Abstract
After cerebral ischemia, angiogenesis, by supplying for the deficient perfusion, may be a beneficial process for limiting neuronal death and promoting tissue repair. In this study, we showed that the combination of Ang-1 and vascular endothelial growth factor (VEGF) provides a more adapted therapeutic strategy than the use of VEGF alone. Indeed, we showed on a focal ischemia model that an early administration of VEGF exacerbates ischemic damage, because of its effects on blood—brain barrier (BBB) permeability. In contrast, a coapplication of Ang-1 and VEGF leads to a significant reduction of the ischemic and edema volumes by 50% and 42%, respectively, in comparison with VEGF-treated mice. We proposed that Ang-1 blocks the BBB permeability effect of VEGF in association with a modulation of matrix metalloproteinase (MMP) activity. Indeed, we showed on both ischemic in vivo and BBB in vitro models that VEGF enhances BBB damage and MMP-9 activity and that Ang-1 counteracts both effects. However, we also showed a synergic angiogenic effect of Ang-1 and VEGF in the brain. Taken together, these results allow to propose that, in cerebral ischemia, the combination of Ang-1 and VEGF could be used early to promote the formation of mature neovessels without inducing side effects on BBB permeability.
Introduction
Angiogenesis is one of the processes required for the formation and function of all organs and is involved in both physiological and pathological situations. In some human diseases such as heart ischemia, this process can be used to restore the vital function of affected organs. Likewise, recent findings argue that angiogenesis takes place in the adult brain after stroke (del Zoppo and Mabuchi, 2003). Accordingly, increased expression of angiogenic factors has been described in both brain and serum of patients after ischemia (Issa et al, 1999; Slevin et al, 2000). In addition, stroke patients with a higher density of brain blood vessels appear to have reduced morbidity and a longer survival (Krupinski et al, 1994). These data underline that stimulating the restoration of a cerebral circulation by enhancing collateral circulation and/or angiogenesis may be a therapeutic approach for recovery in stroke.
During development, vascular endothelial growth factor (VEGF) and angiopoietins (Angs) collaborate to regulate various aspects of this process (Carmeliet, 2003). Although VEGF initiates the formation of neovessels, Ang-1 is required for the stabilization of endothelial contacts with surrounding smooth muscle cells to give mature vessels. Thus, because angiogenesis is a tightly controlled molecular multi-step process, therapeutic proangiogenic strategies based on the use of a single growth factor may be insufficient or inadequate to promote a functional vascular system. In addition, although VEGF is a powerful growth factor for therapeutic angiogenesis in the ischemic limb (Baumgartner et al, 1998) and myocardium (Kastrup, 2003), it displays other effects such as an increase of the endothelium permeability (Bates et al, 2002). This latter effect could lead to vasogenic edema, particularly deleterious in cerebral ischemia. Indeed, an early administration of VEGF exacerbates blood—brain barrier (BBB) leakage, although a late administration of VEGF enhances angiogenesis (Zhang et al, 2000). Accordingly, to limit rapidly the extent of neuronal damage and promote angiogenesis as soon as possible, an early therapeutical intervention based on the use of VEGF alone is not appropriate. In the acute stage of stroke, the permeability effect of VEGF has to be counteracted. Interestingly, systemic delivery of Ang-1 by adenoviral gene delivery causes resistance to vascular leakage induced by VEGF in the skin (Thurston et al, 2000) or in the brain (Zhang et al, 2002b). Furthermore, in the skin, the combined overexpression of Ang-1 and VEGF has an additive effect on vascularization and produced the leakage-resistant vessels typical of Ang-1 overexpression (Thurston, 2002). However, in retina, besides antipermeability effects, Ang-1 exhibits antiangiogenic effects (Nambu et al, 2004). Therefore, in agreement with a specific tissue-dependent angiogenesis control, the effects of Ang-1 may vary depending on the tissue. To date, only one study reports an angiogenic effect of Ang-1 in the brain (Ward et al, 2004). If Ang-1 stabilizes cerebral blood vessels and potentiates neovascularization, it may provide benefit in cerebral ischemia in which BBB permeability contributes to neuronal damage but in which a proangiogenic effect could be beneficial.
Based on the above data, it could be proposed that an early administration of a combination of VEGF and Ang-1 may be beneficial for stroke. This study was undertaken to clarify whether Ang-1 potentiates VEGF angiogenic activities in the brain and counteracts its permeability effects and to gain further insights into the molecular mechanisms involved in the effects of these two factors in the ischemic brain. A consistent body of experimental evidence has shown that in the initial steps of angiogenesis, the matrix metalloproteinases (MMPs) play an important role in the degradation of the basal lamina and the invasion of endothelial cells (Pepper, 2001). In addition, angiogenic factors such as VEGF and Angs could regulate MMP expressions and/or activities (Das et al, 2003; Wang and Keiser, 1998). However, proteolytic enzymes such as MMPs, in particular MMP-2 and MMP-9, could contribute to BBB injury in cerebral ischemia (Rosenberg et al, 1998). Therefore, we examined, in the present work, if Ang-1 could counteract VEGF-induced vascular permeability in cerebral ischemic conditions by regulating MMP activities.
Materials and methods
In Vivo Studies
All experiments were performed on OF1 mice (CERJ, France) under chloral hydrate anesthesia (500 mg/kg). Surgical protocols were approved by the local ethics committee and conformed to national legislation.
Focal Cerebral Ischemia
Focal ischemia was induced by permanent occlusion of the left middle cerebral artery (MCAO), as reported previously (Welsh et al, 1987). In this model, the area of ischemia is restricted to the neocortex. At different times after occlusion, mice were anesthetized and the brains were removed. Coronal brain sections (20 μm) were cut on a cryostat and stained with thionin. The morphometric analysis obtained by using the public domain Image J software allows to evaluate on the same section both edema and ischemic areas. The edema surface corresponds to the difference between ipsilateral and contralateral surface. Total infarct and edema volumes (mm3) were calculated after integration of infarcted and edema areas with the distance (400 μm) between each section level analyzed (Swanson et al, 1990).
Intracerebroventricular Injections
Anesthetized mice were treated by intracerebroventricular injection of 3 μL of vehicle (PBS with 1 mg/mL of bovine serum albumin; Sigma-Aldrich, France) or rHuAng-1, (9 μg/mL, R&D Systems, Europe), or rHuVEGF (3 μg/mL, Sigma Aldrich, France), or both rHuAng-1 and rHuVEGF, (n = 8 per group) just before the MCAO. Animals were placed on a stereotactic frame and injected at coordinates bregma 0 mm, mediolateral 2 mm, and dorsoventral 2 mm. Physiologic parameters remained in the normal range (body temperature (°C): 37 ± 0.3; PaCO2 (mm Hg): 40.9 ± 4.2; PaO2 (mm Hg): 131.73 ± 3.97; pH: 7.08 ± 0.08.
Minipump Infusions of Angiogenic Factors
Osmotic minipumps designed to deliver 0.5 μL/h for 7 days (model 1007D, Alzet osmotic pumps) were filled with rHuVEGF (100 ng/mL) or rHuAng-1 (300 ng/mL), or both rHuVEGF and rHuAng-1 (100 and 300 ng/mL, respectively). The osmotic minipump cannula was implanted in the rat striatum (n = 3 per group) as previously described (Rosenstein et al, 1998).
Determination of Vascular Perfusion
To evaluate vascular perfusion, 100 μL of FITC-dextran (77,000 mol.wt., 10% in saline, Sigma-Aldrich, France) was administrated intravenously via the tail vein and circulated for 1 min. The anesthetized animals were then killed by decapitation. The brains were rapidly removed and placed in 4% paraformaldehyde (PFA) at 4°C for 48 h. Coronal free-floating brain sections (50 μm) were cut on a vibratome.
Determination of Blood—Brain Barrier Permeability by Using Evans Blue
Cerebral ischemia was induced for 6 h. At 3 h after MCAO, mice (n = 4 to 6, depending on the treatment) were injected with 100 μL of 2% Evans blue (Eliceiri et al, 1999), as a marker of a frank increase in permeability. After 3 h, animals were perfused with NaCl (150 mmol/L). The brains were then removed, ischemic/ipsilateral and contralateral cortices were dissected, weighted, homogenized in 500 μL of PBS (0.1 mol/L) and centrifuged. Supernatants were diluted with 500 μL of trichloroacetic acid (TCA, 100%) overnight at 4°C. After a centrifugation at 21,000g for 30 mins, Evans blue was quantified in the samples from the absorbance at 620 nm by using a standard Evans blue solution (from 0 to 6.25 μg/mL) and divided by the weight of each cortex. Results are expressed as the percentage of Evans blue quantity in the contralateral cortex.
Determination of Blood—Brain Barrier Disruption by Using 14Cα Amino Isobutyric Acid
To evaluate subtle changes in vascular permeability, at different times after occlusion, mice (n = 3) received an intravenous administration of one of the most sensitive quantitative tracer 14Cα- amino isobutyric acid (AIB, MW 103, PerkinElmer, Europe; 3 μCi per mouse) 30 mins before anesthesia as described (Diler et al, 2002). Then, vascular compartment was rinsed; blood samples were withdrawn and brains were removed. Ipsilateral and contralateral cortices were weighed, homogenized in 1 mL of solvable® (PerkinElmer, Europe) digested for 4 h at 60°C. Radioactivity was quantified in the samples by adding scintillation liquid and expressed as cpm/g of cortex. Results are expressed as the percentage of variation of radioactivity measured in the ipsilateral (ischemic) side to that of the contralateral side. No variation was observed between the contralateral cortex whatever the ischemia duration. Blood samples were bleached with H2O2 (30%) after adding solvable. Radioactivity was quantified in the blood samples by adding scintillation liquid and expressed as cpm/mL and used as an internal control to verify that each animal received an identical quantity of radioactivity.
Matrix Metalloproteinase Activity Analysis
For analysis of MMP activity in mice (n = 3), brains were removed at different times after MCAO, divided into ipsilateral and contralateral cortices, and homogenized in a 2:1 volume-to-weight ratio of lysis buffer containing Tris (50 mmol/L)-NaCl (150 mmol/L), 0.5% Triton X-100 (Sigma Aldrich, France) (Kim et al, 2003). The protein concentrations of the extracts were determined using BCA protein assay kit (Pierce, France) and samples (30 μg) were assayed by gelatin zymography for MMP activity. For analysis of MMP activity in cell media, supernatants were concentrated using a universal system plus (Savant, France). After electrophoresis, the gel was washed twice with 2.5% Triton X-100 (Sigma-Aldrich), then incubated overnight at 37°C in Tris-buffered saline containing 5 mmol/L CaCl2, after which it was stained with 0.5% Coomassie Brilliant Blue R-250 and destained. To confirm the specificity of the detection of the metalloproteinase activity, gelatin-substrate zymogram was realized in presence of EDTA, which blocked the gelatinase activity (data not shown).
Immunohistochemistry
After MCAO, mice were perfused transcardially with 4% PFA and coronal free-floating brain sections (20 μm) were cut on a vibratome. The immunohistochemical staining was performed with the avidin—biotin—peroxidase technique as described previously (Bernaudin et al, 1999). The following primary antibodies were used: rabbit polyclonal antibodies to laminin (1:1,000, Sigma-Aldrich, France), von Willebrand Factor (1:200, Dako, France), PECAM-1 (1:200, Pharmingen, France), Ki67 (1:100, Novo Castra, France).
Reverse Transcription-Polymerase Chain Reaction
Total RNA was prepared from cerebral cortices by phenol/chloroform extraction using the RNAxel® extraction kit (Eurobio, France). Reverse transcription and amplification by PCR were performed as described previously (Bernaudin et al, 1999). Primers for Ang-1, VEGF, and β-actin have been used as published (Abdulmalek et al, 2001; Marti and Risau, 1998).
Transendothelial Transport Studies in an In Vitro BBB Model
The in vitro BBB model corresponding to a coculture of bovine brain endothelial cells grown on inserts, in presence of rat primary astrocytes cultures, was prepared as previously described (Dehouck et al, 1990). The upper and lower compartments are defined as the luminal and the abluminal side, respectively. During the permeability study, Ringer—HEPES solution (150 mmol/L NaCl, 5.2 mmol/L KCl, 2.2 mmol/L CaCl2, 0.2 mmol/L MgCl2 · 6H2O, 6 mmol/L NaHCO3, 5 mmol/L HEPES, 2.8 mmol/L glucose, pH 7.4) was added to the lower compartment of a six-well plate. After 6 h of treatment with rHuVEGF (75 ng) or rHuAng-1 (225 ng), or both rHuVEGF and rHuAng-1 (75 ng and 225 ng, respectively), filters containing endothelial cells (ECs) were transferred into a six-well plate containing Ringer—HEPES solution. Ringer—HEPES solution (1.5 mL) containing 14C sucrose (Amersham, UK) was placed, at time zero, in the upper compartment. Incubations were performed at 37°C. At 15, 30, and 45 mins, inserts were transferred to other wells to minimize the possibility of passage from the lower compartment to the upper compartment. For each condition, three inserts were used. An aliquot of 500 μL from each lower compartment at each time and 20 μL from the initial solution containing the labeled molecule were placed in a scintillation vial, and radioactivity was determined (Tri Carb 2100 scintillation counter; Packard Instrument Company, Meriden, CT, USA). The endothelial permeability coefficient (Pe) was calculated in centimeters per minute on triplicate samples (from three independent experiments) as described previously (Brillault et al, 2002).
Oxygen Glucose Deprivation Transport Studies
To mimic ischemic conditions, oxygen glucose deprivation (OGD) was used on the in vitro BBB model (Jones et al, 2004; Pellegrini-Giampietro et al, 1999; Wiessner et al, 2000). For OGD studies, Ringer—HEPES solution was first equilibrated with nitrogen and glucose was removed from the medium. On the day of the experiment, ECs with glial cells were exposed during 4 h to OGD conditions by adding low-glucose (0.2 g/L) medium equilibrated with nitrogen (OGD medium). Cytokines, rHuVEGF (75 ng) or rHuAng-1 (225 ng), or both rHuVEGF and rHuAng-1 (75 and 225 ng, respectively) were added during the exposition to OGD. Cells were then placed for defined times in a 37°C oven at 0% O2, 5% CO2, 95% N2 using the Gas Pack Pouch bags (Becton Dickinson, Franklin Lakes, NJ, USA). Oxygen glucose deprivation transport studies were performed on triplicate samples (from three independent experiments) as described above. Transport studies were performed under an airtight glove box filled with nitrogen at 37°C (Forma Scientific, Mountain View, CA, USA).
Study Population
The present study assessed consecutive patients with acute ischemic stroke admitted to the Emergency Room of a teaching hospital within 3 h after symptom onset, who had a nonlacunar stroke involving the vascular territory of the middle cerebral artery (MCA). The final analysis included 41 patients who had an acute cardioembolic stroke in the MCA territory and received t-PA at < 3 h after symptom onset. The clinical protocol has been described elsewhere in detail (Montaner et al, 2003). The study was approved by the Ethics Committee of the hospital and all patients or relatives gave informed consent.
Vascular Endothelial Growth Factor and Matrix Metalloproteinase-9 Immunoassays
Peripheral blood samples were drawn from each patient at study entry (before t-PA administration) and at 12 and 24 h from stroke onset. EDTA tubes were used to collect the blood; plasma was immediately separated by centrifugation at 3,000 r.p.m. for 15 mins and stored at −80°C. Total VEGF (Quantikine® R&D Systems, USA) and MMP-9 (Biotrak Amersham Pharmacia, UK) levels were determined in duplicate by commercially available enzymelinked immunosorbent assay (ELISA) and the mean value of both determinations was used. All ELISAs were performed according to the manufacturer's instructions. Our laboratory reference values for healthy controls are: 12.47 ± 14.96 pg/mL for VEGF (n = 68, 70% females, mean age 43 years, normal range < 27.43 pg/mL) and 41 ± 27.8 ng/mL for MMP-9 (n = 62, 42% females, mean age 43 years, normal range < 97 ng/mL). The mean intraassay coefficients of variation were <10%.
Intercellular Adhesion Molecule-1 and hs-CRP Immunoassays
Serum was used to measure intercellular adhesion molecule-1 (ICAM-1) and human serum C reactive protein (hs-CRP) ICAM-1 levels were determined by commercially available ELISA (Bender MedSystems, Austria). This ELISA was performed in duplicate, according to the manufacturer's instructions, with the mean intraassay coefficients of variation < 10%. Hs-CRP (human serum C reactive protein) levels were obtained by nephelometry, using a Behring Nephelometer Analyzer.
Statistical Analyses
For in vitro and in vivo experiments, statistical analyses were obtained by either Bonferroni—Dunn's or Fischer/PLSD post hoc tests after a significant ANOVA.
For human data, descriptive and frequency statistical analyses were obtained and compared using SPSS 10.0. The Mann—Whitney U-test was used to assess differences between patients and healthy controls. Spearman's rank correlation test was used to test all correlations.
Results
Induction of a few Functional Neovessels in Response to Focal Cerebral Ischemia
To characterize the evolution of the vasculature pattern in response to focal permanent ischemia in mice, we performed immunohistochemistry on brain coronal sections using antibodies raised against laminin, platelet endothelial cell adhesion molecule-1 (PECAM-1) and von Willebrand factor (vWf) at different times after occlusion. An immunodetection with an antibody against laminin revealed an increase in the number of vessels present in the ischemic area from 24 h to 7 days (Figures 1A–1D). These vessels are tortuous and enlarged, particularly 3 to 7 days after occlusion (Figures 1C–1D, large magnification). Moreover, PECAM-1 immunostaining detected on vessels increased from 24 to 48 h after occlusion in the ischemic tissue but strongly decreased at 3 days (Figures 1E–1H). To study whether the neovessels present in the ischemic area result from an angiogenic process, we compared, on adjacent sections, the immunostaining obtained for vWf and for Ki-67, a marker of cell proliferation. Some endothelial cells are codetected with both antibodies suggesting that a process of angiogenesis is initiated in the ischemic tissue (Figures 1I-1J). These immunostainings were observed at the border zone around the infarcted area and in a lesser extent in the ischemic core (Marti et al, 2000). As the reestablishment of blood flow is the final step in angiogenesis, we looked for the existence of perfused vessels in the mouse brain after 3 days of occlusion. Administration of FITC-dextran, a vascular perfusion tracer, revealed the presence of perfused vessels in the ischemic area suggesting the functionality of these neovessels; several of these vessels seem to be also permeable (Figures 1K–1L). Beyond 3 days, functional vascular structures were no more detected in the ischemic area (data not shown).
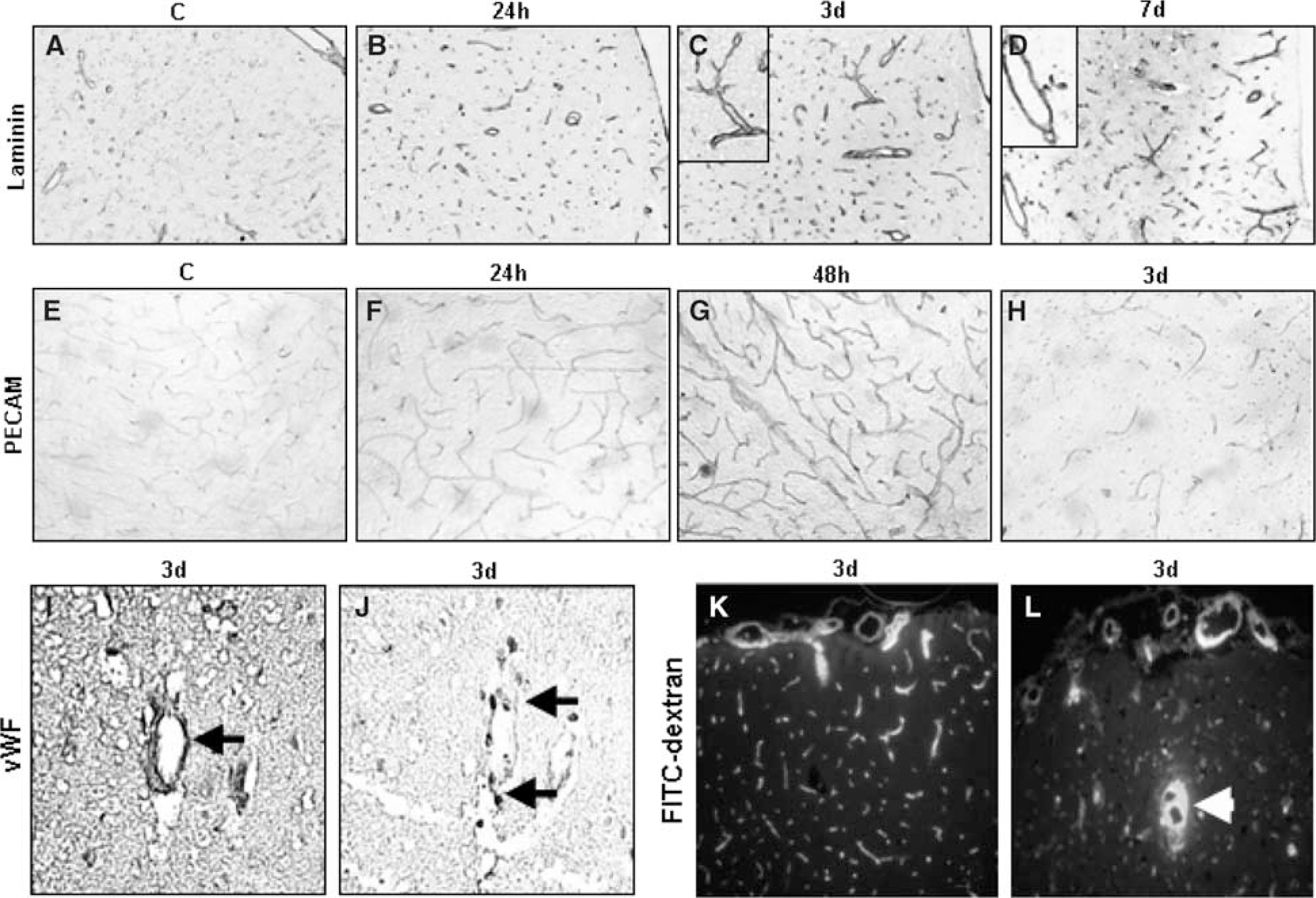
Immunohistochemistry for laminin (
Cerebral Ischemia Induces a Modulation of Angiogenic Factor Expression
To study if the cellular mechanisms of angiogenesis were related to a modulation of angiogenic factors involved in the formation of mature neovessels, we analyzed the time course of VEGF and Ang-1 expressions at mRNA level (Figure 2). The results revealed a significant induction of the two rodent VEGF isoforms (VEGF120 and VEGF164) in ischemic cortices as early as 3 h after occlusion in comparison with contralateral cortices. However, this expression declined from 3 days after occlusion to return to a basal level (Figure 2). The temporal expression of Ang-1 mRNA was inverted in comparison with that of VEGF. Indeed, Ang-1 mRNA expression firstly decreased up to 12 h after occlusion but increased from 24 h to 7 days after MCAO (Figure 2). The different expression patterns of these two major angiogenic factors may explain why, in response to cerebral ischemia, this process is initiated but does not lead to a robust and efficient neovascularization in the ischemic area. These observations raise the question whether an exogenous administration of angiogenic factors could stimulate angiogenesis leading to functional vascular structures in the ischemic brain.
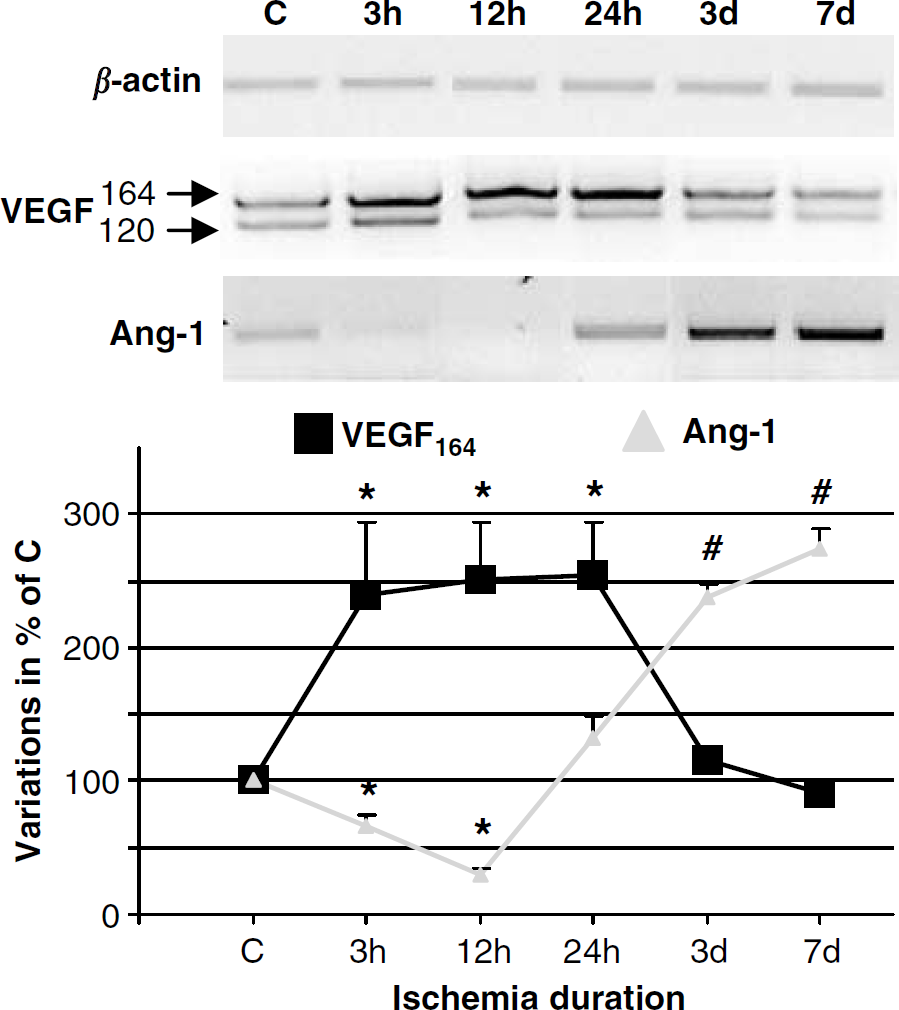
Representative time course of expression profiles of vascular endothelial growth factor (VEGF), Ang-1 in response to cerebral ischemia, obtained by RT-PCR from total RNA isolated from control cortex (C) or ischemic cortices at different time after occlusion (from 3 h to 7 days). β-Actin was used as housekeeping gene. Data represent the mean ± s.e.m. n = 3, *P < 0.05; #P < 0.01 versus control cortex by a Bonferroni-Dunn's post hoc test after a significant ANOVA (P < 0.001).
Ang-1 and Vascular Endothelial Growth Factor Display Synergic Angiogenic Effects in the Brain
Vascular endothelial growth factor, Ang-1, or VEGF/Ang-1 was chronically administrated in the striatum. After 7 days, the vascularization was examined by laminin immunostaining. An exogenous administration of VEGF (Figure 3B) or Ang-1 (Figure 3C) alone weakly increased the number of vessels present in the striatum compared with contralateral tissue (Figure 3A). In addition, only neovessels induced after the VEGF administration are enlarged compared with the basal vasculature or those obtained after Ang-1 infusion. In accordance with a synergic angiogenic effect described in the skin (Thurston, 2002), the coapplication of Ang-1 and VEGF leads to a frank neovascular network (Figures 3B and 3D) with vessels that have smaller diameter compared with those obtained with VEGF alone (Figures 3E and 3F).
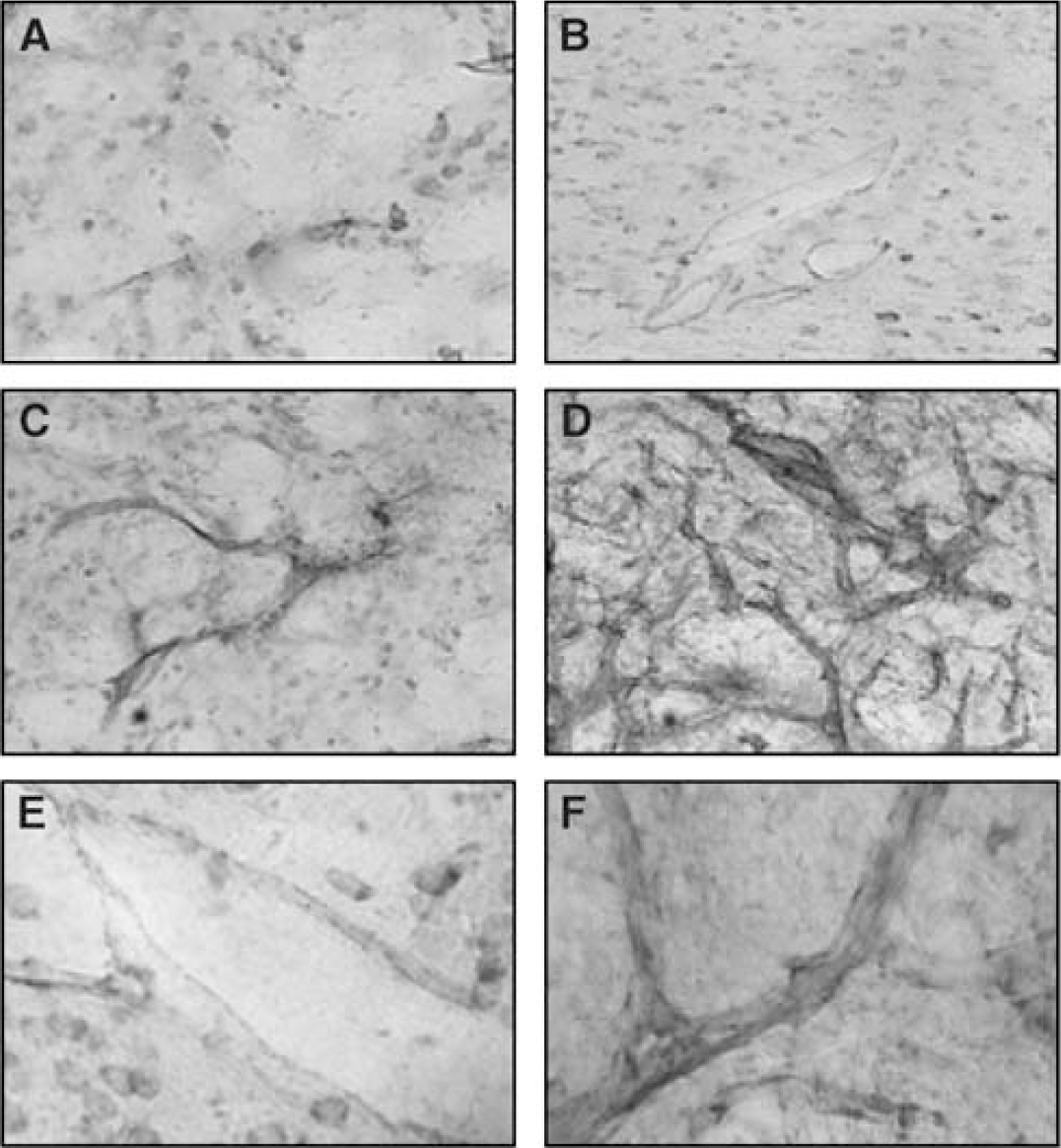
Immunohistochemistry for laminin on brain sections obtained from rats treated for 7 days with a chronic striatal administration of vehicle (
Ang-1 Counteracts the Deleterious Effects of Exogenous VEGF in Cerebral Ischemia
To test the potential benefit of exogenous administration of VEGF and Ang-1 in cerebral ischemia, mice were treated with VEGF or Ang-1 or a combination of VEGF/Ang-1, administrated intra-cerebroventricularly at the time of MCAO. Brain damage was estimated by measuring the cortical infarct volume (mean ± s.d.) 24 h after occlusion (Figure 4). Whereas VEGF injection doubled the ischemic area in comparison with control animals (50.1 ± 9.2 versus 27 ± 6.5 mm3, n = 7, P < 0.001) (Figures 4A and 4B), the administration of Ang-1 alone did not modify the infarct volume (Figure 4B). However, when Ang-1 was coadministrated with VEGF, Ang-1 fully counteracted the deleterious effect of the latter (Figure 4B). Because VEGF is also known to enhance vascular permeability, while Ang-1 displays antipermeability properties, edema volume was also evaluated 24 h after ischemia. Vascular endothelial growth factor injection increased the edema by 100% in comparison with control animals, whereas the presence of Ang-1 decreased VEGF-induced edema formation (Figure 4C). These deleterious effects of VEGF were, nevertheless, no more detectable when this angiogenic factor was injected 6 h after occlusion (data not shown).
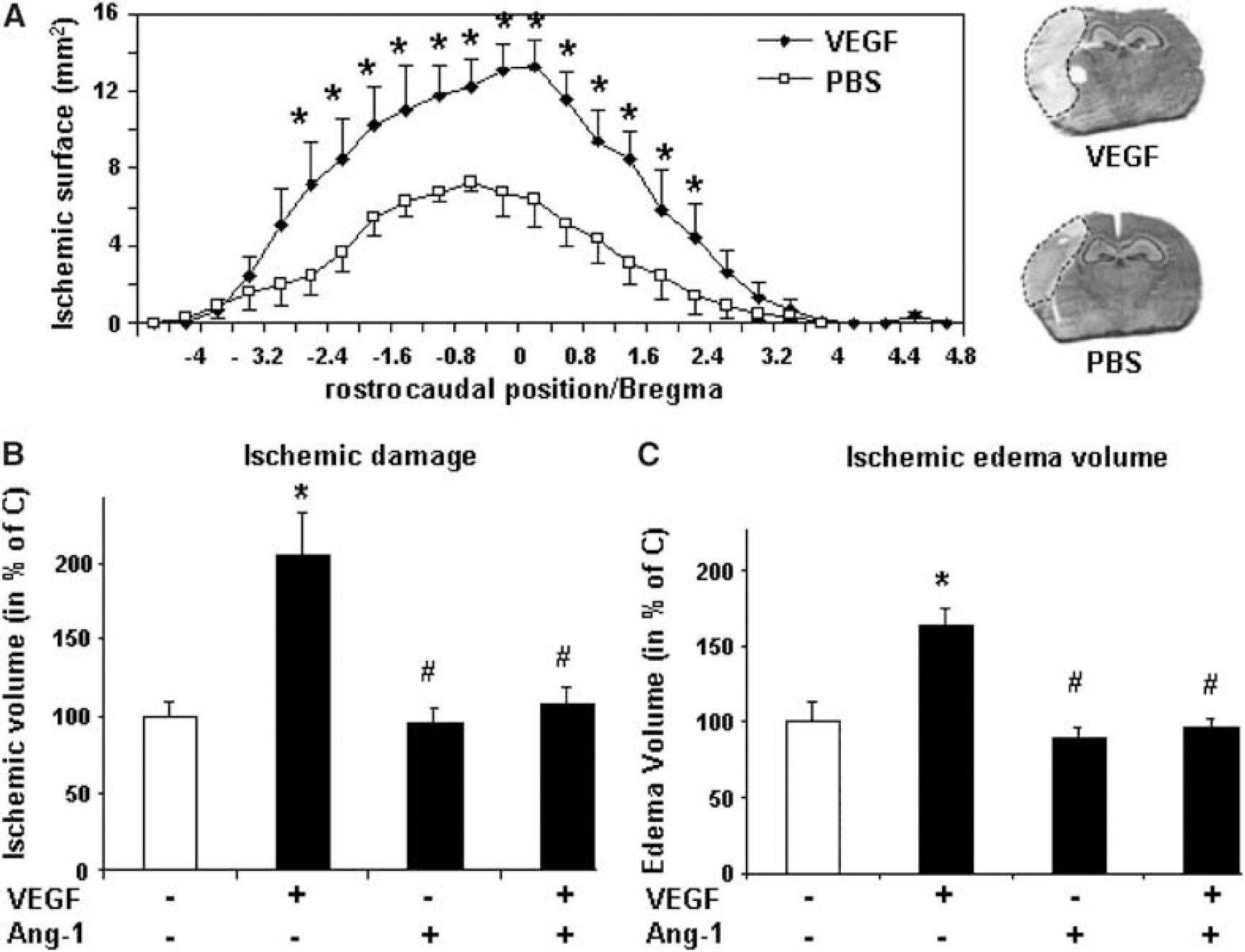
Effects of Ang-1 and vascular endothelial growth factor (VEGF) on ischemic damage. (
Ang-1 Counteracts Vascular Endothelial Growth Factor-Induced Permeability in the Ischemic Brain
We further investigated whether Ang-1 may modulate the vascular permeability induced by VEGF by assessing whether these angiogenic factors affect the BBB leakage. Mice received an intracerebroventricular injection of either vehicle, VEGF, Ang-1, or VEGF/Ang-1 at the time of ischemia induction. At 6 h after occlusion, BBB integrity was evaluated by quantifying the parenchymal Evans blue content. In vehicle-treated animals, the Evans blue content increased by around 50% in comparison with the contralateral cortex (Figure 5A). In agreement with the results obtained for ischemic edema evaluation, VEGF enhanced the BBB opening in the ischemic cortex in comparison with the contralateral cortex (Figure 5A). In contrast, a coadministration of Ang-1 and VEGF led to a decrease of vascular leakage (Figure 5A).
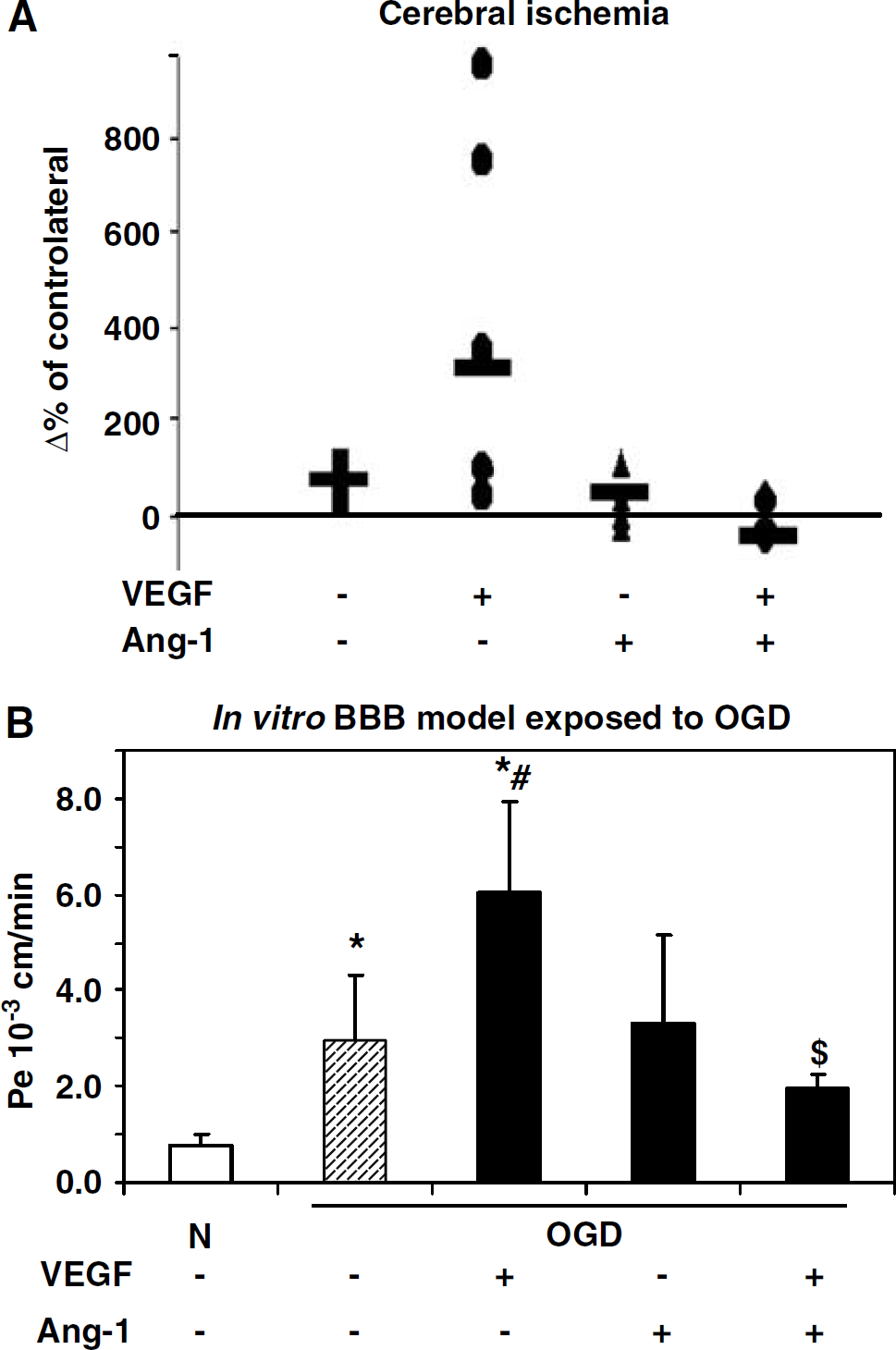
Effect of Ang-1 on vascular endothelial growth factor (VEGF)-induced permeability in ischemic conditions. (
These results were confirmed using an in vitro model of BBB placed in OGD to mimic ischemia (Jones et al, 2004; Pellegrini-Giampietro et al, 1999; Wiessner et al, 2000). Blood—brain barder permeability was evaluated by 14C-sucrose transport through the endothelial monolayer. Oxygen glucose deprivation induced a permeability of the in vitro BBB model (Figure 5B), which was partially reduced by the presence of Ang-1 applied in the luminal compartment. In OGD conditions, the presence of VEGF exacerbated this permeability but Ang-1 totally counteracted this effect (Figure 5B).
Matrix Metalloproteinases are Involved in Blood—Brain Barrier Opening after Cerebral Ischemia
Because MMP-2 and MMP-9 play an important role in the degradation of the basal lamina, a modulation in their activities may be responsible for BBB opening and consequently may contribute to cerebral damage (Rosenberg et al, 1998). To test this hypothesis, MMP-2 and MMP-9 activities were evaluated by gelatin-based zymography and the results were compared with the kinetic of BBB opening measured by extravasation of AIB. An uptake of 14C AIB into the brain parenchyma was observed early after MCAO but was only significant after 24 and 48 h of ischemia (Figure 6A). Matrix metalloproteinase activities were also analyzed for a similar time course after occlusion. As described in a previous study (Rosenberg et al, 1998), MMP-9 (gelatinase B, 92 kDa) activity was undetectable in contralateral cortices (Figure 6B). However, a raise of MMP-9 activity was clearly observed at 12 and 24 h after occlusion and decreased at 36 h. For MMP-2 (gelatinase A, 72 kDa), an activity was particularly observed at 24 h after ischemia. No modification of MMP-2 levels was observed over time in the contralateral cortices. Our results showed a modulation of MMP-9 expression, which paralleled the kinetics of the BBB opening in response to cerebral ischemia.
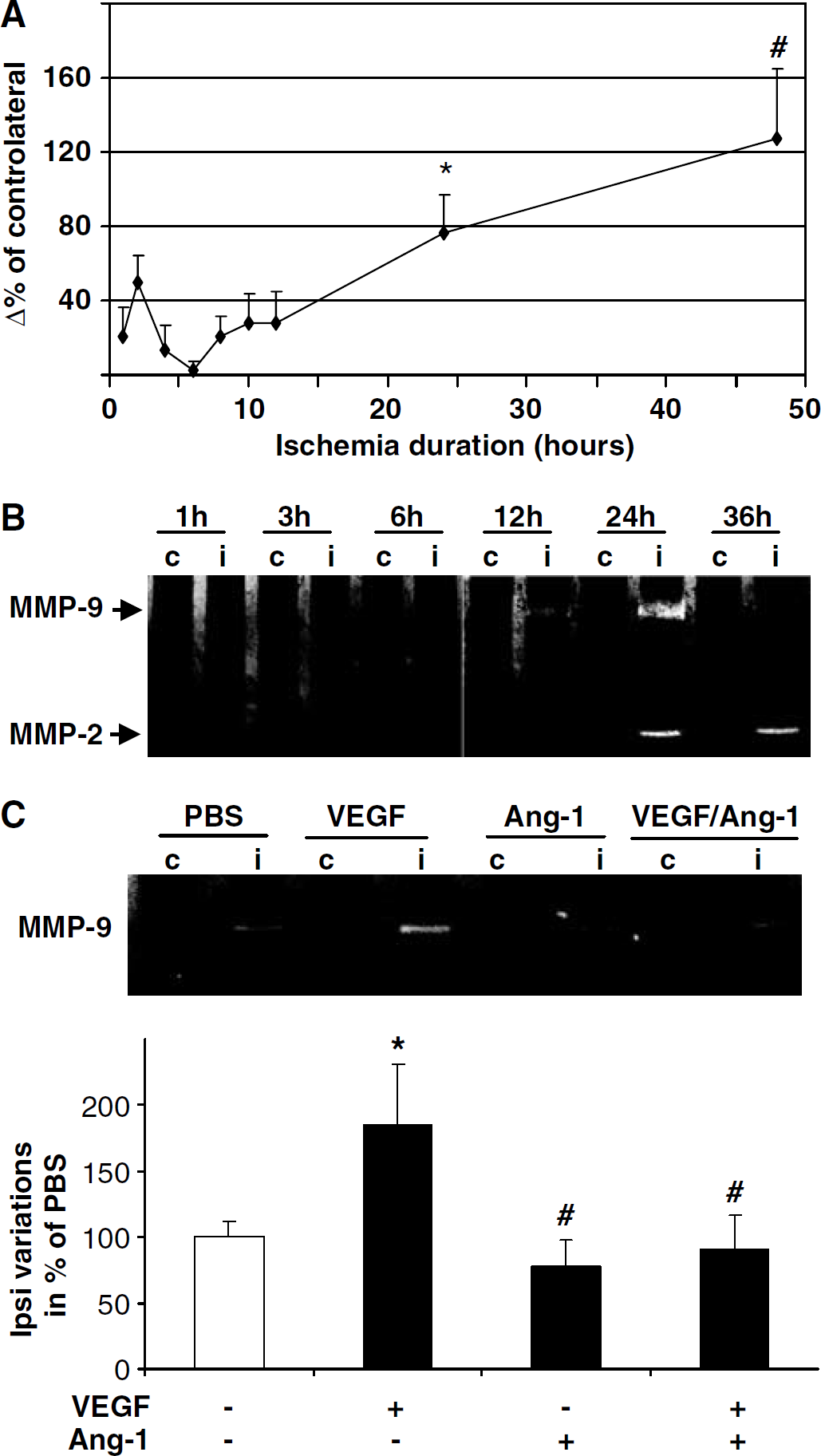
Effect of Ang-1, vascular endothelial growth factor (VEGF) and VEGF/Ang-1 on matrix metalloproteinase (MMP) activity in cerebral ischemia. (
Ang-1 Counteracts Vascular Endothelial Growth Factor-Induced Permeability in Association with a Modulation of Matrix Metalloproteinase-9 Activity
To elucidate the molecular mechanisms underlying the BBB alterations, we hypothesized that VEGF and Ang-1 might control the disruption of the BBB by modulating MMP activities. To confirm this assumption, we studied MMP-2 and MMP-9 activities on brain extracts obtained 12 h after ischemia from mice, treated at the moment of ischemia with an intracerebroventricular administration of either PBS, VEGF, Ang-1, or both. We observed that VEGF exacerbates MMP-9 activity induction (Figure 6C). Conversely, when coadministrated with VEGF, Ang-1 inhibited this gelatinase activity (Figure 6C). In this paradigm, no difference was observed for MMP-2 activity. These results underlined the involvement of VEGF and Ang-1 in MMP-9 activity pathway in ischemic conditions. We next tested whether identical mechanisms can take place in basal conditions. For this purpose, we tested the effect of VEGF, Ang-1, or VEGF/Ang-1 on the in vitro BBB model placed in normoxic conditions. Each culture well was concomitantly evaluated for MMP activities and BBB permeability, 6 h after incubation with angiogenic factors. The application of VEGF increases the BBB permeability and increases MMP-9 activity (Figures 7A and 7B), while the antipermeability effect of Ang-1 is associated with a reduction in MMP-9 activity (Figures 7A and 7B).
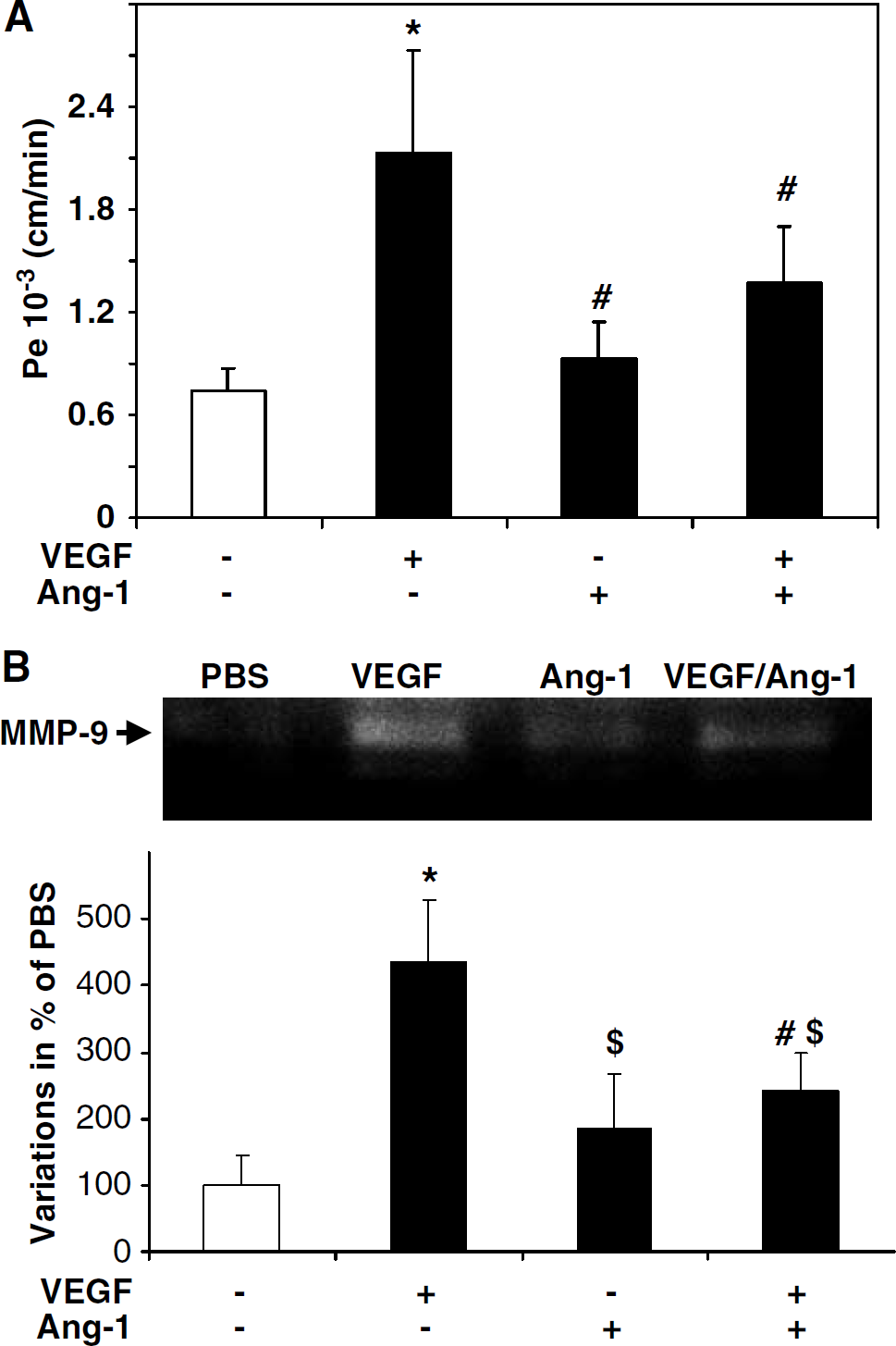
Effect of Ang-1, vascular endothelial growth factor (VEGF), and VEGF/Ang-1 on matrix metalloproteinase (MMP) activity in an in vitro model of blood—brain barrier (BBB) exposed to oxygen glucose deprivation (0GD). (
VEGF and MMP-9 Expression Correlate During the Acute Phase of Stroke
Our results suggest that MMP-9 participates in the physiopathology of cerebral ischemia. These results are supported by clinical data showing that, in human, MMP-9 levels are associated with neurologic deficits and infarct volume (Montaner et al, 2001a). In the present study, we investigated any possible correlation between VEGF and MMP system in the acute phase of ischemic stroke. Vascular endothelial growth factor and MMP-9 levels measured in plasma at baseline and at 12 h from stroke onset were higher in the study patients than a healthy control population (P < 0.001) (Figure 8). The temporal profiles of both molecules during the study period showed the highest level for both VEGF and MMP-9 at baseline (VEGF = 75.73 pg/mL (48.33 to 166.02); MMP-9 = 71.7 ng/mL (37.1 to 220.7)). Interestingly, a positive correlation exists between both molecules at any measured time point (r = 0.373, P = 0.043 at baseline; r = 0.554, P < 0.001 at 12 h, r = 0.403, P = 0.020 at 24 h, data not shown). The specificity and significance of the present findings is supported by the lack of correlation of VEGF or MMP-9 with a marker of general oxidative (hs-C-reactive protein) or a marker of vascular inflammation (ICAM-1). At baseline (hs-C reactive protein-VEGF: r = 0.085, P = 0.661; hs-C reactive protein-MMP-9: r = 0.280, P = 0.185; ICAM-1-VEGF: r = 0.169, P = 0.391; ICAM-1-MMP-9: r = −0.073, P = 0.707)); at 12 h (ICAM-1-VEGF: r = 0.219, P = 0.206; ICAM-1-MMP-9: r = 0.362, P = 0.030); at 24 h (ICAM-1-VEGF: r = 0.000, P = 1.000; ICAM-1-MMP-9: r = −0.054, P = 0.753).
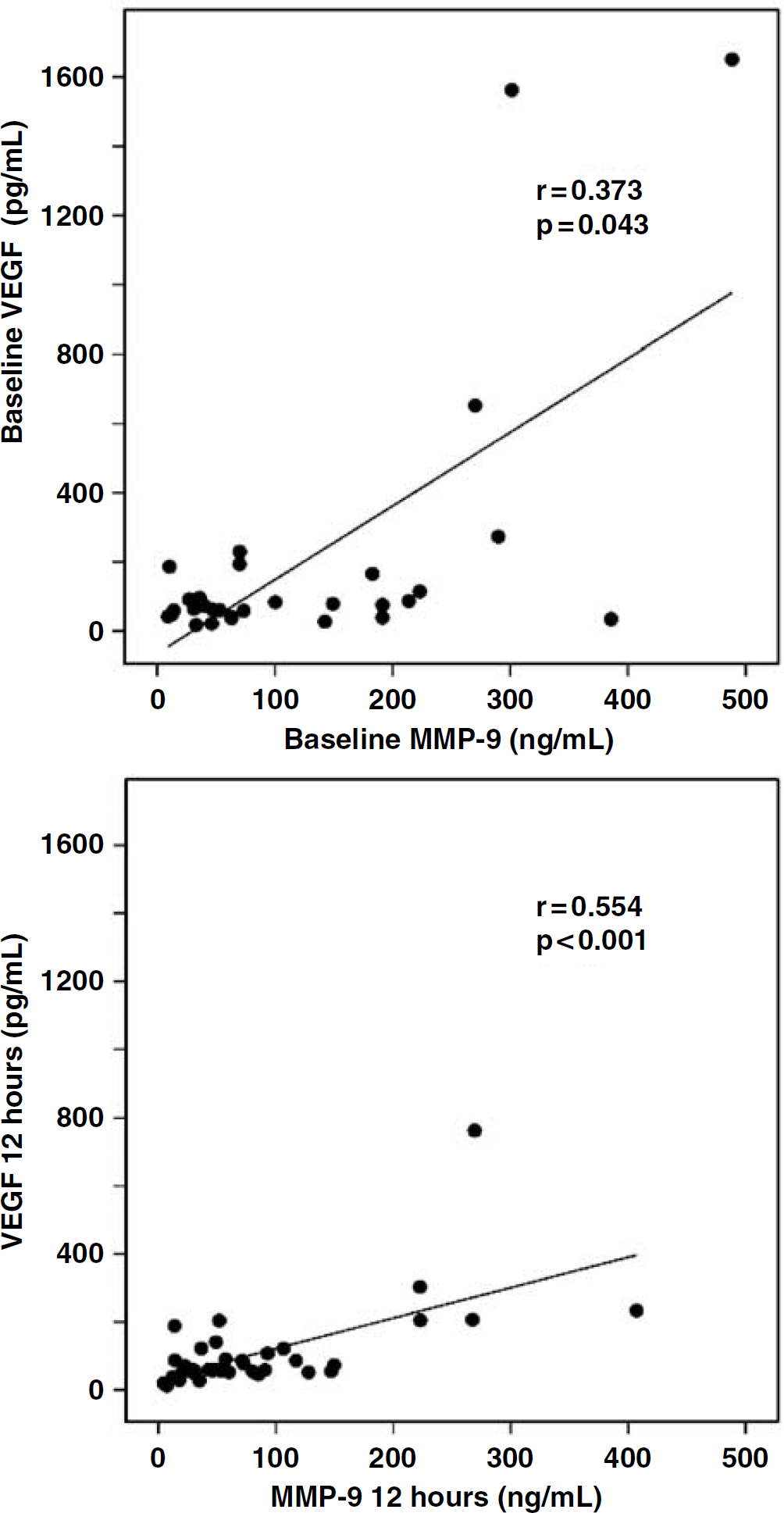
VEGF and matrix metalloproteinase (MMP)-9 correlation during the acute phase of stroke. Plasmatic VEGF and MMP-9 levels are evaluated from each patient at study entry and at 12 from stroke onset. Our laboratory reference values for healthy controls are: 12.47 ± 14.96 pg/mL for VEGF (n = 68, 70% females, mean age 43 years, normal range < 27.43 pg/mL) and 41 ± 27.8 ng/mL for MMP-9 (n = 62, 42% females, mean age 43 years, normal range < 97 ng/mL). The mean intraassay coefficients of variation were < 10%. The Mann—Whitney U-test was used to assess differences between patients and healthy controls. Spearman's rank correlation test was used to test correlations.
Discussion
In the current study, we show that, in cerebral ischemia, exogenous VEGF enhances BBB disruption but Ang-1 is able to antagonize this VEGF deleterious effect, in association with an inhibition of MMP-9 activity. Our results show that in ischemic animals, an administration of VEGF leads to BBB permeability as to an induction of MMP-9 activity. Conversely, the coadministration of Ang-1 and VEGF blocks the BBB permeability and reduces MMP-9 activity. Furthermore, we observe that in combination, these two factors display synergic angiogenic effects in the brain. Collectively, these results indicate that a combination of Ang-1 and VEGF may be more beneficial than VEGF alone to promote vascularization in cerebral ischemia.
Stroke induces angiogenesis, particularly in the ischemic penumbra, which correlates with longer survival in humans (Krupinski et al, 1994). However, the natural course of angiogenesis is not sufficient to fully compensate for the hypoperfusion ischemic state. Therefore, novel therapeutic strategies attempt to enhance this vascular process. Angiogenic growth factors stimulate the development of collateral vascularization in animal models of peripheral ischemia, a concept called therapeutic angiogenesis (Baumgartner et al, 1998; Kastrup, 2003). For instance, the efficacy of therapeutic angiogenesis using VEGF gene transfer has been reported in human patients with critical limb ischemia or myocardial infarction (Vale et al, 2001). Thus, the strategy for therapeutic angiogenesis using angiogenic growth factors should be considered for the treatment of patients with cerebral ischemia. Several angiogenic growth factors, such as fibroblast growth factor, hepatocyte growth factor, and VEGF prevent the extension of focal cerebral ischemic injury in animal models (Li and Stephenson, 2002; Sun et al, 2003; Tsuzuki et al, 2001). However, in the progression to human clinical therapy, optimal paradigms to promote angiogenesis, but also side effects of the selected molecules, should be evaluated.
Although angiogenesis has been commonly suggested to contribute to brain repair in stroke, only few studies characterized this process in animal ischemia models (Manoonkitiwongsa et al, 2001; Sbarbati et al, 1996; Wei et al, 2001). Our study contributes to describe the temporal evolution of vascular remodeling in relation with the expressions of the two central mediators of angiogenesis, VEGF, and Ang-1. Our results show that, in focal permanent cerebral ischemia, angiogenesis is initiated as early as 24 h. A few neovessels are observed in the ischemic area up to day 3; however, some of them are enlarged and leaky. More importantly, these neovessels tend to disappear at 7 days after occlusion.
The expression pattern of angiogenic factors that we described in response to cerebral ischemia may explain, in part, that angiogenesis is not sustained in the infarcted area. Indeed, in accordance with our previous data (Marti et al, 2000), the present study shows that VEGF expression is induced precociously but is not sustained after cerebral ischemia. This transient expression of VEGF may be related to the appearance of neovessels in the ischemic tissue observed up to day 3 after occlusion. However, from day 3, VEGF mRNA decreased and vessels disappeared in the ischemic area. However, a biphasic expression of Ang-1 was observed. Ang-1 mRNA expression firstly decreased but secondarily increased from 7 days after occlusion. This expression kinetic may explain the leaky vessels detected at 3 days after occlusion but also reflects the fact that Ang-1 exerts its function at later stages of vascular remodeling. The kinetic profile of VEGF and Ang-1 expressions that we characterized in permanent cerebral ischemia in mice closely resembles that described on transient cerebral model in mice or rats (for a review see Croll and Wiegand, 2001).
After stroke, a disruption of the BBB occurs acutely and contributes to the development of injury. Free radicals, proteases, and cytokines may mediate the alteration of capillary integrity (Rosenberg et al, 1998). Among cytokines, our results, in accordance with those of a recent study, suggest that VEGF and Ang-1 expressions may be related not only to angiogenesis but also to BBB disruption after focal cerebral ischemia (Zhang et al, 2000). In addition, recombinant VEGF promotes angiogenesis but also enhances BBB permeability, hemorrhage, and ischemic lesions (Zhang et al, 2000). Thus, it is noteworthy to delineate the effects of these angiogenic factors on BBB integrity.
The kinetics of BBB opening has been particularly characterized on transient ischemia and to a lesser extent on permanent models. Ischemia/reperfusion causes a biphasic opening of the BBB corresponding to an initial acute disruption of the BBB occurring between 3 and 5 h after MCAO, and a later, more widespread increase in BBB permeability present at 48 h (Belayev et al, 1996; Rosenberg et al, 1998). In this model of focal permanent ischemia, the BBB opening, evaluated by parenchymal extravasation of AIB, was significantly detected at 24 h. This result is consistent with a previous report that described a BBB breakdown in rat permanent ischemia, detected at 12 h by evaluating albumin transfer (Hatashita and Hoff, 1990). Although insignificant, we also measured an acute AIB uptake between 1 and 4 h after occlusion, which may be because of an increase of transcellular-transport in the early period after ischemia, as it has been proposed for tissue-type plasminogen activator (tPA), an effect reported to be independent of the vascular basement membrane degradation (Yepes et al, 2003).
With therapeutic angiogenesis for the treatment of cerebral ischemia as a goal, we evaluated the effects of an administration of VEGF and Ang-1 in the brain. We showed that a coapplication of Ang-1 and VEGF leads to a frank neovascular network, with a great number of vessels thinner than those obtained with VEGF alone in accordance with a synergic angiogenic effect of VEGF and Ang-1 described in the skin (Thurston, 2002). Beside these beneficial effects, we examined whether these factors displayed deleterious side effects in stroke. An acute cerebral administration of VEGF at the time of MCAO doubles the ischemic area in comparison with control animals, 24 h after occlusion. Furthermore, according to the permeability properties of VEGF, we observed that this factor exacerbates ischemic edema and enhances trans-endothelial sucrose transport on an in vitro BBB model placed in ischemic mimicking conditions (OGD). However, we showed that a coadministration of Ang-1 blocked not only the VEGF-induced BBB permeability as similarly reported (Zhang et al, 2002b) but also the VEGF-induced ischemic damage. These results raised the question of whether VEGF and Ang-1 have direct or indirect effects on the infarcted tissue. Although in the present work, we did not evaluate the biodisponibility of these cytokines, a recent study (Storkebaum et al, 2005) shows that intracerebroventricular administration is a suitable route for the brain by showing that, on systemic delivery, VEGF is rapidly trapped by its soluble receptor Flt-1, whereas after intracerebroventricular delivery, VEGF diffuses and accumulates into the parenchyma until at least 24 h after injection.
We were next interested to identify mechanisms by which Ang-1 counteracts VEGF effects. In cerebral ischemia, previous studies suggested that BBB disruption is because of a proteolytic degradation of the vascular membrane. Among proteases, MMPs, in particular MMP-2 and MMP-9, are able to digest the endothelial basal lamina and therefore may play a major role in promoting BBB permeability. The modulation of MMP-2 depends on model of cerebral ischemia. Some studies report a significant elevation in MMP-2 by 24 h (Romanic et al, 1998) whereas others do not detect any significant changes before 5 days after ischemia (Rosenberg et al, 1998). In our model, the main significant induction of gelatinase activities was observed for MMP-9 as previously reported (Asahi et al, 2001; Fukuda et al, 2004; Gasche et al, 1999; Heo et al, 1999; Mun-Bryce and Rosenberg, 1998; Rosenberg et al, 1996). This significant induction of MMP-9 activity was first observed at 12 h of permanent ischemia, whereas changes in BBB permeability were significantly detected at 24 h after occlusion.
In angiogenesis, vascular destabilization and sprouting are two steps of cellular processes, which may be modulated by MMPs. Regarding the angiogenic effects of VEGF and Ang-1, several studies have evaluated the role of these molecules in the regulation of gelatinases during angiogenesis. In vitro, VEGF increases both expression and release of MMP-2 and MMP-9 (Lamoreaux et al, 1998; Rooprai et al, 2000; Wang and Keiser, 1998). More recently, Ang-1 have been also described to interfere with MMP-2 and MMP-9 expressions (Das et al, 2003; Kim et al, 2000).
By using an exogenous administration, we showed that VEGF effects are associated with MMP-9 induction and that Ang-1 effects are associated with inhibition of this gelatinase activity. Taken together, these results suggest an association between MMP-9 activity, VEGF and Ang-1. These data are supported by those obtained in the present study on human which show a correlation between VEGF and MMP-9 levels in stroke patients but also by those obtained in a previous study demonstrating an association between MMP-9 overexpression, infarct volume and stroke severity (Montaner et al, 2001a, b).
In ischemic animals, an administration of VEGF leads to an increase in BBB permeability concomitant with an induction of MMP-9 activity. Conversely, the coadministration of Ang-1 and VEGF leads to a reduction of both BBB permeability and MMP-9 activity. These results were also observed on the in vitro BBB model exposed to OGD, reinforcing the idea of a relation between angiogenic factors, modulation of MMP activity and BBB alteration. As in ischemic conditions, in normoxia, Ang-1 inhibits concomitantly VEGF-induced permeability and MMP-9 activity induction. The mechanisms by which Ang-1 reduces BBB alteration in the ischemic brain are not yet fully understood. Taken together, our data suggest that MMP-9 is a common pathway in the BBB effects of VEGF and Ang-1. Nevertheless, we can not rule out that others mechanisms might be involved such as a regulation of the tight junction proteins. Indeed, Ang-1 antipermeability effect and VEGF permeability effect have been ascribed, in vitro, to a regulation of the expression of ZO-1 (zonae occludens) and occludin. It is therefore likely that in future, strategies that enhance both early reperfusion and neuroprotection will be investigated. Following this hypothesis, VEGF and Ang-1, by leading to a vascular beneficial effect but also to a neuroprotective effect are of interest. Indeed, an application of VEGF, after transient arterial occlusion, significantly reduces ischemic brain damage and enhances angiogenesis but also neurogenesis in the ischemic area with a significant improvement in neurological recovery (Sun et al, 2003). Furthermore, recent in vitro studies underline the possibility that VEGF may protect neurons against excitotoxic or apoptotic stresses (Jin et al, 2000; Matsuzaki et al, 2001). Concerning Ang-1, we also previously reported, in vitro, a neuroprotective effect of this angiogenic factor against an apoptotic stress (Valable et al, 2003). Thus, Ang-1, in combination with VEGF, is a candidate for treatment of cerebral ischemia. We propose that, besides their antiapoptotic effects on both endothelial cells (Gerber et al, 1998; Papapetropoulos et al, 2000) and neurons (Jin et al, 2000; Valable et al, 2003), a coadministration of VEGF and Ang-1 could be used early to stimulate a functional angiogenesis without inducing side effects on BBB permeability. Based on the above data and on our findings, a novel therapeutic approach based on an early administration of both angiogenic factors leading to a limitation of the extent of neuronal death might be encouraged for the treatment of cerebral ischemia. However, it has been shown that neuroprotection and angiogenesis induced by VEGF monotherapy do not necessarily occur at the same dose of exogenous VEGF (Manoonkitiwongsa et al, 2004). Consequently, it would be necessarily, in the future, to investigate the role of endogenous VEGF and Ang-1 and to determine whether an optimal parenchymal concentration of both agents can safely stimulate both angiogenesis and neuroprotection in relation with an increase in functional outcome (Hori et al, 2004; Iizasa et al, 2002; Wang et al, 2001). However, Ang-1 might be also of interest for thrombolytic therapy for stroke. Indeed, Lo's group shows that, in cerebral ischemia, tPA can increase MMP-9 (Wang et al, 2003). Accordingly, by down-regulating MMP-9 activity, Ang-1 in combination with tPA might reduce tPA-induced BBB deleterious effects and increase the therapeutic window for tPA in stroke (Morris et al, 2001; Zhang et al, 2002a).
Footnotes
Acknowledgements
The authors thank Denis Viven, PhD, for advices and helpful discussions.
