Abstract
We investigated the effects of ionizing radiation on microvessel structure and complexity in the hippocampus. We also assessed neurogenesis and the neurovascular niche. Postnatal day 14 male C57BL/6 mice received a single dose of 8 Gy to the whole brain and were killed 6 hours, 1 week, 7 weeks, or 1 year later. Irradiation decreased the total number of microvessels and branching points from 1 week onwards and decreased the total microvessel area 1 and 7 weeks after irradiation. After an initial increase in vascular parameter densities, concomitant with reduced growth of the hippocampus, the densities normalized with time, presumably adapting to the needs of the surrounding nonvascular tissue. Irradiation decreased the number of neural stem and progenitor cells in the hippocampus. The relative loss increased with time, resulting in almost completely ablated neurogenesis (DCX+ cells) 1 year after irradiation (77% decreased 1 week, 86% decreased 7 weeks, and 98% decreased 1 year after irradiation compared with controls). After irradiation, the distance between undifferentiated stem cells and microvessels was unaffected, and very few dying endothelial cells were detected. Taken together, these results indicate that the vasculature adjusts to the surrounding neural and glial tissue after irradiation, not vice-versa.
INTRODUCTION
Brain tumors account for 20–30% of all childhood cancers, making them the second most common type of pediatric cancers after leukemias.1,2 The Nordic countries have among the highest incidence rates in the world 3 and radiotherapy is commonly used in the treatment of these tumors. However, there are several long-term debilitating effects following radiotherapy in children, including cognitive decline and motor, hormonal and psychological–emotional dysfunctions.4,5 Furthermore, survivors of childhood cancer have an increased risk of long-term cerebrovascular mortality, and the risk of death from cerebrovascular disease increases linearly with the local radiation dose to the prepontine cistern. 6
More than four centuries ago, it was observed that the vascular and the nervous systems show similar anatomical patterning. Vascular outgrowth has been observed to precede neural axon outgrowth in the developing mouse embryo. This robust generation of a vascular plexus in the embryo is later followed by remodeling into fine capillary networks. 7 Paradoxically, it has been suggested that peripheral nerves in turn provide a template for this vascular remodeling and determines the patterning of arterial differentiation and blood vessel branch patterning in the skin. In addition, arteries are specifically aligned with peripheral nerves. 8 It therefore appears as if the vasculature and the nervous system during development and beyond are interdependently regulated and a change in one can alter the other.
Decades ago, it was hypothesized that the damage observed in the brain following irradiation was mainly due to microvessel damage that in turn caused ischemia and necrosis. This hypothesis was supported by alterations to capillaries that preceded general tissue necrosis, such as thickening of the capillary basal lamina. 9 In addition, cranial irradiation has previously been demonstrated to cause widely scattered minute necrotic lesions in the forebrain white matter of adult monkeys subjected to 60 Gy fractionated irradiation. The number of lesions increased with time after treatment and were accompanied by both focal vascular endothelial hyperplasia as well as patches of telangiectasias. 10 The prevailing view was that the radiosensitivity of the brain and spinal cord was mediated in large part by the response of cerebrovascular wall elements and that these responses constituted the primary limitation of radiotherapy. 11 Today, much attention is instead focused on the birth of new neurons in the postnatal brain. 12 As a result, it was revealed that areas in the brain where postnatal neurogenesis occurs are particularly sensitive to radiotherapy and that loss of these stem/progenitor cells may contribute to the cognitive deficits observed after radiation therapy.12,13 A critical, but poorly understood regulator of neurogenesis is the role that blood vessels play in nurturing newborn cells. Proliferating cells in the hippocampus are vascular-associated, that is, they are born in close proximity to blood vessels and blood vessels appear to be critical in providing support for these cells.14,15 In addition, endothelial cells are believed to have direct cell-to-cell contact with stem cells to increase neuronal survival. 16 It is therefore important to consider the effect on the vasculature when investigating radiation-induced outcomes in neurogenesis.
Most studies have focused on how adult and mature brain tissue responds to radiation therapy. Considerably fewer studies have investigated the effects of irradiation on the young, developing brain and very little is known about how microvessels in the hippocampus respond to this type of insult.17–19 Therefore, the primary aim of this study was to investigate how microvessels in the hippocampus were affected by irradiation to the young mouse brain, by quantifying blood vessel parameters at different time points after irradiation. We also sought to investigate the role of the neurovascular niche in the support and survival of neural stem/progenitor cells.
MATERIALS AND METHODS
Animals
Male C57BL/6 mice were delivered on postnatal day 9 (P9) from Charles River Breeding Laboratories (Sulzfeld, Germany). The animals were housed with a reversed 12-hour dark-light cycle (light between 2100 and 0900 hours) at constant temperature (24°C) and relative humidity (50–60%), with
Irradiation Procedure
A linear accelerator (Varian Clinac 600 CD, Radiation Oncology Systems LLC, San Diego, CA, USA) with 4 MV nominal photon energy and a dose rate of 2.3 Gy/min was used to irradiate mice on P14. The mice were anesthetized with an intraperitoneal injection of tribromoethanol (50 mg/kg; Sigma, Stockholm, Sweden), placed on a polystyrene bed in a prone position (head to gantry) and irradiated with a symmetrical 2 × 2 cm radiation field. The head was covered with a 1-cm tissue equivalent material to obtain an even radiation dose in the recipient tissue. Source to skin distance was ~99.5 cm and the irradiated tissue received a single absorbed dose of 8 Gy with a dose variation of ± 5%. The animals were kept on a warm bed (36°C) both before and after irradiation to maintain body temperature. Control animals were anesthetized but did not receive any irradiation.
Tissue Preparation
Animals (six controls and six irradiated for each time point) were deeply anesthetized with sodium pentobarbital (Pentothal; Electra-box Pharma, Tyresö, Sweden) and transcardially perfused with 0.9% saline, followed by 4% paraformaldehyde, 6 hours, 1 week (8 days), 7 weeks, or 62 weeks (in the text referred to as the ’1-year’ group) after irradiation. The brains were removed and postfixed for 24 hours in paraformaldehyde at 4°C before being transferred to a 30% sucrose solution (30% sucrose in 0.1 M phosphate buffer).
After equilibration in sucrose, the brains were fixed with a cryo-gel (Tissue-Tec O.C.T compound) to a dry ice-cooled copper block and one hemisphere was sagitally cut into 25-μm sections with a sliding microtome (Leica SM2000R, Leica Microsystems, Nussloch, Germany). Serial sections were collected in a series of 10 (1-week group) or 12 (6 hours, 7 weeks, and 1-year groups) tubes containing TCS (tissue cryoprotectant solution; 25% ethylene glycol, 25% glycerin, and 0.05 M phosphate buffer), and stored at 4°C until used for immunohistochemistry.
Immunohistochemistry
Stainings for Stereological Quantifications.
Stainings for stereological analysis were performed on every 10th (1-week group) or 12th (7 weeks and 1-year groups) section and rinsed in Tris-buffered saline (TBS; 0.08 mol/L Trizma-HCl, 0.016 mol/L Trizma-Base, 0.15 mol/L NaCl, pH 7.5) before staining. Antigen retrieval was performed on sections for doublecortin (DCX) staining, consisting of 30 minutes incubation in 80°C sodium citrate (10 mM, pH 9.0). Sections for cluster of differentiation 31 (CD31, also called platelet endothelial cell adhesion molecule-1 [PECAM-1]) staining did not undergo any pretreatment. After rinsing in TBS, the sections underwent two subsequent steps to avoid unspecific binding. First, endogenous peroxidase activity was blocked by incubating the sections in 30% hydrogen peroxide (H2O2) for 30 minutes, followed by rinsing in TBS. This was followed by 30 minutes incubation in TBS with 3% donkey serum and 0.1% Triton X-100 (blocking solution) to avoid unspecific antigen binding. Sections were incubated with primary antibodies diluted in blocking solution at 4°C for either 24 hours (monoclonal rat anti-mouse CD31, 1:2,000, BD Bioscience Pharmigen, Franklin Lakes, NJ, USA) or 72 hours (polyclonal goat anti-DCX, 1:125, Santa Cruz Biotechnology, Santa Cruz, CA, USA). After rinsing in TBS, sections were incubated at room temperature for 1 hour with biotinylated secondary antibodies diluted 1:1,000 in blocking solution; donkey anti-rat IgG or donkey anti-goat IgG (Jackson ImmunoResearch Laboratories, West Grove, PA, USA). This was followed by rinsing and amplification with avidin-biotin enzyme complex (ABC kit, Vectastain Elite, Vector Laboratories, Burlingame, CA, USA) for 1 hour, and finally incubation in detection solution (0.25 mg/mL diaminobenzidine, 0.009% H2O2, 0.04% NiCl2) for visualization.
Immunofluorescence.
For double and triple stainings, every 12th section was used to quantify numbers of dying endothelial cells (CD31, active caspase-3, and TO-PRO) and the distance between stem cells and nearest microvessel (CD31 and brain lipid-binding protein (BLBP)). Sections were rinsed in TBS and incubated in blocking solution for 30 minutes, followed by rinsing and incubation for 72 hours at 4°C with primary antibodies: rat anti-mouse CD31 (1:500), rabbit anti-BLBP (1:500, Millipore, Temecula, CA, USA) and rabbit anti-active caspase-3 (1:250, BD Bioscience Pharmigen). Sections were then rinsed and incubated with appropriate fluorescent secondary antibodies for 2 hours: donkey anti-rat Alexa 488, donkey anti-rabbit Alexa 647, donkey anti-rabbit Alexa 555 (all 1:1,000, Invitrogen/Molecular Probes, Carlsbad, CA, USA), and TO-PRO-3 Iodide (1:4,000, Invitrogen/Molecular Probes).
Stereological Principles for Microvessel Analysis
To quantify and characterize microvessels in the brain, the following parameters were analyzed:
area covered by microvessels
number of microvessels
diameter of the microvessels
number of microvessel branching points
This was assessed in one neurogenic hippocampal area (i.e., the granule cell layer; GCL) and two adjacent nonneurogenic hippocampal areas (the hilus and the molecular layer; ML), using a Leica DM6000B microscope (Leica, Wetzlar, Germany) and the
The counting frame had an overlay of crosses every 5 μm to estimate the total area covered by blood vessels (10 × 10 crosses for ML (Figure 1A) and 5 × 15 crosses for GCL and hilus). Every cross that touched a vessel was marked and the total number of marked crosses was summed up. Each cross represented an area of 25 μm2 (5 × 5 μm). The total area covered by vessels was calculated by first multiplying the total number of marked crosses with the corresponding area of each cross (25 μm2). This was then divided with the fraction of the sampled area and the total traced areas, and finally multiplied by the series (10 or 12). The sampled area is the total number of analyzed counting frames multiplied with the counting frame area (1,875 μm2 (75 × 25 μm) for the GCL and hilus and 2,500 μm2 (50 × 50 μm) for the ML). The total number of vessels was quantified by counting the number of vessels that crossed any of the four borders of the counting frame (Figure 2A). This number was then multiplied by the fraction of the sampled and the traced areas, multiplied by the series. The diameter of all the counted vessels was measured using the
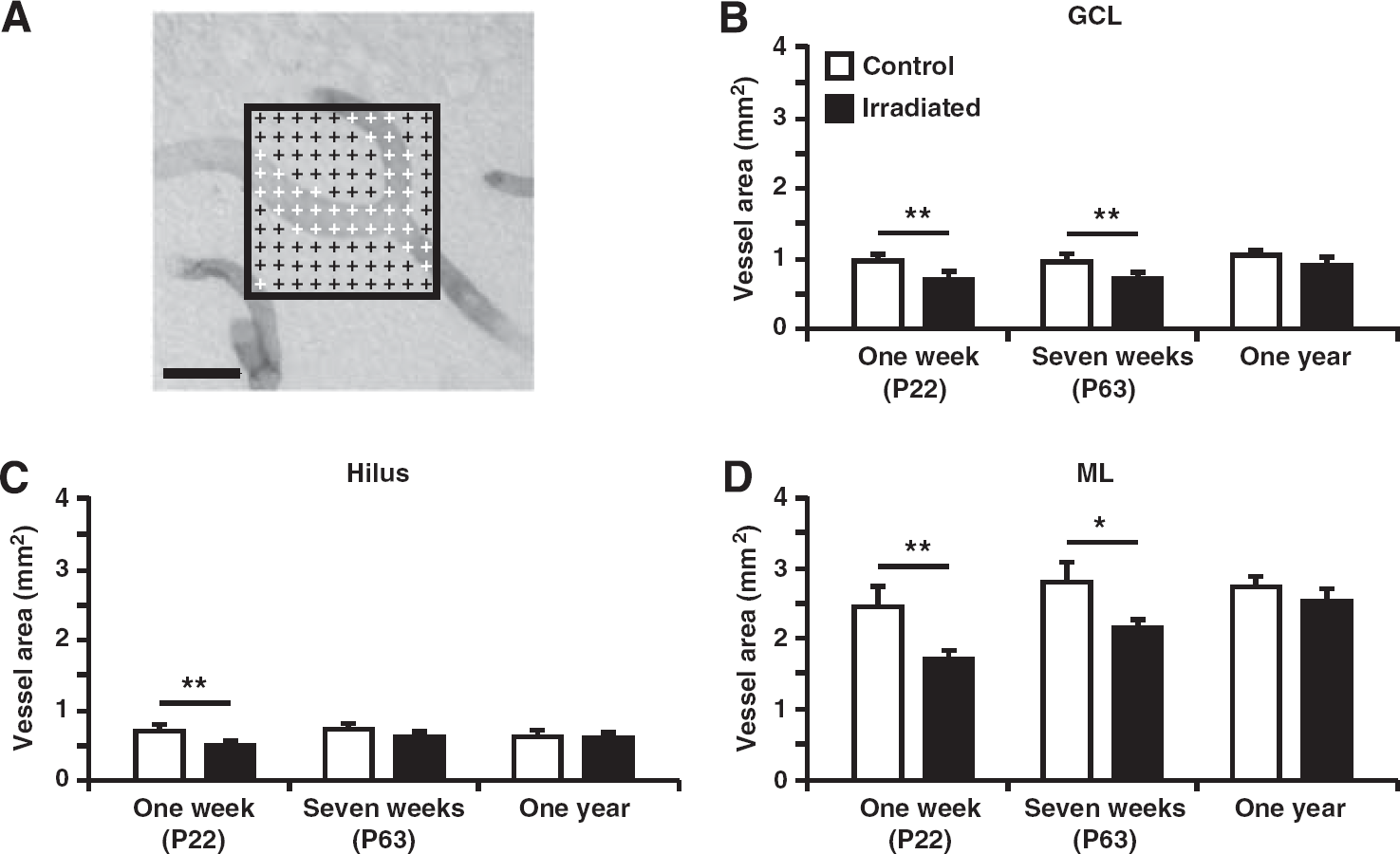
The total area covered by microvessels in the granule cell layer (GCL), hilus, and molecular layer (ML) of the dentate gyrus. (
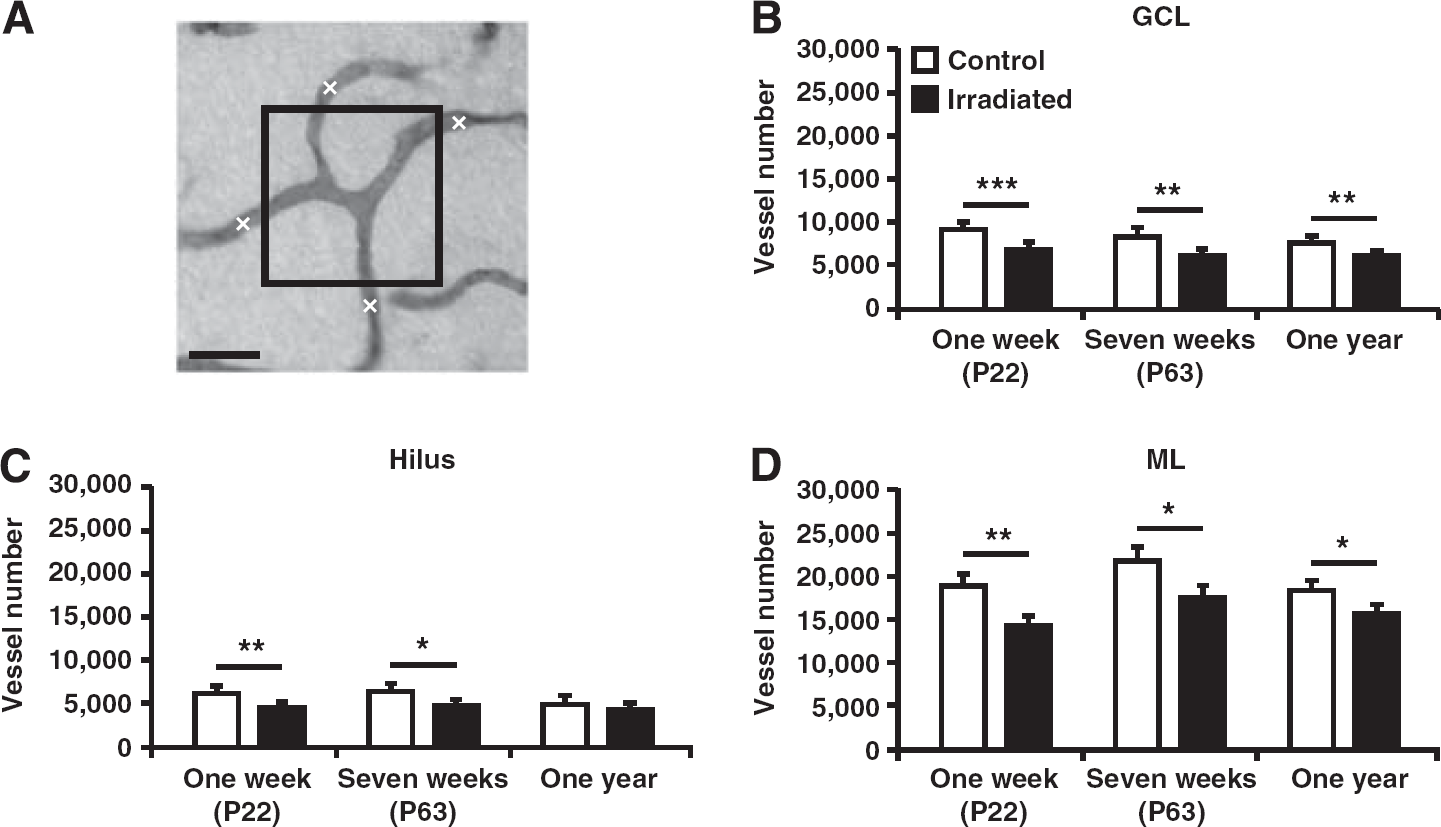
Quantification of the number of microvessels in the granule cell layer (GCL), hilus, and molecular layer (ML) of the dentate gyrus. (
Quantification of Neuronal Precursor Cells
Stereo Investigator and the previously mentioned Leica microscope were used to trace the GCL at × 10 magnification and count the DCX-positive cells in the GCL plus two cell layers (one cell diameter defined as 6 μm) into the hilus at × 40 magnification. The number of counted cells was then multiplied with the series to obtain the total number of cells.
Cell Death Analysis
To evaluate whether irradiation induced endothelial cell death, sections from 6 hours after irradiation where scanned with a confocal Leica TCS SP2 microscope at × 20 magnification to visualize cells double-positive for CD31 and active caspase-3 in the GCL, hilus, and ML. Active caspase-3-positive cells were further magnified and examined in their full z-dimension. The analysis was performed on every 12th section, with an average of four to five analyzed sections per hippocampus.
Analysis of the Neurovascular Niche
A confocal Leica TCS SP2 microscope was used to measure the distance between BLBP-positive neural stem cells and CD31-positive microvessels. Confocal images were acquired of a minimum of 40 cells per subgranular zone and sections for analysis were picked randomly. The distance was measured in the
Statistical Analysis
All data are presented as mean ± s.e.m. The data were compared using independent samples
RESULTS
Irradiation Altered Microvessel Structure and Complexity in Both Neurogenic and Nonneurogenic Hippocampal Areas
The main focus of this study was to investigate how microvessels in both neurogenic (i.e., the GCL) and nonneurogenic (i.e., the hilus and the ML) hippocampal areas respond to whole-brain irradiation. At the age of P14, mice were irradiated with a single dose of 8 Gy. Microvessels were then histologically visualized through a staining with cluster of differentiation 31 (CD31, also called PECAM-1), which is present in the intercellular junctions of both human and murine endothelial cells where it contributes to cell adhesion.20,21
First, the total area covered by microvessels was quantified (Figure 1A). A reduction in the total microvessel area was found in irradiated mice compared with control mice in all investigated structures with an overall decrease of 25–30% 1 week after irradiation. In the GCL, a decrease was observed 1 week (0.92 ± 0.05 versus 0.70 ± 0.04 mm2;
Second, we found that irradiation led to a decrease in the total number of microvessels (Figure 2A). This was seen in both the neurogenic (Figure 2B) and the nonneurogenic areas (Figures 2C and 2D). The reduction in the GCL persisted from 1 week (9,218 ± 362 versus 6,920 ± 263;
Third, the diameter of the microvessels (capillaries) was measured and a smaller microvessel diameter was observed in the hilus of irradiated mice compared with control mice 1 week after irradiation (4.80 ± 0.06 in controls versus 4.28 ± 0.13 μm in irradiated mice;
To evaluate whether irradiation affected microvessel complexity,
22
we quantified the total number of branching points (Figure 3A). Irradiated mice had a reduced number of branching points compared with control mice at all three time points in the GCL (1 week; 617 ± 42 versus 502 ± 17;
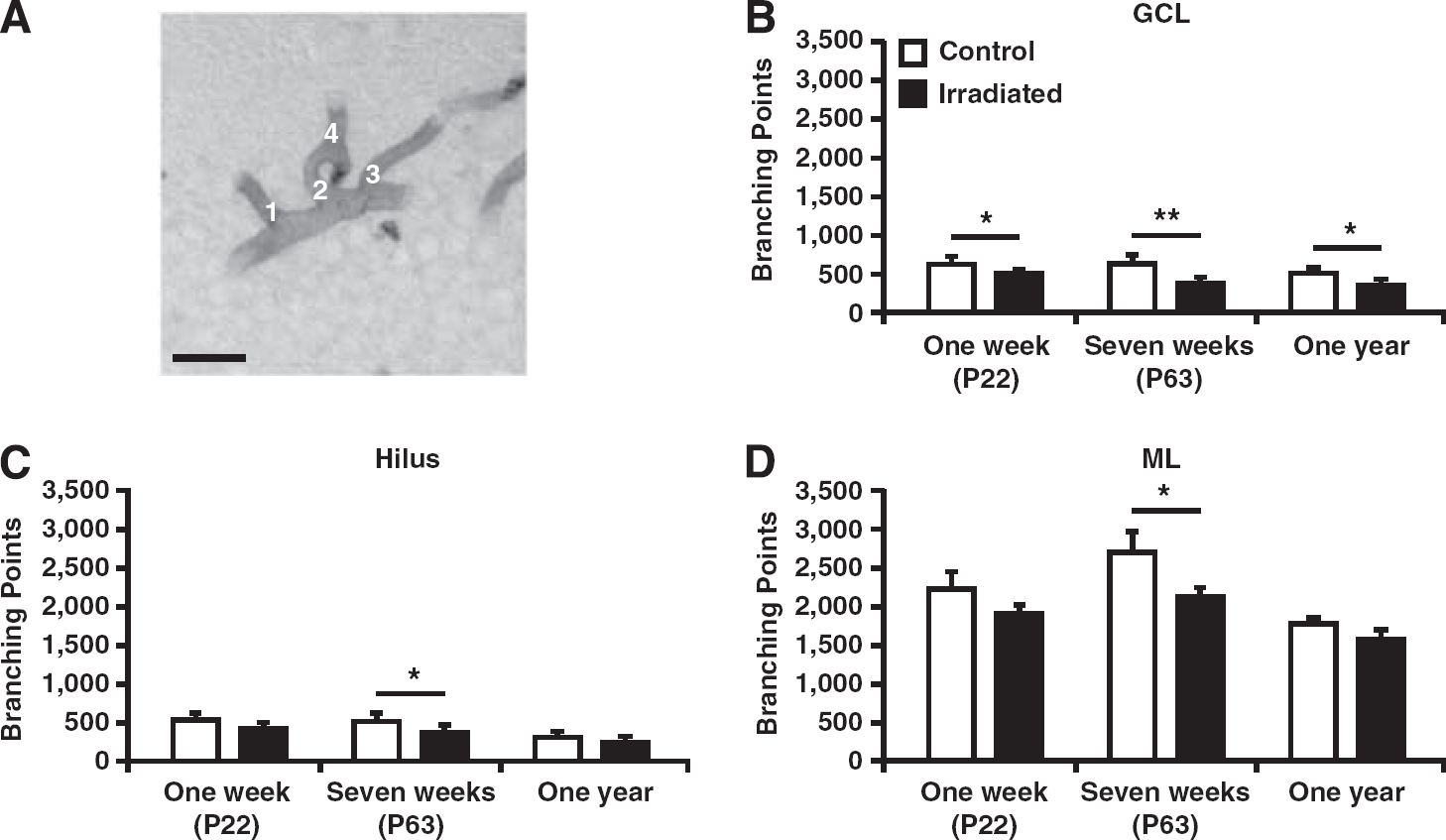
All branching points were counted in the granule cell layer (GCL), hilus, and molecular layer (ML) of the dentate gyrus. (
Persistent Volume Changes in the Hippocampal Granule Cell Layer and Molecular Layer up to 1 year After Irradiation
Alterations in brain growth after irradiation to the developing brain have been demonstrated earlier23–25 and might have negative effects on hippocampal structure and function. We therefore assessed if irradiation affected the total volumes of the GCL, hilus, or ML.
Acutely, 6 hours after irradiation, no differences were observed between irradiated and control mice in any subregion (left panel, Figures 4B–4D). A smaller GCL volume was found at all other investigated time points (1 week; 0.21 ± 0.01 versus 0.14 ± 0.006 mm3;
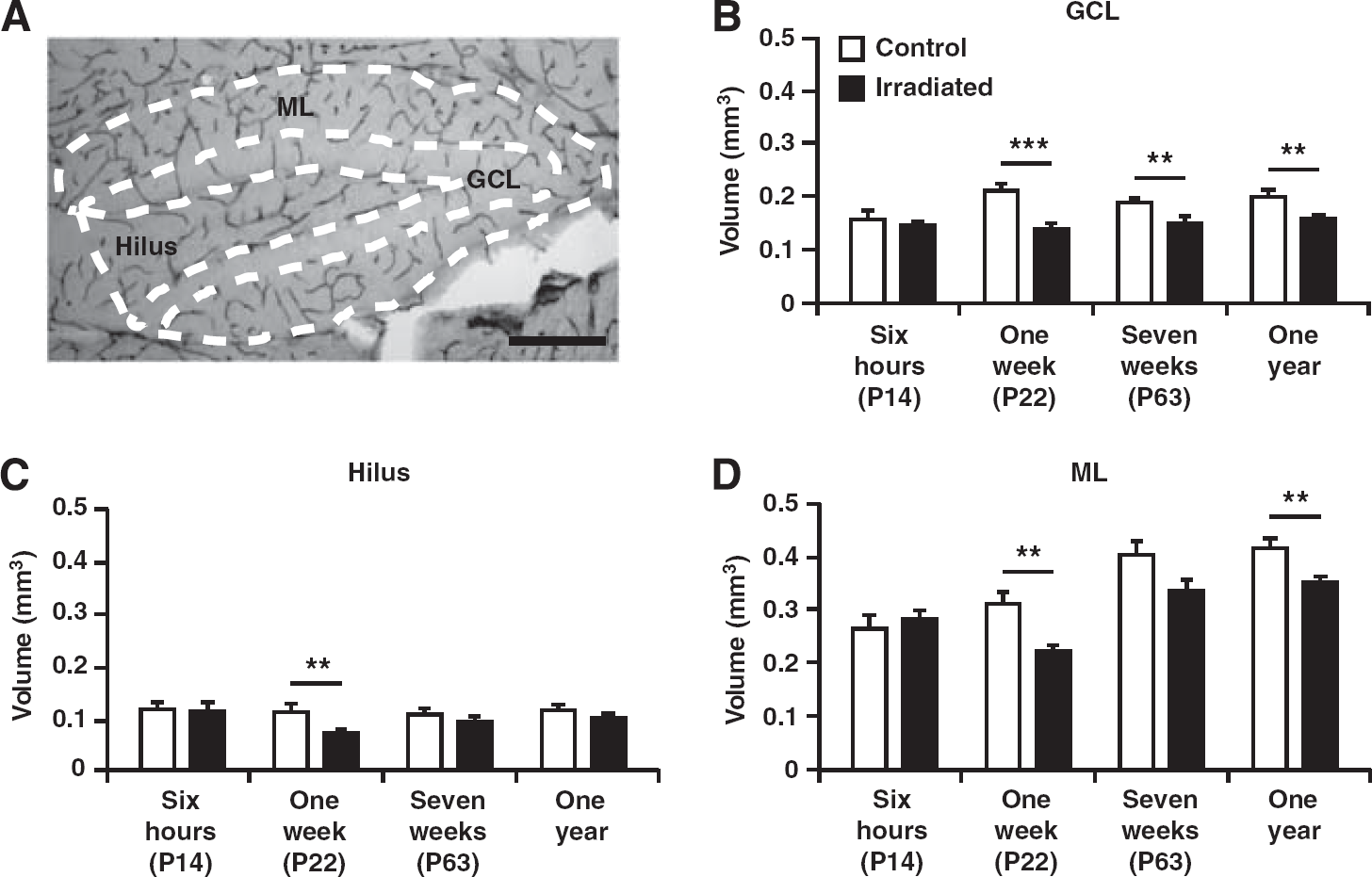
Volume of the granule cell layer (GCL), hilus, and molecular layer (ML) of the dentate gyrus. (
Microvessel Density Increased Early and Transiently in Irradiated Brains
As all quantifications of microvessel parameters and volume measurements followed the same trend, that is, the reduction in irradiated compared with control mice diminished with time, we also wanted to calculate the densities. To our surprise, we found that irradiated animals displayed a significantly higher density of branching points in the GCL 1 week after irradiation, followed by a reduced density of branching points in irradiated animals at 7 weeks after irradiation (Table 1A). Also in the hilus, irradiated animals displayed an increased density of branching points 1 week after irradiation, followed by a decreased density of microvessels 7 weeks after irradiation in irradiated animals (Table 1B). Similarly, irradiated animals in the ML displayed an increased density of branching points at 1 week after irradiation (Table 1C). At 1 year after irradiation, no differences were observed between control and irradiated mice for any of the microvessel density parameters.
Densities (number/mm3) for the vessel number and number of branching points presented in the GCL (A), hilus (B), and ML (C) 1 week, 7 weeks, and 1 year after irradiation

Abbreviations: GCL, granule cell layer; ML, molecular layer. Postnatal day (P),
Neurogenesis Was Decreased both Short-Term and Long-Term After Irradiation
After analyzing the structure and complexity of the vasculature, we sought to verify the detrimental effects of irradiation on neurogenesis, as shown in earlier studies.25,26 Sections were stained for DCX to identify immature neuronal precursor cells and the total number of DCX-positive cells in the GCL including the subgranular zone (SGZ) was quantified. Irradiation drastically decreased the number of DCX-positive cells 1 week (28,297 ± 1,201 versus 6,520 ± 1,054;
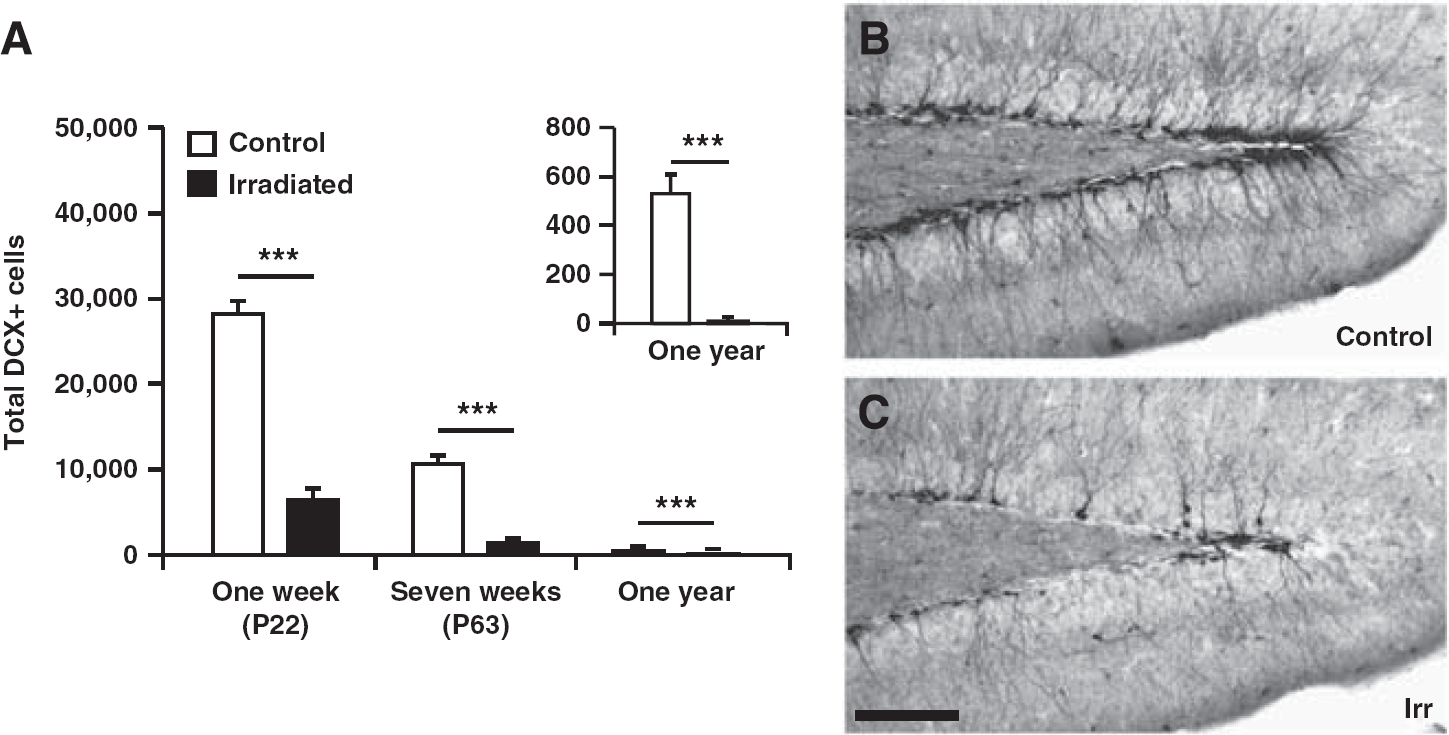
(
Irradiation to the Young Brain Did Not Alter the Neurovascular Niche
To address what role microvessels may have in the support and maintenance of hippocampal neurogenesis and if this neurovascular niche is affected by irradiation to the young brain, we measured the distance between undifferentiated neural stem cells and the nearest microvessel.
16
The distance to the nearest microvessel (CD31-positive structure) from at least 40 stem cells (BLBP-positive) per SGZ was measured 7 weeks after irradiation (Figure 6A). The average distance was not different between controls and irradiated brains (8.47 ± 0.59 versus 9.96 ± 0.44 μm;
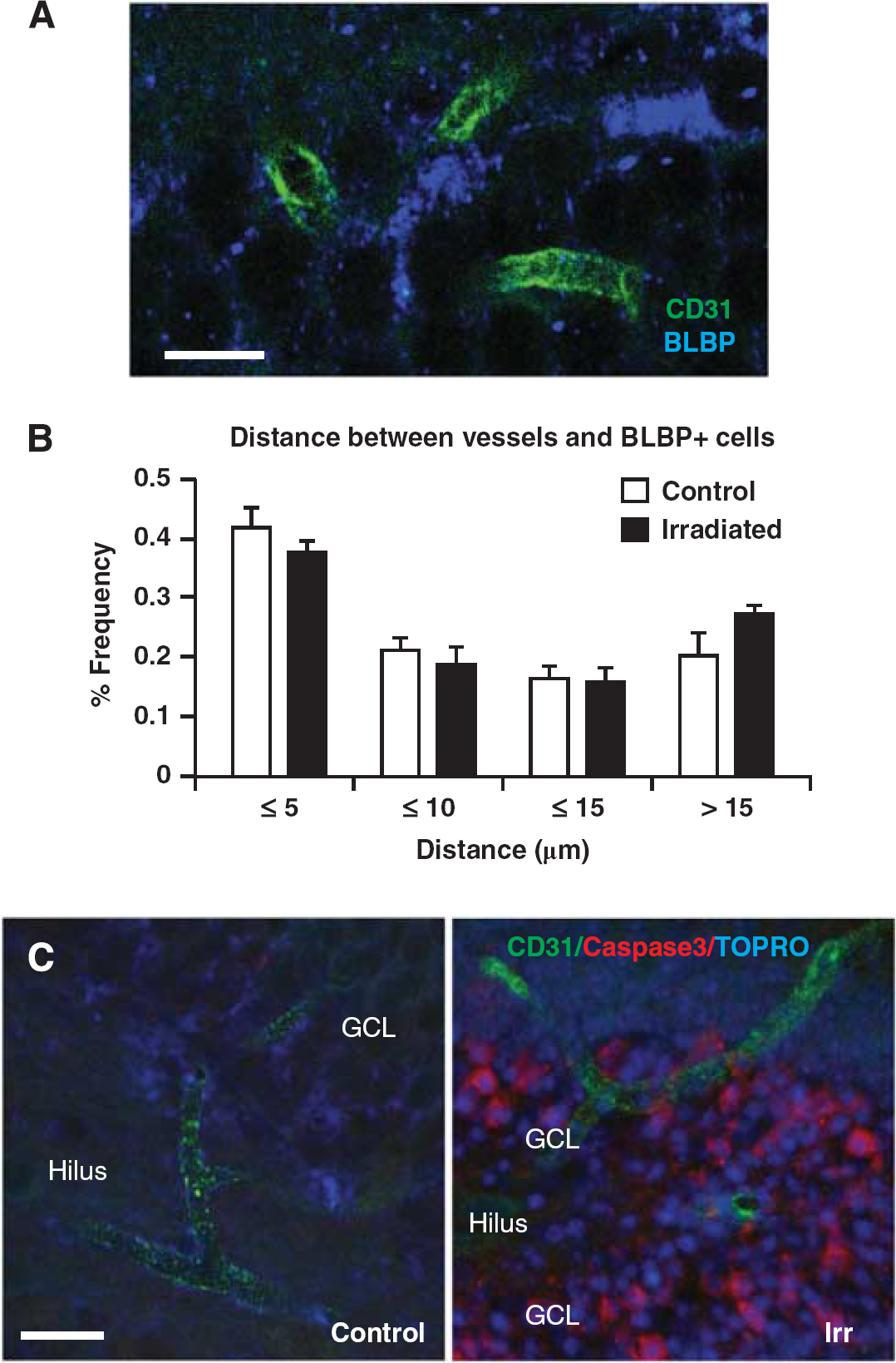
Irradiation to the young brain did not disrupt the vascular niche or cause acute endothelial cell death. (
DISCUSSION
In the present study we demonstrated that a single radiation dose of 8 Gy to the young, still developing mouse brain reduced microvessel area, number of microvessels and the number of microvessel branching points in the hippocampus at three different time points after irradiation. Early (1 week) after irradiation, the absolute numbers of microvessels and branching points were decreased. However, the densities of microvessel branching points were increased at this time point, indicating that the surrounding neural and glial tissue initially was more affected than the vascular tissue. At the later time points, the densities were gradually normalized, demonstrating the microvasculature's capacity to adapt to the surrounding tissue. We also confirmed and extended previous studies reporting that irradiation reduces the pool of neural progenitor cells and leads to a persistently decreased level of neurogenesis.16,23,25,27,28 In addition, we verify both the natural decline in hippocampal neurogenesis with age, 29 as well as clearly demonstrate the detrimental effect of irradiation on immature neurons, such that neurogenesis deteriorated more rapidly after irradiation and showed no signs of recovery up to 1 year after irradiation.
Irradiation Caused Growth Arrest During Normal Development of the Brain
Our results show significant volume differences in the GCL and ML up to 1 year after irradiation. A general brain volume loss in children, 9 as well as specifically smaller hippocampal volumes in both rats 24 and mice25,30 have previously been reported following irradiation. In humans, as well as in rodents, the brain, and particularly the hippocampus, continues to grow postnatally and shows an exponential growth in early childhood until a plateau phase and maximal brain volume is reached at adolescence.31,32 Hence, irradiation in this model of juvenile mice targets the brain in a phase when it is still continuously developing and growing. The smaller volume observed after irradiation is therefore most likely explained by arrested growth, resulting in reduced volumes in irradiated brains compared with control brains. 26 Cell death may also contribute to the smaller volumes through an acute loss of cells,24,30 and the corresponding volume. This is most evident in areas where postnatal neurogenesis occurs. Here, we demonstrate that also neighboring, nonneurogenic regions (the hilus and the ML), where no overt cell death occurs, display growth retardation. Irradiation has previously been demonstrated to cause a more profound effect on hippocampal volume in the young (immature) brain, as compared with irradiation to the brains of older animals. 23 In addition, the relative differences between irradiated and control brains in the current study decreased with time, indicating a partial volume recovery (Figure 4).
The Vasculature Adjusted to the Surrounding Neural Tissue
When calculating the densities for the blood vessel parameters, we found that irradiated animals had a significantly higher density of branching points in all three regions investigated 1 week after irradiation. This suggests that even though the total volume of the vascular bed was reduced, the surrounding tissue displayed an even more pronounced volume loss and growth arrest. However, 7 weeks after irradiation, all densities except branching points in the GCL and microvessels in the hilus, were normalized compared with controls, indicating that the vasculature had in fact adjusted to the new environmental conditions at this time point. This was confirmed by the latest time point, 1 year after irradiation, when all vascular densities were normalized. Taken together, this indicates that the surrounding tissue regulates the dynamics of the vasculature. In contrast, Warrington
Irradiation and the Neurovascular Niche
Neurogenesis has previously been shown to be persistently reduced in the hippocampus after irradiation.13,23,26–28 We found a robust decrease in the number of neuronally committed progenitors in the hippocampal GCL at all three time points investigated. Furthermore, the difference between irradiated and control mice increased with time. Monje
Implications of the Study
Survivors of childhood leukemia and brain tumors, particularly those with brain tumors treated with radiation therapy at doses of ≥30 Gy, are at an increased risk of stroke due to subsequent vasculopathies with the risk of stroke being dose-related.38,39 Compared with unaffected siblings, the relative risk of stroke is almost 10 times higher in survivors of all childhood cancers, 40 but the underlying mechanisms are poorly understood. Our research group has previously demonstrated that the immature and still growing rat hippocampus is more susceptible to irradiation, 23 and it has been proposed that even very low doses of irradiation during infancy may produce cognitive deficits that persist into adulthood. 41 It is therefore important to investigate the acute and late effects of radiation to the brain. Specifically, understanding the roles that blood vessels play in intrinsic support and maintenance of stem cell populations may provide novel ways to improve the hippocampal microenvironment and could thereby provide a neurobiological basis for protective strategies after irradiation to the young brain. If we can mitigate the adverse side-effects of radiation therapy in the increasing number of survivors of childhood cancer, we could improve their quality of life.
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
Footnotes
ACKNOWLEDGEMENTS
The authors are grateful to the skillful technical assistance of Rita Grandér. The authors thank Michelle Porritt for fruitful input. The funding agencies had no influence on the study design.
