Abstract
Resveratrol may be a powerful way of protecting the brain against a wide variety of stress and injury. Recently, it has been proposed that resveratrol not only reduces brain injury but also promotes recovery after stroke. But the underlying mechanisms are unclear. Here, we tested the hypothesis that resveratrol promotes angiogenesis in cerebral endothelial cells and dissected the signaling pathways involved. Treatment of cerebral endothelial cells with resveratrol promoted proliferation, migration, and tube formation in Matrigel assays. Consistent with these pro-angiogenic responses, resveratrol altered endothelial morphology resulting in cytoskeletal rearrangements of β-catenin and VE-cadherin. These effects of resveratrol were accompanied by activation of phosphoinositide 3 kinase (PI3-K)/Akt and Mitogen-Activated Protein Kinase (MAPK)/ERK signaling pathways that led to endothelial nitric oxide synthase upregulation and increased nitric oxide (NO) levels. Subsequently, elevated NO signaling increased vascular endothelial growth factor and matrix metalloproteinase levels. Sequential blockade of these signaling steps prevented resveratrol-induced angiogenesis in cerebral endothelial cells. These findings provide a mechanistic basis for the potential use of resveratrol as a candidate therapy to promote angiogenesis and neurovascular recovery after stroke.
Introduction
It is increasingly recognized that neurovascular responses have a critical role in the ability of the brain to recover after stroke. Signals and substrates of neurogenesis and neuroplasticity are tightly coregulated with angiogenesis and vascular remodeling (Arai et al, 2009). This may not be surprising since the fundamental mechanisms of neurogenesis and angiogenesis are evolutionarily conserved so that similar mediators are involved in both phenomena (Carmeliet and Tessier-Lavigne, 2005). After stroke, newborn neuroblasts migrate along perivascular routes (Thored et al, 2007) and promotion of angiogenesis enhances neurogenesis and functional recovery (Ohab et al, 2006; Taguchi et al, 2004). Neurovascular responses have a central role as the damaged central nervous system transitions from initial injury into repair (Lo, 2010). Hence, therapies that enhance neurovascular remodeling should provide new opportunities for stroke recovery (Xiong et al, 2010).
Accumulating data suggest that resveratrol is a potent neuroprotectant against stroke (Dong et al, 2008; Sakata et al, 2010; Shin et al, 2010; Sun et al, 2010). The underlying mechanisms are pleiotropic, including modulation of sirtuin signaling, upregulation of pro-survival mediators such as FOXO and MnSOD, dampening cell death signals such as bax and caspases (Della-Morte et al, 2009; Pallàs et al, 2009). More recently, however, it has been proposed that besides acute neuroprotection, resveratrol may also have beneficial effects on stroke recovery (Dong et al, 2008). But the mechanisms that might mediate these phenomenon remain unclear.
In the present study, we test the hypothesis that resveratrol can promote angiogenesis in cerebral endothelial cells. Our findings suggest that resveratrol activates phosphoinositide 3 kinase (PI3-K)/Akt and Mitogen-Activated Protein Kinase (MAPK)/ERK signaling pathways that upregulate endothelial nitric oxide synthase (eNOS) and increases extracellular levels of vascular endothelial growth factor (VEGF) and matrix metalloproteinases (MMPs). These data suggest that resveratrol may be a candidate therapy for stroke recovery by promoting angiogenesis via a programmed NO/VEGF/MMP signaling axis.
Materials and methods
Reagents and Cells
Resveratrol (> 99%) was purchased from Sigma-Aldrich (St Louis, MO, USA). A stock solution of resveratrol was made in dimethyl sulfoxide at a concentration of 10 mM. The following pharmacologic agents were used: MEK inhibitor—U0126 (Calbiochem, San Diego, CA, USA), a PI3-K inhibitor—LY294002 (Calbiochem), VEGF antagonist (sFlt-1) (Calbiochem), VEGF receptor blocker—SU1498 (Calbiochem). A human cerebral microvascular endothelial cell line (Callahan et al, 2004) was seeded at 60% to 70% confluence and kept at 37°C in 5% CO2. Culture media comprised RPMI 1640 containing 10% fetal bovine serum, 10% Nu-Serum, 2 mM
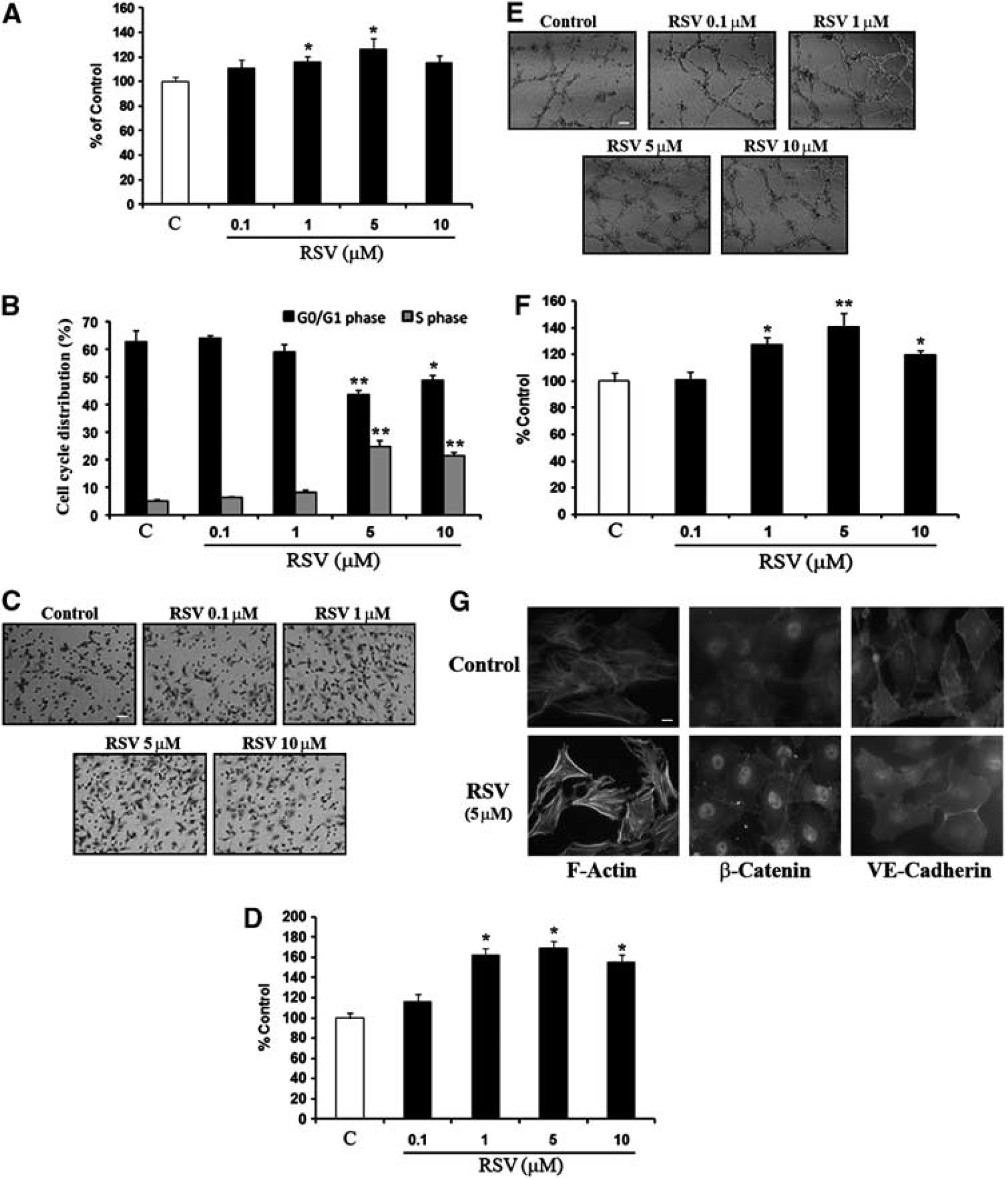
Resveratrol stimulates brain endothelial cell proliferation, migration, and tube formation. (
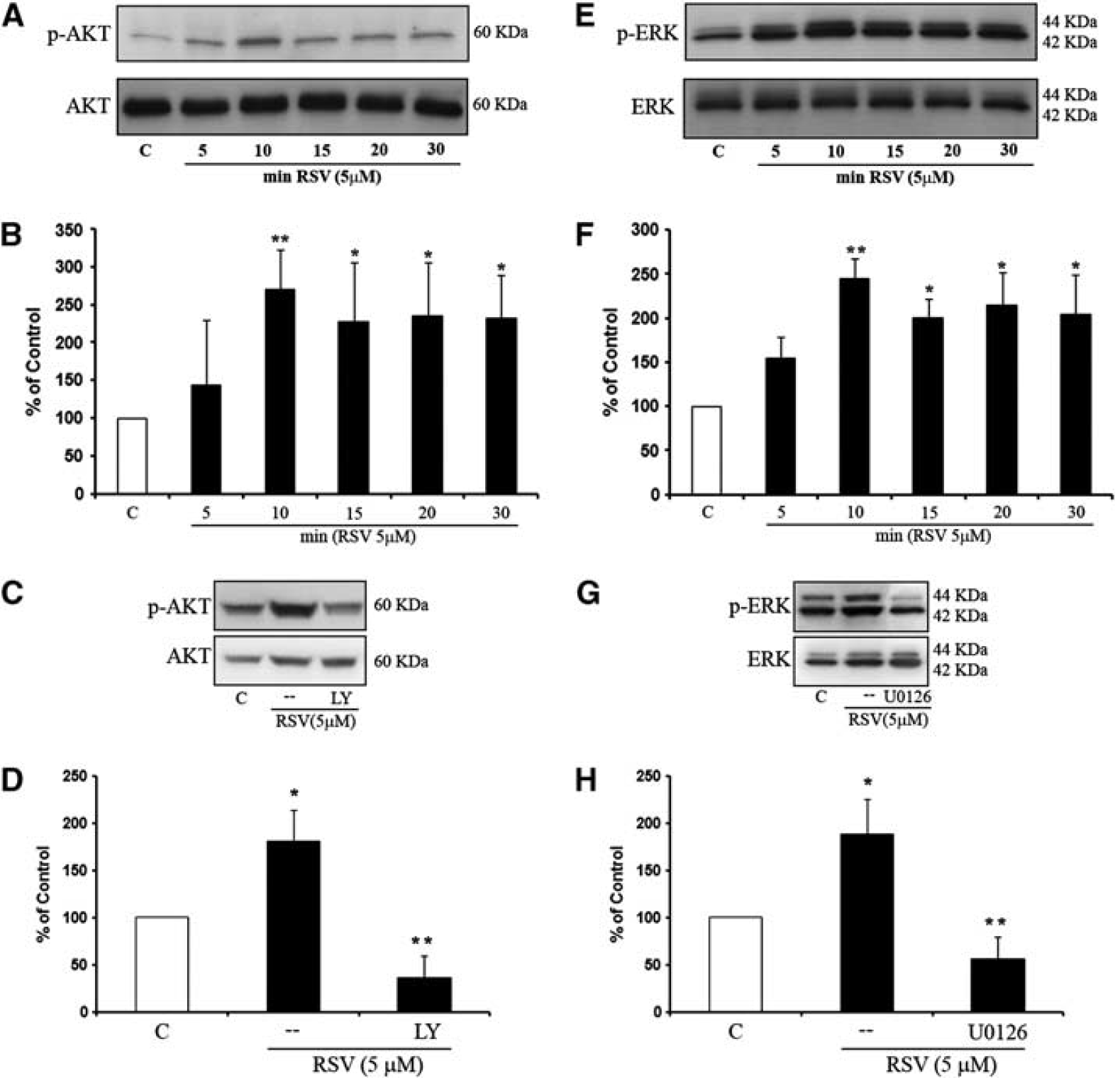
Regulation of phosphoinositide 3-Kinase (PI3-K) and Mitogen-Activated Protein Kinase (MAPK) signaling pathway by resveratrol. (
Proliferation Assay
For in vitro proliferation assays, brain endothelial cells were seeded into 96-well (1 × 105 cells/well) flat bottom plates in triplicate with medium alone (control) or medium containing different concentrations of recombinant resveratrol (0.1 to 10 μM). Cell proliferation was determined with the WST proliferation assay kit (Dojindo, Japan). Briefly, serum-starved cells were treated with resveratrol (0.1 to 10 μM) for 24 hours. Following resveratrol treatment, 10 μL WST reagent was added to 100 μL fresh culture medium in each well. Absorbance was recorded at 490 nm (EL800, Bio-Tek Instruments, Winooski, VT, USA), and calculated as a ratio against untreated cells.
Sodium Dodecyl Sulfate Polyacrylamide Gel Electrophoresis Zymography
Conditioned media were collected and concentrated via centricon (Millipore, Bedford, MA, USA). Equal amounts were mixed with 2 × sample buffer and loaded on a 10% polyacrylamide gel incorporated with 0.1% gelatin for electrophoresis. The MMP-2 and MMP-9 zymographic standards were used as positive controls (Chemicon, Bedford, MA, USA). After electrophoresis, gels were renaturated and then incubated with developing buffer at 37°C for 72 hours, and stained for 1 hour with 0.1% Coomassie blue. Gelatinolytic activity was quantified via densitometry.
Matrigel Tube Formation Assay for Angiogenesis
The standard Matrigel assay was used to assess the spontaneous formation of capillary-like structures in vitro. Cells (8 × 104 cells/well) were seeded in 24-well plates in serum-free media previously coated with Growth Factor-Reduced Matrigel Matrix (BD Bioscience, San Jose, CA, USA), and incubated at 37°C for 18 hours. The degree of tube formation was determined by counting the number of tubes in four random fields from each well. Data were analyzed as tube percentage versus untreated control wells.
Endothelial Cell Migration Assay
Cells (1 × 105) in RPMI containing 0.5% FBS were plated onto the upper transwell chamber (6.5 mm, 8 μM pore Corning Costar, Cambridge, MA, USA) coated with 0.1% collagen Type I (BD Bioscience). The bottom chamber contained 700 μL of medium (serum free) alone or medium containing different concentrations of resveratrol. The cells were incubated for 18 hours at 37°C and cells that did not pass through the membrane pores were removed. Migrated cells were stained with crystal violet and counted in five random fields (× 200).
Nitric Oxide and Vascular Endothelial Growth Factor Assays
Nitric oxide concentrations in conditioned media were determined using the QuantiChrom Nitric Oxide Assay Kit (BioAssay Systems, Hayward, CA, USA). Vascular endothelial growth factor levels in supernatants were determined using a human VEGF ELISA kit (R&D System, Minneapolis, MN, USA). Known concentrations of VEGF (0 to 500 pg/mL) were used to generate standard curves.
Immunoblotting
Cells were lysed in lysis buffer (Cell Signaling Technology, Danvers, MA, USA) and clarified, and protein concentration was determined with the Bradford assay (Bio-Rad, Hercules, CA, USA). Total cell lysates (50 to 100 μg per lane) were separated in precast 4% to 12% NuPage gels (Invitrogen, Carlsbad, CA, USA), and proteins were transferred to nitrocellulose membranes (Invitrogen). After blocking with 0.2% I-block (Tropix, Applied Biosystem, CA, USA), membranes were incubated overnight at 4°C with primary antibodies, and 1 hour at room temperature with horseradish peroxidase-conjugated secondary antibodies (Amersham, Piscataway, NJ, USA). The immune complexes were visualized by enhanced chemiluminescence (Amersham). All immunoblots were repeated for at least three independent experiments.
Statistical Analysis
All of the experiments were performed in duplicate or triplicate, and repeated three to four times independently. Quantitative data were expressed as mean ± s.d. and analyzed with analysis of variance followed by Tukey's test for multiple comparisons. Differences of P < were considered significant. All experiments were performed under an institutionally approved protocol.
Results
Resveratrol Augments Proliferation, Migration, and Tube Formation
Cerebral endothelial cells were incubated with different concentrations of resveratrol (0.1 to 10 μM) over 24 hours. Proliferation, migration, and matrigel tube formation assays were examined as in vitro markers of angiogenesis. Resveratrol induced a dose-dependent increase in endothelial proliferation (Figure 1A). Flow cytometry analysis confirmed that resveratrol induced a significant increase in the proliferative S phase while decreasing the resting G0/G1 phase of the cell cycle (Figure 1B). Along with increased proliferation, resveratrol also enhanced endothelial cell migration as quantified with a transwell assay (Figures 1C and 1D). Matrigel assays showed that resveratrol induced tube formation in a dose-dependent manner (Figures 1E and 1F). In all proliferation, migration, and tube formation assays, the most effective concentrations of resveratrol appeared to peak around 5 μM. Similar results were obtained with a rat brain endothelial cell line RBE4 (Supplementary Figure 2).
Immunostaining confirmed that resveratrol induced morphological responses that were consistent with a pro-angiogenic phenotype in cerebral endothelial cells. In control untreated cells, normal resting state profiles were observed with F-actin mostly found in unorganized fibers and membrane structures, β-catenin localized to the cytoplasm, and VE-cadherin localized at intercellular junctions (Figure 1G). After resveratrol treatments, cytoskeletal rearrangements were observed with edge ruffling and F-actin stress fiber formation, relocation of β-catenin to the nucleus, and movement of VE-cadherin from membrane junctions into the cytoplasm (Figure 1G).
Resveratrol Upregulates Nitric Oxide via PI3-Kinase/Akt and MAPK/ERK Signaling
Resveratrol robustly activated PI3-kinase signaling in cerebral endothelial cells, leading to a rapid increase in phospho-Akt levels (Figures 2A and 2B). Activation of this pathway was significantly blocked by the specific PI3-kinase inhibitor LY294002 (Figures 2C and 2D). Additionally, the ERK MAP kinase pathway was also triggered by resveratrol, with clear increases in phospho-ERK levels (Figures 2E and 2F). ERK signaling was pharmacologically confirmed with the MEK/ERK inhibitor U0126, which significantly lowered resveratrol-induced phospho-ERK levels (Figures 2G and 2H).
Since Akt and ERK pathways are known to be related to NO signaling, we next examined the role of eNOS in resveratrol-induced angiogenesis. Incubation of cerebral endothelial cells with resveratrol rapidly increased phosphorylation of eNOS (Figures 3A and 3B). Activation of eNOS occurs downstream of PI3-K/Akt and Mitogen-Activated Protein Kinase/ERK signaling since blocking these pathways with LY294002 or U0126 significantly decreased resveratrol-induced phospho-eNOS levels (Figures 3C and 3D). Consistent with the increased phosphorylation of eNOS, resveratrol amplified NO levels in endothelial-conditioned media (Figure 3E). Cotreatment with the NOS inhibitor
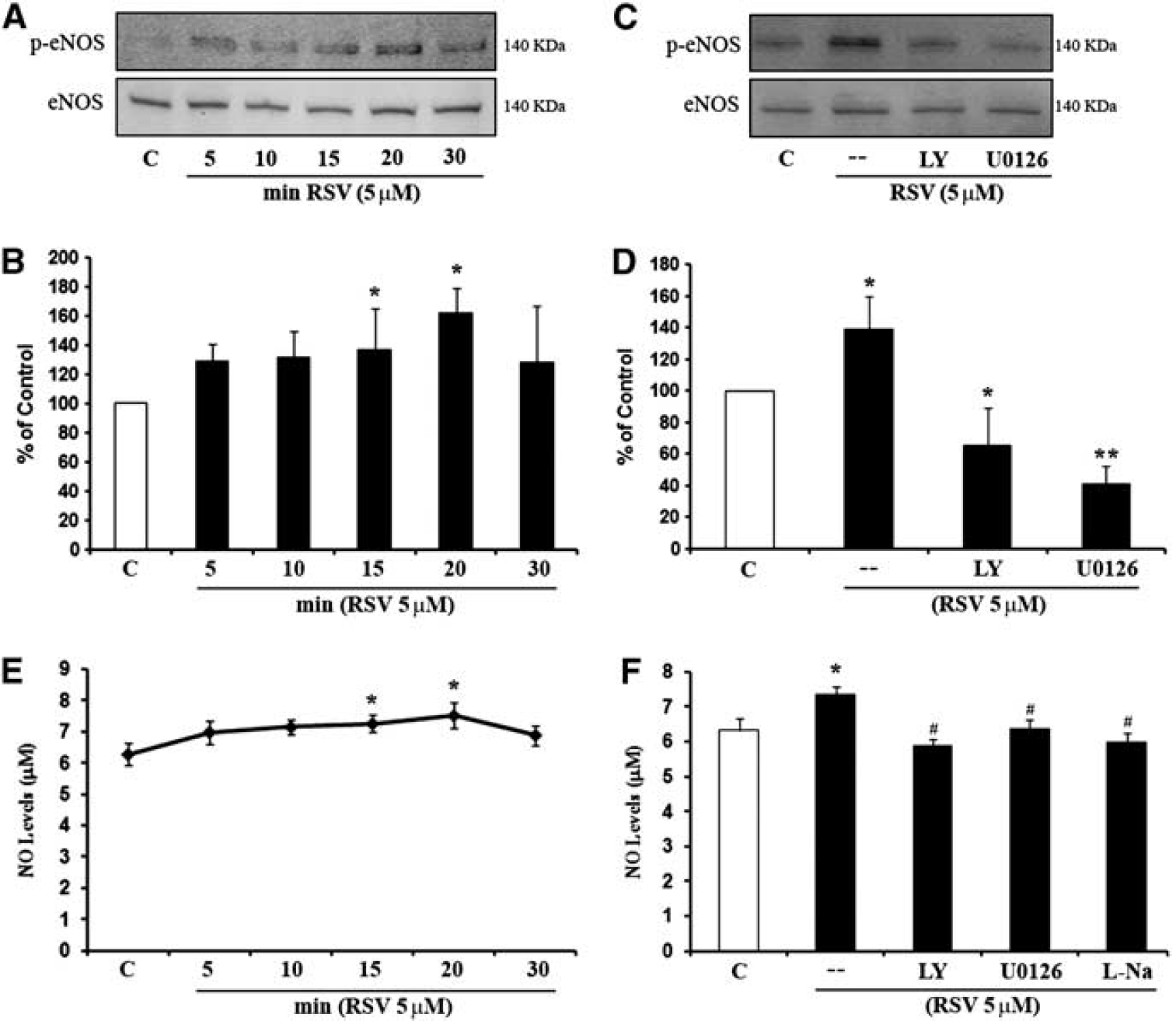
Endothelial nitric oxide synthase (eNOS) is regulated by resveratrol and downstream of phosphoinositide 3-Kinase (PI3-K) and Mitogen-Activated Protein Kinase (MAPK) signaling pathway. (
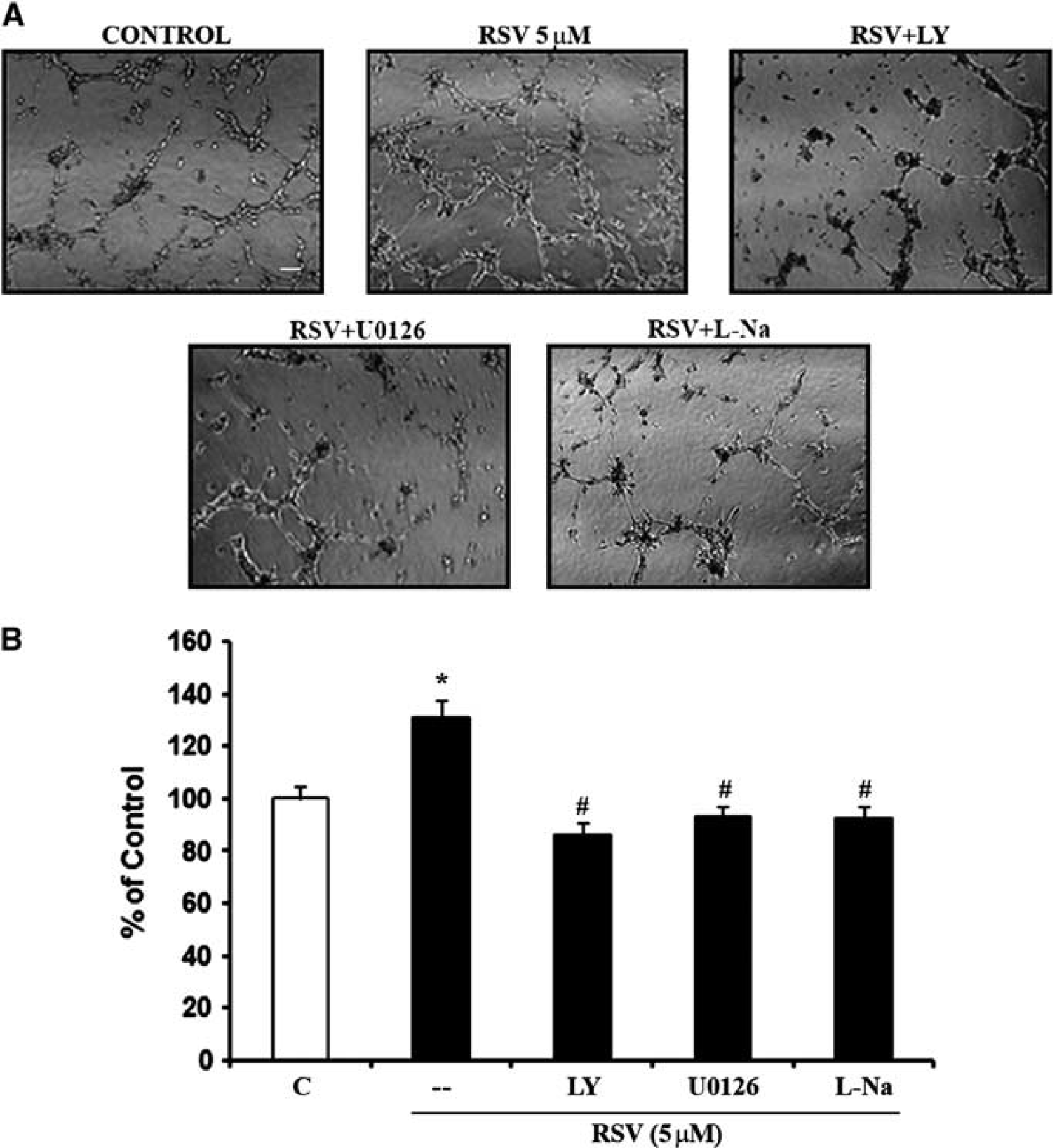
Involvement of phosphoinositide 3-Kinase (PI3-K), Mitogen-Activated Protein Kinase (MAPK), and nitric oxide synthase (NOS) signaling pathway in resveratrol-induced capillary tube formation. (
Resveratrol Increases Vascular Endothelial Growth Factor and Matrix Metalloproteinases Via Nitric Oxide-Dependent Signaling
To directly link resveratrol-induced signaling with angiogenesis, we assessed the well-established pro-angiogenic mediator VEGF. Immunostaining (Figure 5A) and Western blots (Figures 5B and 5C) showed that VEGF expression in cerebral endothelial cells was strongly upregulated by resveratrol. This effect was dependent on NO. Inhibition of NOS with
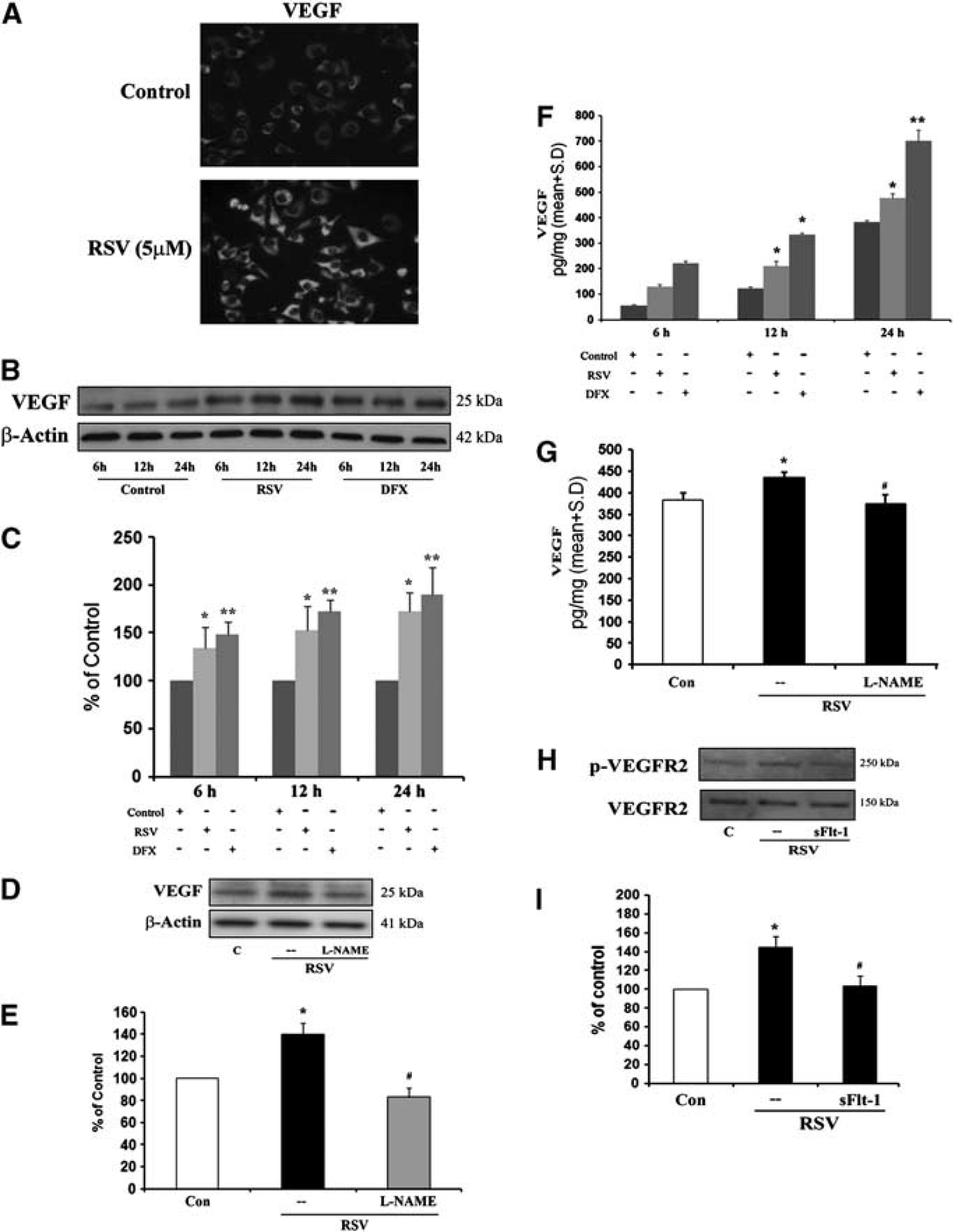
Vascular endothelial growth factor (VEGF) induced by resveratrol is nitric oxide synthase (NOS) dependent. (
Cellular upregulation of VEGF was accompanied by a release of protein into extracellular space. Vascular endothelial growth factor levels in endothelial-conditioned media were clearly increased by resveratrol (Figure 5F). Extracellular VEGF release was also NO dependent since
Consistent with elevated protein levels, activation of VEGF signaling was detected in resveratrol-treated endothelial cells. Levels of the phosphorylated receptor phospho-VEGFR2 was increased by resveratrol, indicating that active signaling was indeed taking place (Figures 5H and 5I). These pathways involved autocrine signaling since blockade of the VEGF with sFlt-1 dampened the ability of resveratrol to activate the VEGF pathway and phosphorylate VEGFR2 (Figures 5H and 5I).
Besides VEGF, another major class of pro-agiogenic mediators comprise MMPs. Exposure of cerebral endothelial cells to resveratrol upregulated both MMP-2 and MMP-9 (Figure 6A). Induction of MMPs was also dependent on NO since the inhibitor
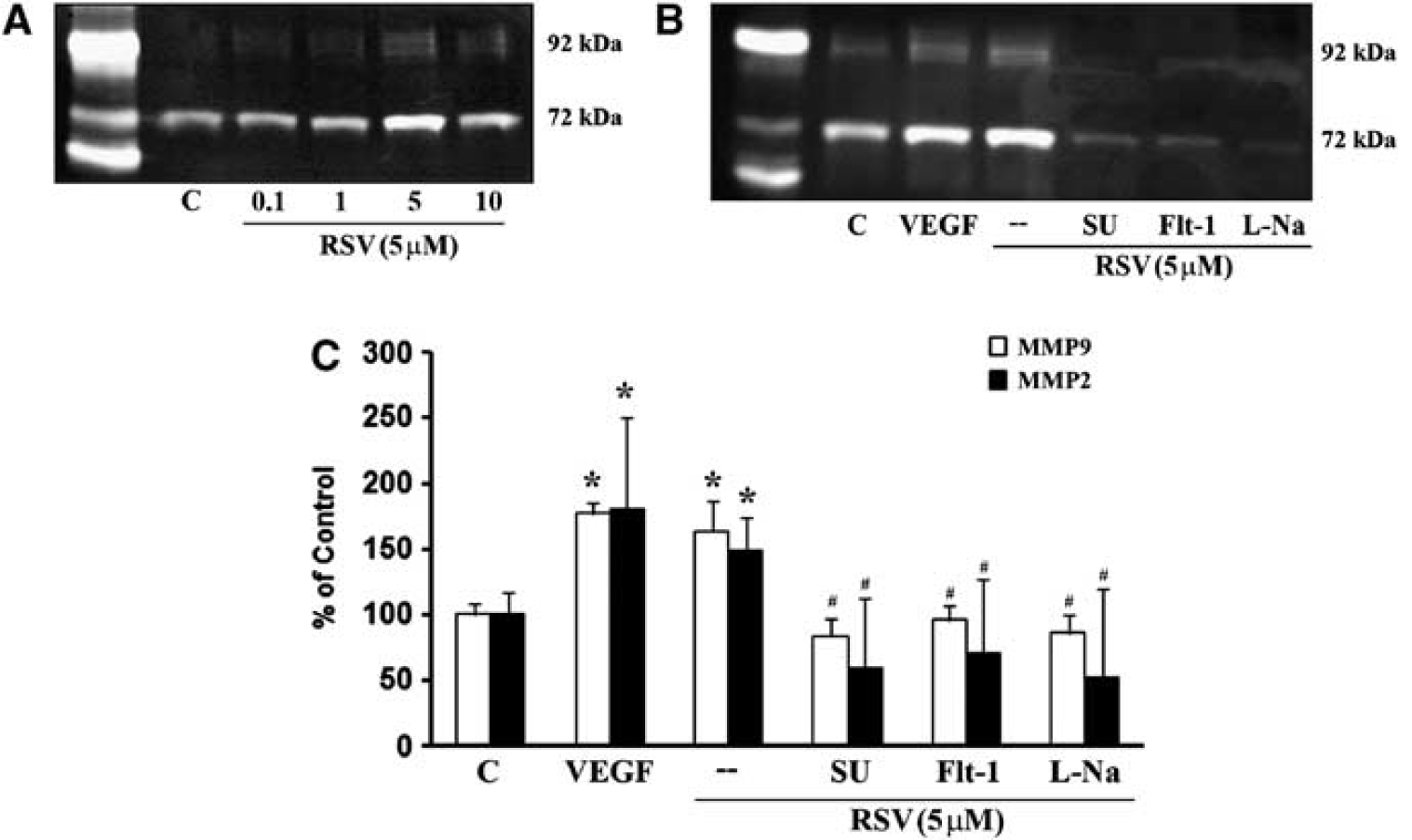
Effect of resveratrol on matrix metalloproteinase (MMP)-2 and MMP-9 secretion. Gelatin zymograph analysis (
Crosstalk between VEGF and MMP signaling may exist. Exposing cerebral endothelial cells to VEGF directly upregulated MMP levels (Figures 6B and 6C). And the ability of resveratrol to induce MMPs can be prevented by either antagonizing VEGF with sFlt-1 or a blocking the VEGFR2 receptor with SU1498 (Figures 6B and 6C). Taken together, these data demonstrated that resveratrol strongly activates both VEGF and MMP pathways.
Resveratrol-Induced Angiogenesis Is Dependent on Vascular Endothelial Growth Factor and Matrix Metalloproteinases
Next, we asked whether resveratrol-induced angiogenesis in cerebral endothelial cells was indeed dependent on the control of VEGF and MMP mechanisms. As expected, resveratrol increased tube formation in matrigel assays. Blocking either VEGF or MMP signaling potently suppressed these resveratrol-induced effects. Cotreatments with the VEGF antagonist sFlt-1, or the VEGFR2 blocker SU1498, or the broad spectrum MMP inhibitor GM6001, all significantly decreased resveratrol-induced tube formation (Figures 7A and 7B).
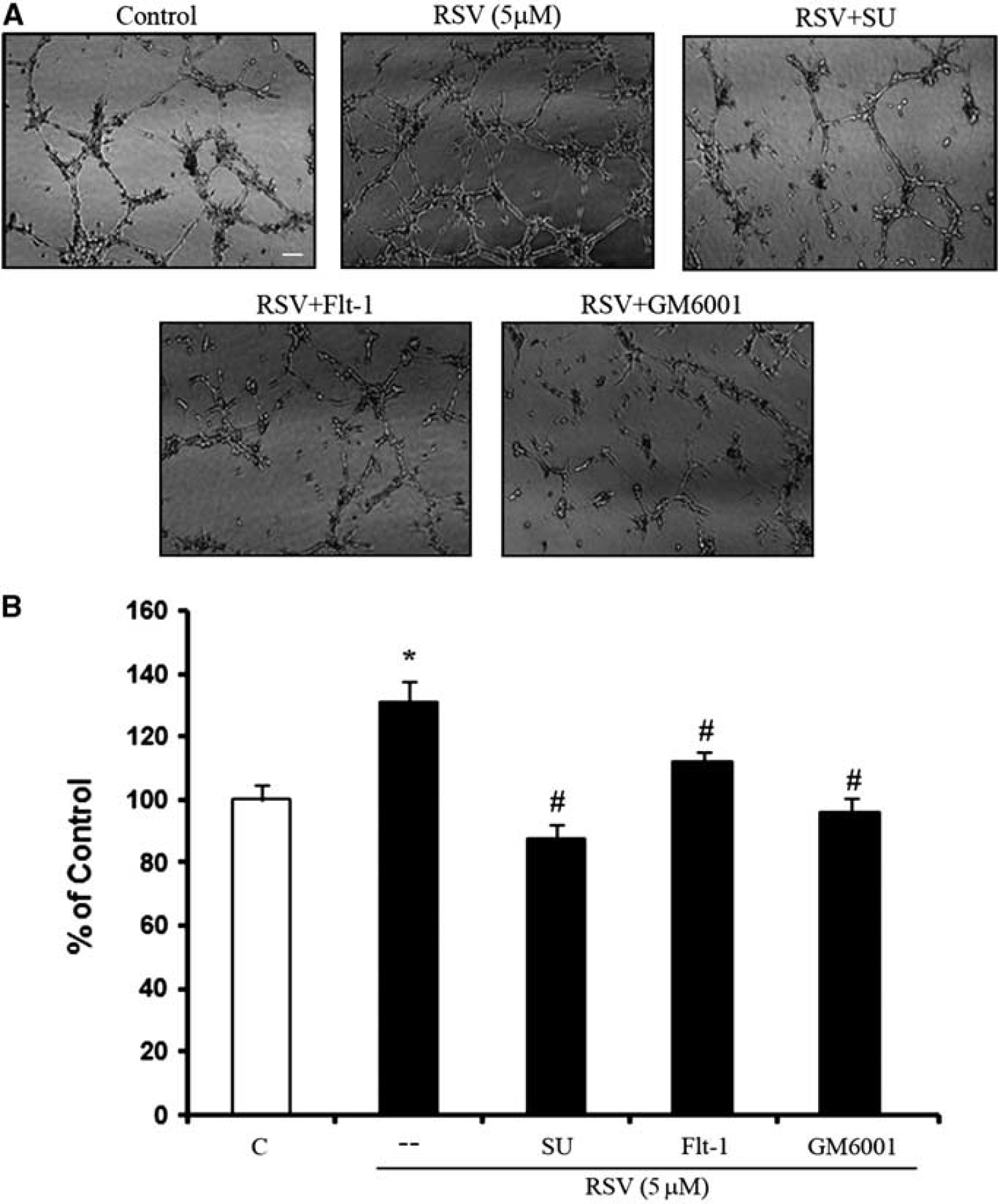
Involvement of vascular endothelial growth factor (VEGF), VEGFR2, and matrix metalloproteinases (MMPs) signaling pathway in resveratrol-induced capillary tube formation. (
Discussion
Resveratrol is a potent neuroprotectant that has been shown to reduce neuronal death in many experimental models of stroke and brain injury (Dong et al, 2008; Sakata et al, 2010; Shin et al, 2010; Sun et al, 2010). But emerging data now suggest that resveratrol may also be used to promote recovery after stroke (Dong et al, 2008). The major finding of the present study is that resveratrol can promote angiogenesis in cerebral endothelial cells. The mechanisms of this phenomenon appear to involve upstream control of NO via PI3-kinase/Akt and ERK signaling, and downstream induction of VEGF and MMPs (Figure 8). These data provide a mechanistic basis for the potential application of resveratrol as a candidate therapy for neurovascular repair.
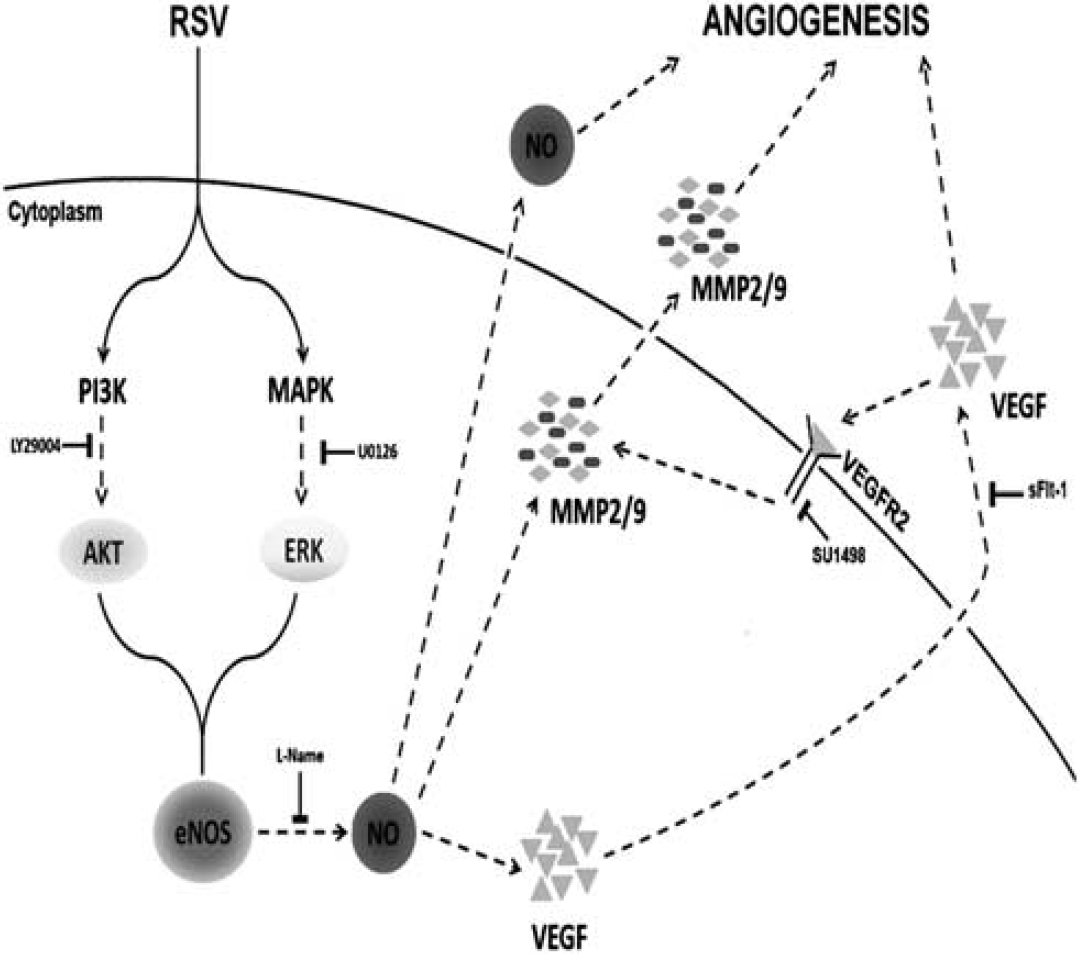
A schematic diagram showing the proposed mechanism by which intracellular signaling pathway involved in resveratrol-induced brain endothelial cell angiogenesis.
Although several groups have previously reported generation of NO by resveratrol (Andriambeloson et al, 1997; Bruder et al, 2001; Hsieh et al, 1999; Hung et al, 2000; Wallerath et al, 2002); the precise signaling pathways are not well established. Resveratrol upregulated eNOS expression and NO generation in Human Umbilical Vein Endothelial Cells (HUVEC) (Wallerath et al, 2002). But in our study, the major effects of resveratrol appeared to take place via phosphorylation rather than absolute alterations of protein levels. Endothelial nitric oxide synthase was constitutively expressed in cerebral endothelial cells but the basal level of phosphorylation was minimal. Resveratrol increased ser1177 phosphorylation of eNOS within minutes. Ser1177 phosphorylation is critical for eNOS activity (Fulton et al, 1999). In our experiments, resveratrol-induced eNOS phosphorylation led to prompt generation of NO. Of note, this response was rapid but transient so downstream effectors may be required to extend the biologic effect.
To dissect how resveratrol regulates eNOS phosphorylation, we assessed two major kinase pathways, PI3-kinase and ERK MAP kinase. Incubation of endothelial cells with the PI3-kinase inhibitor LY294002 or the MEK/ERK inhibitor U0126, both abrogated eNOS phosphorylation induced by resveratrol. It is interesting to note that activation of PI3-kinase and ERK are required for angiogenesis, since either PI3-kinase or ERK inhibitors reduced resveratrol-induced tube formation. Functionally, the balance between these two signals may allow fine-tuning of eNOS activation, and the redundancy of the two signaling pathways may ensure robust NO generation. Collectively, our observations highlight the importance of PI3-kinase/Akt and ERK pathways in resveratrol-induced NO production, and both appear to be necessary for cerebral endothelial angiogenesis.
It is well known that many important mediators execute the angiogenic program downstream of NO. Acting as a messenger molecule, NO mediates the majority of endothelium-dependent responses in the brain (Faraci and Heistad, 1998). Treatment of cells with NO donors increases VEGF (Zhang et al, 2003), and inhibitors of NO synthase such as
Besides VEGF, neurovascular proteolysis via MMPs represent another major mechanism for angiogenesis (Karagiannis and Popel, 2005; Moses, 1997; Segarra et al, 2005). To form new blood vessels, activated endothelial cells must degrade the basement membrane and remodel the extracellular matrix around neovascular sites. Our findings reveal that resveratrol directly upregulated MMP-2 and MMP-9 production in cerebral endothelial cells, whereas inhibition of MMPs significantly reduced resveratrol-mediated angiogenesis. Of course, the inhibitor used here, GM6001, is a broad spectrum agent. So we cannot exclude the possibility that besides MMP-2 and MMP-9, other MMPs may also be involved. Taken together, these findings support a causal link between resveratrol-induced MMP responses and angiogenesis in cerebral endothelial cells. Furthermore, MMPs are known to be regulated by a number of cytokines and growth factors (Moses, 1997). In our cerebral endothelial system, crosstalk between MMPs and VEGF was apparent. Inhibition of VEGF signaling potently suppressed resveratrol-induced MMPs, and prevented angiogenesis.
Taken together, our findings suggest that resveratrol may be a novel way to promote angiogenesis in cerebral endothelial cells. But there are several important caveats to keep in mind. First, although our data provide cellular and pharmacologic proof-of-principle for resveratrol in cerebral angiogenesis, in vivo validation of these mechanisms remain to be obtained. The pro-angiogenic utility of resveratrol as a potential stroke recovery therapy should be explored in future experiments. An important limitation, however, may be related to the poor bioavailability of oral resveratrol dosing that can only achieve 10 to 50 ng/mL levels in plasma. Our 5 μM concentrations here would correspond to almost 1 μg/mL levels. Second, our findings are only focused on endothelium. But it is now recognized that multicellular interactions between endothelial cells and astrocytes and pericytes are required for neurovascular homeostasis (Abbott et al, 2006). Further dissection of how resveratrol affects cell—cell signaling in the entire neurovascular unit is warranted. Third, although our data define downstream effects, we do not know what receptors of resverstrol might be involved. Resveratrol can activate a wide range of receptors including androgen/estrogen receptors, EGF receptors, and integrins etc. (Han et al, 2006). Fourth, our studies focused on VEGF and MMP mechanisms. But angiogenesis is known to recruit complex networks of genes and proteins (Muñoz-Chápuli et al, 2004). More powerful proteomic approaches may be required to fully catalog the effects of resveratrol on cerebral endothelium. Finally, our studies only examined the pro-angiogenic effects of resveratrol in normal cells. In the context of stroke, brain injury or neurodegeneration, future studies should ask how resveratrol may operate in the milieu of a damaged neurovascular system.
In summary, the present study provides mechanistic evidence that resveratrol promotes angiogenesis in cerebral endothelial cells via NO, VEGF, and MMP signaling. Further in vivo and clinical exploration of these pathways is warranted to validate these experimental findings and develop resveratrol as a potential neurovascular repair therapy for stroke and brain injury.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
