Abstract
Perturbations in cerebral blood flow and abnormalities in blood vessel structure are the hallmarks of cerebrovascular disease. While there are many genetic and environmental factors that affect these entities through a heterogeneous group of disease processes, the ultimate final pathologic insult in humans is defined as a stroke, or damage to brain parenchyma. In the case of ischemic stroke, blood fails to reach its target destination whereas in hemorrhagic stroke, extravasation of blood occurs outside of the blood vessel lumen, resulting in direct damage to brain parenchyma. As these acute events can be neurologically devastating, if not fatal, development of novel therapeutics are urgently needed. The zebrafish (
ZEBRAFISH CHARACTERISTICS
Vasculature
Drug discovery involves a stepwise series of processes that typically begin with biochemical and cellular assays to screen for agents of potential value, which are later validated in animal models, and ultimately in human subjects.
1
The process is costly, resource intensive, and time consuming. The use of zebrafish (
Genetics
For zebrafish models of disease to be pertinent to the pathophysiology of humans, they must share genetic underpinnings, not just merely a common phenotype. Although the zebrafish appears as a relatively simple organism, comparison of the two genomes has demonstrated a high degree of conservation in genes implicated in processes ranging from oncogenesis to angiogenesis.12, 13, 14 There is also a high degree of conservation between humans and zebrafish with respect to drug responses, indicative of a high degree of amino-acid sequence identified at protein-active sites where many drugs bind. 15 Although the genetic sequence of zebrafish is highly conserved with humans, it is more readily manipulated for the purposes of experimentation. Several tools exist in the armamentarium of the zebrafish biologist that can be used to dissect pathophysiologic pathways. Broadly speaking, the use of ‘forward’ genetic screens can be used where chemicals with both known and unknown functions, along with other mutagens, can be administered to zebrafish and their phenotype can be characterized. 16 Conversely, ‘reverse’ genetic screens can be performed where the gene of interest is manipulated precisely with one of the several methods and then the phenotype is observed. Examples of this include morpholino oligonucleotide knockdown17, 18, transcription activator-like effector nucleases19, 20, zinc-finger nucleases21, 22, and the clustered, regularly interspaced, short palindromic repeats (CRISPR)—CRISPR-associated (Cas) systems 23 , each of which can induce targeted genetic modifications in zebrafish embryos.
Phenotype Screening
The size of the zebrafish facilitates a large experimental scale size that is not possible with other vertebrates. Adult zebrafish pairs can generate up to 300 embryos at each mating, allowing for experiments with thousands of organisms at a time. Because the zebrafish embryo is much smaller than 1 mm in diameter, experiments can be performed in 96 or 384-well plates. Most small molecules readily diffuse into zebrafish, requiring only a dilution of the drug into the fish water to effectively ‘treat’ organisms. Tens of thousands of known and unknown compounds are commercially available, including libraries specifically generated with FDA-approved compounds. 2 The zebrafish's microscopic size, high fecundity, and ease of drug administration, coupled with ease of phenotype recognition, make it well suited for high-throughput screening. 24
ZEBRAFISH STROKE MODELS
Cerebral Amyloid Angiopathy
Cerebral amyloid angiopathy is a cause of potentially fatal lobar intracranial hemorrhage, particularly in the elderly.
25
Histologically, the diagnosis is characterized by deposition of amyloid peptides around brain vessels, resulting in fibrinoid necrosis, microaneurysm development, and ultimately, vascular rupture.
26
Effective therapeutics do not currently exist that have been shown to alter the natural history of the disease and the exact pathogenesis of the condition remains elusive. One of the leading hypothesis underlying cerebral amyloid angiopathy-related hemorrhage is that beta amyloid deposition leads to endothelial dysfunction (early senescence).
27
Emerging work in a zebrafish model has been used to explore the pathogenic mechanisms of the disease.
28
In these experiments, beta amyloid peptide was administered via diffusion in fish water and a senescence phenotype was assessed by measuring beta-galactosidase activity and the cyclin-dependent kinase inhibitor p21 expression (
Cerebral Arteriovenous Malformation
Arteriovenous malformations (AVMs) of the brain are vascular anomalies of children and adults who carry a high risk of hemorrhage, about 2% to 4% per year over the patient's lifetime.
29
The primary pathological phenotypic character of AVMs is a direct communication between arteries and veins without an intervening capillary bed. Treatment of these lesions, either with surgery, endovascular embolization, or external beam radiation (stereotactic radiosurgery), carries significant risks, especially when lesions are large or located in eloquent brain areas. No specific medical therapies currently exist and their pathogenesis is incompletely understood. Despite a robust understanding of vasculogenesis and angiogenesis30, 31, 32, 33, 34, 35, 36, 37, 38, 39, the mechanisms behind the formation of discrete AVMs are not well known. It is thought that abnormalities in blood vessel formation and segregation during embryonic development are thought to be responsible, although
Arteriovenous malformations occur sporadically or much more rarely, in the context of a hereditary syndrome. One such syndrome in humans, hereditary hemorrhagic telangiectasia type 2 (OMIM phenotype ID 600376), is caused by a mutation in the gene encoding activin receptor-like kinase 1, a type 1 transforming growth factor beta receptor in the BMP signaling pathway.
43
This known mutation provides an opportunity to study the pathogenic mechanisms of AVMs in vertebrate models.44, 45 Another syndromic form of cerebral AVM development, such as CM-AVM (OMIM phenotype ID 608354), is caused by a mutation in
Several mammalian models of cerebral AVMs exist, but are limited in their applicability for drug discovery. For example, cerebral AVMs have been created in swine, but this method requires a surgical intervention to generate a lesion that mimics the human condition.
53
Other animal models, including mice, have been used, although they require angiogenic stimulation with vascular endothelial growth factor, in addition to genetic manipulation to generate lesions.54, 55, 56 In comparison, zebrafish are an attractive model in that their endothelium is visualized easily with fluorescent proteins and their entire cranial circulation can be observed
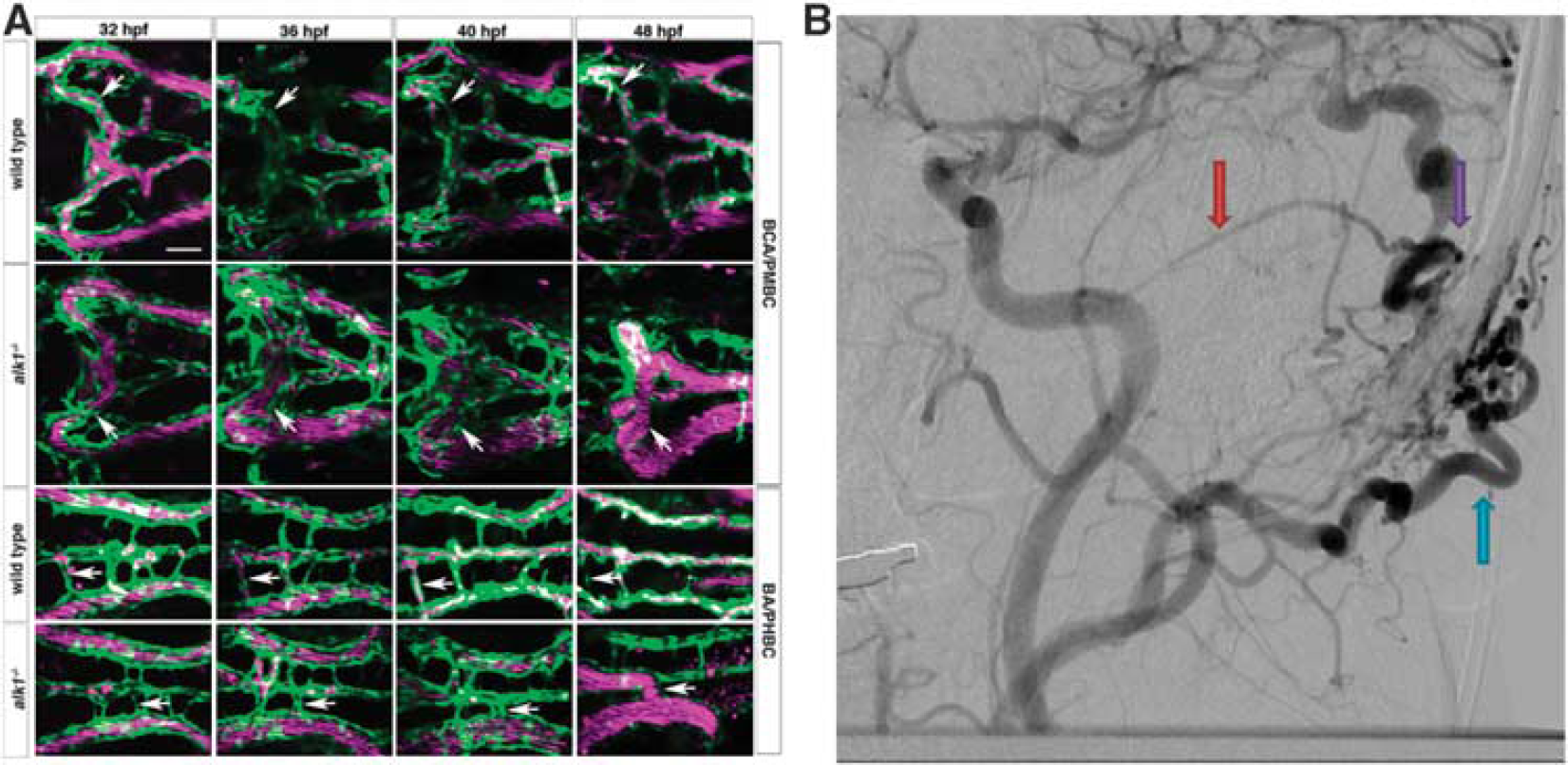
Phenotype comparison of zebrafish and human arteriovenous malformations (AVM). (
Cerebral Aneurysm
Aneurysms are lesions of the cerebral vasculature that have a typical phenotypic characteristic of an outpouching of a blood vessel wall as a result of an inherent weakness. As aneurysms enlarge, their propensity for catastrophic rupture increases.
60
Treatment of these lesions is optimally performed prior to an hemorrhagic event, either with microsurgical obliteration or endovascular techniques.
61
Their pathogenesis is generally thought to result from the interaction between genetic and epigenetic factors (such as cigarette smoking and hypertension).62, 63 Evidence for the genetic aspects of aneurysm pathogenesis is well established from powerful population based studies, such as the familial intracranial aneurysm study.
64
In addition, informative studies, known as genome-wide linkage studies, have been performed on rare families that are affected with aneurysms.65, 66 What is known from these studies is that there are many genetic susceptibilities implicated in the development of familial aneurysms.
67
Furthermore, cerebral aneurysms can occur in the setting of autosomal dominant polycystic kidney disease (OMIM phenotype ID 601313), a hereditary condition caused by a mutation in the
The understanding of aneurysm development is best examined experimentally where
Cerebral Cavernous Malformations
Cerebral cavernous malformations, also known as cavernous angiomas or cavernomas, are one of the few causes of stroke known to reliably be caused by one of at least three genetic mutations. 76 Histologically, they consist of enlarged capillary cavities (low flow, low pressure) without any intervening brain parenchyma. These lesions can result in seizure and/or hemorrhage, and therefore, the treatment is typically recommended for when they are symptomatic.77, 78 Currently, no therapeutics exist outside the realm surgery79, 80 or focused external beam radiation (stereotactic radiosurgery).81, 82, 83 Treatment becomes challenging, carries significant risk, or can even be considered impossible for lesions situated deep within critical brain structures. In addition, lesion multiplicity also complicates treatment suitability and is more commonly seen with familial cases. 84
While both Mendelian inheritance and sporadic cases exist, mutations in
The zebrafish exome shares a striking similarity with humans, and the orthologs of the three genes (
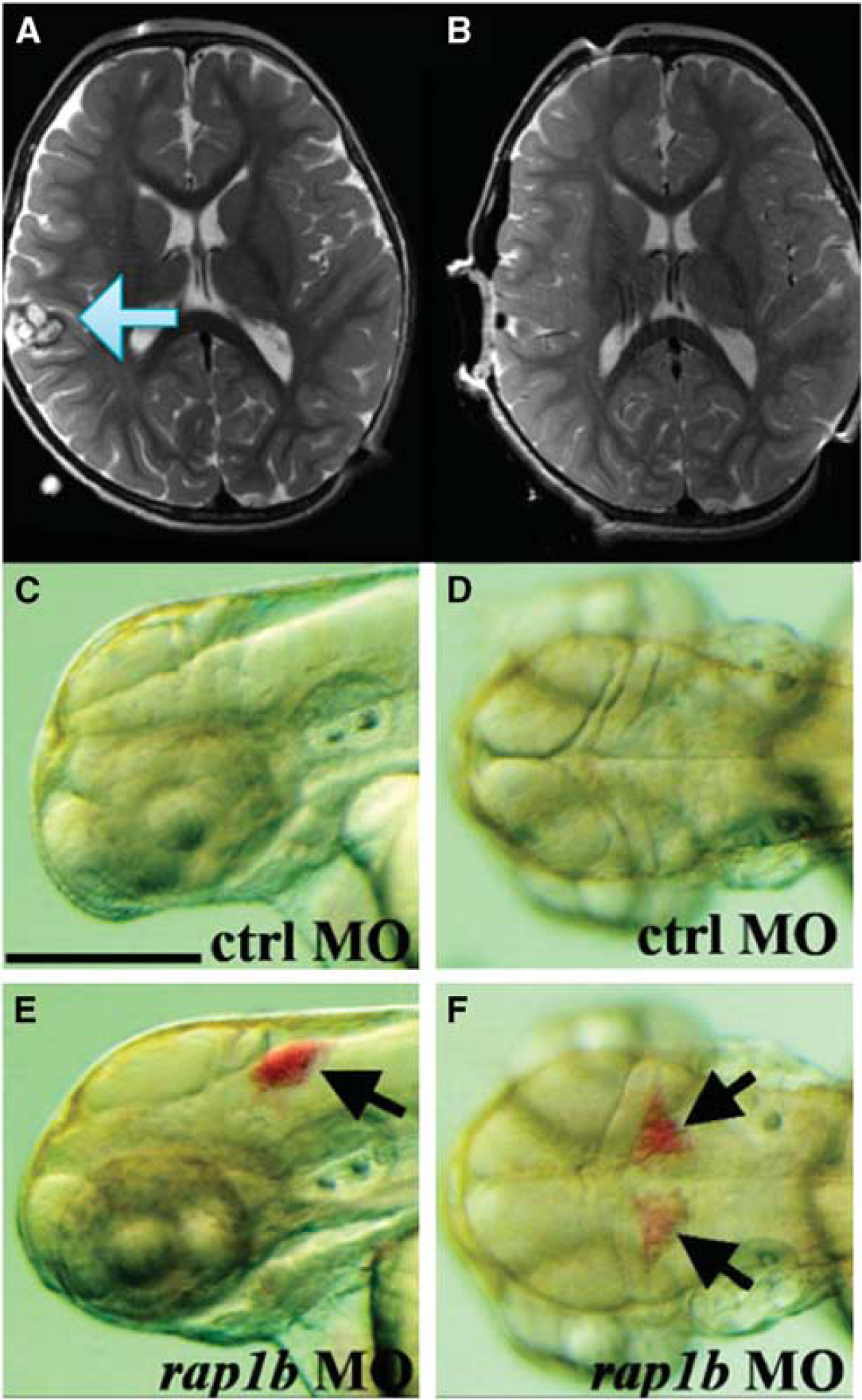
Phenotype comparison of zebrafish and cerebral cavernous malformation. In an magnetic resonance imaging of a human (
Moyamoya Disease
Moyamoya disease is a life-threatening cerebrovascular disease that predisposes patients to both ischemic and hemorrhagic stroke. 98 The key angiographic feature that defines the condition is progressive stenosis of the intracranial internal carotid arteries and their branches. The classic configuration of their intracranial circulation is described as a ‘puff of smoke’, with network of abnormally dilated collateral vessels that attempt to compensate for the lack of blood flow through the normal conduits in the circle of Willis. A variety of direct and indirect surgical revascularization procedures are used to treat the condition, each with varying rates of success.99, 100, 101 No pharmacologic therapy has been shown to alter the natural history of the disease. Although several susceptibility loci have been identified102, 103, the pathogenesis of the condition remains elusive.
Zebrafish have proven to be valuable tools in this condition by allowing further investigation of gene function. Following genome-wide linkage analysis of affected families, a candidate gene
Ischemic Stroke
Ischemic stroke, resulting from cerebral vascular occlusion, is a major cause of death and disability worldwide. With the exception of tissue plasminogen activator, there are no targeted medical therapies available, highlighting the need for accelerated drug discovery. The use of zebrafish in the study of ischemic stroke lags behind hemorrhagic stroke, and only relatively few preliminary research efforts have been published on the subject. A notable study describes the establishment of a zebrafish model of hypoxic–ischemic injury
107
, with a follow-up report from the same group describing the neuroprotective effects of a zinc chelator using the same model.
108
Furthermore, certain hereditary forms of ischemic stroke in humans, such as cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy have been modeled in the zebrafish.
109
This model, generated by mutations in the
Even though limited work has been performed with ischemic stroke models, zebrafish represent a vast platform to investigate gene function. In particular, genes that control the expression of ion transport channels are central importance in understanding the pathophysiologic sequelae that follows an initial ischemic insult.
110
For example, the cation-chloride co-transporter NKCC1 (sodium-potassium-chloride co-transporter 1) is one of the key ion channels that contributes to the development of cytotoxic and ionic edema following ischemia.110, 111, 112, 113, 114 This ion channel is well described in the zebrafish and has been shown to be important in the regulation of endolymph volume in the otic vesicle and swim bladder volume.
115
Even though an inhibitor of this ion channel is available in humans (bumetanide), it is limited by low blood–brain barrier penetration
116
and lack of specificity at high concentrations.
117
The zebrafish represents an opportunity to discover another more selective NKCC1 inhibitor with better blood–brain barrier penetration, with potential as therapy to preempt post-ischemic cytotoxic and ionic edema. Several other channels implicated in dysregulation of the neurogliovascular unit following ischemic stroke, such as the
In addition to mitigating the secondary effects from ischemic stroke, such as hemorrhagic transformation and cerebral edema formation, an effort to understand the mechanisms of post-stroke recovery has the potential to uncover novel therapeutic targets. Specifically, a focus on adult neurogenesis and the migration of regenerating neurons in the post-injury recovery period is an ongoing area of research.120, 121, 122 These processes are being studied in the zebrafish, using adult fish as model organisms.123, 124, 125 Further refinements in experimental injury will allow for the study of brain recovery from injury, whether it is from stroke or trauma.
In any zebrafish model of cerebrovascular disease, it should be noted that many physiologic aspects of the cerebral circulation in zebrafish are not yet well known. Factors that have proven important in human ischemic stroke, such as collateral circulation 126 and autoregulatory capacity 127 , are yet to be evaluated in the zebrafish.
SUMMARY
Zebrafish have been used as model organisms in the investigation of both hemorrhagic and ischemic stroke. They have been shown to be useful not only in the investigation of gene function, but also as a high-throughput drug discovery screening platform. With the conservation of many molecular mechanisms of disease among vertebrates, zebrafish experiments are poised to result in a better understanding and new therapeutics for human cerebrovascular disease.
Footnotes
The authors declare no conflict of interest.
