Abstract
Cerebral angiogenesis is an important adaptive response to hypoxia. As the αvβ3 integrin is induced on angiogenic vessels in the ischemic central nervous system (CNS), and the suggested angiogenic role for this integrin in other systems, it is important to determine whether the αvβ3 integrin is an important mediator of cerebral angiogenesis. αvβ3 integrin expression was examined in a model of cerebral hypoxia, in which mice were subject to hypoxia (8% O2) for 0, 4, 7, or 14 days. Immunofluorescence and western blot analysis revealed that in the hypoxic CNS, αvβ3 integrin was strongly induced on angiogenic brain endothelial cells (BEC), along with its ligand vitronectin. In the hypoxia model, β3 integrin-null mice showed no obvious defect in cerebral angiogenesis. However, early in the angiogenic process, BEC in these mice showed an increased mitotic index that correlated closely with increased α5 integrin expression.
Introduction
Cerebral angiogenesis is a well-described adaptive response to cerebral hypoxia (LaManna et al, 1992) or ischemia (Krupinski et al, 1994). When the brain is starved of oxygen, a molecular cascade is initiated that exerts it effect to promote new blood vessel growth. The precise sequence of events underlying this process has yet to be fully elucidated, though evidence suggests it includes the transcription factor hypoxia-inducible factor-1α, and the growth factors vascular endothelial growth factor (VEGF) and angiopoietin-2 (LaManna et al, 1998). Growth of new cerebral blood vessels has been reported in cerebral hypoxia and ischemia, both in animal models (Chen et al, 1994; Hayashi et al, 2003; Wei et al, 2001) and in the human brain (Krupinski et al, 1994). Following focal cerebral ischemia, growing capillaries are first seen 7 days after the insult, and are localized to the area immediately surrounding the ischemic core, or penumbra (Chen et al, 1994; Hayashi et al, 2003; Wei et al, 2001). Neural cells in the ischemic penumbra are hypoxic but still viable, suggesting that these cells could be rescued if adequate support is delivered. Thus, interventions that promote new vessel sprouting in the ischemic penumbra represent an attractive therapeutic target in the treatment of ischemic stroke, making it imperative to identify the key molecular mechanisms promoting angiogenesis in response to hypoxia/ischemia.
The major pathways that promote angiogenesis are well described. These include the growth factors VEGF (Millauer et al, 1993), basic fibroblast growth factor (Klein et al, 1997), transforming growth factor-β1 (Roberts et al, 1986), and the angiopoietins (Sato et al, 1995). The present working model suggests that in the hypoxic central nervous system (CNS), hypoxia triggers hypoxia inducible factor-1α production (Chavez et al, 2000), which leads to increased expression of VEGF (Kuo et al, 1999) and angiopoietin-2 (Pichiule and LaManna, 2002), leading to angiogenic sprouting.
Extracellular matrix (ECM) proteins have a pivotal role in regulating angiogenesis and vascular remodeling (Eliceiri and Cheresh, 2001; Stromblad and Cheresh, 1996). In particular, fibronectin and its cognate integrin receptors are essential for angiogenesis, as shown by a failure of angiogenesis in mice mutants lacking the fibronectin gene (George et al, 1993) or fibronectin-specific integrins (Yang et al, 1993, 1995). In cerebral angiogenesis, the precise role of ECM–integrin interactions has not been fully explored, though several lines of evidence suggest an important role. During development, angiogenic vessels are surrounded by a fibronectin-rich ECM, and in the postnatal CNS, angiogenesis is associated with a switch in the expression of ECM proteins and endothelial cell β1 integrins, from fibronectin to laminin-mediated pathways (Milner and Campbell, 2002a). In addition, we recently showed that in the adult hypoxic CNS, angiogenic vessels show marked upregulation of fibronectin and endothelial cell α5β1 integrin expression (Milner et al, 2008a). Previous studies have described induction of the αvβ3 integrin on cerebral microvessels after cerebral ischemia (Abumiya et al, 1999; Okada et al, 1996; Wei et al, 2001). Furthermore, retroviral delivery of the proangiogenic transcription factor HoxD3 to the CNS resulted in blood vessel sprouting and a concomitant induction of the αvβ3 integrin on angiogenic capillaries (Chen et al, 2004). Taken with our previous observation that fibronectin promotes angiogenic behavior in brain endothelial cells (BEC) through α5β1 and αvβ3 integrins (Wang and Milner, 2006), the data suggest that both these integrins may be important in cerebral angiogenesis. In this study, we aimed to test the hypothesis that the αvβ3 integrin plays an instructive role in cerebral angiogenesis, by (1) examining expression of the αvβ3 integrin and its ECM ligands in a mouse model of hypoxia-induced cerebral angiogenesis, and (2) examining the angiogenic response in β3 integrin-null mice.
Materials and methods
Animals
The studies described have been reviewed and approved by The Scripps Research Institute Institutional Animal Care and Use Committee. Wild-type C57Bl/6 and β3 integrin-null mice (backcrossed >10 times on the C57Bl/6 background) were maintained under pathogen-free conditions in the closed breeding colony of The Scripps Research Institute. Heterozygous β3 integrin-null mice were bred and offspring were genotyped using previously described protocols (Hodivala-Dilke et al, 1999), to generate homozygous β3 integrin-null (β3−/−) mice and wild-type littermate controls (β3+/+).
Chronic Hypoxia Model
β3 integrin-null or wild-type littermate control mice, 8 to 10 weeks of age, were housed 4 to a cage, and placed into a hypoxia chamber (Biospherix, Redfield, NY, USA), maintained at 8% oxygen for up to 14 days. Littermate controls of each strain were also kept in the same room under similar conditions except that they were kept at normal oxygen levels (normoxia) for the duration of the experiment. Every few days, the chamber was opened for cage cleaning and food and water replacement as needed.
Immunohistochemistry and Antibodies
Immunohistochemistry was performed as described previously (Milner and Campbell, 2002a) on 10 μm frozen sections of cold saline-perfused brains taken from mice subjected to either normoxia (control) or hypoxia for 4, 7, or 14 days. Each slide contained mouse brains representing the four different time points of hypoxia, to ensure consistent antibody incubation times across different time points. The following monoclonal antibodies were obtained from BD Pharmingen (La Jolla, CA, USA): rat monoclonal antibodies reactive for the integrin subunits α5 (clone 5H10-27; MFR5), αv (clone RMV-7), α6 (clone GoH3), CD31 (PECAM-1, clone MEC13.3), clone MECA-32, the hamster monoclonal antibody reactive for the β3 integrin subunit (clone 2C9.G2), and isotype control antibodies; rat anti-KLH (A110-2), and hamster anti-TNP-KLH (G235-1). The monoclonal anti-β1 integrin antibody (MB1/2) was obtained from Chemicon (Temecula, CA, USA). Rabbit polyclonal antibodies against the following proteins were used in this study: vitronectin (Molecular Innovation, Southfield, MI, USA), fibronectin (Sigma, St Louis, MO, USA), Ki67 (Vector Laboratories, Burlingame, CA, USA), α5 integrin (Chemicon), β3 integrin (Cell Signaling Technology, Danvers, MA, USA), VEGF (Santa Cruz Biotechnology, Santa Cruz, CA, USA), and β-actin (Neomarker, Fremont, CA, USA). The Cy3-conjugated secondary antibodies against rat IgG, mouse IgG, and rabbit IgG were all obtained from Jackson Immunoresearch (West Grove, PA, USA). Alexa Fluor 488-conjugated secondary anti-rabbit and anti-rat IgGs were obtained from Invitrogen (Carlsbad, CA, USA).
Quantification of the number of capillaries positive for the different antigens was performed by taking images using a × 20 objective on a Zeiss Axio Observer A1 microscope and Zeiss Axiocam MRC digital camera (Thornwood, NY, USA). These images were used to determine the number of positive events within this field of view. For each animal, several images of the brain stem and cerebral cortex were taken for each antigen, and the mean number of events was calculated. Each experiment was performed with three different animals per condition, and the results are expressed as the mean ± s.e.m. of the number of capillaries positive for each antigen per field of view. Statistical significance was assessed using the Student's
Western Blot Analysis
Western blot analysis was used to determine levels of the α5 and β3 integrins, vitronectin, and VEGF in brain lysates, as previously described (Milner et al, 2007). Within each brain sample, levels of these proteins were first normalized to the level of β-actin, and then expressed as the fold-increase over the level present within the brain of normoxic animals.
Brain Endothelial Cell Culture
Pure cultures of mouse BEC were obtained as described previously (Milner et al, 2008b), with the modification that puromycin (4 μg/mL; Alexis GmbH, Grunberg, Germany) was included in culture media between days 1 and 3 to remove contaminating cell types. Endothelial cell purity was >99% as determined by CD31 in flow cytometry. BEC were used only for the first passage.
Flow Cytometry
Integrin expression of β3 integrin-null or wild-type littermate control BEC was examined as described previously (Milner et al, 2008b). The fluorescent intensity of labeled cells was analyzed with a Becton Dickinson FACScan machine (San Diego, CA, USA), with 10,000 events recorded for each condition. For each integrin subunit, the mean fluorescent intensity (MFI) of β3 integrin-null BEC was compared with wild-type BEC. Each experiment was repeated three times and the data are expressed as the mean ± s.e.m. Statistical significance was assessed using Student's
Cell Adhesion Assays
Adhesion assays were performed as previously described (Milner and Campbell, 2002b). Adhesion was quantified by phase microscopy by counting all attached cells within five fields of view per condition. Within each experiment each condition was performed in duplicate; the results represent the mean ± s.e.m. of three experiments. Statistical significance was assessed using Student's
Proliferation Assays
Glass coverslips were coated with ECM substrates and BEC cultured until cells reached ∼50% confluence. BEC were then cultured overnight in the presence of 5-bromo-2-deoxyuridine (BrdU; Invitrogen), fixed in acid/alcohol, and analyzed for BrdU incorporation according to the manufacturer's instructions. BrdU-positive cells were expressed as the percentage of total cells (Hoechst staining), and the results expressed as the mean ± s.e.m. of three experiments. Statistical significance was assessed using Student's
Results
Cerebral Hypoxia Induced β3 Integrin Expression on BEC
To examine whether BEC expression of the αvβ3 integrin is regulated during angiogenesis in the adult CNS, we examined these events in a mouse model of chronic hypoxia, in which exposure to mild hypoxia (8% O2) induces a strong angiogenic response in the CNS (Kanaan et al, 2006; LaManna et al, 1992). Four groups of mice were studied: normoxia (control), or hypoxia for 4, 7, or 14 days (
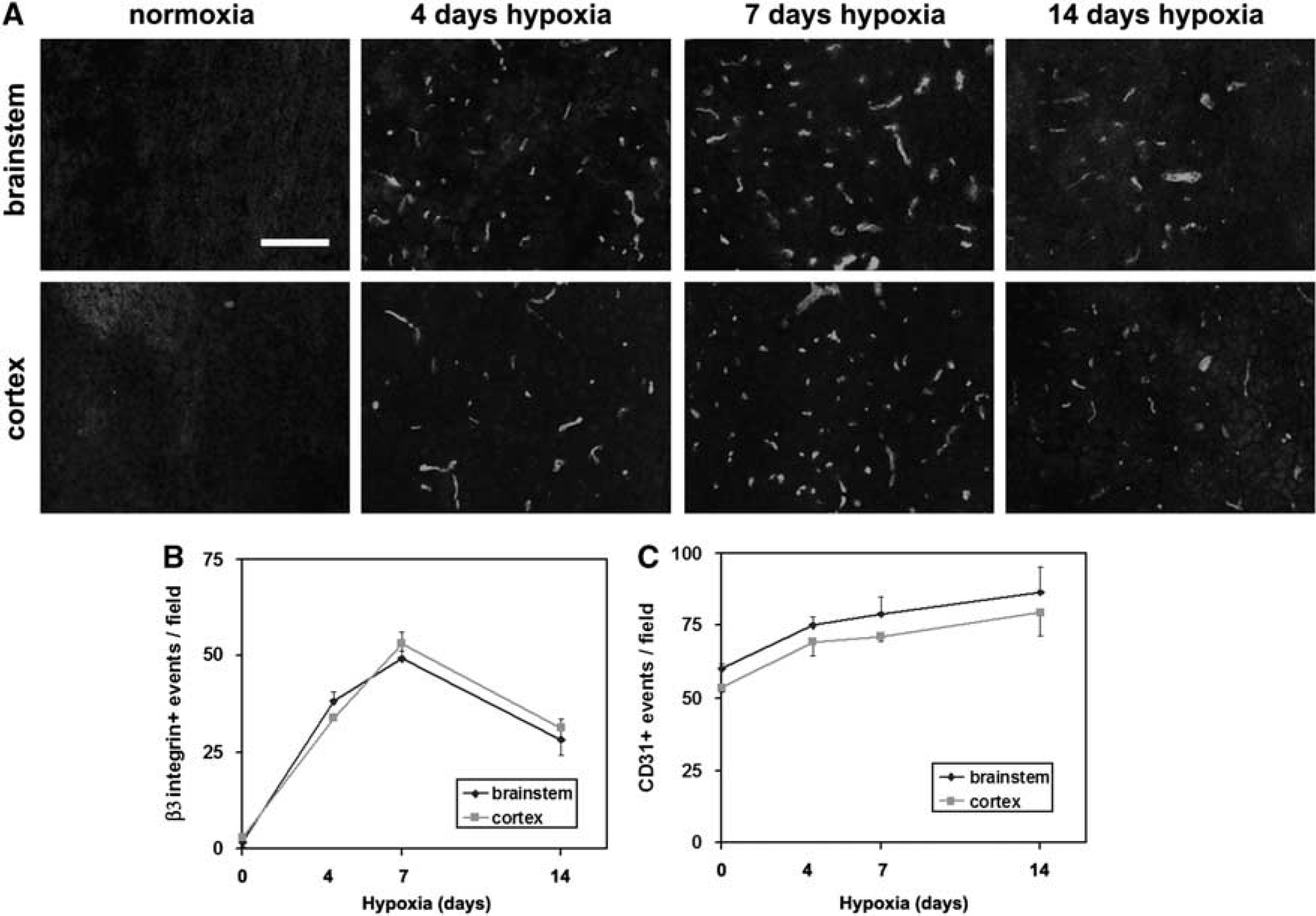
Hypoxic induction of β3 integrin expression on cerebral blood vessels. (
In the Hypoxic CNS, the αvβ3 Integrin is Expressed by Angiogenic BEC
Close examination of the number of blood vessels positive for CD31 or β3 integrin suggested that at any one time, only a fraction of the capillaries express β3 integrin. To confirm this relationship, we performed dual-color IF for CD31 and β3 integrin in the 4-day hypoxic CNS (Figure 2A). This revealed two points: first, that colocalization of CD31 and β3 integrin identifies BEC as the cells expressing β3 integrin, and second, not all CD31-positive structures are β3 integrin positive, showing that the αvβ3 integrin is expressed by only a subset of capillaries in the hypoxic CNS. The MECA-32 antigen is expressed by endothelial cells in the CNS up to embryonic day 15, and then switched off. After this time it cannot be detected on BEC, implying that MECA-32 labels only immature BEC (Hallman et al, 1995). Previously, we showed that MECA-32 is absent in the normoxic adult brain, and then re-expressed briefly after hypoxia (peaked at 4 days hypoxia), consistent with the idea that MECA-32 can be used as a marker of angiogenic immature BEC (Milner et al, 2008a). To examine whether angiogenic capillaries in the hypoxic CNS express the β3 integrin, we next performed dual-IF staining for MECA-32 and β3 integrin. As shown in Figure 2B, all capillaries expressing MECA-32 antigen also expressed β3 integrin, suggesting that angiogenic capillaries specifically induce the αvβ3 integrin.
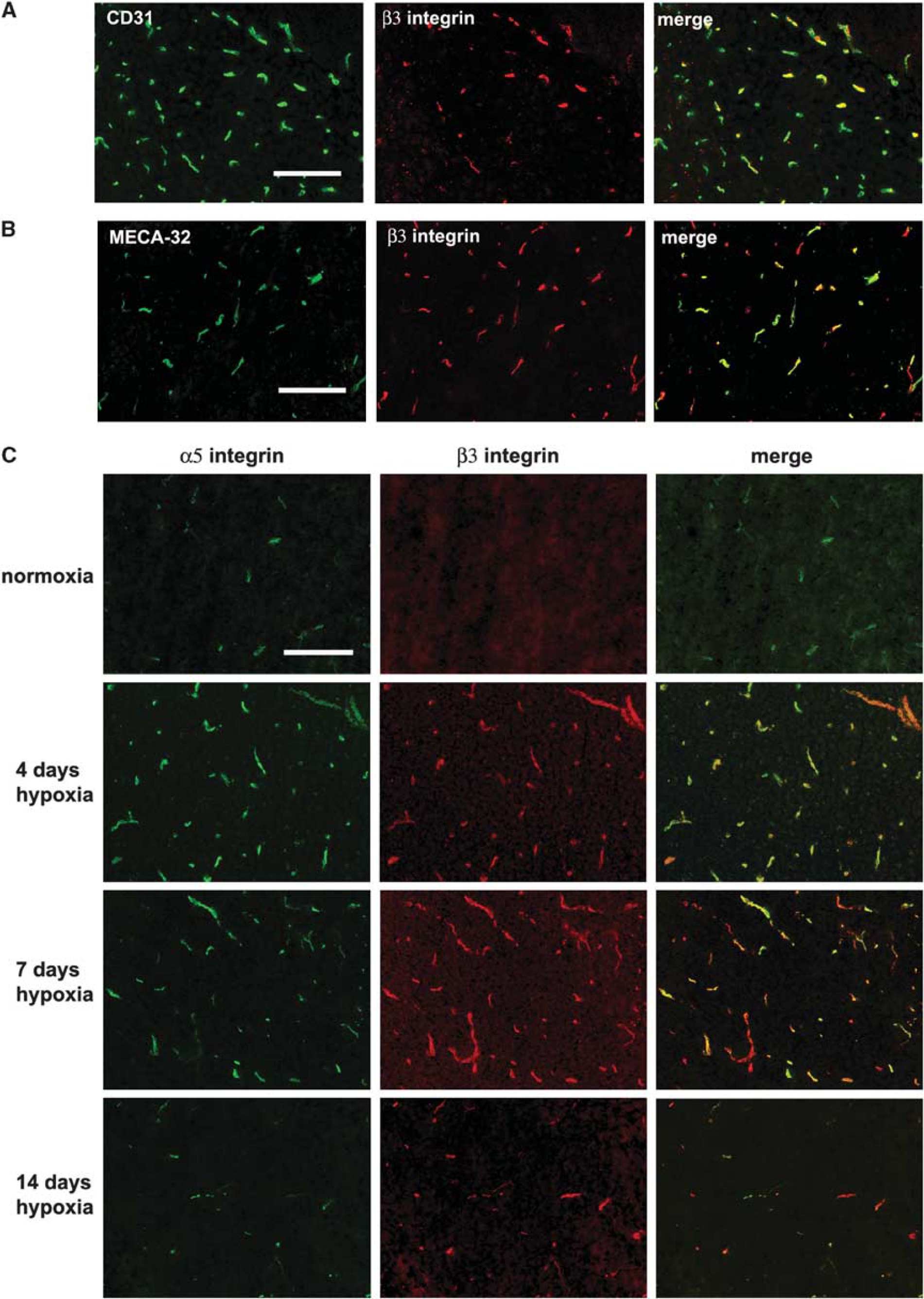
Expression of the β3 integrin on angiogenic capillaries in the hypoxic central nervous system (CNS). Dual-immunofluorescence (IF) was performed on frozen sections of cerebral cortex from normoxic or hypoxia-exposed mice using monoclonal antibodies specific for: (
Both α5β1 and αvβ3 Integrins are Upregulated on Angiogenic Endothelial Cells but the Temporal and Spatial Expression Patterns only Partially Overlap
As α5β1 integrin is also induced by angiogenic BEC in the hypoxia model (Milner et al, 2008a), we next performed dual-IF to examine whether the α5 and β3 integrin subunits show similar expression profiles. As shown in Figure 2C, in the normoxic brain, a few α5 integrin-positive capillaries were always present, whereas β3 integrin-positive capillaries were rarely detected. Hypoxia promoted strong induction of both α5 and β3 integrins on capillaries, and largely speaking, the two receptors were co-expressed by the same capillaries. However, consistent with previous findings, capillary expression of the α5 integrin was quickly induced, peaked at day 4, and then declined. In contrast, expression of the β3 integrin followed a slower time-course, reaching a peak after 7 days hypoxia. This shows that although hypoxia increases BEC expression of α5β1 and αvβ3 integrins, these responses follow a different time-course.
Hypoxia also Induces Brain Capillary Expression of the αvβ3 Ligand Vitronectin
We have shown that hypoxia leads to increased expression of fibronectin around cerebral capillaries, reaching maximal level after 4 days hypoxia, before declining at later time points (Milner et al, 2008a). Now, we examined whether expression of other αvβ3 ligands also shows changes in expression. The αvβ3 integrin is a promiscuous receptor, binding vitronectin, osteopontin, and fibronectin (Eliceiri and Cheresh, 2001; Stromblad and Cheresh, 1996). As shown in Figure 3, although vitronectin-positive capillaries were weakly present in the normoxic brain in both brainstem and cortex, cerebral hypoxia markedly increased expression levels. Quantification showed that in both the brainstem and cerebral cortex, the number of vitronectin-positive capillaries was maximal after 7 days hypoxia, and then declined at later time points. Seven days hypoxia increased the number of vitronectin-positive vessels per field of view, from 39.5 ± 0.6 to 56.3 ± 2.3 in the brainstem (
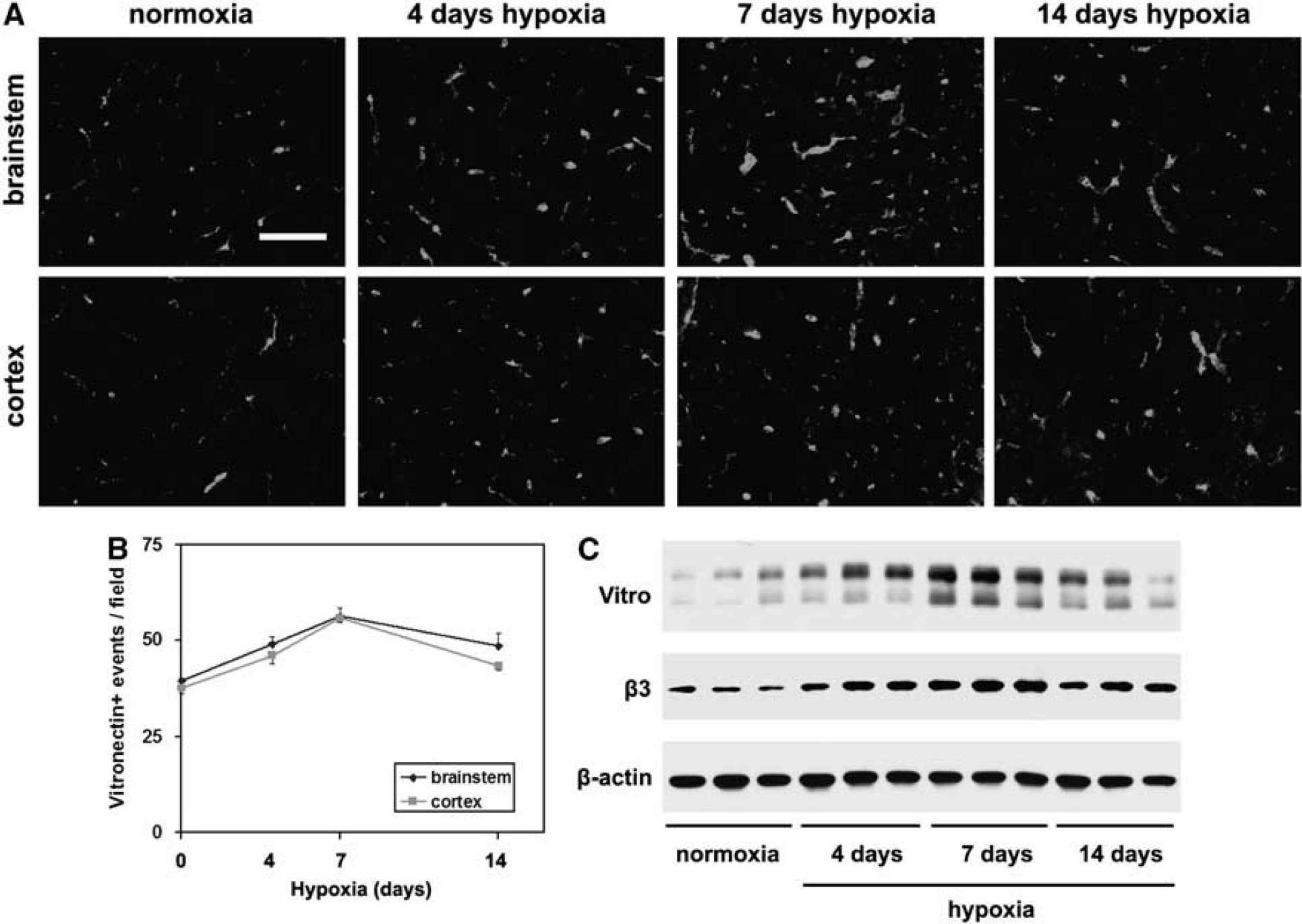
Hypoxic induction of vitronectin on cerebral blood vessels. (
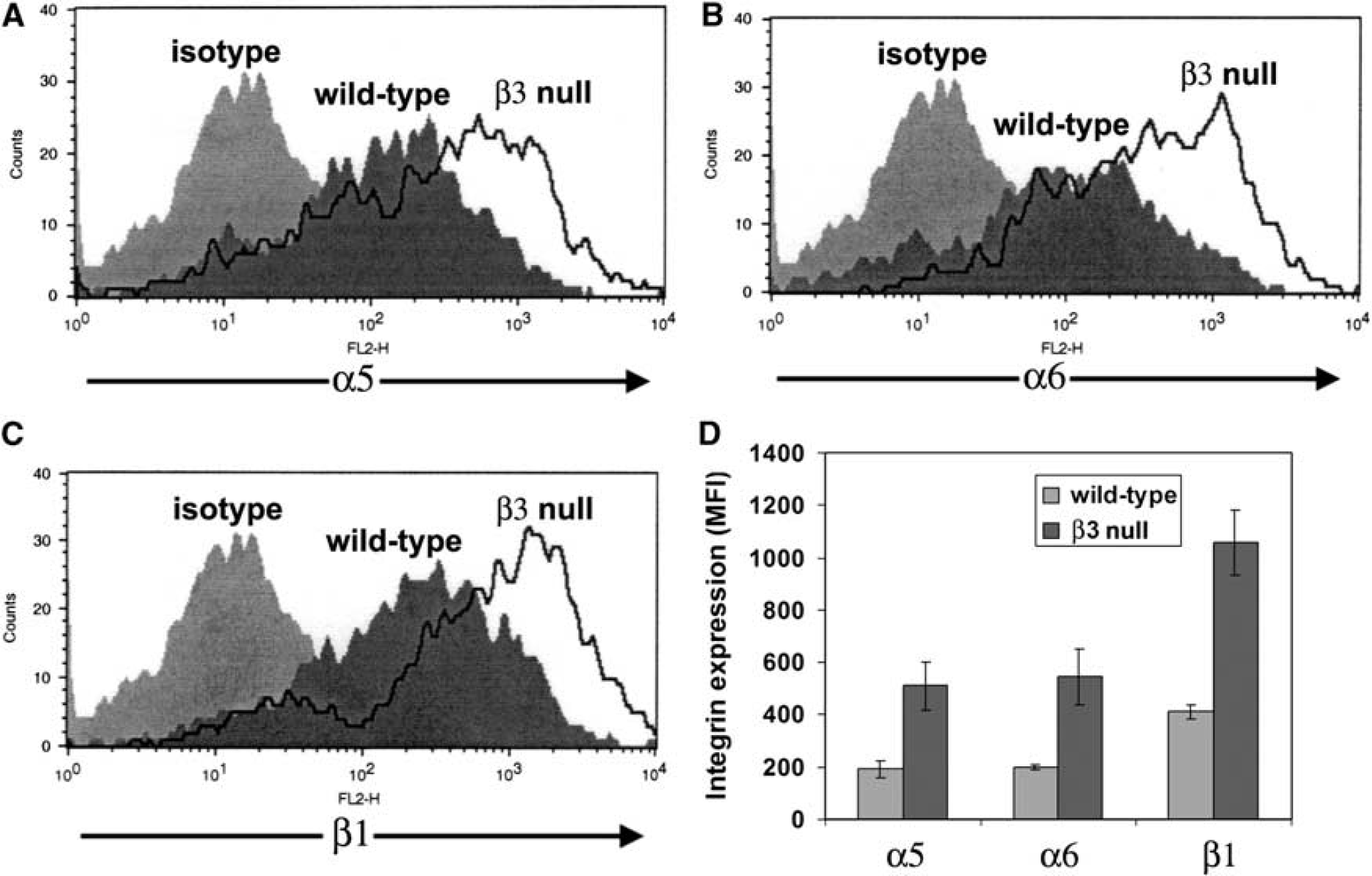
BEC from β3 Integrin-Null Mice Express no αv Integrins
To investigate the potential angiogenic role of the αvβ3 integrin, we studied β3 integrin-null mice. First we characterized the αv integrin expression profile of BEC derived from β3 integrin-null or wild-type littermate control mice by flow cytometry. As shown in Figure 4A, BEC from wild-type mice expressed high levels of the β3 integrin subunit, whereas in contrast, no β3 integrin expression was detected on β3 integrin-null BEC. Next, to exclude the possibility that BEC express other αv integrins, or that β3 integrin-null BEC show compensatory induction of other αv integrins, we examined the expression of the αv integrin subunit (Figure 4B). While wild-type BEC expressed the αv subunit, β3 integrin-null BEC showed absolutely no expression. This confirms that BEC express just one αv integrin, αvβ3, and also shows that β3 integrin-null BEC show no compensatory upregulation of other αv integrins. To confirm the absence of the β3 integrin subunit
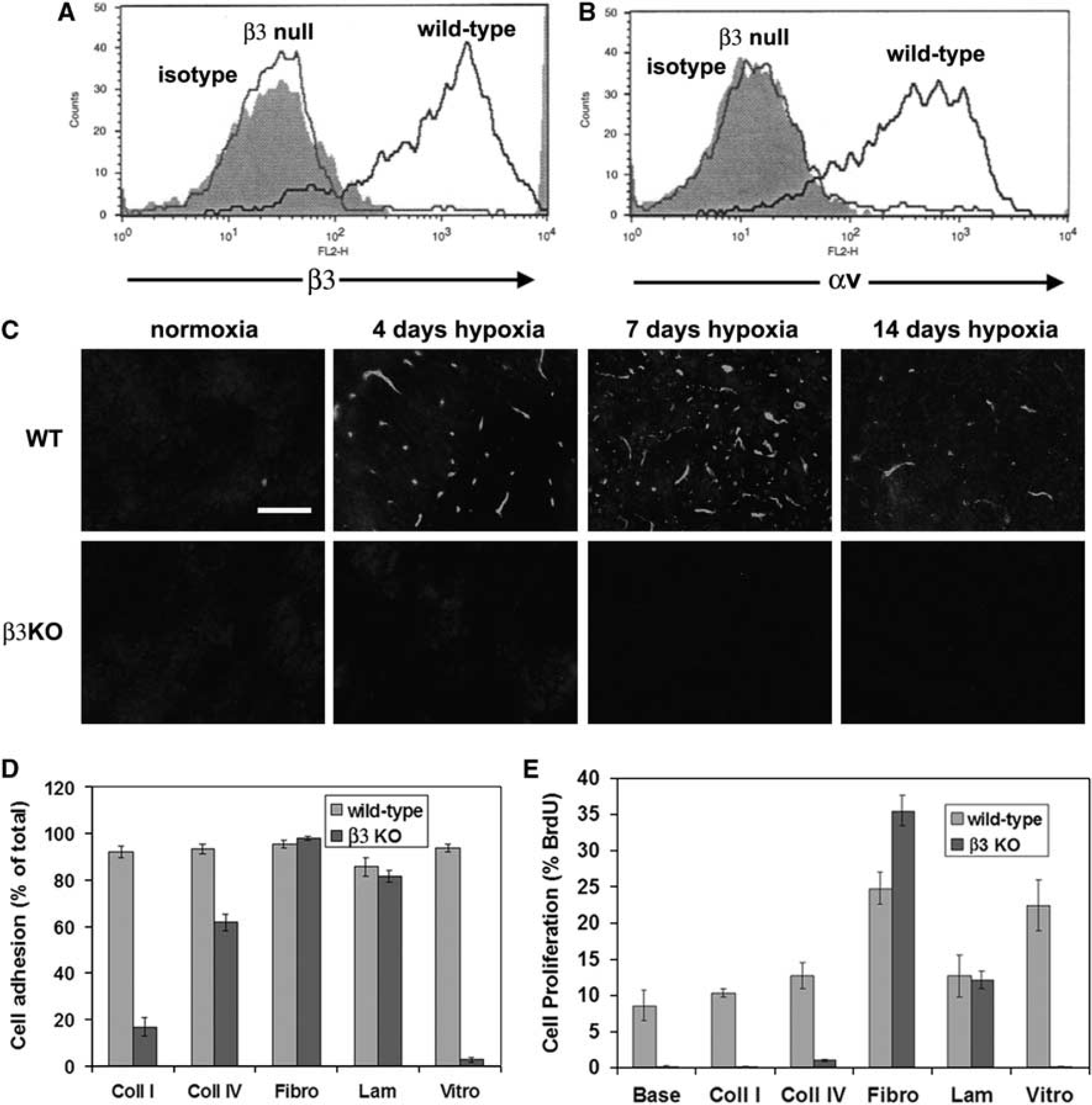
Characterization of brain endothelial cells (BEC) derived from β3 integrin-null mice. (
β3 Integrin-Null BEC Show Reduced Adhesion and Proliferation on Vitronectin and the Collagens I and IV, but Enhanced Proliferation on Fibronectin
Next, we examined whether β3 integrin-null BEC showed any defects in their response to ECM ligands. Wild-type BEC attached to all ECM proteins tested, including collagens I and IV, fibronectin, laminin, and vitronectin. In contrast, β3 integrin-null BEC showed marked adhesive defects to vitronectin and collagen I, and a mild defect to collagen IV, though adhesion to fibronectin and laminin was no different from wild-type cells (Figures 4D and 5). The adhesive defect of β3 integrin-null BEC was most profound on vitronectin, with almost no cells adhering to vitronectin (2.6% ± 1.1% compared with 93.8% ± 1.7% wild-type,
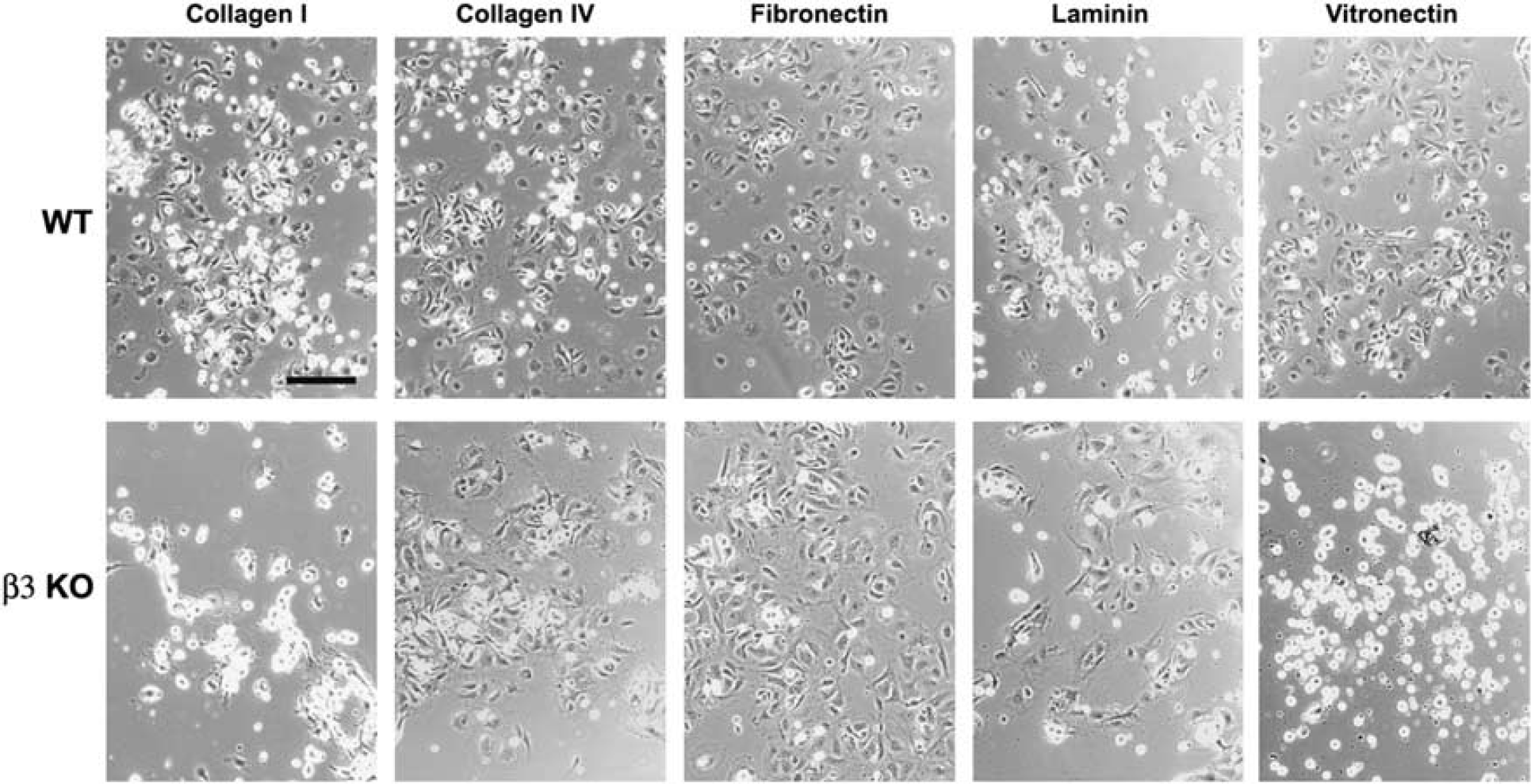
Analysis of the adhesion defects in β3 integrin-null brain endothelial cells (BEC). Wild-type or β3 integrin-null BEC were plated onto the extracellular matrix (ECM) proteins, collagen I, collagen IV, fibronectin, laminin, or vitronectin. After 3 h, photographs were taken. Scale bar = 50 μm. Note that wild-type BEC attached and spread on all ECM proteins tested. In contrast, β3 integrin-null BEC showed marked adhesive defects on vitronectin and collagen I, and a mild defect on collagen IV, though adhesion to fibronectin and laminin was no different from wild-type cells.
Next, the role of αvβ3 integrin in mediating BEC proliferation on different ECM proteins was examined using BrdU incorporation. Consistent with previous findings (Wang and Milner, 2006), fibronectin (24.8% ± 2.2%) and vitronectin (22.4% ± 3.3%) significantly promoted proliferation of wild-type BEC, relative to uncoated glass (8.6% ± 2.1%,
β3 Integrin-Null Mice Show an Altered Angiogenic Response to Cerebral Hypoxia
To determine whether the αvβ3 integrin has an important role in mediating the angiogenic response to cerebral hypoxia, we examined this response in β3 integrin-null mice. As an increased capillary density is the end product of angiogenesis, we first examined capillary density at the different time points. CD31 IF showed that cerebral hypoxia promoted an increased capillary density in the brains of β3 integrin-null and wild-type littermate control mice, with no significant differences observed between the two groups at any time point (Figures 6A and 6B). Next we examined cell proliferation by Ki67 IF, as this is a sensitive early indicator of angiogenesis that peaks after 4 days cerebral hypoxia (Li
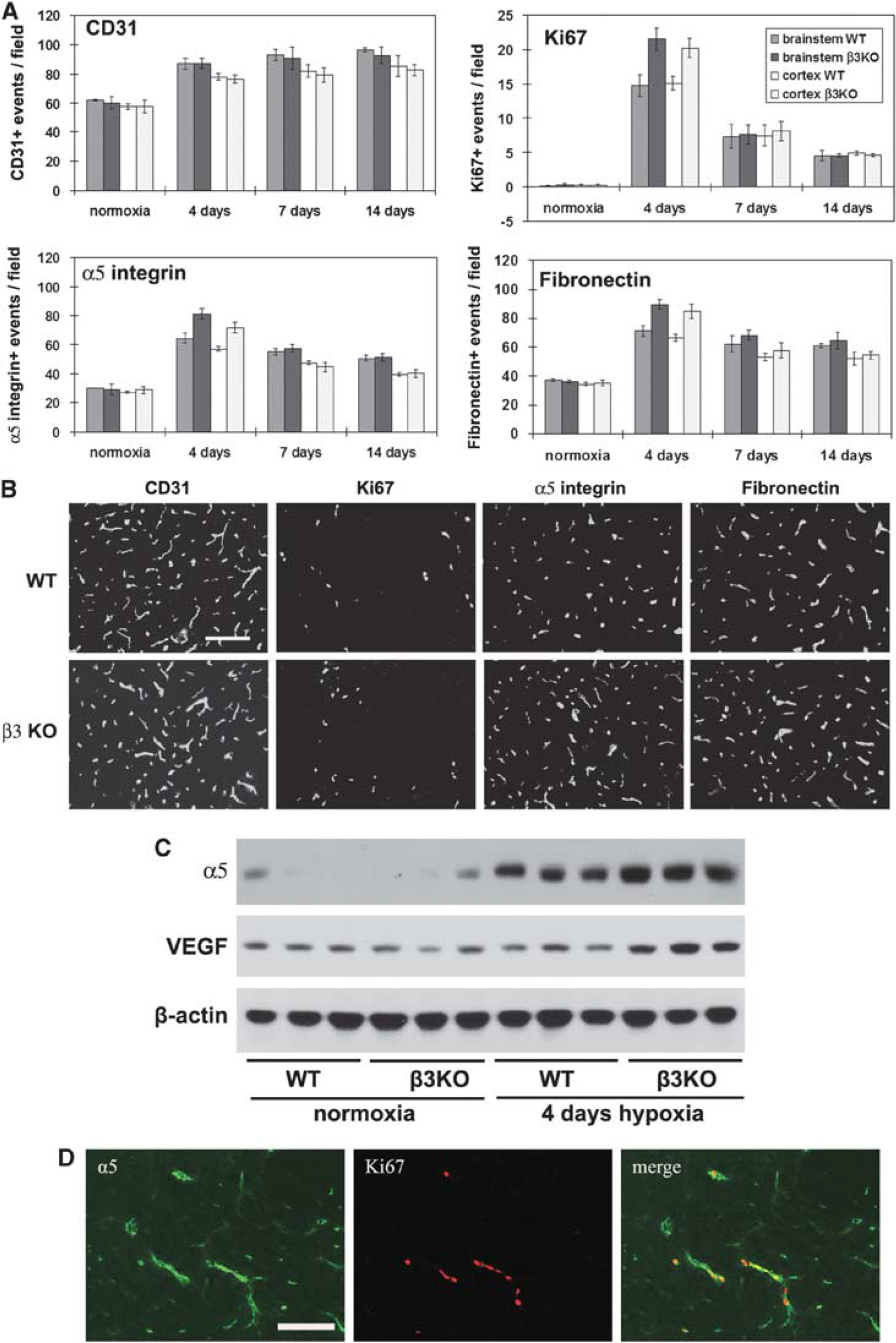
Comparison of the hypoxic-induced angiogenic response, in the wild-type and β3 integrin-null central nervous system (CNS). β3 integrin-null and wild-type littermate control mice were maintained at normoxia or exposed to 8% hypoxia for 4, 7, or 14 days before frozen brain sections immunostained to assess blood vessel density (CD31), cell proliferation (Ki67), and expression of α5 integrin and fibronectin. (
β3 Integrin-Null BEC also Show Upregulated Expression of β1 Integrins In Vitro
To quantify the compensatory upregulation of α5 integrin in β3 integrin-null BEC, and also examine whether this mechanism operates
Compensatory upregulation of β1 integrin expression on β3 integrin-null brain endothelial cells (BEC). Wild-type or β3 integrin-null BEC were cultured on fibronectin. On reaching confluence, flow cytometry was used to quantify BEC expression of the integrin subunits α5 (
Discussion
In this study, we examined the expression and potential angiogenic role of the αvβ3 integrin in the hypoxic CNS. We found that the αvβ3 integrin and its ECM ligand vitronectin were strongly upregulated on angiogenic capillaries, with the effect maximal after 7 days hypoxia. Surprisingly, in the cerebral hypoxia model, β3 integrin-null mice showed no obvious defect in cerebral angiogenesis. However, at an early stage of the angiogenic process, BEC in these mice showed an increased rate of proliferation, which correlated closely with increased expression of α5 integrin. Flow cytometry confirmed strong compensatory upregulation of α5 integrin on β3 integrin-null BEC, which correlated with an increased BEC proliferation rate on fibronectin. These studies confirm hypoxic induction of αvβ3 integrin on angiogenic vessels, and show that in β3 integrin-null BEC, parallel increases occur in α5 integrin expression and cell proliferation, both
Induction of the αvβ3 Integrin on Angiogenic Cerebral Blood Vessels
In this study, we showed that αvβ3 integrin was strongly upregulated on angiogenic capillaries in the hypoxia CNS. This observation is consistent with previous findings from studies of focal cerebral ischemia, both in the primate (Abumiya et al, 1999; Okada et al, 1996) and in the rat (Wei et al, 2001). Previously, we described upregulation of the α5β1 integrin and its ligand fibronectin in the cerebral hypoxia model (Milner et al, 2008a), and upregulation of α5 integrin on hypoxic BEC
Behavior of β3 Integrin-Null BEC In Vitro
Flow cytometry revealed that β3 integrin-null BEC express no αv integrins. This makes two points: first, consistent with our previous biochemical studies (Wang and Milner, 2006), BEC appear to express only one αv integrin, αvβ3, and second, β3 integrin-null BEC show no compensatory induction of other αv integrins. Interestingly however, β3 integrin-null BEC showed strong upregulation of β1 integrins, including the α5, α6, and β1 subunits, all of which showed a 2.5-fold increase. This is the first demonstration that β3 integrin-null cells show compensatory upregulation of other integrins, though it is in keeping with findings from other systems that show increased α2 integrin levels in renal cells of α8 integrin-null mice (Haas et al, 2003), and elevated levels of α6 integrin in cerebral vessels from α7 integrin-null mice (Flintoff-Dye et al, 2005). Upregulation of the α5 integrin subunit is particularly significant, as aside from αvβ3, the α5β1 integrin is the main fibronectin receptor expressed by BEC (Wang and Milner, 2006), thus it is logical that β3 integrin-null BEC would upregulate α5β1 integrin in an attempt to maintain their responses to fibronectin. Consistent with this, proliferation of β3 integrin-null BEC on fibronectin was greater than wild-type cells, highlighting a tight correlation between BEC α5 integrin expression and proliferation rate. β3 integrin-null BEC did show marked adhesive defects on vitronectin and the collagens, culminating in a lack of proliferation on these substrates. The lack of adhesion to vitronectin is consistent with studies in other cell types (Reynolds et al, 2002, 2005), and highlights the importance of αvβ3 as a major vitronectin receptor.
Role of αvβ3 in Angiogenesis In Vivo
Induction of αvβ3 integrin on angiogenic endothelial cells is well established (Abumiya et al, 1999; Eliceiri and Cheresh, 2001; Okada et al, 1996; Wei et al, 2001). What is less clear is whether this integrin is essential for angiogenesis. Studies examining the angiogenic role of αvβ3 integrin in other systems have generated conflicting results, with function-blocking approaches supporting an angiogenic role (Brooks et al, 1994a,
b
), but studies in β3 integrin-null mice refute this role (Hodivala-Dilke et al, 1999; Reynolds et al, 2002). The results of our study show that the αvβ3 integrin is not essential for hypoxia-induced cerebral angiogenesis. In fact its absence led to accelerated BEC proliferation, which correlated with elevated levels of VEGF and BEC expression of the alternate fibronectin receptor, α5β1 integrin. These observations are consistent with other studies, which describe increased tumor-induced angiogenesis, and accelerated wound-healing in β3 integrin-null mice, events that correlate with compensatory increases in VEGF and transforming growth factor-β1-mediated signaling, in these two respective models (Reynolds et al, 2002, 2005). Exactly what accelerates BEC proliferation in β3 integrin-null mice is unclear. The most likely reason is that the αvβ3 integrin has an important, though redundant adhesive function on BEC, and that absence of this function leads to upregulation of other angiogenic mechanisms, including the α5β1 integrin and VEGF, which exert their effects to promote BEC proliferation. Compensatory upregulation of the α5 integrin was observed both
Implications for the Role of α5β1 Integrin in Cerebral Angiogenesis
The results of our study point to the conclusion that of the two BEC fibronectin receptors, αvβ3 is not essential for cerebral angiogenesis, whereas the tight relationship between α5β1 and BEC proliferation strongly suggests a proangiogenic role for this integrin. This conclusion is consistent with recent data obtained in our laboratory, which show a very close correlation between α5β1-fibronectin expression levels and BEC proliferation, during the angiogenic response to hypoxia (Li
Footnotes
The authors declare no conflict of interest.
