Abstract
Physical activity may influence cerebrovascular function. The objective of this study was to determine the impact of life-long aerobic exercise training on cerebral vasomotor reactivity (CVMR) to changes in end-tidal CO2 (EtCO2) in older adults. Eleven sedentary young (SY, 27 ± 5 years), 10 sedentary elderly (SE, 72 ± 4 years), and 11 Masters athletes (MA, 72 ± 6 years) underwent the measurements of cerebral blood flow velocity (CBFV), arterial blood pressure, and EtCO2 during hypocapnic hyperventilation and hypercapnic rebreathing. Baseline CBFV was lower in SE and MA than in SY while no difference was observed between SE and MA. During hypocapnia, CVMR was lower in SE and MA compared with SY (1.87 ± 0.42 and 1.47 ± 0.21 vs. 2.18 ± 0.28 CBFV%/mm Hg, P < 0.05) while being lowest in MA among all groups (P < 0.05). In response to hypercapnia, SE and MA exhibited greater CVMR than SY (6.00 ± 0.94 and 6.67 ± 1.09 vs. 3.70 ± 1.08 CBFV1%/mm Hg, P < 0.05) while no difference was observed between SE and MA. A negative linear correlation between hypo- and hypercapnic CVMR (R2 = 0.37, P < 0.001) was observed across all groups. Advanced age was associated with lower resting CBFV and lower hypocapnic but greater hypercapnic CVMR. However, life-long aerobic exercise training appears to have minimal effects on these age-related differences in cerebral hemodynamics.
INTRODUCTION
Cerebral blood flow (CBF) is sensitive to changes in partial pressure of arterial carbon dioxide (PaCO2) such that hypercapnia increases and hypocapnia decreases CBF. 1 The responses of CBF to changes in PaCO2, termed cerebral vasomotor reactivity (CVMR), have been measured extensively in both clinical and research settings to assess cerebrovascular function. 2
Notably, CVMR is likely to be determined by both the local effects of PaCO2 on the cerebral vasculature as well as the systemic effects on arterial blood pressure (ABP) (e.g., chemoreceptor-mediated blood pressure changes). 3 Thus, the assessment of CVMR should take into account the effects of concomitant changes in arterial pressure on CBF.
Advancing age is associated with reduction in basal CBF due to decreased cerebral metabolic rate and/or the presence of cerebrovascular abnormalities.4–6 However, the effects of aging on CVMR are not well understood and previous findings are inconsistent.7–9 Moreover, recent studies suggest that aerobic exercise training from several months to a year improves cerebrovascular function.10,11 However, the impact of life-long aerobic exercise training on cerebrovascular function is not well understood. In this regard, Masters athletes are a unique group of older adults who have participated in life-long high volume, high intensity exercise training and can be used as a model to investigate the effects of exercise training on age-related changes in cerebrovascular function. 12
Accordingly, the purpose of this study was to determine the impact of life-long aerobic exercise training on age-related changes in CVMR. Based on previous studies,10,11 we hypothesized that cerebral CO2 reactivity to both hypo- and hypercapnia would be reduced in the sedentary elderly compared with young adults. Furthermore, we hypothesized that age-related reduction in cerebral CO2 reactivity would be attenuated in Masters athletes.
MATERIALS AND METHODS
Subjects
Twelve healthy sedentary young (27 ± 4 year, five women), 10 healthy sedentary elderly (72 ± 4 year, three women), and 11 Masters athletes (72 ± 6 year, two women) participated. A sedentary lifestyle was defined as not engaged in regular exercise training over the past 2 years. Sedentary subjects were excluded if they were exercising for ≥ 30 minutes, 3 times a week. Masters athletes were recruited from the race records of the United States Masters Athletes-sanctioned events demonstrating consistent age-group place winners at regional and national races. Masters athletes ultimately recruited were runners and who had participated in endurance training with a weekly running mileage of about 20–50 miles or equivalent cycling or swimming for more than 15 years and still engaged in exercise training and competitions at the time of this study.
Subjects were screened rigorously to exclude vascular disease and hypertension using 12-lead electrocardiogram, 24 hour blood pressure recordings, and echocardiograms. A brain MRI was performed to exclude cerebral infarct or other focal lesions. A detailed medical history questionnaire was used to exclude history of transient ischemic attack, stroke, diabetes, or other major medical problems. All subjects signed an informed consent to study protocols approved by the Institutional Review Boards of the UT Southwestern Medical Center and Texas Health Presbyterian Hospital Dallas.
Instrumentation and Data Acquisition
Heart rate (HR) was recorded using a 3-lead electrocardiogram system (Hewlett-Packard, Palo Alto, CA, USA). Brachial blood pressure was intermittently measured from the right upper arm by an electrosphygmomanometer (Suntech, Morrisville, NC, USA). Continuous ABP was monitored in the middle finger of the left hand with a Finapres (Finapres Medical Systems, Amsterdam, The Netherlands). The finger pressure transducer was fixed at the heart level during the study. End-tidal CO2 (EtCO2), an estimate of PaCO2, and breathing frequency were monitored with a nasal cannula using a capnograph (Carpnogard, Novamatrix, Wallingford, CT, USA). Arterial blood oxygen saturation was measured using a pulse oximeter (Biox 3700, Ohmeda Monitoring Systems, Boulder, CO, USA). Cerebral blood flow velocity (CBFV) was measured from the middle cerebral artery (MCA) by transcranial Doppler (TCD, Multi-Dop X2, Compumedics/DWL, Singen, Germany) using a 2-MHz probe. The probe was securely attached to the cranium using a mold that was casted individually to fit the facial bone structure to keep the position and angle of the probe unchanged during the study. 13 All signals were stored on a computer and analyzed using a data acquisition and analysis software (Acknowledge, BIOPAC Systems, Goleta, CA, USA) with a sampling frequency of 1000 Hz.
Experimental Procedures
All experiments were performed in an environmentally controlled laboratory with an ambient temperature of 22 °C. Subjects refrained from high intensity exercise, caffeinated beverages, or alcohol at least 24 hours before the test.
Of importance, previous literature has demonstrated that hypo- and hypercapnic cerebral CO2 reactivity is asymmetric and that hypocapnic cerebral vasoconstriction immediately before hypercapnic stimuli may influence ensuing vasodilation.14–16 For these reasons, cerebral CO2 reactivity to hypocapnia (hyperventilation) and hypercapnia (rebreathing) was assessed separately in this study.
After the subject had rested in the supine position for at least 10 minutes, a nose clip was placed and subjects breathed through a mouthpiece with a Y-valve, with one end open to room air and the other end connected to a 5 L rebreathing bag. Baseline CBFV, electrocardiogram, HR, ABP, and EtCO2 were recorded simultaneously for 3 minutes. During these measurements, subjects were instructed to breathe normally and to avoid body movement or Valsalva-like maneuvers. After baseline data collection, subjects were instructed to perform voluntary hyperventilation for 20 seconds (1 breath/s) to induce a brief period of hypocapnia. Following hyperventilation, a 5-minute recovery period was provided to allow cerebral hemodynamics to return to the baseline. Then, a modified rebreathing protocol was used to induce hypercapnia. 17 Briefly, at the end of a deep inspiration, the Y-valve of the mouthpiece was switched to the rebreathing bag for 3 minutes to induce a progressive increase in arterial CO2 and then followed by a recovery period for 4 minutes. 17 During rebreathing, a small amount of oxygen was added to the rebreathing bag based upon each subject's basal metabolic rate (estimated using the Harris-Benedict formula) to maintain constant arterial blood oxygen saturation. 17 Intermittent cuff arterial pressure was measured at baseline and during rebreathing to corroborate finger arterial pressure. The rebreathing protocol was well tolerated by all subjects.
Data Analysis
Baseline data were obtained by averaging a 3-minute data under resting condition. Cerebrovascular conductance index (CVCi) was calculated as mean CBFV divided by mean ABP to account for the effects of changes in blood pressure on CBFV during hypo- or hypercapnia. 17 The magnitude of changes in CBFV, CVCi, ABP, HR, and EtCO2 during hypo- and hypercapnia are presented as ΔCBFV, ΔCVCi, ΔABP, ΔHR, and ΔEtCO2 respectively. Percentage changes in CBFV and CVCi were calculated relative to the baseline values.
During hyperventilation, ΔCBFV, ΔCVCi, ΔABP, ΔHR, and ΔEtCO2 were calculated as the differences between the baseline and maximal changes of these variables (averaged from three breath cycles) after reduction in EtCO2 reached its nadir (Supplementary Figure 1). CVMR was calculated as the ratio of ΔCBFV% to ΔEtCO2 (i.e., ΔCBFV%/ΔEtCO2) or ΔCVCi% to Δ EtCO2 (i.e., ΔCVCi%/ΔEtCO2). 17 Of note, linear regression analysis of breath-by-breath data was not performed because rapid transient changes in EtCO2 during hyperventilation may not accurately reflect transient changes in PaCO2. Thus, maximal reductions in CBFV% and CVCi% in response to the maximal reduction in EtCO2 were used to assess cerebral CO2 reactivity when these variables reached steady state regardless of the time delay (Supplementary Figure 1). Cardiovascular reactivity to changes in EtCO2 was calculated as the ratio of ΔABP to the corresponding changes in EtCO2 (i.e., ΔABP/ΔEtCO2).
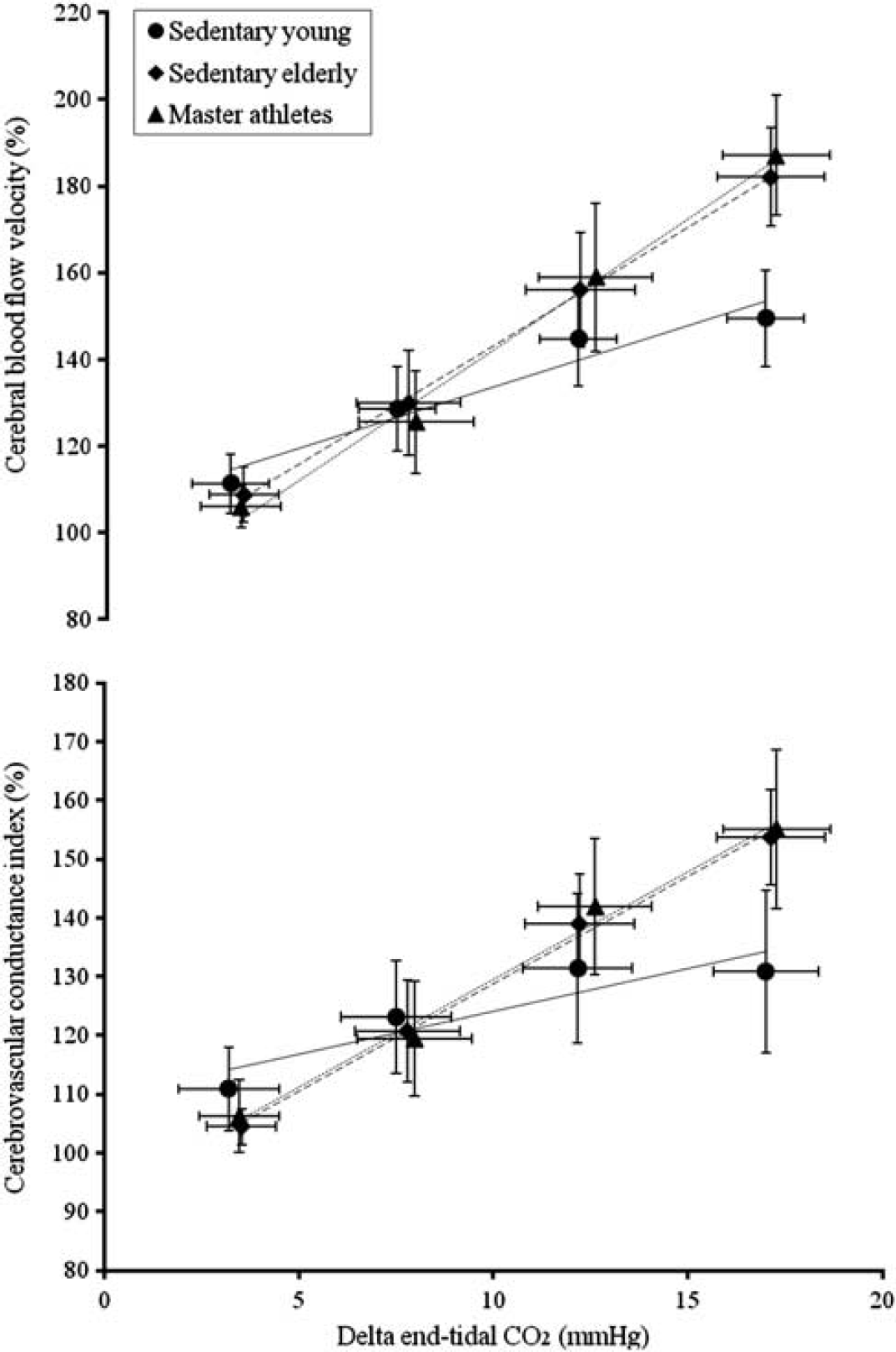
Linear regression analysis of group-averaged data showing the associations among the changes in cerebral blood flow velocity (CBFV%), cerebrovascular conductance index (CVCi%), and end-tidal CO2 (EtCO2) during hypercapnic rebreathing in the sedentary young (SY), the sedentary elderly (SE), and Masters athletes (MA). The error bars represent standard deviations. SY: CBFV% = 105.44 + 2.82* ΔEtCO2, R2 = 0.94, P < 0.001; CVCi% = 109.54 + 1.46*ΔEtCO2, R2 = 0.83; P < 0.001. SE: CBFV% = 88.94 + 5.44*ΔEtCO2, R2 = 0.99, P < 0.001; CVCi% = 92.32 + 3.65*ΔEtCO2, R2 = 0.99; P < 0.001. MA: CBFV% = 82.39 + 6.01*ΔEtCO2, R2 = 0.99, P < 0.001; CVCi% = 92.79 + 3.67*ΔEtCO2, R2 = 0.99; P < 0.001.
For hypercapnia, breath-by-breath data were measured.17,18 Notably, due to a deep inspiration performed immediately before rebreathing, a brief reduction in both EtCO2 and CBFV was observed at the very beginning of rebreathing (Supplementary Figure 1). Hence, only those data of CBFV from its recovery to the baseline level and to the maximal increases during rebreathing were used to assess cerebral CO2 reactivity. Notably, a time interval of ∼ 15 seconds between the onset of rebreathing and recovery of CBFV to the baseline level would allow equilibrium to be reached between changes in EtCO2 and PaCO2. Linear regressions of ΔCBFV% and ΔCVCi% vs. ΔEtCO2 were performed for each individual subject and then group averaged for statistical analysis. The slopes of these regression lines were used as the estimates of cerebral CO2 reactivity during hypercapnia. 17 Of note, the rebreathing protocol used in the current study was shorter than that of ours and other previous studies and induced moderate increases in EtCO2 and CBFV and thus an approximately linear relationship between these variables (Figure 1). 17 For data presentation, changes in EtCO2 from the baseline (ΔEtCO2) in each bin of 5 mm Hg were averaged, and the corresponding changes in ABP, CBFV%, and CVCi% were calculated for each individual subject. These data were averaged within each group and presented using linear regression. Cardiovascular reactivity to ΔEtCO2 was assessed by the slope of linear regression between ΔABP and ΔEtCO2.
Finally, cerebral vasomotor capacity from the hypocapnic minimum to hypercapnic maximum was assessed by the ratio of maximal changes in CBFV% and CVCi% to the corresponding changes in EtCO2. Consistently, ΔABP and ΔHR were calculated as the maximal differences between the hypo- and hypercapnic conditions. Cardiovascular reactivity to changes in EtCO2 was calculated as the ratio of maximal changes in ABP to EtCO2.
Statistics
One-way analysis of variance was used to compare group differences in baseline data and the magnitude of changes in hemodynamic variables during hyperventilation and rebreathing. χ-Analysis was used to test group difference in the sex distribution. Analysis of covariance was used to test group differences after the adjustment for the effect of sex. For a significant F-value, LSD post hoc analysis was used to determine group differences. Maximal changes in CBFV, CVCi, ABP, HR, and EtCO2 at the end of hyperventilation or rebreathing were compared with the baseline values using paired t-tests. The results of linear curve fitting were examined by the coefficient of determination (R2) and by the model residual analysis of individual data. Furthermore, a multivariate linear regression model (i.e., CBFV% = a*ΔEtCO2 + b*ΔABP) was constructed to examine the contributions of hypercapnic changes in ΔEtCO2 and ΔABP to ΔCBFV%. 19 The unstandardized regression coefficients for EtCO2 (a) and ABP (b) were compared among the groups using analysis of variance. Pearson's product-moment and partial correlation analyses were used to examine the relationship between hypo- and hypercapnic cerebral CO2 reactivity. Data are presented as mean ± s.d. An α-level of 0.05 was set as the criterion for statistical significance. All statistical analyses were performed using SPSS 19.0 (SPSS, Chicago, IL, USA).
RESULTS
Baseline Hemodynamics
Data from one young subject and one Masters athlete were excluded because of the poor-quality of TCD recordings. Demographic characteristics of subjects and baseline hemodynamics are shown in Table 1. There were no group differences in sex, body mass index, ABP, EtCO2, and arterial blood oxygen saturation. Systolic blood pressure was higher in the sedentary elderly and Masters athletes than in young subjects; however, this group difference disappeared after adjustment for sex. As expected, Masters athletes showed lower resting HR than young and old sedentary subjects (P < 0.05), indicating an effect from aerobic exercise training. Systolic, diastolic, mean, and pulse CBFV and CVCi were all lower in the sedentary elderly and Masters athletes than in young subjects.
Demographic characteristics of subjects and baseline cerebral and systemic hemodynamics
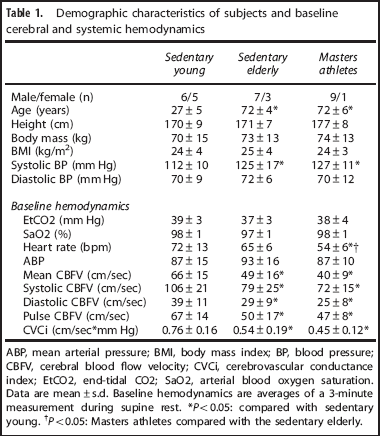
ABP, mean arterial pressure; BMI, body mass index; BP, blood pressure; CBFV, cerebral blood flow velocity; CVCi, cerebrovascular conductance index; EtCO2, end-tidal CO2; SaO2, arterial blood oxygen saturation. Data are mean±s.d. Baseline hemodynamics are averages of a 3-minute measurement during supine rest. *P < 0.05: compared with sedentary young. *P < 0.05: Masters athletes compared with the sedentary elderly.
Cerebral and Cardiovascular Reactivity to Hypocapnia
Representative data of changes in CBFV, ABP, HR, and EtCO2 during hyperventilation is presented in Supplementary Figure 1. End-tidal CO2 and CBFV rapidly decreased with hyperventilation, reached a steady state at the end of the breathing maneuver, and remained low until spontaneous breathing was recovered. Group-averaged data of maximal and percentage changes in EtCO2, ABP, HR, CBFV and CVCi are presented in Table 2 and individual data are presented in Data Supplementary Table 1. During hyperventilation, EtCO2, ABP, CBFV, and CVCi decreased while HR increased from the baseline. Notably, despite similar reductions in EtCO2 across all groups, reduction in ABP and elevation in HR were attenuated in the sedentary elderly and Masters athletes compared with young subjects. Consistently, cardiovascular reactivity calculated as ΔABP/ΔEtCO2 was lower in the sedentary elderly and Masters athletes than in young subjects. The magnitude of reduction in ΔCBFV% was attenuated in the sedentary elderly and Masters athletes compared with young subjects. Interestingly, the reduction in ΔCBFV% was further attenuated in Masters athletes than the sedentary elderly. Correspondingly, CVMR was lowest in the Masters athletes.
Cerebral and systemic hemodynamics during hypocapnia
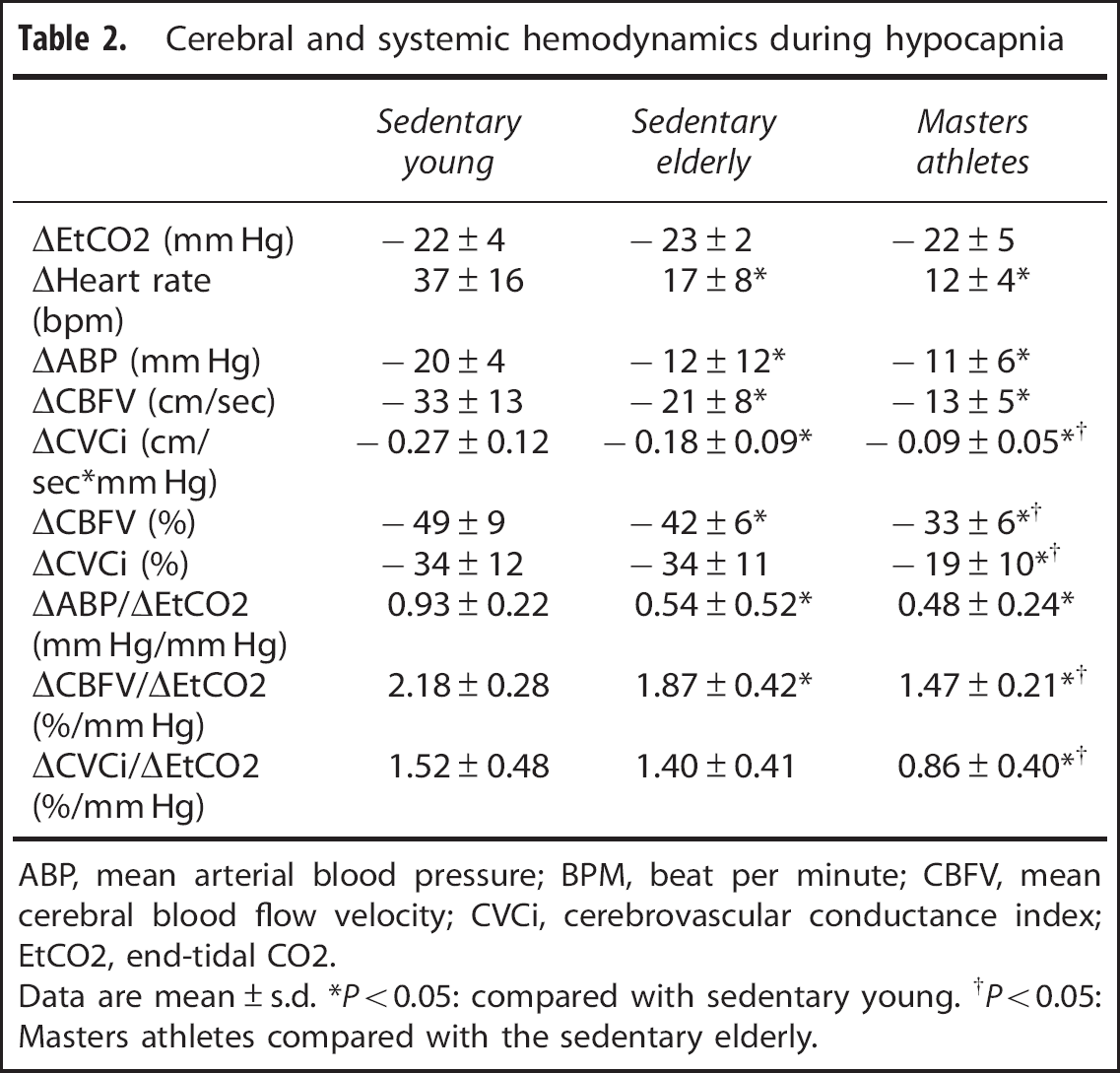
ABP, mean arterial blood pressure; BPM, beat per minute; CBFV, mean cerebral blood flow velocity; CVCi, cerebrovascular conductance index; EtCO2, end-tidal CO2.
Data are mean ± s.d. *P < 0.05: compared with sedentary young. †P < 0.05: Masters athletes compared with the sedentary elderly.
Cerebral and Cardiovascular Reactivity to Hypercapnia
Representative data of changes in CBFV, ABP, HR, and EtCO2 during rebreathing is presented in Data Supplementary Figure 1. Group-averaged data of maximal and percentage changes in EtCO2, ABP, HR, CBFV, and CVCi are summarized in Table 3 and individual data are presented in Data Supplementary Table 1. During rebreathing, EtCO2, HR, ABP, CBFV, and CVCi increased from the baseline. Despite the similar increase in EtCO2, elevations in δCBFV% and δCVCi% were greater in the sedentary elderly and Masters athletes compared with young subjects. Correspondingly, CVMR measured from the slopes of both δCBFV% and δCVCi% vs. δEtCO2 were steeper in the sedentary elderly and Masters athletes than in young subjects. Goodness of line fit for δCBFV% and δCVCi% vs. δEtCO2 yielded the excellent coefficients of determination (R2 = 0.90 ±0.13 and 0.80 ±0.27, respectively). These data were also consistent with the group-averaged data in Figure 1. Cardiovascular reactivity to EtCO2 measured from the slope of δABP vs. δEtCO2 did not show group differences although there was great individual variability regardless of age and exercise status (Data Supplementary Figure 2).
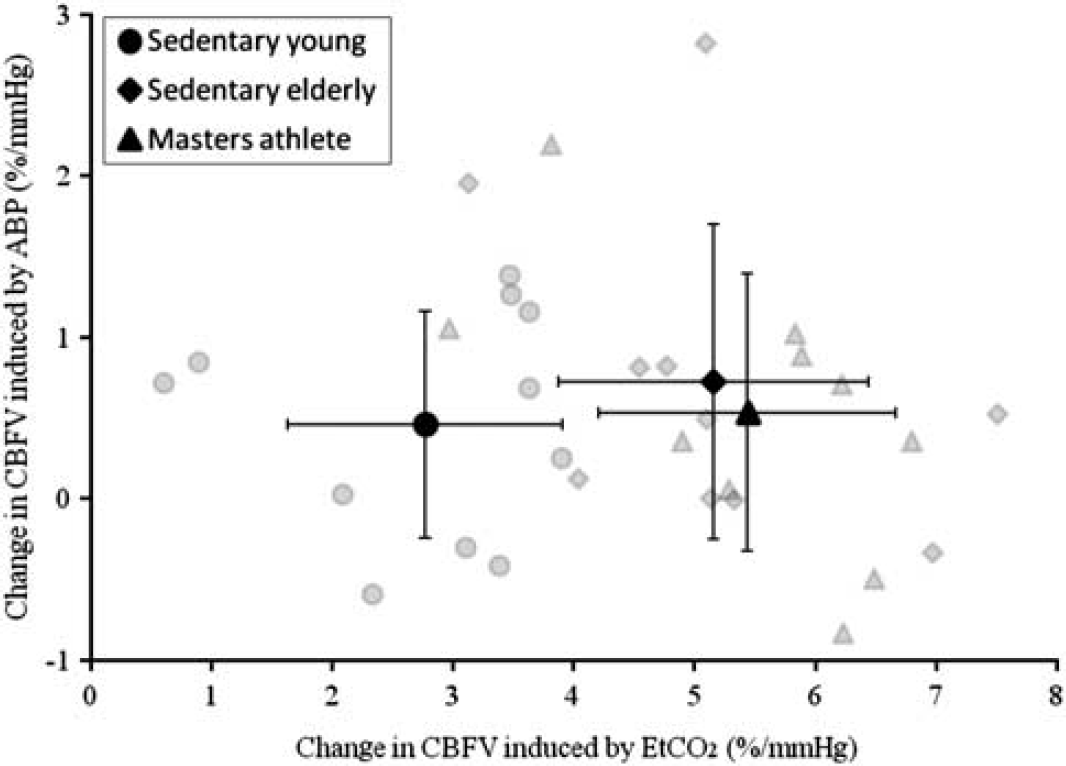
Multivariate linear regression analysis of group-averaged and individual data exhibiting the contributions of hypercapnic changes in end-tidal CO2 and arterial blood pressure to cerebral blood flow velocity in the sedentary young, sedentary elderly, and Masters athletes. The error bars represent standard deviations.
Cerebral and systemic hemodynamics during hypercapnia
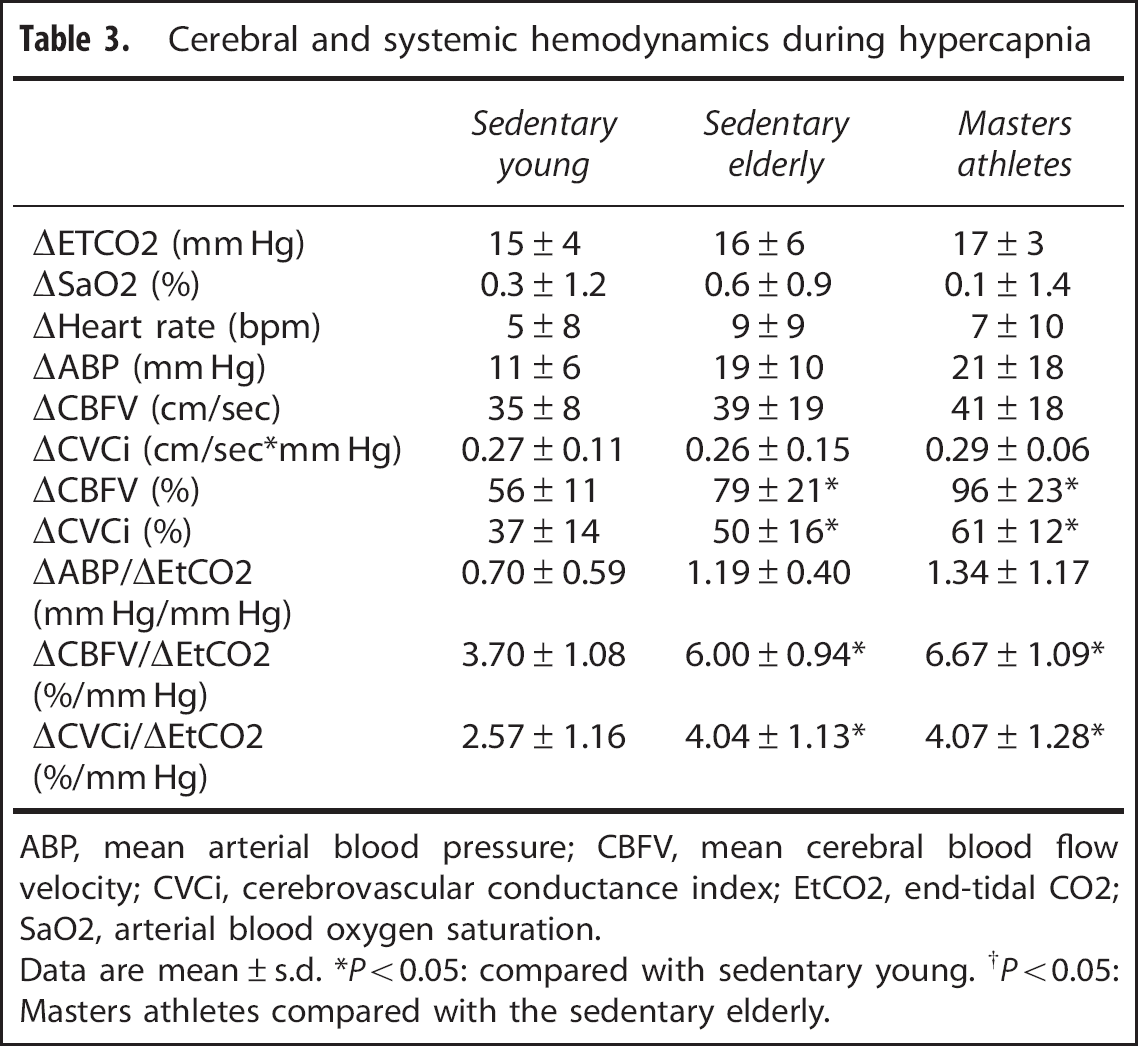
ABP, mean arterial blood pressure; CBFV, mean cerebral blood flow velocity; CVCi, cerebrovascular conductance index; EtCO2, end-tidal CO2; SaO2, arterial blood oxygen saturation.
Data are mean ± s.d. *P < 0.05: compared with sedentary young. †P < 0.05: Masters athletes compared with the sedentary elderly.
Multivariate linear regression analysis demonstrated a greater contribution of δEtCO2 to δCBFV% in the sedentary elderly and Masters athletes than in the sedentary young subjects (5.16 ± 1.28 and 5.44 ± 1.23 vs. 2.77 ± 1.14 CBFV%/mm Hg, respectively) while no group difference was observed by the contribution of δABP (0.73 ±0.98, 0.54 ± 0.86, and 0.46 ±0.70 CBFV%/mm Hg, respectively) (Figure 2), suggesting that the observed group differences in CVMR are not due to age- or exercise-related differences in cerebral pressure autoregulation.
Relationship between Hypo- and Hypercapnic Cerebral Vasomotor Reactivity
There was a negative correlation between hypo- and hypercapnic CVMR measured as CBFV% to the corresponding changes in EtCO2 (R2 = 0.37, P < 0.001) across all groups (Figure 3). Of note, this relationship in fact indicates that greater reductions in CBFV during hypocapnia were associated with smaller increases in CBFV during hypercapnia and vice versa. In addition, this correlation remained statistically significant after controlling the differences in baseline CBFV, which was included in the calculation of CVMR.
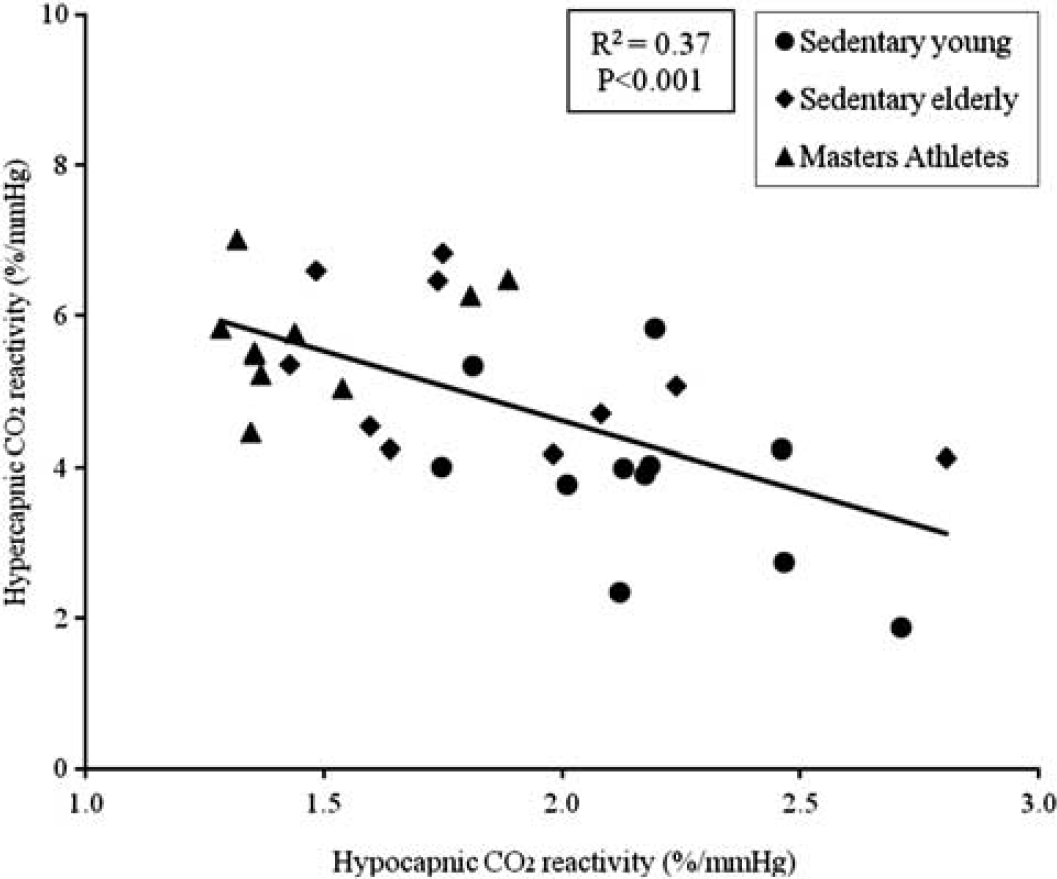
Linear correlation between hypo- and hypercapnic cerebral CO2 reactivity from each subject. Both reactivity measurements were calculated as the ratio of maximal changes in cerebral blood flow velocity (%) in response to the corresponding changes in end-tidal CO2 (mmHg).
Maximal changes in EtCO2, ABP, CBFV%, and CVCi% from hypocapnia to hypercapnia did not show group differences (Supplementary Table 2). Consistently, there were no group differences in cardiovascular reactivity to the changes in EtCO2. Cerebral CO2 reactivity calculated as δCBFV%/δEtCO2 from the hypocapnic minimum to hypercapnic maximum did not show age-related group difference between the sedentary young and older subjects while higher in Master athletes. The maximal CVMR calculated as δCVCi%/δEtCO2 did not show group differences, suggesting the preservation of cerebral vasomotor capacity with advanced age.
DISCUSSION
The primary findings from the present study are as follows. First, we found that CBFV and CVCi under resting condition were lower in the sedentary elderly and Masters athletes than in young subjects. Second, CVMR to hypocapnia was attenuated in the sedentary elderly and Masters athletes compared with young subjects while Masters athletes exhibited the lowest reactivity. Third, in contrast to our hypothesis, CVMR to hypercapnia was enhanced in both sedentary elderly and Masters athletes compared with young subjects while no differences were observed between sedentary elderly and Masters athletes. Interestingly, there was a negative correlation between hypo- and hypercapnic CVMR while the vasomotor capacity was similar across all groups. Collectively, these findings suggest that although there were significant age-related differences in CVMR, the effects of life-long aerobic exercise training on these age-related differences are likely to be minimal.
Effects of Age and Exercise on Basal Cerebral Blood Flow
The effect of age on CBF has been investigated extensively over the last several decades.4,5 Most studies have demonstrated a gradual decline in CBF after the late young adulthood.5,20 Consistent with previous findings, the current study demonstrated the reductions in systolic, diastolic, and mean CBFV in the sedentary elderly, although the lack of MCA diameter data does not provide us the actual volumetric flow measurement.6,21 However, it should be highlighted that given the age range of 45 years between our young and old sedentary subjects and a reduction of mean CBFV byB25% in this study, the rate of reduction in CBFV would be B0.6% per year (assuming a constant rate of decline). These results are consistent with previous findings of age-related decline in brain perfusion measured with positron emission tomography or phase-contrast MRA.5,22
Engagement in physical activity or aerobic exercise training may ameliorate age-related reduction in brain perfusion.21,23 A recent study showed that CBFV was elevated in the physically fit individuals over a lifespan from 18-79 years. 21 In addition, in a study of elderly retirees, CBF was preserved in those who either participated in regular physical activities or continued to work, but was reduced in inactive retirees in a 4-year follow-up study. 23 These studies have led us to hypothesize that age-related decline in CBF may be ameliorated in the Masters athletes who have participated in life-long high volume, high intensity aerobic exercise training. However, contrary to our hypothesis, Masters athletes exhibited reductions in CBFV resembling the sedentary elderly.
There are at least two possibilities, which may explain these controversial findings. First, the cross-sectional nature of the present study cannot shed light on any potential cause-effect relationship between exercise training and CBF or CBFV. Second, a mechanism of hormesis may underpin these apparent inconsistent findings. It is possible that life-long high volume, high intensity aerobic exercise training may not be optimal for protecting brain perfusion from the age-related decline.24,25 In this regard, our recent study demonstrated that 3 months of moderate aerobic exercise training increased global CBF in the elderly women over 60 years. 26 Thus, further longitudinal studies are needed to determine if indeed there is a ‘dose–response’ relationship between exercise training and brain perfusion.
Effects of Age and Exercise on Cerebral Vasomotor Reactivity
In the present study, CVMR during hypocapnia was reduced in the sedentary elderly compared with young subjects, consistent with previous studies under similar conditions.9,27 In contrast, hypercapnic cerebral CO2 reactivity was not reduced but enhanced in the sedentary elderly and Masters athletes compared with young subjects.8,20,28,29 Of note, the magnitude of absolute increases in CBFV and CVCi during hypercapnic rebreathing was similar across all groups (Table 3). Moreover, hypo- and hypercapnic CVMR were negatively correlated (Figure 3) while the maximal vasomotor capacity was similar across all groups (Supplementary Table 2). These observations may collectively be explained by a down-and-rightward shift of the EtCO2-CBFV curve with age, which would predict an attenuation of hypocapnic but enhancement of hypercapnic CVMR in older adults associated with a reduction in resting CBFV (operating point) (Figure 4).
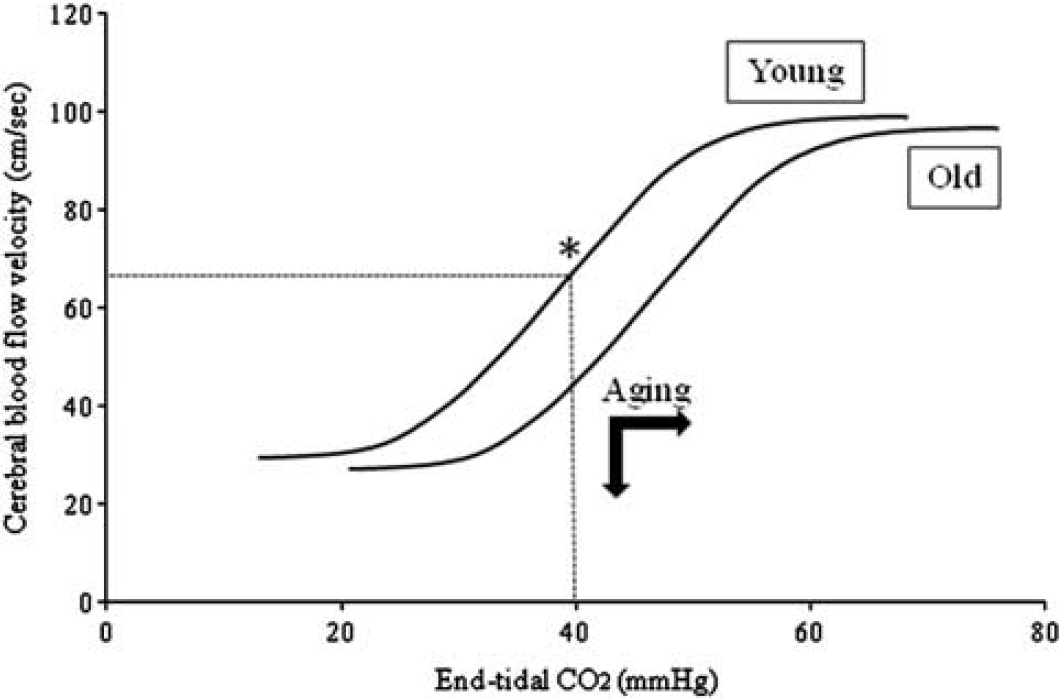
A hypothetical drawing to illustrate an age-related down-and-rightward shift of cerebral blood flow velocity (CBFV) and end-tidal CO2 (EtCO2) curve. During hypocapnic hyperventilation, cerebral vasomotor reactivity (slope of the curve) would be reduced in old when compared with young because reduction in basal CBFV would lead the operating point (*) being closer to the level of maximal vasoconstriction. Conversely, during hypercapnic rebreathing, the slope of the curve in the young would be attenuated when EtCO2 reached approximately 50 mm Hg while CBFV may continue to increase in old leading to higher cerebral vasomotor reactivity.
Only few studies have previously addressed the effect of exercise training on cerebral CO2 reactivity.11,30 An earlier study of aerobic training of 18 weeks in heart failure patients found no changes in cerebral CO2 reactivity although the improvements in physical and cognition were observed. 30 In contrast, a recent study of stroke survivors reported that cerebral CO2 reactivity was improved after 6 months of aerobic training, although resting CBFV did not change. 11 In the present study, we found minimal effects of life-long aerobic exercise training on age-related differences in CVMR. Masters athletes only demonstrated a smaller hypocapnic CVMR when compared with the sedentary elderly. These data suggest that life-long exercise training may protect brain from hypoperfusion during hypocapnia-induced cerebral vasoconstriction.
Methodological Considerations and Study Limitations
Several methodological issues need to be discussed. First, different from our own previous studies and others using the CO2 rebreathing methods, CVMR was assessed separately during hypocapnic hyperventilation and hypercapnic rebreathing because of its asymmetric nature under these conditions.4,15,17,31-33 Previous studies of aging effects on cerebral CO2 reactivity often were conducted either during hypocapnia or hypercapnia separately.8,9,20,28,29 Thus, findings of this study can be compared directly with those conducted under similar conditions.8,9,20,28,29
Second, different from previous studies which have used prolonged rebreathing protocols (∼ 5–7 minutes) to induce a wide range of changes in arterial CO2, a relatively short period of 3 minutes was used in this study to induce a moderate increase in EtCO2 and linear increases in CBFV to facilitate data analysis.16,17,34 A shorter rebreathing protocol also reduced the discomfort of subjects, which may occur during prolonged rebreathing associated with high level of CO2.16,17 Of note, arterial blood oxygen saturation was maintained constant during rebreathing to minimize any potential effects of hyperoxemia or hypoxemia on CBF responses to CO2. 35
Finally, CVMR measured during transient changes in EtCO2 may be different from that obtained under steady-state conditions. Previous studies reported that CVMR was higher during steady state than transient changes in EtCO2. 16 While cautions should be taken to compare measurements obtained under different conditions, our preliminary study showed no significant differences in cerebral CO2 reactivity to transient or steady-state changes in EtCO2. 36
Several study limitations also deserve discussion. First, changes in CBFV reflect changes in blood flow only if the insonated MCA diameter remained unchanged. Direct measurement of the MCA diameter during moderate changes in arterial pressure and CO2 during craniotomy did not show significant changes. 37 Thus, changes in CBFV during hyperventilation and rebreathing most likely reflected changes in volumetric blood flow. However, age-related group differences in absolute CBFV may reflect individual or group differences in the MCA diameter. Recent magnetic resonance angiography studies reported a slight increase in the MCA diameter in older adults over 60 years on the right side but not on the left side compared with young subjects aged 18-29 years. 38 Such age-related differences in the MCA diameter may explain lower CBFV in our older subjects. However, the reported differences in the MCA diameter were small (∼ 0.04 mm) and well below the imaging voxel size used in this study (0.5 mm × 0.5 mm × 0.8 mm). 38 Future studies need to determine the potential contributions of age-related changes in the MCA diameter to CBFV in the elderly.
Second, like most of previous studies of effects of aging on CVMR, a potential cause-effect relationship between aging and CVMR cannot be determined in this study. Moreover, previous studies suggested that age-related differences in CVMR were more prominent in women than in men. 29 Although the current study did not find such sex-related effects on CVMR, cautions should be taken due to the small sample size and exploratory nature of this study. Future studies with larger sample size are needed to confirm the observation of this study.
Third, assessment of CVMR is likely to be influenced by both systemic and local hemodynamic factors such as changes in arterial pressure and sympathetic neural activity during CO2 stimuli.3,39,40 Indeed, we observed large individual variability in cardiovascular reactivity (δABP/δEtCO2) to changes in EtCO2 during both hyperventilation and rebreathing (Tables 2 and 3, and Data Supplementary Table 1). However, assessment of changes in CVCi showed similar pattern as changes in CBFV during both hypo- and hypercapnic stimuli. In addition, no group differences in arterial pressure responses during hypercapnic rebreathing were observed (Supplementary Figure 2). Moreover, multivariate linear regression analysis showed no group differences in the contribution of ABP to CBFV% during hypercapnia (Figure 2). Thus, age-related differences in CVMR observed in this study cannot be attributed mainly to systemic factors.
In summary, this study demonstrated significant age-related differences in cerebral hemodynamics under resting condition and during hypo- and hypercapnia. We found that CBFV and CVCi under resting condition were substantially reduced in the sedentary elderly and Masters athletes compared with young subjects. Furthermore, CVMR to hypocapnia was reduced while it was enhanced during hypercapnia in the sedentary elderly and Masters athletes. However, life-long aerobic exercise training appears to have minimal effects on the age-related differences in CVMR.
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
Footnotes
ACKNOWLEDGEMENTS
The authors would like to thank the study participates for their willingness, time, and effort to make this project possible and Daniel Cresson and Kyle Armstrong for their technical support. This study was supported in part by the Texas Health Research & Education Institute Pilot Study Award, R01HL102457-01 and R01AG033106-01.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
