Abstract
The precise biologic mechanisms involved in functional recovery processes in response to stroke such as dopaminergic neurotransmission are still largely unknown. For this purpose, we performed in parallel
Keywords
INTRODUCTION
Stroke, a leading cause of mortality and serious long-term disability in adults,
1
is the result of a prolonged interruption of blood flow in a cerebral vessel. Brain activity is dramatically sensitive to the decrease in blood supply leading to a loss of function in the vascular territory. Brain dysfunction underlying stroke may be triggered by well-known pathophysiological mechanisms such as excitotoxicity,
2
inhibition of protein synthesis,
3
oxidative stress,
4
apoptosis,
5
brain barrier breakdown,
6
and inflammation.
7
Despite the loss of brain function early after stroke, a behavioral recovery in response to brain injury has been described after stroke in both humans
8
and animals.
9
Some of the mechanisms suggested for the functional recovery include neo-formation of brain vessel, brain plasticity, and functional reorganization through recruitment of new brain regions and neuromodulation to the opposite hemisphere.
10
Recently, postischemic thalamic hyperperfusion has also been proposed as a novel type of remote plasticity, which might support removal of necrotic brain tissue and aid functional recovery.
11
Supporting these findings, Martin
Positron emission tomography (PET) with [ 11 C]raclopride has been widely used to investigate the availability of striatal dopamine D2 receptors 20 related to several neurologic and psychiatric disorders. This imaging method has been proven to be helpful in elucidating the role of dopaminergic mechanisms in pathologies such as schizophrenia, 21 Huntington's disease, 22 and Parkinson's disease. 23 Nevertheless, the role of dopaminergic neurotransmission in stroke evolution using PET imaging has been scarcely explored to date.
The purpose of the present study was to investigate the late postischemic dopamine D2 receptor changes in the rat brain after transient cerebral ischemia using PET with [ 11 C]raclopride. In particular, we were interested in clarifying the relationship of the dopaminergic system with (1) cerebral glucose metabolic activity and (2) the recovery of long-term brain function underlying experimental stroke. The results reported here might have significant practical importance as they could provide novel information about the neurotransmitters involved in the recovery of brain function and may ultimately contribute to the development of new treatments and imaging biomarkers for the evaluation of brain plasticity after stroke.
MATERIALS AND METHODS
Animals and Surgery
Adult male Sprague-Dawley rats (300 g body weight; Janvier, France) (
Magnetic Resonance Imaging
Magnetic resonance imaging (MRI) experiments were performed to examine the extent of brain infarction in those animals included in the nuclear studies. T2-weighted (T2W) MRI scans were performed in ischemic animals (
Magnetic Resonance Imaging Image Analysis
The T2W images were used to calculate the lesion volume. Regions of interest were manually defined using the Open Source software 3D Slicer image analysis software (Version 3.6.3; www.slicer.org) for each rat on the region of increased signal in the ipsilateral hemisphere. The volume of the infarcted region in a slide was calculated by multiplying the area of the delimited region by the slice thickness. The total lesion volume was calculated by summing the volume of the infarcted region of all slices affected by the lesion.
Radiochemistry
For the production of [
11
C]raclopride, [
11
C]CH4 was directly generated in an IBA Cyclone 18/9 cyclotron and transferred to a TRACERlab FXC Pro synthesis module (GE Healthcare, Waukesha, WI, USA) where [
11
C]methyl iodide was generated. At the end of the process, [
11
C]CH3I was distilled under continuous helium flow (20 mL/min) and introduced in a 2-mL stainless steel reaction loop, precharged with a solution of
Positron Emission Tomography Scans and Data Acquisition
The PET scans were repeatedly performed in ischemic (
Positron Emission Tomography Image Analysis
The PET images were analyzed using PMOD image analysis software (PMOD Technologies Ltd, Zürich, Switzerland). To verify the anatomic location of the signal, PET images were coregistered to the anatomic data of an MRI rat brain template. Two type of volumes of interest (VOIs) were established as follows: (1) A first set of VOIs was defined to study the evolution of metabolism by using [ 18 F]FDG. A VOI was manually defined in the entire ipsilateral hemisphere that contained the cortical and striatal regions and then mirrored to the homologous contralateral hemisphere using an MRI (T2W) rat brain template. The mean value and the standard deviation (mean ± standard deviation) of each VOI corresponding to the uptake at 30 minutes for [ 18 F]FDG was expressed as percentage of injected dose per body weight (%ID/g). The % of ID/g for both ischemic (ipsilateral) and the homologous contralateral region, as well as the ipsi-to-contralateral ratio was considered. (2) A second set of VOIs was automatically generated in striatums and cerebellum, by using the regions proposed by the PMOD rat brain template, to study the evolution of [ 11 C]raclopride binding potential (BP) after cerebral ischemia. The simplified reference-tissue model 26 from the PMOD software was used to assess BP of the tracer to the tissue in both ipsilateral and contralateral striatums. This model relies on a two-tissue reversible compartment for a target region (here ipsilateral or contralateral VOI) and a single-tissue compartment for a reference region (here cerebellum). The BP values calculated by the simplified reference-tissue model are referred as BPND (nondisplaceable), which is the BP of the tracer to the tissue and refers to ratio of specifically bound radioligand to that of nondisplaceable radioligand in tissue at equilibrium.
[ 3 H]raclopride Binding
A group of 32 rats were used at control (
Immunohistochemistry
Immunohistochemical staining was performed for control and at day 7 after ischemia. Animals were terminally anesthetized and killed by decapitation, and the brain was removed from the skull. The brains were frozen and 5-
Assessment of Histologic Damage
Brain damage was evaluated using Hematoxyline-eosine (H-E) and 2,3,5-triphenyltetrazolium chloride (TTC) staining at 1 and 7 days, respectively. For H-E stain, rat brains were frozen and 10-
Neurologic Assessment
The assessment of deficits and neurofunctional recovery induced by cerebral ischemia was based on previously reported tests. 27 Four consecutive tests were performed on every ischemic animal at 1, 3, 7, 14, 21, and 28 days after MCAO and at control (nonoperated) rats as follows: (1) spontaneous activity (moving and exploring = 0, moving without exploring = 1, and no moving or moving only when pulled by the tail = 2); (2) left drifting during displacement (none = 0, drifting only when elevated by the tail and pushed or pulled = 1, spontaneous drifting = 3, circling without displacement, or spinning = 4), (3) parachute reflex (symmetrical = 0, asymmetrical = 1, and contralateral forelimb retracted = 2), and (4) resistance to left forepaw stretching (stretching not allowed = 0, stretching allowed after some attempts = 1, and no resistance = 2). Total score could range from a 0- (normal) to a 9- (highest handicap) point scale.
Statistical Analyses
Behavioral data have been compared with Mann–Whitney
RESULTS
Glucose metabolism and dopamine D2 receptor binding were explored by PET imaging after a 2-hour MCAO in rats. All the images were quantified in standard units, i.e., percent of the injected dose per volume tissue for [ 18 F]FDG and BP for [ 11 C]raclopride PET. Hence, the measurements made at different time points in the VOIs of the ischemic area and the mirror contralateral area can be compared directly with the measurements made in control animals. The images with normalized color scales illustrate the evolution of the signal over time (Figure 1).
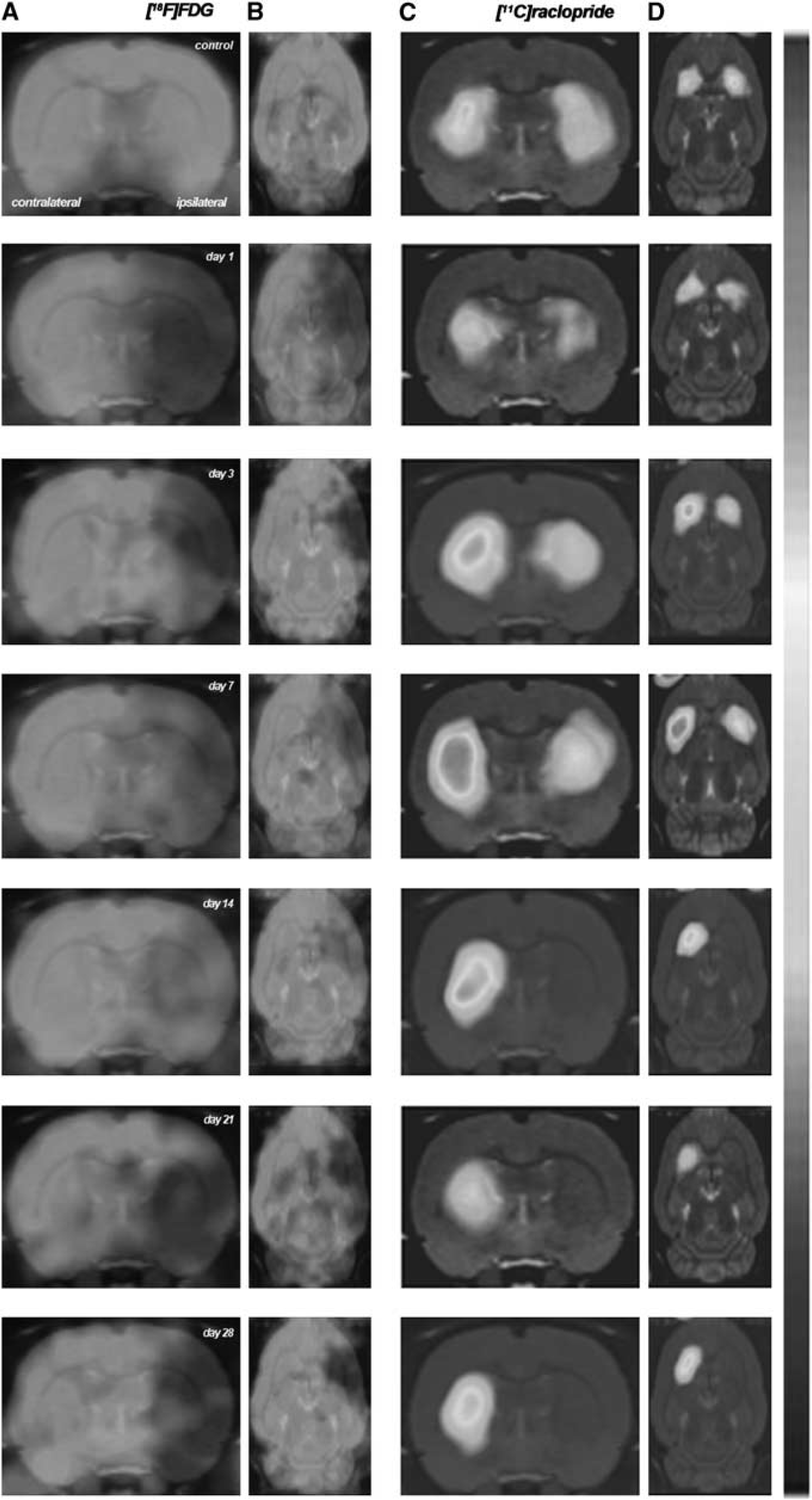
Serial images of [
18
F]fluorodeoxyglucose ([
18
F]FDG) and [
11
C]raclopride positron emission tomography (PET) at control, day 1, day 3, day 7, day 14, day 21, and day 28 after middle cerebral artery occlusion (MCAO). Normalized axial and coronal PET images of [
18
F]FDG (
Brain Damage Assessment After Cerebral Ischemia
The extent of brain damage after cerebral ischemia was assessed using T2W MRI at 1 day after ischemia onset. Hyperintensities of T2W images showed similar infarct extents as well as locations affected. All ischemic rats subjected to nuclear studies showed corticostriatal MRI alterations (mean ± s.d. = 290 ± 55 mm3).
[ 18 F]fluorodeoxyglucose Positron Emission Tomography After Middle Cerebral Artery Occlusion
The time course of cerebral glucose metabolism was evaluated using [
18
F]FDG PET at 1, 3, 7, 14, 21, and 28 days after MCAO (Figure 2). The [
18
F]FDG PET signals were significantly lower (–50%) in the cerebral territory of the occluded MCA than in the same region of control animals at day 1 after reperfusion. This was followed by a slight increase in metabolism to 25% of day 1 at days 7 to 14 that did not reach control values. Subsequently, [
18
F]FDG PET signal decreased again from day 21 to 28 after MCAO (
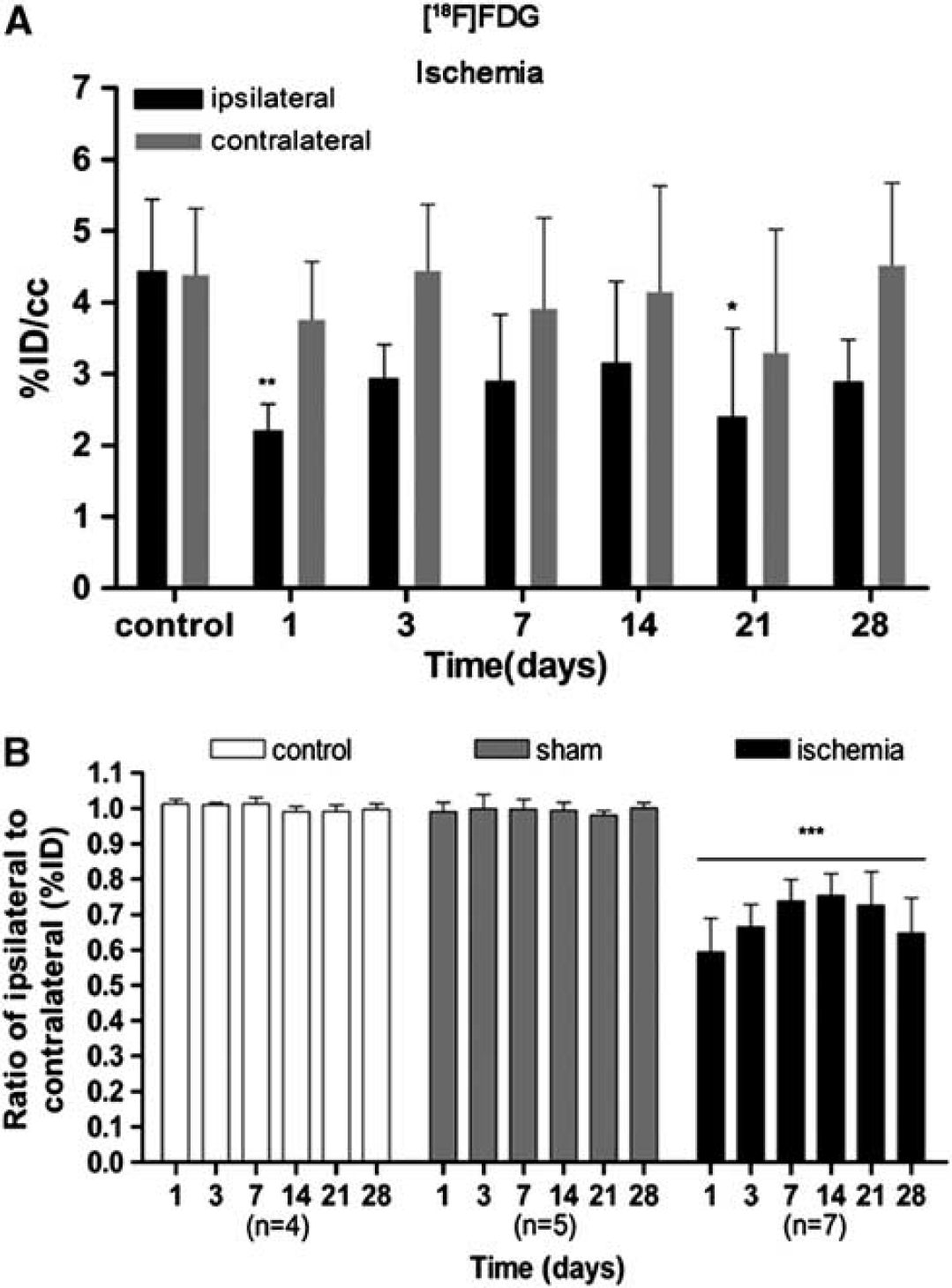
Time course of the progression of the [
18
F]fluorodeoxyglucose ([
18
F]FDG) positron emission tomography (PET) signal after cerebral ischemia. The percentage of injected dose per cubic centimeter of [
18
F]FDG (%ID/cc, mean ± s.d.) was quantified in two volumes of interest (VOIs) defined for each animal over the entire ipsilateral (containing the infarcted area) and the contralateral hemispheres. Rats were repeatedly examined by using PET at 1, 3, 7, 14, 21, and 28 days after ischemia or sham operation. Nonoperated rats were also repeatedly scanned at the same time points and were used as controls. The ipsilateral and contralateral PET values within the ischemic group are shown in (
The ratio of metabolic values in the ipsilateral (occluded MCA) over contralateral (nonoccluded MCA) showed a statistically significant decrease in the signal in the ischemic group in relation to control groups (nonoperated and sham-operated rats) (
Time course of [ 11 C]raclopride Positron Emission Tomography after Middle Cerebral Artery Occlusion
The time course of striatal dopamine D2 receptor was evaluated using [
11
C]raclopride PET at 1, 3, 7, 14, 21, and 28 days after MCAO (Figure 3A). The BPND of [
11
C]raclopride showed quasi-normal values from day 1 to day 7 after reperfusion in the striatum occluded by the MCA. After 7 days, however, [
11
C]raclopride PET signal decreased sharply from 75% at day 14 to ca. 80% at days 21 to 28 of the control values (
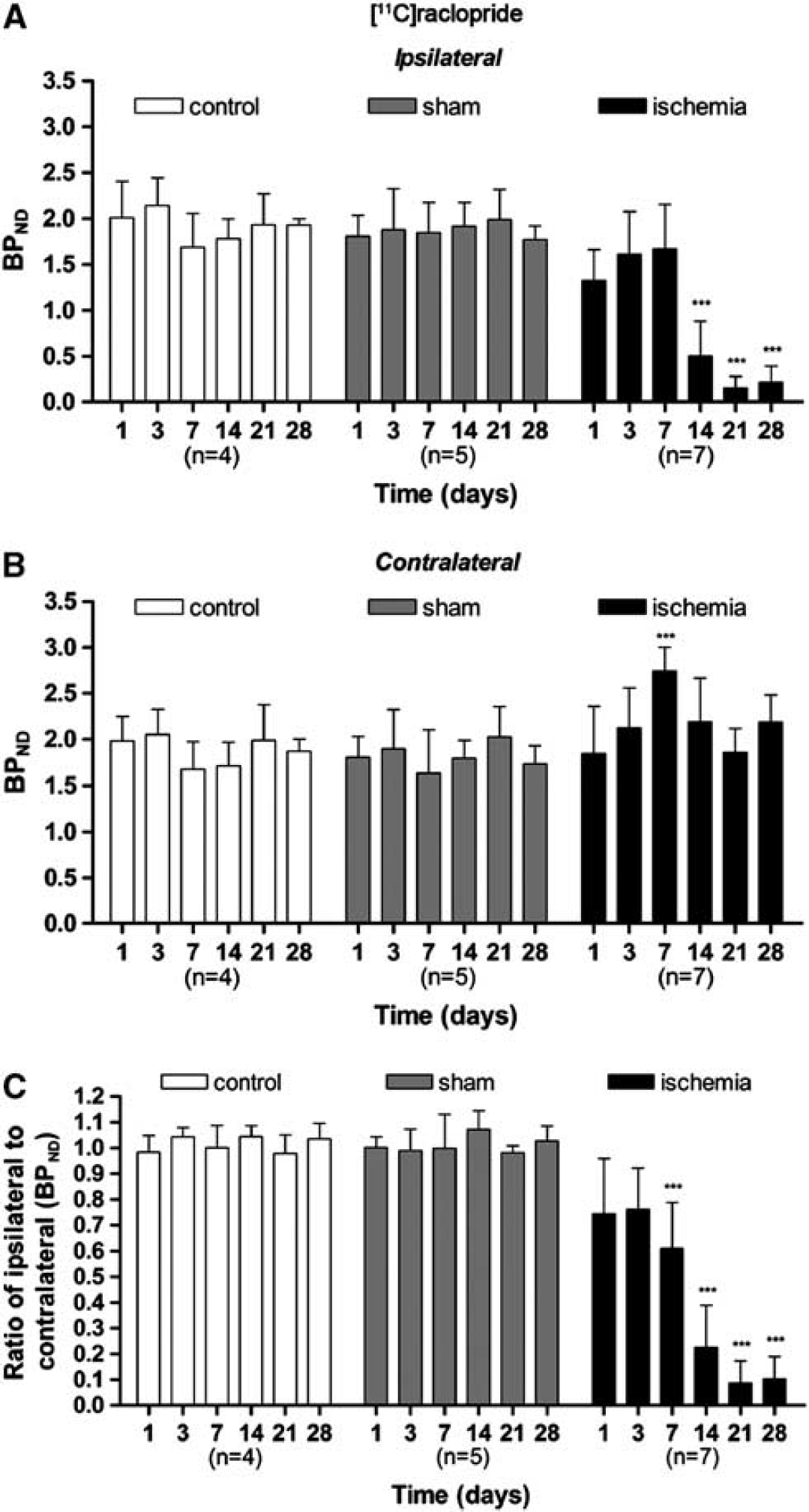
Time course of the progression of the [
11
C]raclopride positron emission tomography (PET) signal after cerebral ischemia. The binding potential (BP) nondisplaceable of [
11
C]raclopride (BPNP, mean ± s.d.) was quantified in two volumes of interest (VOIs) defined automatically for each animal over the entire ipsilateral (
Interestingly, changes in [
11
C]raclopride PET signal were also observed in the contralateral, nonischemic area. Contralateral BP of [
11
C]raclopride showed control values at 1 day after reperfusion, then PET signals rose steadily at day 3, and overshoot to ca. 40% of control values at 7 days after MCAO (
Accordingly, the ratio of BPND values in the ipsilateral (occluded MCA) over contralateral (nonoccluded MCA) areas varied from 0.7 at days 1 and 3 to 0.6 at day 7 after MCAO. By day 14, [
11
C]raclopride PET signal ratios decreased again from 0.2 to 0.1 at day 14 to day 21 (
Autoradiography
The time course of [
3
H]raclopride binding tissue was performed with brain sections of rats killed at 1, 3, 7, 14, 21, and 28 days after the induction of cerebral ischemia, to verify the results obtained
[
3
H]raclopride signal in the ischemic striatum considerably decreased at days 14, 21, and 28 with respect to control (
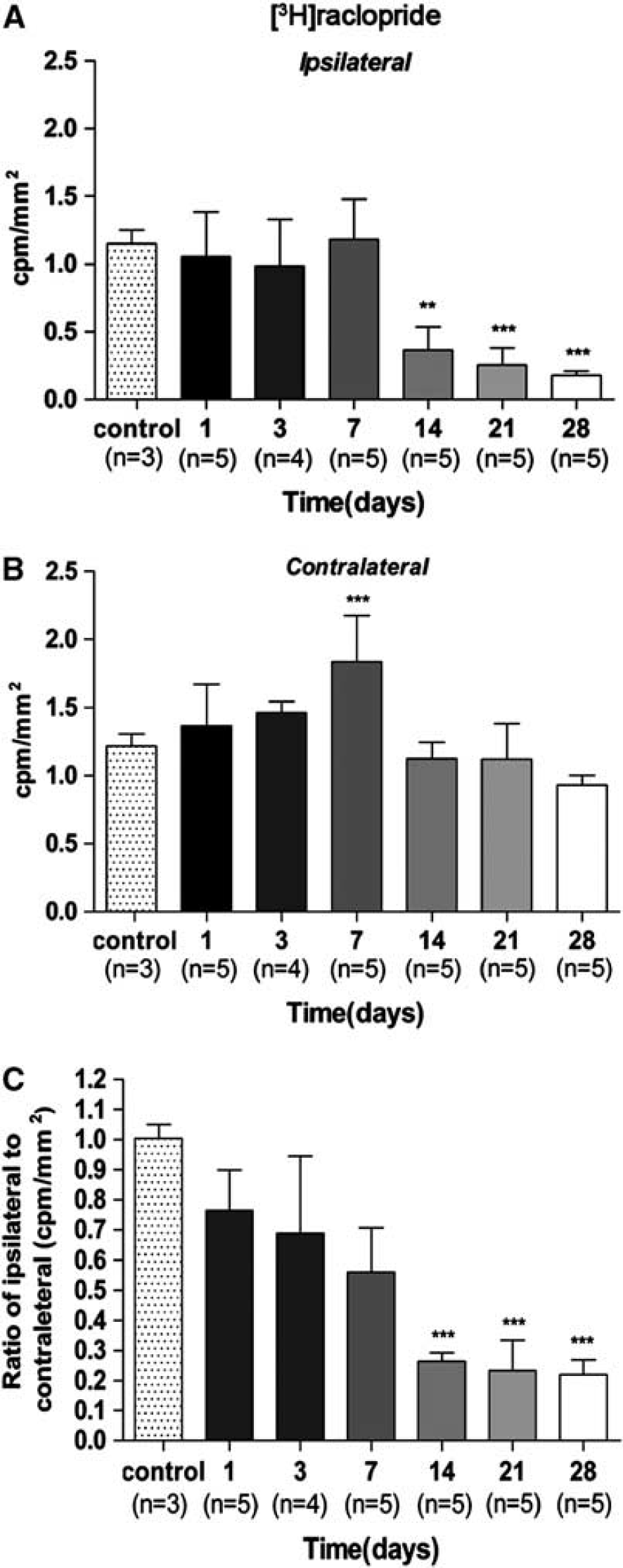
Time course of the progression of [
3
H]raclopride binding after ischemia. The [
3
H]raclopride binding (c.p.m./mm2, mean ± s.d.) was quantified in two volumes of interest (VOIs) defined manually for each animal over the entire ipsilateral (
As a consequence, the ratio of [
11
H]raclopride signal ratio evidenced a trend of progressive decrease from day 1 to day 7 followed by a further decrease at day 14 and onwards. The ratio of [
3
H]raclopride signal showed a significant decrease versus that in the controls at days 14, 21, and 28 after ischemia (
Time Course of Neurologic Score After Experimental Stroke
Ischemic animals showed the major neurologic impairment at 1 day after MCAO in relation to control animals. After day 3, rats showed a trend to a progressive functional recovery over time. The neurologic impairment showed significant increase versus that in the controls at day 1, 3, 7, 14, 21, and 28 (
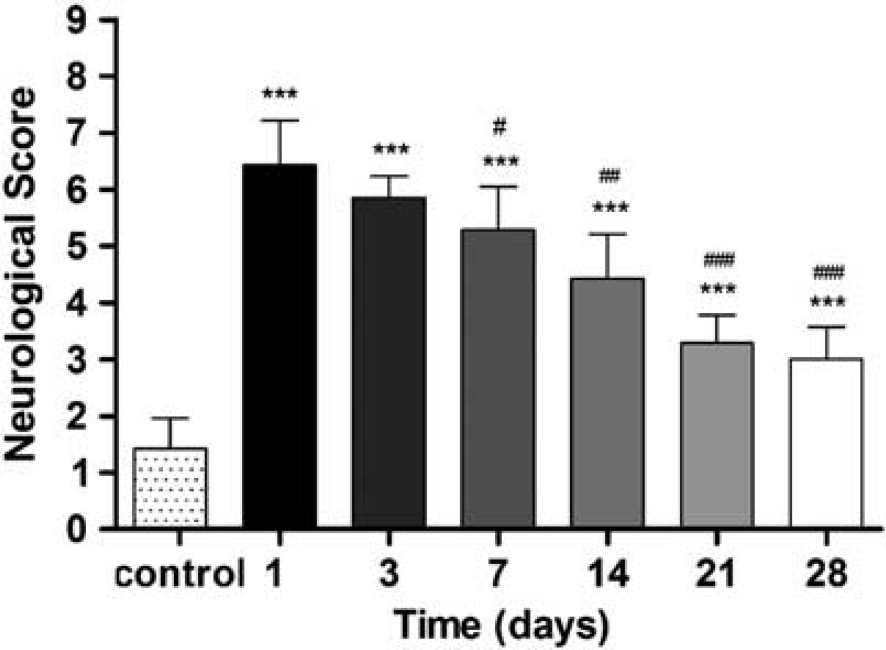
Neurologic outcome after cerebral ischemia. The neurologic score shows an improvement over time. Statistical analyses were performed by using Mann–Whitney
Evolution of Brain Lesion After Experimental Stroke
At 1 day after MCAO, T2W MRI evidenced hyperintensities in the territory irrigated by the MCA (Figure 6A). Cortical and striatal MRI alterations coincided with the presence of histopathologic features at 24 hours after ischemia. Histopathologic outcome using H-E staining showed a massive necrosis in the area affected by the blood interruption (Figure 6B). The TTC staining showed infarction evolution (pale area) over the first week after reperfusion, evidencing the hemorrhagic transformation of brain infarction at day 7 after MCAO (Figure 6C). Immunohistochemistry for TH and neuron nuclear antigen (NeuN) illustrated neurons expressing TH in the striatum at control animals (Figure 6D). Surprisingly, in the territory irrigated by MCA, TH and NeuN showed a dramatic decrease at day 7 after ischemia onset. Despite these findings, in the contralateral striatum TH staining did not show an apparent change of the signal at 7 days after ischemia in relation to control rat brain (Figure 6E).
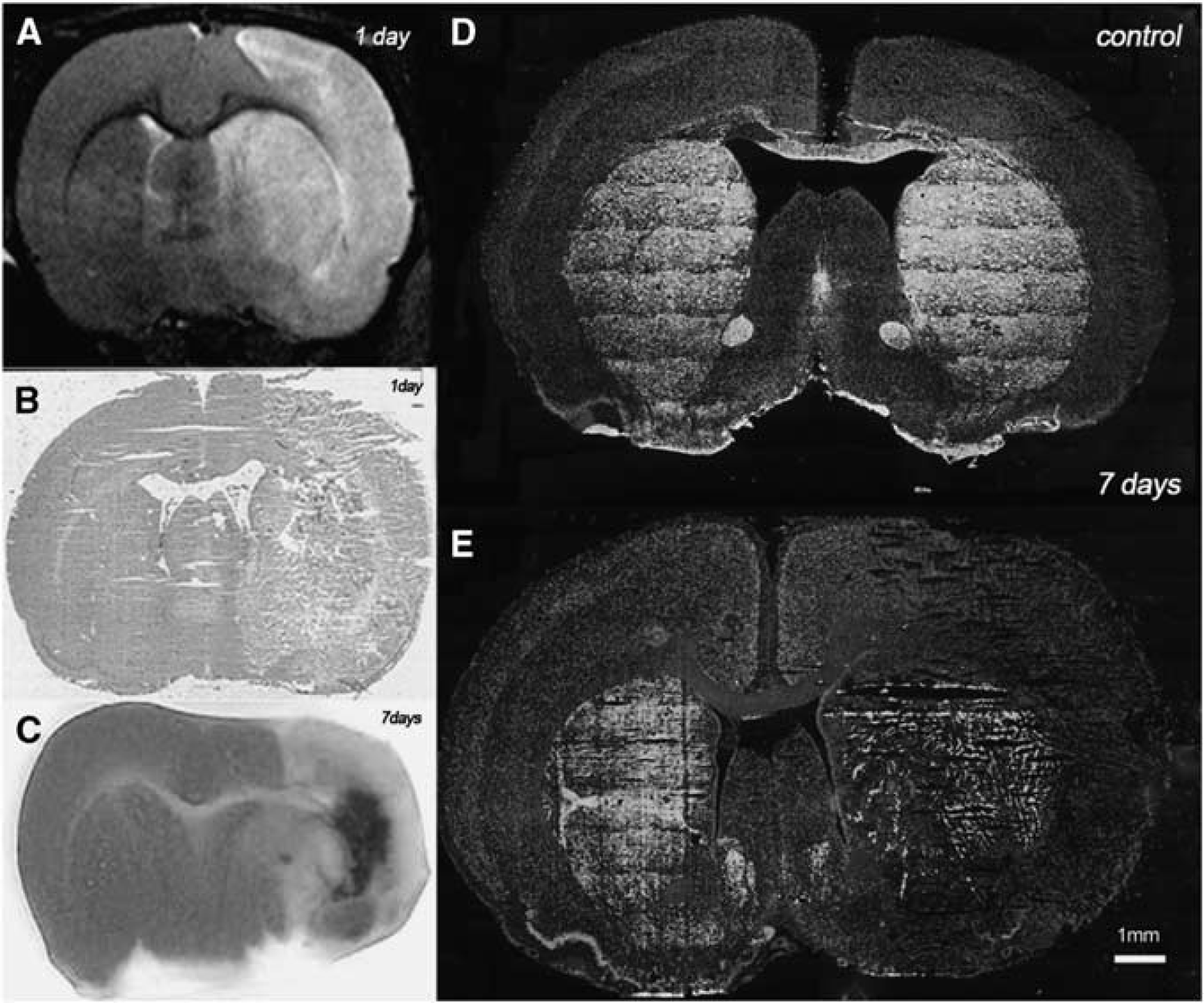
Brain lesion evolution after cerebral ischemia. magnetic resonance imaging (MRI) alterations in T2-weighted (T2W) signal (
DISCUSSION
Clinical observations using PET with [
11
C]raclopride, a selective striatal dopamine D2 receptor antagonist, have provided more insight into the role that dopaminergic system has in the pathophysiology of major neurologic and psychiatric disorders. 28–31 The modulation of these receptors using pharmacological agents has also provided benefits on learning and motor formation memory in both the subacute state after clinical stroke and chronic period, when less therapeutic options are available.15,32 Despite these findings, PET imaging of dopaminergic receptor density using [
11
C]raclopride has not been explored neither experimental nor clinical stroke to date. Because of this, we evaluated in parallel
Glucose Metabolic Changes After Cerebral Ischemia
Positron emission tomography with FDG is a well-validated technique used for the assessment of glucose metabolism that is commonly used in clinical imaging for oncologic, cardiac, and neurologic applications.
33
The PET imaging with [
18
F]FDG has been shown to be sensitive to metabolic changes that facilitate the detection of functional impairment after focal cerebral ischemia in rats.
34
The area irrigated by MCA showed a dramatic decrease in [
18
F]FDG uptake at 1 day after 2-hour MCAO that ran parallel to the T2W-MRI hyperintensities observed at this same time point after ischemia. This reduction was followed by a slight recovery of the signal from day 3 to day 7, reaching maximum uptake values at day 14 after ischemia onset evidencing and increase in metabolic activity in the injured territory. Our results are in good perspective with those of Schroeter
In Vivo and Ex Vivo Dopaminergic Changes After Cerebral Ischemia
Experimental studies have shown that dopaminergic neurotransmission is strongly affected by cerebral ischemia;
36
and the modulation of this neurotransmission system has stressed a recovery of movement after stroke in both animals
19
and human.32,37 Likewise, the striatal transplantation of embryogenic stem cells has shown an improvement of rat dopaminergic dysfunction after focal cerebral ischemia in rats.
38
Thus, since these findings suggest a key role of dopaminergic neurotransmission in neural plasticity and functional recovery after stroke, we sought to evaluate for the first time the striatal dopamine D2 receptor evolution using
In the ischemic territory, [ 11 C]raclopride PET showed a (1) nonsignificant decrease in BP at 1 day, (2) followed by a slight recovery, reaching pseudo-control values, and (3) a decrease again to roughly one-third of control values from day 14 to day 28 after ischemia. The [ 11 C]raclopride binding uptake from day 1 to day 7 may be because of the presence of nondegraded dopamine D2 receptors present in necrotic neurons, which showed a particular resistance to proteolytic processes after cerebral ischemia. These events run in parallel with a progressive [ 11 C]raclopride binding increase in the contralateral hemisphere to the lesion at 7 days after cerebral ischemia. Interestingly, the contralateral area to the MCAO showed a time-dependent increase in the BPND of [ 11 C]raclopride from day 1 to day 3, followed by a peak of [ 11 C]raclopride binding at day 7 after MCAO. This result was followed by a slight decrease from day 14 to day 28, reaching again control values. Therefore, these results evidenced an effect of cerebral ischemia over dopamine D2 receptors not only in the impaired tissue, but also in regions remote to the lesion. These results are in agreement with the dopamine D1 receptor increase observed in the contralateral side to the lesion at days 4 and 7 after focal cerebral ischemia in rats. 36 Accordingly, the ratio of the BPND in the lesion over the contralateral hemisphere varied from roughly 0.75 to 0.6 from day 1 to day 7 and followed by a reduction to 0.1 at days 21 to 28 after cerebral ischemia. Control and sham-operated animals did not show significant differences over the different time points, evidencing a minimum contribution of the stress caused by the surgical process underlying MCAO in the density of dopamine D2 receptors.
Simplified reference-tissue model is a model that simplifies the experimental setting needed for full kinetic modeling at the cost of assuming certain conditions as follows: (1) the distribution volume is the same for the tissue of interest and the reference tissue and (2) the kinetics in the receptor-rich tissue of interest is such that it is difficult to distinguish between the specific and the free/nonspecific compartment. Therefore, it may seem unrealistic to assume such conditions after cerebral ischemia where the vascularization, neuron viability, and blood–brain barrier are hardly compromised. Nevertheless, the results obtained by
Immunolabelling showed an evident decrease in TH, the enzyme responsible for catalyzing the conversion of L-tyronise to L-DOPA (a precursor for dopamine) in the injured territory 7 days after ischemia. This decrease evidenced the loss of terminal presynaptic dopaminergic terminals in the striatum. Furthermore, this result supported the posterior loss of postsynaptic dopamine neurons observed by the
Likewise, the decrease in TH could be responsible for the decrease in dopamine levels which might as well be the reason for the nonsignificant increase in [ 11 C]raclopride PET binding observed in the infarcted striatum 7 days after ischemia.
Functional Recovery After Stroke
There is clinical evidence showing that optimal functional recovery might appear around 6 months after stroke depending on the effectiveness of neural plasticity and both lesion topography and size. 8 In the present study, ischemic animals showed the highest functional impairment at 24 hours after focal cerebral ischemia followed by a progressive functional recovery over time. At 4 weeks after stroke, animals experienced an optimal recovery of the neurologic function showing similar skills to that presented by control animals. Despite this, the precise mechanisms and biologic basis involved in the recovery after cerebral ischemia are still largely unknown. Clinical and preclinical studies have revealed a major contribution of the hemisphere homolog to the lesion in the compensations of impaired functions after stroke.9,39,40 Likewise, long-term increase in microglial activation and cerebral blood flow have been observed in remote regions to the infarction after MCAO in rats.7,12 Supporting these findings, we observed a significant increase in dopamine D2 receptor density in the contralateral striatum at 7 days after occlusion. Despite these changes, cerebral glucose metabolism did not experience significant variations in the territory homolog to the lesion over the entire study. Therefore, the fact that the increase in dopamine D2 receptor density is not accompanied by an increase in neuronal/astrocytic metabolism in the same region supports the specificity of dopaminergic changes after cerebral ischemia. Therefore, based on previous studies showing that modulation of dopaminergic systems using levodopa (1) improved sensorimotor function and (2) enhanced brain plasticity in human15,37 and also in rat brain after stroke, 14 it is tempting to hypothesize that dopamine D2 receptor might contribute to the recovery of brain function after cerebral ischemia.
Summary and Conclusions
In summary, we report here for the first time the PET imaging of dopaminergic system using [ 11 C]raclopride and its relationship with glucose metabolism and functional recovery after transient focal ischemia in rats. These results should contribute to better understand the role of dopaminergic neurotransmission on stroke evolution and to establish an effective clinical treatment targeted on the healthy hemisphere for the enhancement of the recovery of impaired functions and to provide patients with a better quality of life.
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
Footnotes
ACKNOWLEDGEMENTS
The authors would like to thank M González and A Leukona for technical support in the radiosynthesis; A Arrieta and M Puigivila for technical assistance in the PET studies, and the Departamento de Educación, Universidades e Investigación del Gobierno Vasco (PO2011-3) and the Departamento de Industria, Comercio y Turismo Vasco for financial support.
