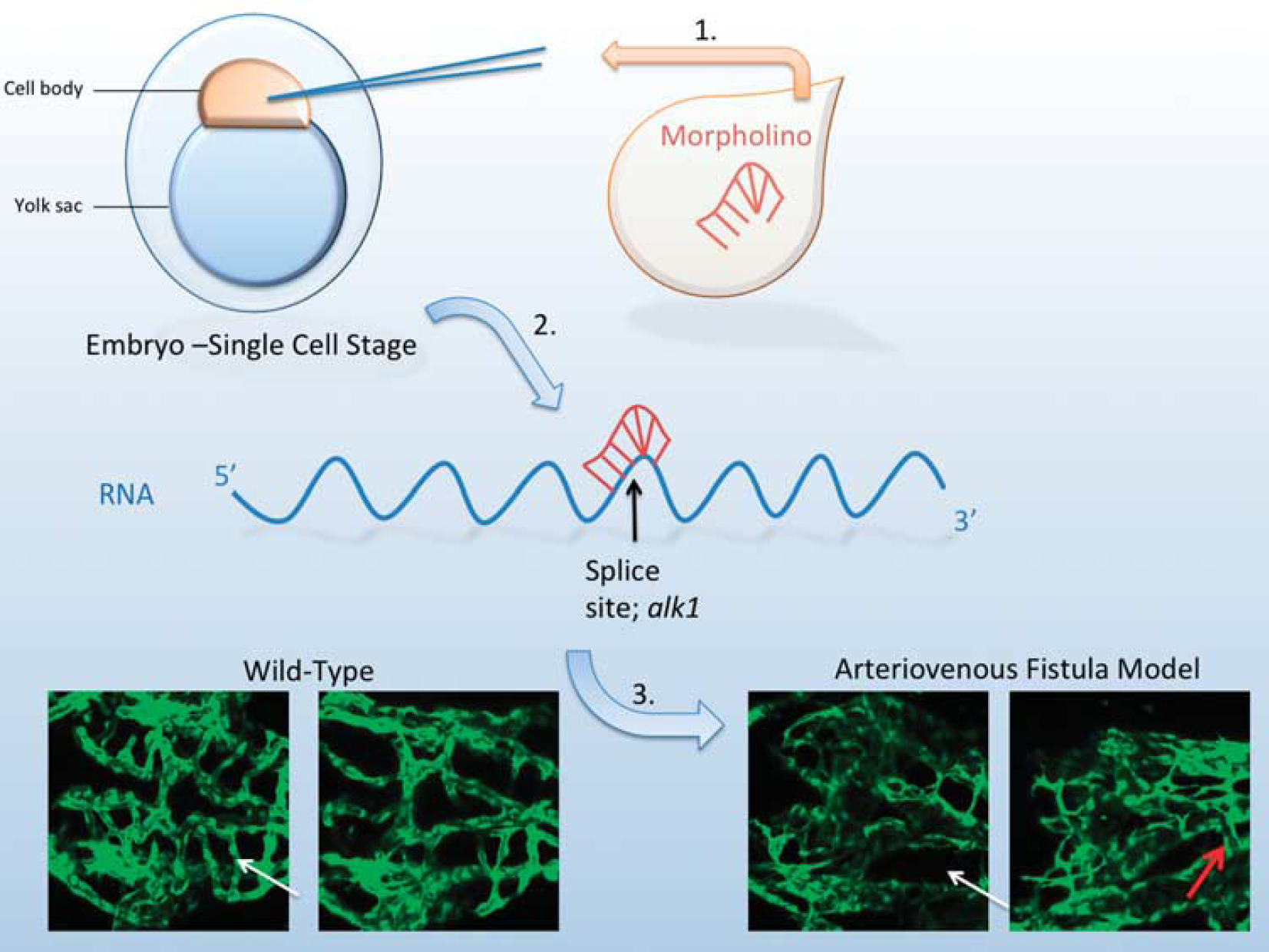
Experimental design and arteriovenous malformation (AVM) characteristics. (1) An
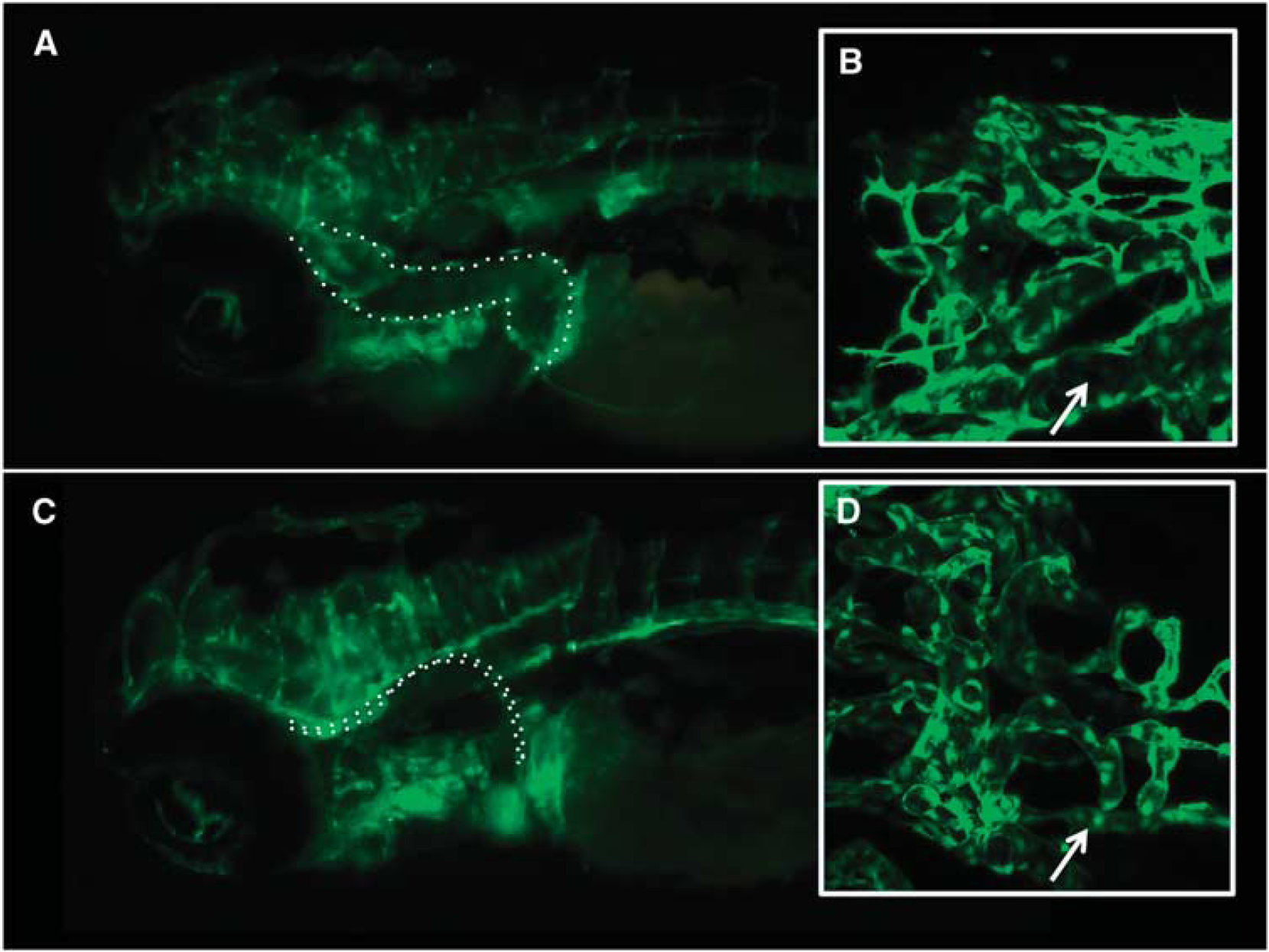
Epifluorescence and confocal microscopy of
Compound Selection
Losartan was selected for testing in the zebrafish AVM model given its demonstrated biologic activity seen in various aspects of TGF-beta superfamily pathways in humans. It is a widely used, orally administered FDA-approved medication for hypertension with an excellent safety profile. 15 Although its primary target is the angiotensin II type 1 receptor (AT1), it is also known to indirectly attenuate canonical TGF-beta signaling in the vasculature. 16 Additionally, its vascular modifying effects are also the current focus of a phase 3 clinical trial (ClinicalTrials.gov identifier: NCT00763893; Efficacy of Losartan on Aortic Dilatation in Patients with Marfan Syndrome).
Treatment with Losartan Attenuates Abnormal Vasculature in Zebrafish Cerebral Arteriovenous Malformation Morphants
In
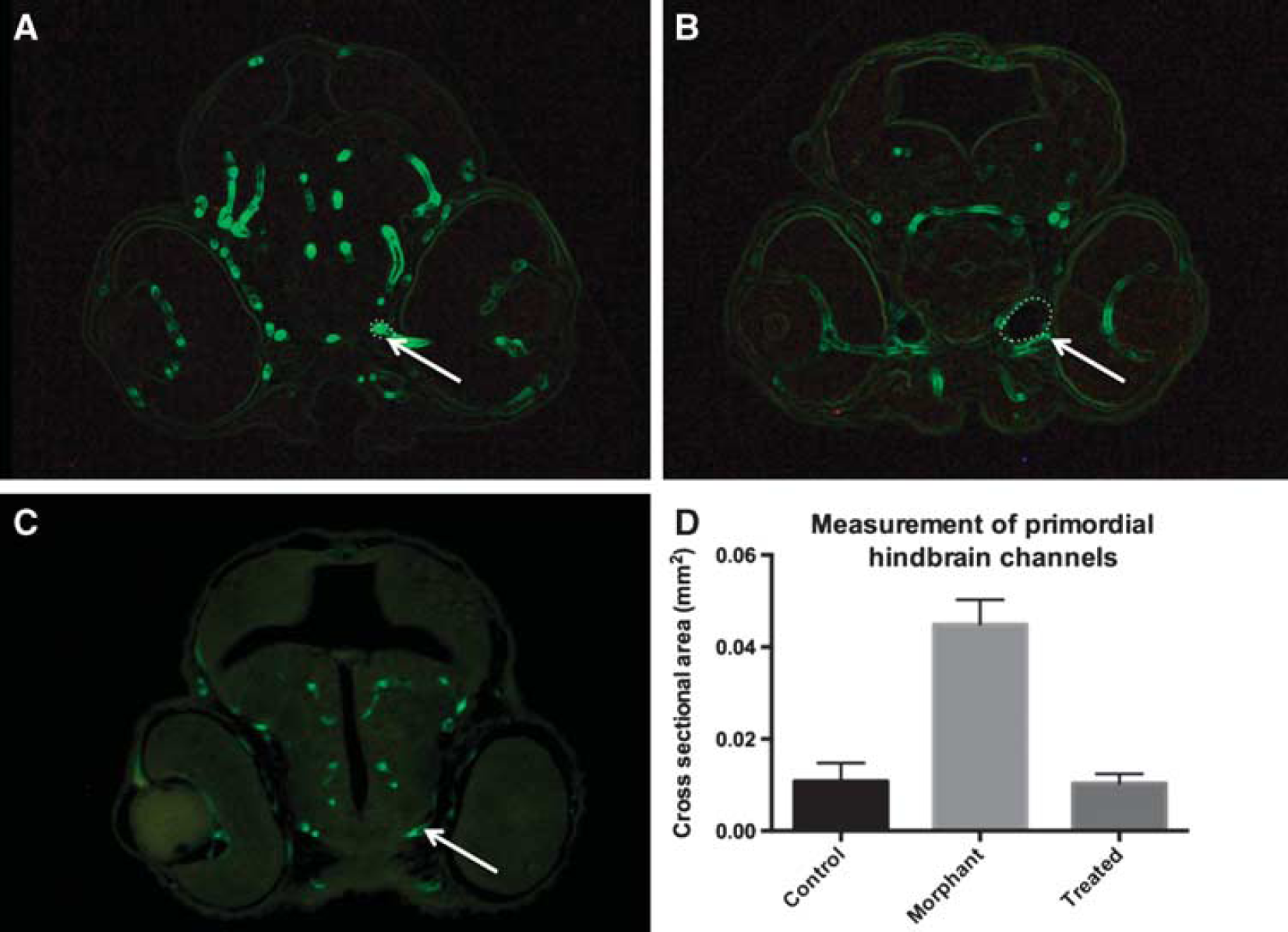
Histologic assessment of cerebral vasculature shows dilated cranial blood vessels in an arteriovenous malformation (AVM) model. 4-dpf zebrafish were embedded in JB-4 resin, serially sectioned, and visualized at the level of the mid-eye in the coronal plane. Measurements of the primordial hindbrain channels were significantly smaller in the (
Treatment with Losartan Attenuates the Systemic Organism-Level Phenotype in Zebrafish Cerebral Arteriovenous Malformation Morphants
In addition to vessel architecture, the hemodynamic properties of experimental organisms were measured, using whole organism morphometric characteristics as a surrogate. In uninjected organisms the cranial–caudal angle was measured to be 176.0 ± 0.6488 degrees, whereas it was significantly more acute in the morphant-control group 97.50 ± 4.987,
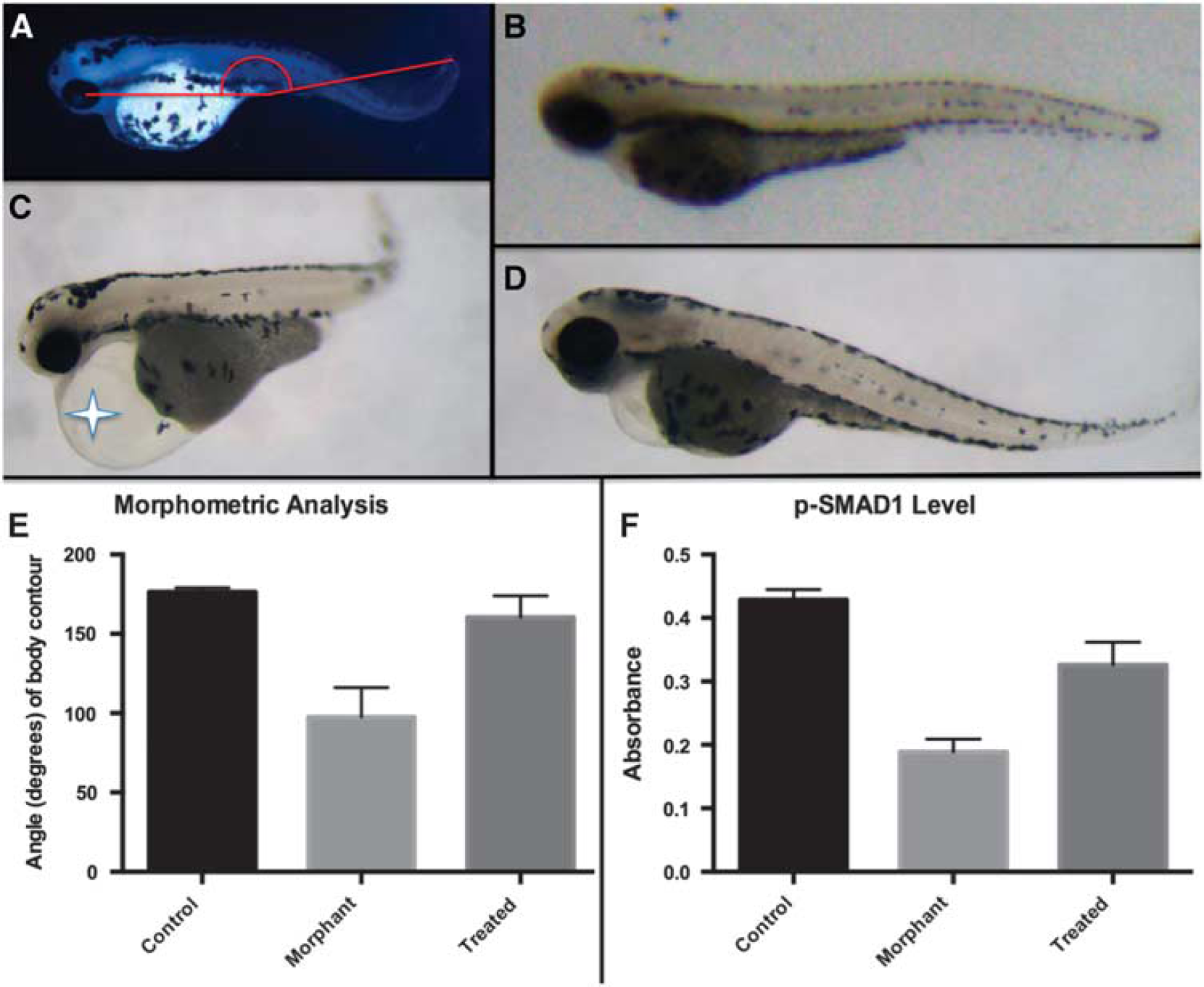
Morphometric and suppression of canonical transforming growth factor (TGF)-beta signaling changes in
Functionally, hemodynamics were directly observed between the three experimental groups using light microscopy. The qualitative absence (or near absence) of blood flow in the body and tail region of any organism was identified corresponding to the
Treatment with Losartan Partially Normalizes Phospho-SMAD1 Levels Caused by alk1 Knockdown
It was hypothesized that the ability of losartan to rescue the vascular and morphologic effects of
DISCUSSION
Currently, there are no FDA-approved pharmacologic treatments for cerebral AVMs in humans. One attractive approach for developing such therapies is repurposing of existing drugs, which could theoretically reduce the developmental and regulatory burden required for new drug approval. Therefore, an AVM model that allows rapid testing of drug candidates could be useful for identifying existing drugs with activity against AVMs.
Several mammalian models of cerebral AVMs exist, but are limited in their applicability for drug discovery. For example, cerebral AVMs have been created in swine, but this method requires a combination of surgical intervention and hemodynamic response to generate a lesion that mimics the human condition.
17
Additionally, mice have been used as model organisms, although they require angiogenic stimulation with vascular endothelial growth factor, in addition to genetic manipulation of
By using morpholino technology, we were able to manipulate the gene expression of
Targeted evaluation of losartan, an FDA-approved antihypertensive medication in widespread clinical use, resulted in a modest attenuation of the zebrafish AVM phenotype. This could be seen in a trend toward normalization of the abnormal cranial vasculature and hemodynamic pertubations seen with the experimental model. Furthermore, the loss of SMAD1 phosphorylation associated with
The effects of losartan on zebrafish AVMs are not the first example of its beneficial effects on a congenital vascular condition. Losartan has been shown to prevent aortic aneurysm development
24
and progression
25
in a mouse model of Marfan syndrome. On the basis of these and other data, several active clinical trials are now investigating the efficacy of losartan in treating aortic aneurysms in humans. The central hypothesis underlying these trials is that losartan antagonizes TGF-beta signaling,26,27 likely by inhibition of the AT1 receptor or other pathways independent of a TGF-beta receptor itself.
16
However, less is known about the influence of AT1 on the opposing BMP signaling pathway (the pathway directly influenced by mutations in
It was suspected that the molecular mechanism underlying the rescue mechanism of losartan treatment is related to restoration of BMP signaling. Conceptually, the TGF-beta and BMP pathways balance each other through opposing mechanisms to regulate gene expression (Figure 5).28,29
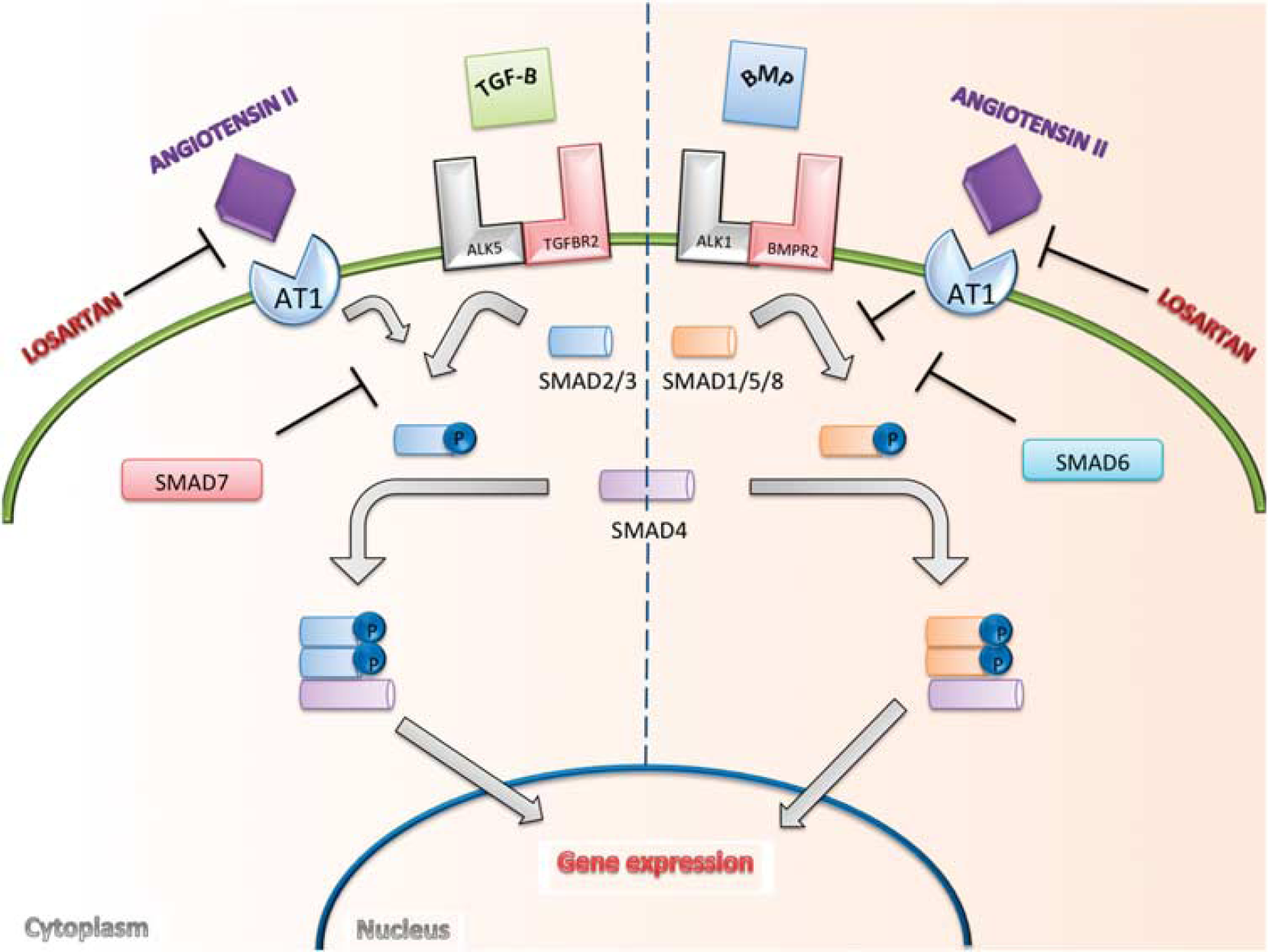
Transforming growth factor (TGF)-beta signaling pathway. Canonical signaling by TGF-beta is typically thought of in terms of two opposing, but interconnected, signaling pathways. Ligands in the TGF-beta or BMP family bind to specific Ser/Thr kinase type I (such as ALK1 and ALK5) and type II receptors (such as TGFBR2 and BMPR2) on the cell surface. Activation leads to phosphorylation of either SMAD2/3 or SMAD1/5/8, the intracellular effectors of the TGF family. These activated SMADs form complexes with SMAD4 that accumulate in the nucleus, where they go on to regulate the expression of target genes.31,32 We propose that losartan has an effect on the canonical signaling cascade, independent of type I and type II TGF-beta receptors. It has been previously shown that angiotensin II type 1 receptor (AT1) blockade antagonizes AT1 receptor stimulation of TGF-beta, decreasing activation of SMAD2. 16 The presented data suggest that activation of SMAD1/5/8 occurs after antagonism of the AT1 receptor with the use of losartan.
If losartan treatment is able to counteract loss of
Footnotes
The author declares no conflict of interest.
ACKNOWLEDGMENTS
The author thanks Dr Randall T Peterson, PhD for assistance in designing the experiments used in this study, for material support, and for thoughtful review of the submitted manuscript.
