Abstract
In rodent stroke models, investigation of deficits in spatial memory using the Morris watermaze may be confounded by coexisting sensory or motor impairments. To target memory specifically, we devised a watermaze protocol to minimize the impact of sensory and motor impairments in female Lister-hooded rats exposed to proximal electrocoagulation of the middle cerebral artery (MCAO). Rats were trained in a reference-memory task comprising 4 trials/day; trial 1 being a probe trial (platform absent for the first 60 seconds). Training ended once animals reached a strict criterion based on the probe-trial performance. Memory retention was tested 1, 7, and 28 days later. The MCAO did not affect the number of days to reach criterion during acquisition or the time spent in target quadrant during retention testing, compared with sham or unoperated rats. However, MCAO rats showed slightly poorer accuracy in crossing the platform location and increased thigmotactic swimming compared with controls. Our results show that spatial memory deficits are minimal in this rodent stroke model, and suggest that previously published watermaze impairments are attributable to sensory and motor deficits but not memory deficits. We recommend using probe trials and training to a predetermined performance criterion in future studies assessing watermaze memory deficits in rodent stroke models.
Introduction
The middle cerebral artery (MCA) is the blood vessel most commonly affected by cerebrovascular accident (Mohr et al, 1986) and infarcts in MCA territory can lead to diverse neurologic deficits including memory impairments. Understanding these functional deficits is essential for development of future therapies. Many experimental studies have used watermaze tasks to better understand spatial learning and memory deficits after MCA occlusion (MCAO) and to assess potential future therapies (Hayase et al, 2009; Kawanishi et al, 2010; Markgraf et al, 1992; Noh et al, 2011; Pavlichenko et al, 2008; Sadamoto et al, 1998; Shinoda et al, 1996; Smith et al, 1997; Wang et al, 2010; Xu et al, 2009; Yonemori et al, 1996; Yun et al, 2007; Zhang et al, 2010). However, in rodent experimental stroke models, investigation of deficits in spatial memory using the Morris watermaze may be confounded by coexisting sensory and motor impairments.
In the rat, the MCA supplies the frontal, sensorimotor, auditory and occipital cortices, and the striatum. The blood supply to the hippocampus, recognized for its key role in spatial memory, is not derived from the MCA (Scremin, 1995). Spatial memory deficits after MCAO reported using the watermaze may be explained by denervation of the hippocampus due to damaged projections from infarcted cortex, thalamic atrophy, or denervated basal nucleus cholinergic fibers to the cortex (Fujie et al, 1990; Kataoka et al, 1991). Alternatively, neocortical sites, some of which lie within the MCA territory, are known to be involved in spatial learning and memory (Frankland and Bontempi, 2005). However, MCAO-induced deficits identified in the watermaze may be secondary to sensory or motor impairments or motivational deficits. For example, an increase in path length or escape latency in the watermaze may actually be due to sensory or motor deficits including limb paresis, increased circling behavior, or thigmotactic swimming (swimming alongside the walls of the pool), rather than a disruption of learning or memory per se (see Cain and Boon, 2003). The confounding issue of coexisting sensory, motor, motivational, and memory deficits when interpreting watermaze deficits prompted us to design a protocol that minimizes the influence of sensory or motor deficits. We then reevaluated rats exposed to MCAO using this protocol to determine if focal ischemia in the MCA territory led to memory deficits.
First, spatial training was preceded by cued pretraining to enable the rats to acquire nonspatial components of task performance before entering the spatial phase of training. Second, the number of trials per day was kept low, because sensorimotor impairments can be exacerbated by the use of massed training protocols (see Martin et al, 2000). Third, probe trials were conducted at the start of each training day, providing a more informative measure of spatial memory than can be obtained using escape latencies alone. Fourth, rats were trained to a specific performance criterion, allowing task acquisition to be assessed in terms of forward and backward learning curves. The forward learning curve comprised the first 5 days of training and the backward learning curve comprised the last 5 days of training up to and including the day on which criterion was reached. Fifth, after reaching criterion, and hence at a common starting point independent of the rate of acquisition, long-term memory was assessed in probe trials conducted 1, 7, and 28 days later, a procedure that was intended to engage the putative cortical mechanisms of longer term memory storage. Finally, Lister-hooded rats were used, an appropriate strain for spatial navigation studies due to their visual acuity, compared with albino rat strains (Tonkiss et al, 1992).
Materials and methods
Animals
Female Lister-hooded rats (Charles River (UK), aged 10 weeks) were individually housed with ad libitum access to food and water, and maintained on a 12-hour light/12-hour dark cycle. This manuscript is written in accordance with the ARRIVE guidelines and all experiments were performed under a Project License from the UK Home Office, with the approval of the local Ethical Review Panels, and subject to the Animals (Scientific Procedures) Act, 1986.
Rats were randomly allocated to one of the three groups. Group 1 underwent a proximal permanent left MCAO (n=10). A standard model of MCAO (the electrocoagulation Tamura model; Tamura et al, 1981) used in previous watermaze studies (Markgraf et al, 1992; Pavlichenko et al, 2008; Sadamoto et al, 1998; Shinoda et al, 1996; Smith et al, 1997; Yonemori et al, 1996) was adopted where ischemia encompasses frontal, parietal, and perirhinal cortices and striatum, as described previously (Bingham et al, 2005). Anesthesia was induced with 5% halothane and the animals were then mechanically ventilated and anesthesia maintained with 1% to 2% halothane in 100% oxygen. The temporalis muscle was retracted, skull exposed, a craniectomy was performed with a saline-cooled drill and the dura removed to expose the MCA. The MCA was electrocoagulated from the origin of the lenticulostriate arteries to a point just distal to the inferior cerebral vein and then transected before the wound was sutured for recovery of the animal. Group 2, the sham group (n=10), underwent the whole procedure with the exception of the occlusion of the MCA. Group 3 were unoperated animals (n=9) that were not exposed to anesthesia or surgery. Both MCAO and sham groups received antibiotics (Amfipen LA; Intervet UK Ltd, Milton Keynes, UK) presurgery and postsurgery to prevent wound infection and were maintained postsurgery on a diet of softened lab chow. The animals were allowed to recover for 7 to 10 days before the watermaze training and testing started.
Morris Watermaze
The pool was 2 m in diameter, filled with water with latex liquid at 25±1°C to a depth of 60 cm. It was located in the center of a room containing prominent extramaze cues and had a recessed video camera mounted on the ceiling to monitor swim paths. The video signal was relayed to a computer running specialized data acquisition software (‘Watermaze’; Actimetrics, Wilmette, IL, USA) that records the path taken by the animal.
Visible-Platform Training
The visible platform was black, ∼10 cm in diameter, and protruded ∼1 cm above the level of the water and so that it was easily visible to the rat. All three groups of animals underwent 3 days of visible-platform training, with four intercalated 90-second trials per day (intertrial interval=∼45 minutes), generating data for 12 trials in total. For each rat, the platform location and starting position were changed quasi-randomly on each trial. Extramaze cues were blocked from view by pulling a white curtain around the pool. The visible trials were used to assess any motor, sensorimotor, or motivational deficits found in MCAO and sham animals compared with unoperated animals.
Hidden-Platform Training
Three days later, hidden-platform training began with extramaze cues visible. Four intercalated trials were conducted on each day (intertrial interval=∼45 minutes). The first trial of the day was always a standard probe trial in which the rats were put into the pool to swim for 60 seconds with no platform visible or below the surface to escape onto. Animals that have learnt the location of the platform focus their search in the correct vicinity. The extent to which this occurs is a measure of the strength of the memory concerning platform position. After 60 seconds, an ‘Atlantis’ platform rose from the bottom of the pool to the usual hidden position 1 cm beneath the water surface (Spooner et al, 1994). In this way, this first trial of the day started as a spatial memory probe but was also rewarded by allowing the animals to escape from the water at the correct location rather than being removed from the pool after 60 seconds.
Three regular trials were then conducted with the hidden platform. At the start of a trial, rats were placed gently into the water facing the pool wall; the starting position was changed quasi-randomly between trials. All trials lasted for a maximum of 90 seconds; if rats failed to escape within this period, then the experimenter indicated the platform's position by placing a hand above the correct location. After escaping from the water, rats remained on the platform for 30 seconds before being removed from the pool. To eliminate the effects of a possible search bias, platform locations (the center of the north-east or south-west quadrants) were randomly assigned.
The primary measure of learning was the percentage time spent in the target quadrant (containing the Atlantis platform) during the first 60 seconds of each probe trial, and training continued until a rat reached a criterion level of performance—spending at least 50% of each probe trial in the target quadrant for 3 consecutive days, up to a maximum period of 20 days. Other parameters recorded included latency to find the platform, path length, swim speed, and thigmotaxis (time spent swimming within 15 cm of the pool walls). During probe trials, additional measures of search behavior included time spent within a zone (radius=20 cm) around the Atlantis platform, latency to reach the Atlantis platform location, and the number of times the Atlantis platform was crossed.
Longer Term Memory Retention
Conventional, unrewarded probe trials 60 seconds in duration were performed 1, 7, and 28 days after each animal reached criterion.
Harvesting of Tissue
Thereafter, animals were deeply anesthetized and the brain perfusion fixed with 4% paraformaldehyde in phosphate-buffered saline followed by postfixing for 24 hours in the skull. Brains were stored in 30% sucrose solution at 4°C until they sank, and then frozen in isopentane (−42°C) and 30 μm coronal sections cut on a cryostat. Tissue loss associated with MCAO was assessed by measuring the areas of the contralateral and ipsilateral hemisphere on hematoxylin and eosin (H&E)-stained sections taken at eight predetermined coronal levels throughout the MCA territory (10.5, 8.92, 7.19, 6.06, 5.15, 3.75, 2.18, and 1.02 mm relative to the interaural line; see Figure 1A) using an MCID image analyser (Imaging Research, St. Catherines, ON, Canada). Area under the curve (end points for integration 12.5 mm anterior to and 0.05 mm posterior to the interaural line) was used to calculate hemispheric volumes as described previously (Bingham et al, 2005). The ipsilateral hemispheric volume was subtracted from the contralateral volume then divided by the contralateral volume to give the percentage of tissue loss (Swanson et al, 1990).
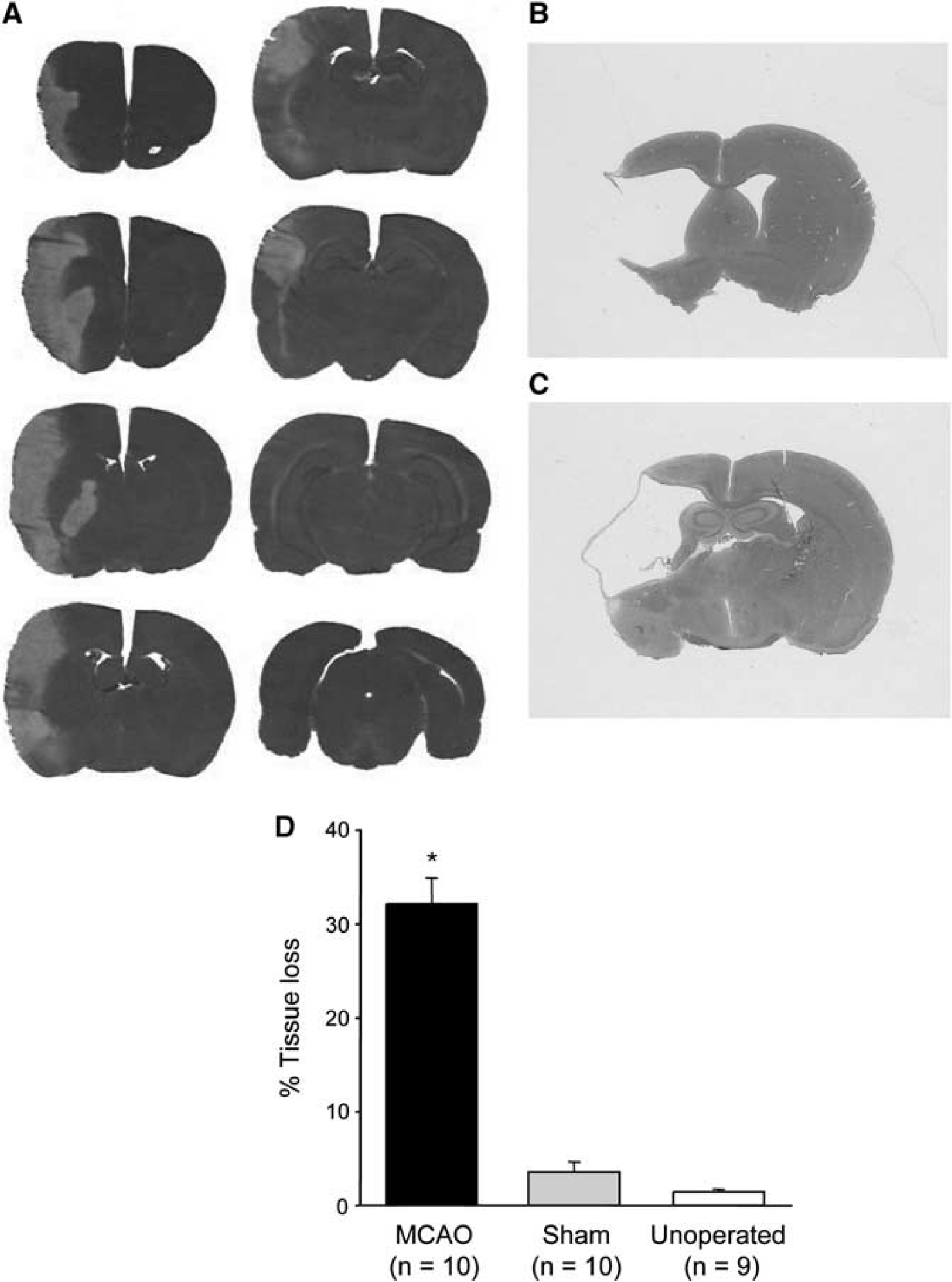
(
Statistics
Data were analyzed with SPSS (IBM, Armonk, NY, USA) using univariate or repeated-measures analysis of variance as appropriate, and post hoc comparisons were performed using the Tukey–Kramer procedure for unequal sample sizes unless otherwise stated. Differences between sham and unoperated groups are not reported unless statistically significant. All data are expressed as mean±s.e.m.
Results
Location and Extent of Tissue Loss
The frontal, parietal, and perirhinal cortical plus striatal regions were lost after electrocoagulation of the MCAO (Figures 1A–1D). Tissue loss in sham animals was small and most likely due to heat-induced damage at the craniectomy site.
Visible-Platform Training
On the first day, rats began by swimming around the sidewalls of the pool, before exploring the interior and locating the visible platform. Performance improved across days (F(2,52)=72.46; P<0.001), and on day 3 sham and unoperated rats typically swam directly to the platform in <10 seconds (Figure 2A). However, there was a significant main effect of group (F(2,26)=8.00; P<0.01), and MCAO rats exhibited longer escape latencies compared with both control groups (MCAO versus sham: P<0.02; MCAO versus unoperated: P<0.01). Nonetheless, the improvement in escape latency across days was similar in all groups, indicated by the absence of a significant group × day interaction (F(4,52)=0.55; P>0.7). An analysis of swim speeds (Figure 2B) revealed that MCAO rats swam slightly faster than sham animals (P<0.05) and these, in turn, swam faster than unoperated rats (P<0.05); the overall group difference was highly significant (F(2,26)=13.97; P<0.001). Owing to this small difference in swim speeds, path length was calculated in addition to escape latency (Figure 2C). As with escape latency, there was an overall group difference in path length (F(2,26)=7.82; P<0.01), with MCAO rats exhibiting longer path lengths than those of sham (P<0.02) and unoperated animals (P<0.01). An analysis of percentage time spent within 15 cm of the pool walls revealed increased thigmotactic swimming in the MCAO group (Figure 2D). Over the 3 days, thigmotactic swimming declined in all groups (F(2,52)=48.44; P<0.001), but the overall main effect of group was significant (F(2,26)=8.86; P<0.002), with MCAO rats exhibiting significantly higher levels of thigmotaxis compared with the control groups (P<0.01 in both cases).
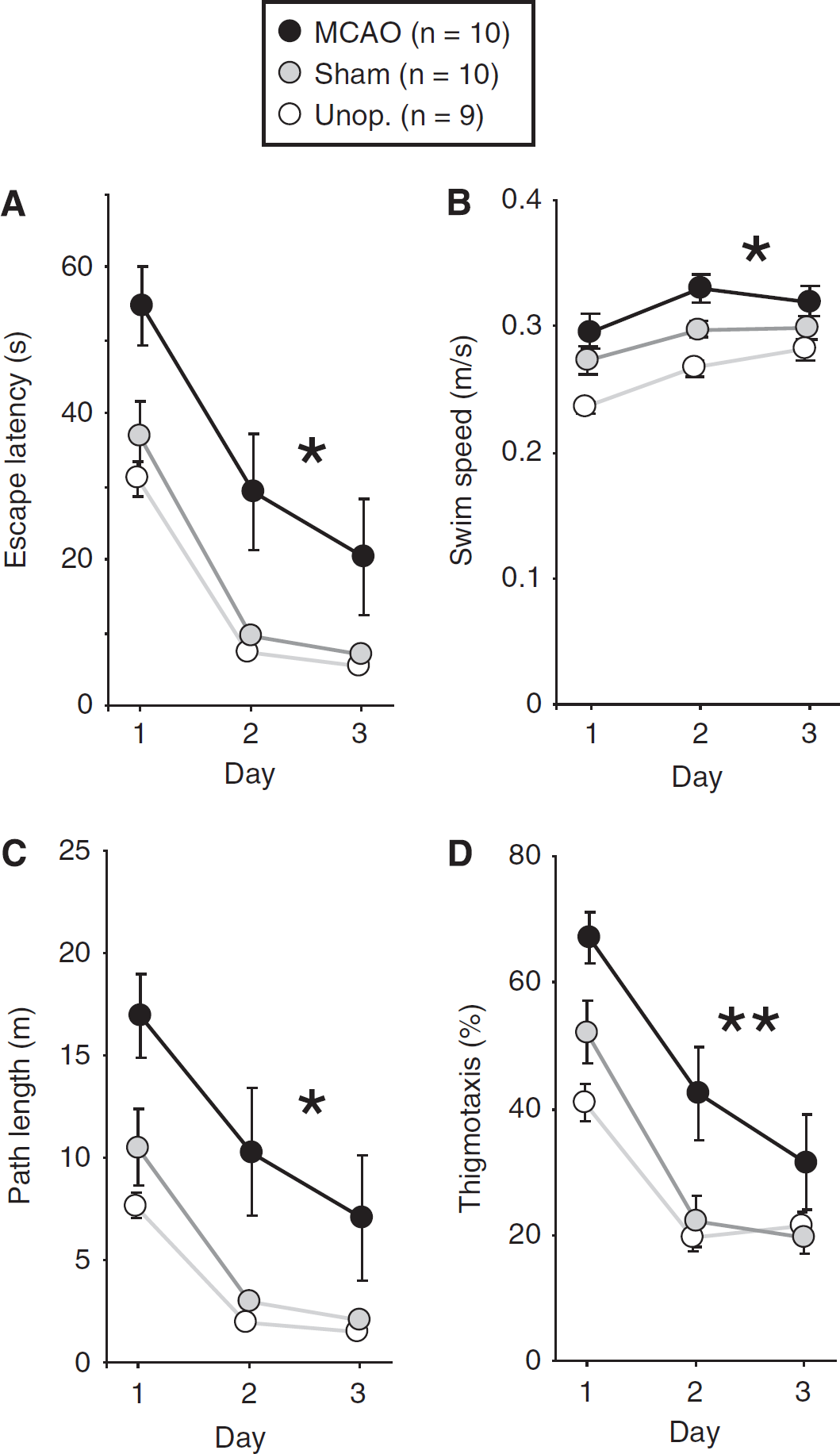
Performance during visible-platform training. (
Spatial Training—Escape Latency and Path Length
Figure 3A shows daily mean escape latencies averaged across the four trials of each day. On probe trials, the time taken to reach the platform location for the first time was used as the latency measure. As animals were no longer tested once criterion was met, the number of animals tested was not constant across days. Accordingly, acquisition data are plotted as forward and backward learning curves, the forward comprising the first 5 days of training (at which point rats began to reach criterion), and the backward comprising the last 5 days of training—up to and including the day on which criterion was reached. Over the training period, rats rapidly learned to locate and climb onto the hidden platform to escape from the pool, indicated by an overall reduction in latencies across days (first 5 days: F(4,104)=25.40; P<0.001; last 5 days: F(4,104)=5.25; P<0.002). However, MCAO rats took slightly longer to escape from the pool: group differences in escape latencies were significant (first 5 days: F(2,26)=9.24; P<0.002; last 5 days: F(2,26)=11.66; P<0.001), with MCAO rats performing more poorly than control groups over both periods (P0.01 in all cases). This impairment was evident from the very first day of acquisition, and was characterized by an upward shift in the learning curve. However, there was no group difference in the rate of learning, indicated by the absence of a significant group × day interaction over the first 5 days (F(8,104)=0.85; P>0.5), or the last 5 days of training (F(8,104)=0.51; P>0.8).
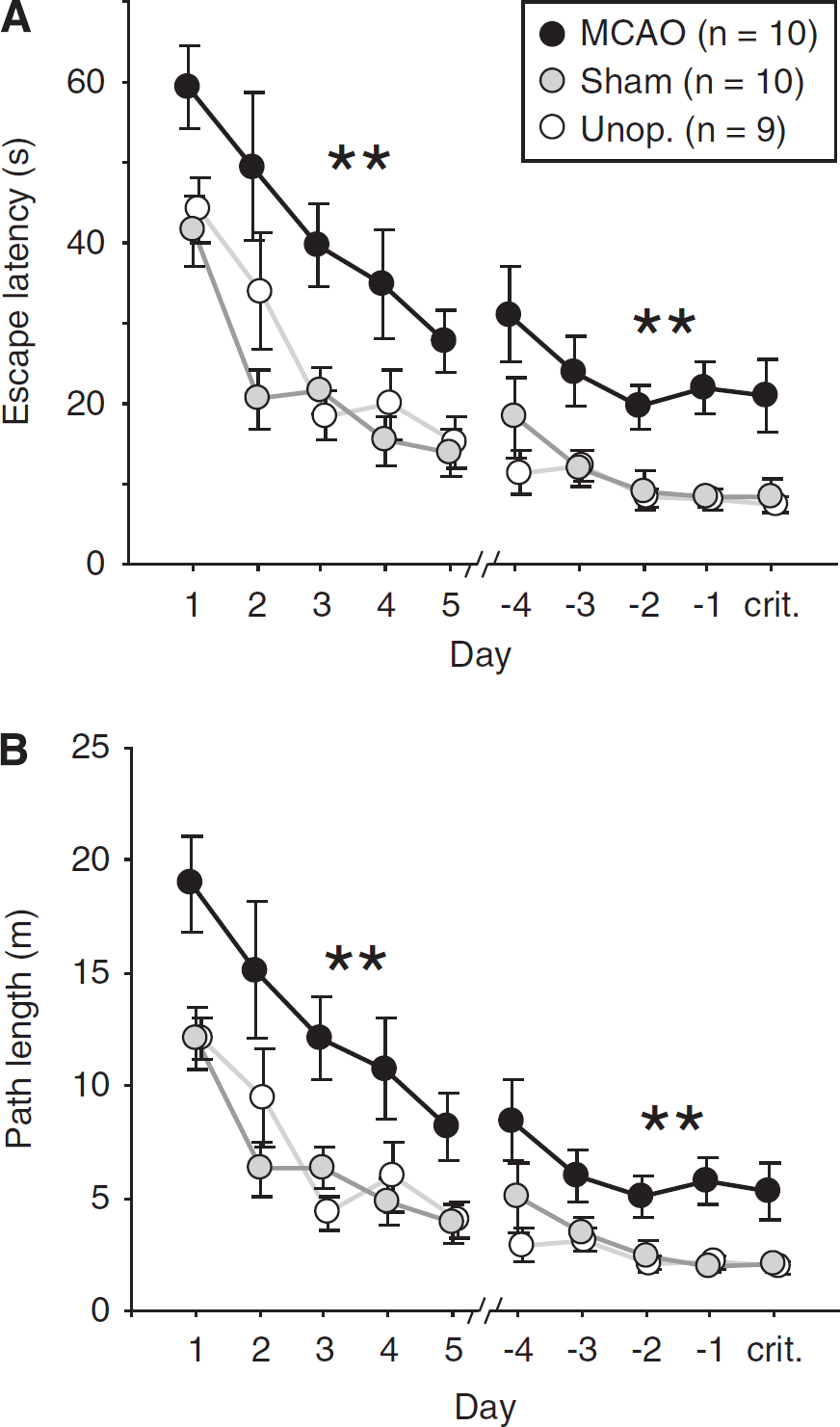
Performance during spatial training. Data are plotted as forward learning curves from the onset of training, and backward learning curves from the day on which each rat reached the performance criterion. As the first trial of each day was always a probe trial, the latency or distance swum before the first crossing of the platform location was used in place of a ‘real’ measure. (
An overall difference in swim speed was evident during all the training trials over the first 5 days (F(2,26)=4.38; P<0.05). In fact, both MCAO and sham groups swam slightly faster than unoperated rats (mean swim speed: MCAO: 0.291±0.014 m/s; sham: 0.286±0.005 m/s; unoperated: 0.255±0.009 m/s; MCAO versus sham: P>0.8; MCAO versus unoperated: P<0.05; sham versus unoperated: P=0.073). No difference was observed over the last 5 days of training (MCAO: 0.253±0.012 m/s; sham: 0.263±0.008 m/s; unoperated: 0.264±0.011 m/s; F(2,26)=0.42; P>0.6).
Owing to the slight difference in swim speed, an analysis of path length was conducted in addition to escape latency (Figure 3B). Again, the MCAO group exhibited longer paths than those of the other groups (first 5 days: P<0.01 in both cases; last 5 days: P<0.01 in both cases) resulting in a significant effect of group (first 5 days: F(2,26)=8.68; P<0.002; last 5 days: F(2,26)=9.11; P<0.002). However, as with escape latencies, the path lengths of all groups decreased in parallel despite the overall impairment in the MCAO group, and no group × day interaction was observed over the first 5 days (F(8,104)=0.90; P>0.5), or the last 5 days (F(8,104)=0.67; P>0.7).
Spatial Training—Probe-Trial Performance
Animals in the three groups took similar numbers of days (∼11 to 12) to reach the performance criterion of 50% time spent in the target quadrant for 3 consecutive daily probe trials (F<1; Figure 4). As for escape-latency and path-length data above, probe-trial performance was plotted as forward and backward learning curves. As training progressed, rats gradually learned to focus their searching in the correct location; in all groups, time in the training quadrant (first 60 seconds only) increased from ∼25% on the very first probe trial to around 60% on the criterion day (Figure 5A). This increase was significant over both the first (F(4,104)=12.65; P<0.001) and the last 5 days (F(4,104)=45.86; P<0.001), and there was no difference between groups (first 5 days: F(2,26)=1.22; P>0.3; last 5 days: F<1). Representative swim paths for MCAO and unoperated rats are included in Figure 5B for days 1, 5, and the criterion day. Both show a normal search strategy on the criterion day, focusing their search predominantly within the target quadrant.
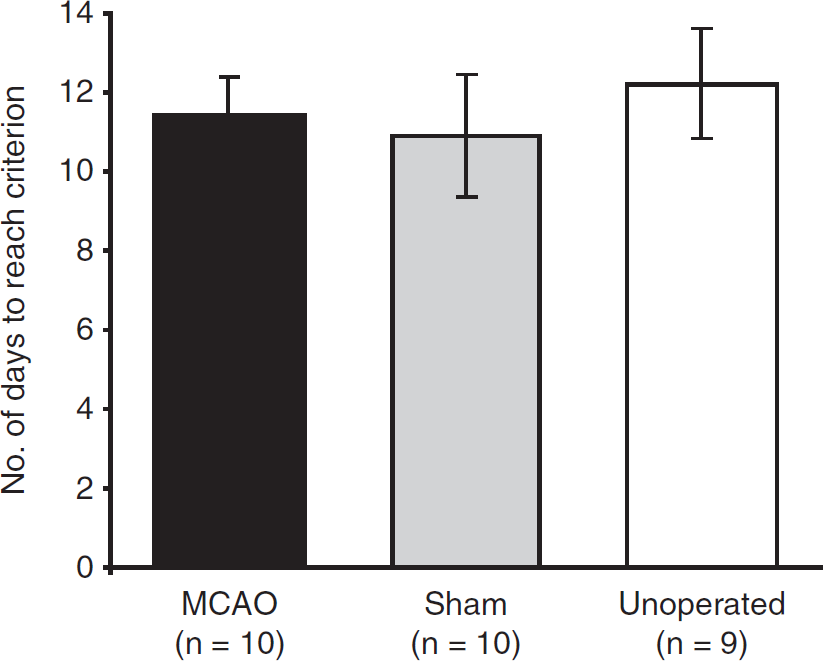
Number of days to reach the performance criterion where 50% time was spent within the training quadrant over 3 consecutive daily probe trials during task acquisition (mean±s.e.m.).
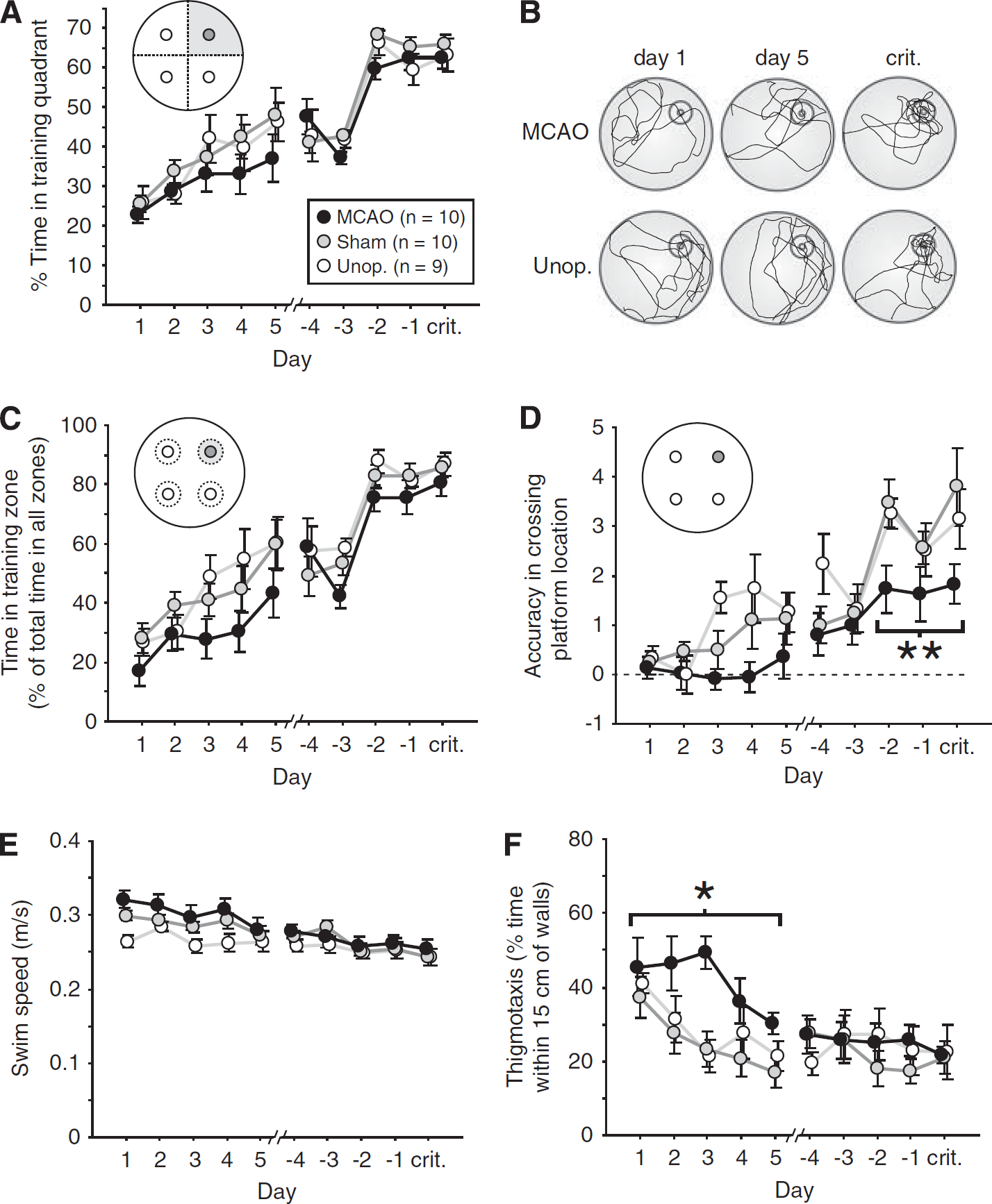
Performance during daily probe trials (the first trial of each day). Data are plotted as forward learning curves from the onset of training, and backward learning curves from the day on which each rat reached the performance criterion (crit.). (
A more sensitive measure of search accuracy than quadrant time is provided by the time spent within a 20-cm zone (training zone) of the (lowered) platform location. Zone data were qualitatively similar to quadrant data, with all rats spending increasing amounts of time in the target zone as training progressed (Figure 5C). However, the MCAO rats were slightly less accurate than controls over the first 5 days: the overall group difference was significant (F(2,26)=3.47; P<0.05), and MCAO rats exhibited a trend toward poorer performance compared with the two control groups (MCAO versus sham: P=0.099; MCAO versus unoperated=0.063). No group difference was evident over the final 5 days, however (F(2,26)=1.64; P>0.2). A measure of accuracy at a still smaller spatial scale is provided by an analysis of the number of times a rat crosses the platform location during probe trials. According to this measure, MCAO rats showed slightly poorer performance in comparison with the unoperated groups over both the first and last 5 days of training (Figure 5D). For the first 5 days, there was a significant group difference (F(2,26)=3.80; P<0.05), with MCAO rats performing significantly more poorly than unoperated rats (P<0.05). Over the final 5-day period, the group difference was again significant (F(2,26)=4.00; P<0.05) with a trend toward poorer performance in MCAO versus sham groups (P=0.063), and a significant difference between MCAO and unoperated rats (P<0.05). This impairment in accuracy was particularly pronounced over the final 3-day ‘criterion run’ (see Figure 5D) during which rats were required to spend >50% of their time within the target quadrant; in contrast to the near-identical quadrant and zone scores observed over the same period (Figures 5A and 5C), the group difference in platform-crossing accuracy was highly significant (F(2,26)=6.46; P<0.01), with MCAO rats performing more poorly than both shams (P<0.01) and unoperated controls (P<0.05).
The MCAO animals swam slightly faster during the forward but not backward period in probe trials compared with unoperated animals (Figure 5E). An analysis of variance of swim speed revealed a main effect of group over the first 5 days (F(2,26)=3.88; P<0.05), with a significant difference between MCAO and unoperated group (P<0.05); no group difference was observed over the final 5 days (F(2,26)=0.39; P>0.6). Similarly, MCAO rats exhibited increased thigmotactic swimming during the forward probe trials but not backward probe trials by which time thigmotaxis was low in all groups (Figure 5F). The overall group difference was significant over the first 5 days (F(2,26)=5.08; P<0.02), with the MCAO group exhibiting significantly more thigmotaxis than shams (P<0.02), and a nonsignificant trend relative to unoperated controls (P=0.069). No group difference was observed over the final 5 days (F(2,26)=0.12; P>0.8).
Longer Term Retention of Memory
Figure 6 shows the time spent in the target quadrant during probe trials conducted after criterion had been reached. Consistent with normal memory loss, performance worsened as the interval between the end of training and retention testing was increased (F(2,52)=18.31; P<0.001), but no group difference was observed (F<1); overall performance remained at above chance levels, even on the 28-day probe trial (P<0.001; one-sample t-test).
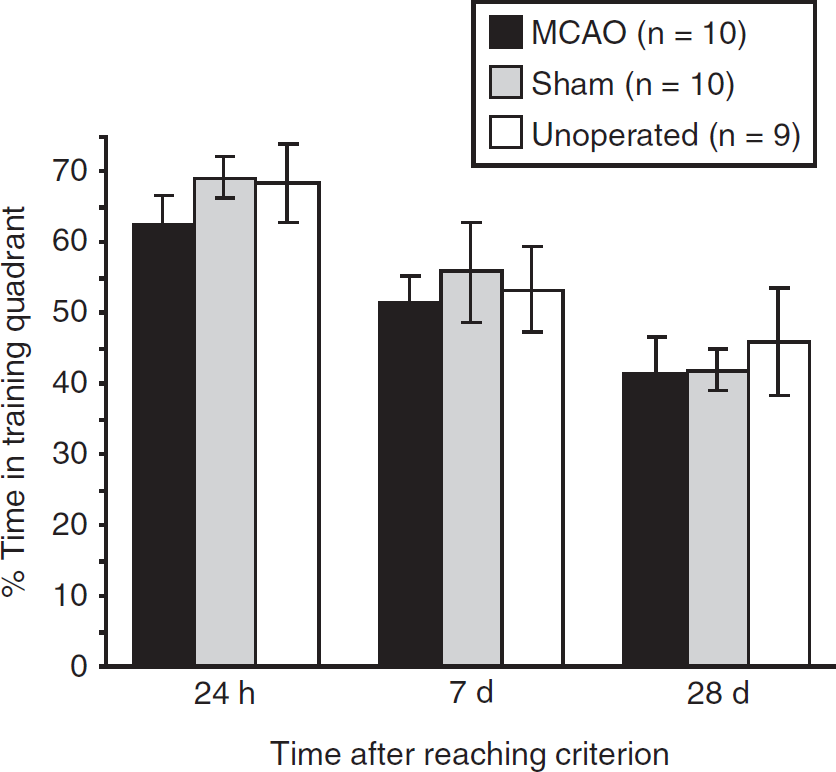
Percentage time spent in the target quadrant during long-term retention testing (mean±s.e.m.).
Discussion
Consistent with previous studies, MCAO resulted in an increased escape latency and path length relative to sham and unoperated animals in a watermaze spatial reference-memory task (Markgraf et al, 1992; Pavlichenko et al, 2008; Sadamoto et al, 1998; Shinoda et al, 1996; Smith et al, 1997; Wang et al, 2010; Xu et al, 2009; Yonemori et al, 1996; Yun et al, 2007; Zhang et al, 2010). However, from close scrutiny of the behavior of MCAO animals in the watermaze, the present study indicates that maintained focal ischemia within the MCA territory has little effect on the acquisition and retention of spatial reference memory. This conclusion is based on three key findings: (1) There were no significant differences between MCAO, sham, and unoperated groups in time spent in the training quadrant during daily probe trials, or in the number of days to reach a specific performance criterion based on this measure. (2) There were no significant differences in long-term memory retention between MCAO, sham, and unoperated groups in time spent in the target quadrant during retention tests conducted up to 28 days after reaching criterion. (3) Increased escape latency and increased path length were accompanied by increased swim speed and increased thigmotaxis, two indications of sensory and motor deficits.
Concerning swim speed, MCAO rats swam slightly faster than unoperated animals. However, an elevation in swim speed was sometimes observed in sham rats, perhaps reflecting the nonspecific consequences of anesthesia and surgery. Concerning thigmotaxis, the MCAO group exhibited substantially more thigmotactic swimming compared with both sham and unoperated controls in the early days of training, but search strategies returned to normal by the last 5 days. This transient deficit is unlikely to be a consequence of surgery-induced injury to the ipsilateral eye since sham rats would also have experienced these. Rather, increased thigmotaxis may be a direct result of the striatal damage induced by this model of MCAO (Devan et al, 1999; although in that study the lesion was bilateral). After unilateral MCAO, thigmotactic swimming may be a result of an imbalance in the output of basal ganglia systems of the rat. However, we observed no obvious bias toward circling in one particular direction in MCAO rats; rather, even when high levels of thigmotactic swimming were evident, rats often changed direction both within and between trials. Other potential explanations are that motor impairments such as weakness of contralateral limbs affect the ability of the rats to control the precise direction of their swimming or that loss of sensory and motor function gives rise to increased anxiety in MCAO rats in the watermaze which in turn may result in increased swim speed and increased thigmotaxis in an effort to escape. Therefore, increased anxiety after MCAO could also at least in part explain impaired performance in many previous studies where simple escape latencies were used as the dependent measure.
In the present study, the combination of increased escape latencies and equivalent time spent in target quadrant might appear puzzling but can be explained by the finding that the MCAO group exhibited a minor deficit in search accuracy exhibiting slightly fewer crossings of the platform location during probe trials. It is possible that this decrease in accuracy might reflect a slight navigational impairment resulting from parietal damage (see Nitz, 2009). However, taken together with the lack of difference in number of days to reach criterion, and considering the above-mentioned MCAO-induced motor and sensorimotor impairments and the known consequences of striatal damage, it may be imprudent to attribute this small reduction in search accuracy to a failure of memory per se.
A number of watermaze studies have been performed to determine the effect of MCAO on learning and memory deficits using either the Tamura model of permanent MCAO by electrocoagulation (e.g., Markgraf et al, 1992; Pavlichenko et al, 2008; Sadamoto et al, 1998; Shinoda et al, 1996; Smith et al, 1997; Yonemori et al, 1996) or the intraluminal thread model (e.g., Hayase et al, 2009; Kawanishi et al, 2010; Noh et al, 2011; Xu et al, 2009; Yun et al, 2007). It is important to note that with intraluminal thread models, particularly in mice, filament insertion can lead to loss of blood supply to the hippocampus (McColl et al, 2004), therefore although intraluminal thread is used as a model of MCAO, it is in fact capable of producing more widespread ischemia affecting other major blood vessel territories. Using the electrocoagulation model confines the ischemia to the MCA territory.
Yonemori et al (1996) trained rats before electrocoagulation of the proximal MCA and reported a memory deficit when retention was tested 8 weeks later, on the basis that the MCAO group exhibited longer latencies and path lengths to reach the hidden platform, compared with shams. However, a neurologic test battery revealed sensorimotor impairments such as abnormal posture and hemiparesis at the time of watermaze testing. During watermaze acquisition after MCAO, an increased latency to reach the platform (Pavlichenko et al, 2008; Sadamoto et al, 1998; Shinoda et al, 1996) or an increased latency plus an increased path length (Smith et al, 1997; Markgraf et al, 1992; Yonemori et al, 1999) have been observed as evidence of memory deficits. Smith et al (1997) and Yonemori et al (1999) showed extensive thigmotactic swimming in the MCAO group compared with the sham group. In other studies (Yonemori et al, 1996; Markgraf et al, 1992; Shinoda et al, 1996; Sadamoto et al, 1998; Pavlichenko et al, 2008), thigmotaxis was not measured. Excessive thigmotactic swimming can increase escape latency and thereby lead to mistaken conclusions concerning spatial memory (Cain and Boon, 2003).
Essentially, the results of these stroke studies are in agreement with our own, only our analysis has allowed us to draw different conclusions in light of evidence of sensory or motor impairments, the equivalent rate of learning and lack of difference in days to reach criterion. The concerning issue is that impaired performance in the watermaze after MCAO have been interpreted as cognitive deficits or a deficit in learning ability in the literature. For example, pharmacological or stem-cell treatments that reverse deficits in the watermaze after MCAO, may be demonstrating either efficacy in reversing motor or sensorimotor impairments (which in itself is useful information for stroke), or reversing spatial memory deficits, or both, and if used appropriately, then the watermaze provides the means to discriminate between influences on sensorimotor function and memory.
Importantly, many of the above-mentioned studies did not include visible-platform training or probe trials to assess memory retention. Data from probe trials with the platform absent are effective in dissociating improvements in performance that are related to nonspatial strategies (such as swimming at the correct distance from the pool walls to reach the platform) from the development of accurate and focused searching. The visible platform task is useful for identifying sensory or motor impairments specifically related to watermaze performance and swimming ability. This control is particularly relevant when using the models that require a craniectomy and proximal MCAO, owing to the possibility of damage to the cranial nerves and possible loss of sight in the ipsilateral eye. In the present study, for example, the combination of increased latency to reach a visible platform and thigmotactic swimming, with near-normal acquisition and long-term retention of spatial memory, is consistent with a sensory or motor account of MCAO-induced impairments.
In addition, our use of cued pretraining before spatial acquisition might be an important factor in minimizing the impact of sensorimotor abnormalities when investigating memory impairment. Nonspatial pretraining, similarly in extent to the cued training conducted in the present study, can eliminate the learning impairments caused by damage to several cortical areas (regions of the parietal and prefrontal cortices, for example; Hoh et al, 2003). This suggests that MCAO-induced impairments in watermaze tasks are possibly more associated with difficulties in the procedural aspects of task performance rather than spatial learning.
Some stroke studies report no influence of MCAO on watermaze performance as presented in Andersen et al (1999) which used an electrocoagulation MCAO model. However, in that study, only a small segment of MCA was electrocoagulated producing small cortical lesions confined to the piriform and insular cortices. Factors that may contribute to the discrepancy in results, other than the tasks used, could include lesion location that can influence behavioral results (Shigeno et al, 1985; Bederson et al, 1986).
There are many straightforward explanations for the lack of any substantial spatial learning and memory deficits after MCAO. First, in the present and other MCAO studies, the lesion was unilateral, raising the possibility that spared functions are mediated by intact structures in the contralateral hemisphere. Second, the electrocoagulation method of MCAO causes no loss of blood supply to the hippocampus. Spatial learning in the watermaze is typically very sensitive to hippocampal damage (e.g., Morris et al, 1982) although rats with hippocampal lesions can reach substantial levels of reference memory performance with extensive training, suggesting that extrahippocampal regions can make a key contribution to performance (Morris et al, 1990; Bast and Morris, 2006). However, disruption of hippocampal function typically retards the rate of learning when training protocols similar to the present one are used (Morris and Inglis, 2003; Bast and Morris, 2006). In addition, watermaze tasks can be accomplished without the full complement of highly processed information from cortical regions surrounding the hippocampus as shown from cortical lesioning studies (Burwell et al, 2004; see Aggleton et al, 2000 for review). Thus, the absence of a marked impairment in spatial memory after MCAO in the present study is broadly consistent with the limited effects of cortical lesions on acquisition of spatial memory, especially since the contralateral hemisphere was undamaged.
Damage to other cortical sites affected by MCAO—such as frontal and temporal regions—has, however, been implicated in the retention of remote spatial memory (Frankland and Bontempi, 2005). It is therefore perhaps more surprising that long-term memory retention was unaffected in the present study; but again, the sparing of contralateral tissue is likely to be a critical issue. The possibility that the hippocampus might have a lasting or even permanent role in spatial memory under some circumstances should also be considered (see Nadel and Moscovitch, 1997).
In summary, using our watermaze protocol that limits the impact of sensory or motor deficits we find minimal deficits in the acquisition of, or long-term memory for, place information in the watermaze after unilateral proximal MCAO. We suspect that many of the deficits reported in previous studies of MCAO and watermaze performance might reflect the consequences of ischemia on sensory or motor processes and procedural learning, rather than a disruption of spatial memory per se. In fact, most of the deficits that we have observed are consistent with the known effects of striatal damage on performance. Consequently, the watermaze procedures that we have used might provide useful assays for the success of therapeutic interventions targeting striatal systems, but other behavioral tasks might be more suitable for the assessment of functional rescue or recovery in damaged regions of the neocortex. We recommend that if groups are investigating cognitive/memory deficits after experimental stroke in rodents using the watermaze, that they follow our protocol using probe trials and training to a predetermined performance criterion to reduce the danger of misinterpreting the data.
Footnotes
Acknowledgements
The authors would also like to acknowledge Research into Ageing and the William Ramsey Henderson Trust Travelling Scholarship for their support and Prof Richard GM Morris for his expertise and advice on the manuscript.
Disclosure/conflict of interest
The authors declare no conflict of interest.
