Abstract
Early identification of the potentially salvageable penumbra is critical for the determination of therapeutic intervention strategies in acute focal cerebral ischemia. This study differentiates the ischemic penumbra from the core on the basis of the dynamics of lactate formation. This was tested in a rat model of focal cerebral ischemia by infusion of [1-13C]-glucose, using lactate-edited magnetic resonance spectroscopic imaging techniques. The authors detected essentially no enrichment of lactate with 13C-label from the infused 13C-glucose in the ischemic core. However, in borderzone areas, 13C was incorporated into lactate, which could point toward compromised but potentially viable tissue. The authors' findings suggest that this combination of 13C-glucose infusion with the proposed magnetic resonance methods may aid in differentiating the penumbra from the core in cerebral ischemia.
In the acute stages of focal cerebral ischemia, the brain lesion encompasses an irreversibly injured core and a peripheral zone, where tissue is at risk but potentially viable (i.e., the penumbra) (see for review Hossmann, 1994). Without therapeutic intervention, the penumbra eventually will be recruited into the process of infarction. Therefore, early identification of this area is of crucial importance.
Recently, magnetic resonance imaging (MRI) and spectroscopy methods have demonstrated their sensitivity to pathophysiologic alterations in brain ischemia as early as in the hyperacute stages (see for review Fisher et al., 1995). Diffusion-weighted MRI allows the detection of the ischemia-induced acute reduction of the apparent diffusion coefficient (ADC) of tissue water, which can be associated with the development of cytotoxic edema (Moseley et al., 1990). 1H-NMR spectroscopic imaging (MRSI) provides a means to spatially assess the metabolic status of ischemic tissue from the detection of metabolites such as the neuronal marker
This study evaluates the potential of MRSI of lactate to distinguish the penumbra from the ischemic core in acute focal ischemic brain tissue. We specifically tested the implicit hypothesis that regions in which the ADC of water continues to decline sustain a significant increase of lactate. To that end, we combined diffusion-weighted MRI with measurements of the distribution and time course of the incorporation of 13C-label into lactate during infusion with [1-13C]-glucose, by use of 1H-MRSI, tailored for the exclusive detection of lactate.
METHODS
Male Fisher rats (225 to 325 g) (n = 8) were anesthetized by a subcutaneous injection of a mixture of 0.55 mL/kg fentanyl citrate (0.315 mg/mL) and fluanisone (10 mg/mL), and 0.55 mL/kg midazolam. After endotracheal intubation, animals were mechanically ventilated with O2/N2O (1:2). Body temperature was maintained at 37.5 ± 0.5°C by means of a feedback-controlled heating pad. The left femoral artery was cannulated for continuous recording of blood pressure and blood sampling. Plasma glucose levels and blood gas tensions were frequently measured. The left femoral vein was cannulated for infusion with D-[1-13C]-glucose (Omicron, Biochemicals, Inc., South Bend, IN, U.S.A.) (6.7 g/kg in 5 mL of saline). Permanent focal cerebral ischemia was induced by unilateral tandem occlusion of the common carotid artery and the middle cerebral artery, as described by Brint and others (1988). Infusion with [1-13C]-glucose (Patlak and Pettigrew, 1976) was started immediately after middle cerebral artery occlusion (MCAO), after which the rats were positioned in the magnet.
The MRI measurements were performed in a 4.7-T SISCO/Varian NMR spectrometer (Palo Alto, CA, U.S.A.) using a 1H/13C surface coil (1H: circular [1.2-cm diameter]; 13C: butterfly [1 × 2 cm loop dimensions]). During the MRI protocol, anesthesia was maintained by ventilation with 2% isoflurane. Diffusion-weighted MRI (spin-echo sequence; repetition time 2 seconds; echo time 100 milliseconds; one [
Data are presented as mean ± SD. Statistical analysis was done by one-way analysis of variance with repeated measures. Groups were compared by use of a paired Student's
RESULTS AND DISCUSSION
Intravenous infusion of [1-13C]-glucose resulted in a significant increase of plasma glucose from 6.6 ± 0.5 mmol/L to 34.5 ± 7.2 mmol/L (
Middle cerebral artery occlusion induced a focal cerebral ischemic lesion, which was delineated as a clear ipsilateral hyperintense area on diffusion-weighted images (Fig. 1A). Calculated maps of the tissue water ADC revealed normal ADC values (0.7 to 0.8 × 10−3 mm2/s) in contralateral cortices. The ADC, as measured from volumes-of-interest (4.5 μL) positioned in the periphery of the lesion (i.e., the primary motor cortex) and the ischemic core (i.e., the primary somatosensory cortex), revealed a significant decline from 0.47 ± 0.05 to 0.38 ± 0.04 × 10−3 mm2/s (
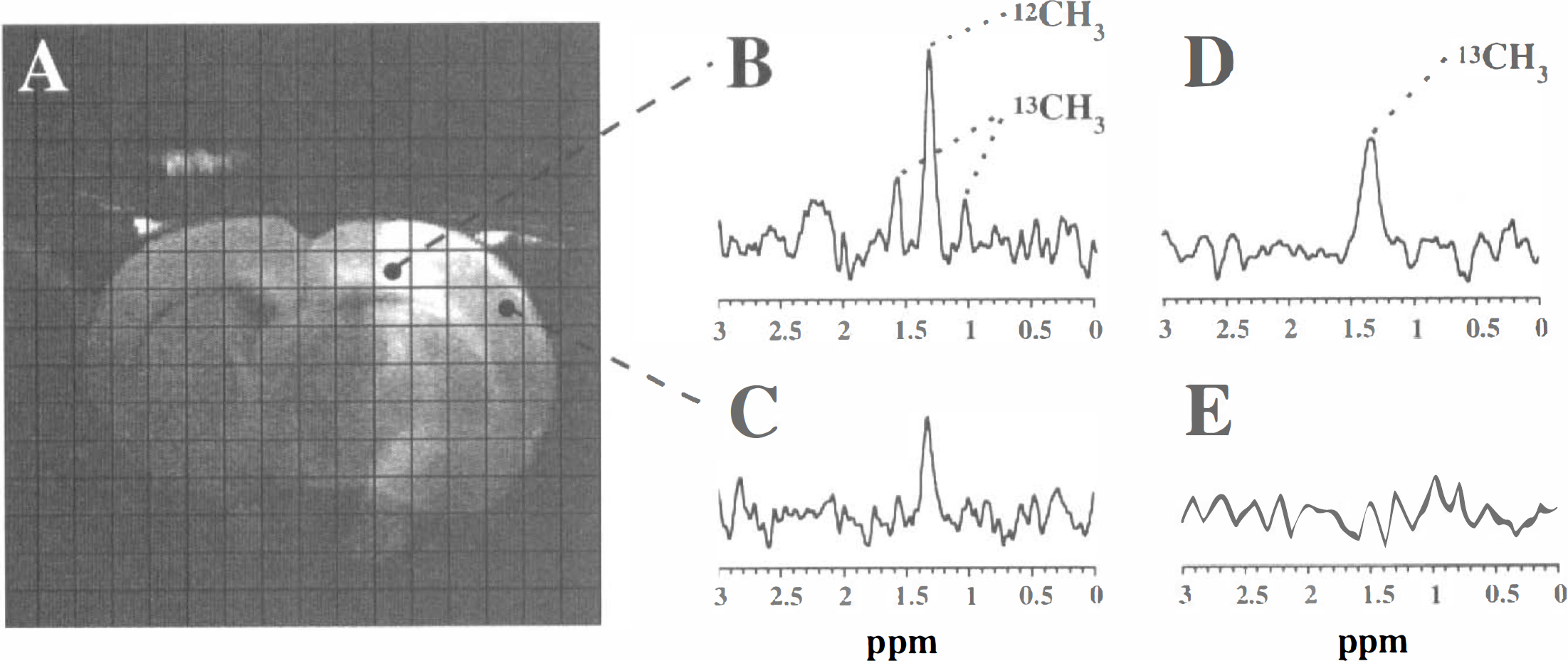
Homonuclear lactate-edited MRSI revealed exclusively lactate peaks, which were detected only in voxels within the ischemic region. Figure 1B and C show spectra from voxels in the ischemic borderzone and core, respectively. Typically, at the boundaries of the lesion, the center signal from [3-12C]-lactate methyl protons at 1.33 ppm was clearly flanked by [3-13C]-lactate methyl proton signals, which were split into a doublet by heteronuclear spin-spin coupling (
The presence or absence of 13C-lactate satellite peaks, as observed with homonuclear lactate-edited MRSI, was invariably confirmed with heteronuclear 1H-observed/13C-edited MRSI from the presence or absence of signal from the 13C-lactate methyl protons (Fig. 1D and E).
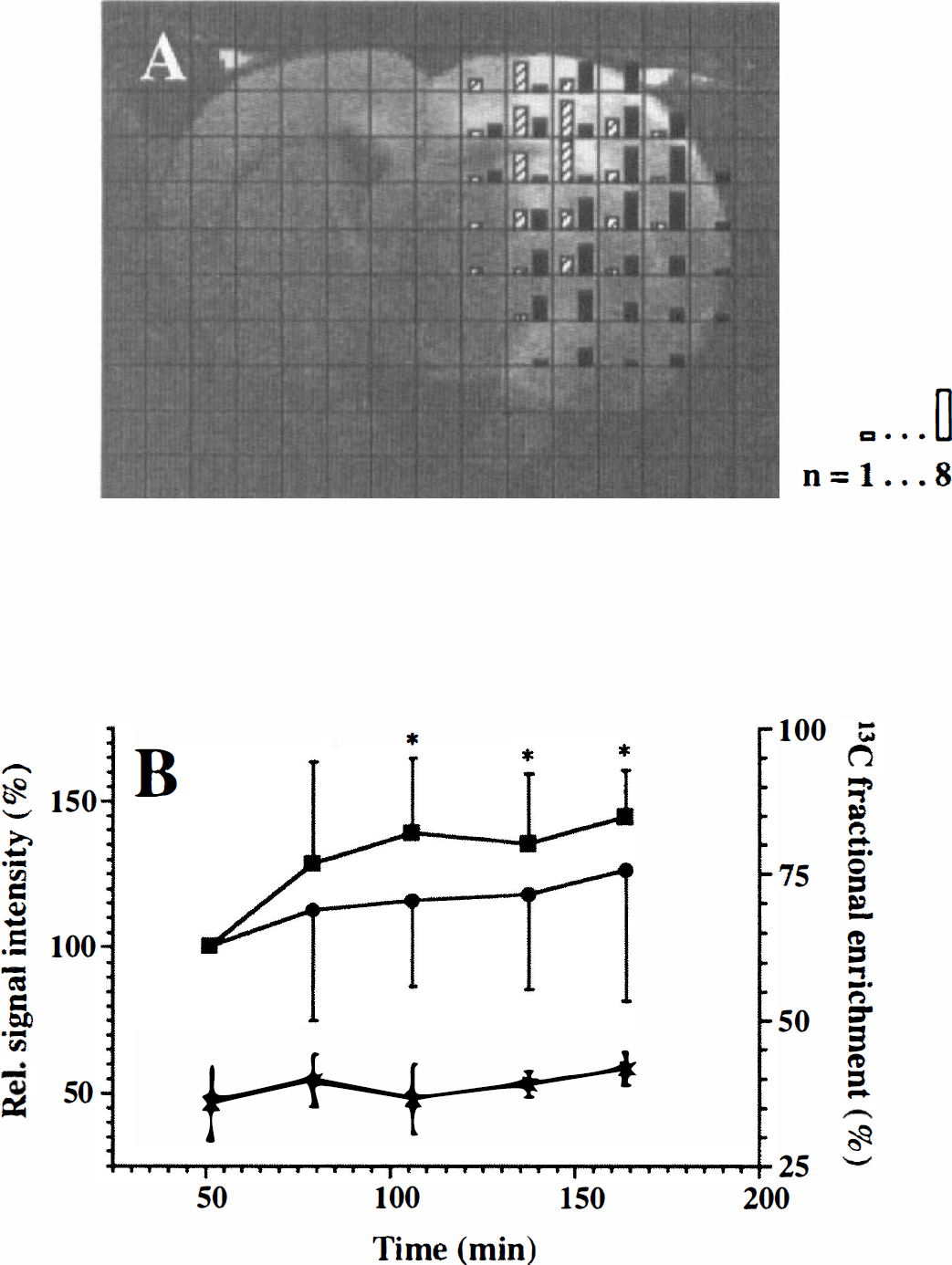
In the borderzone, the lactate concentration increased dynamically and was significantly elevated after 2 to 3 hours compared with the situation after 0.5 to 1 hours (Fig. 2B). In the core, the modest lactate concentration increase was not significant (Fig. 2B). The 13C-lactate fraction in the borderzone already was 30% to 40% after 0.5 to 1 hours and remained at this level up to 3 hours (Fig. 2B). Since theoretically the maximal fractional enrichment is 50% (see Rothman et al., 1991), our results suggest that the lactate present in these areas was almost completely derived from blood glucose during infusion.
In this study, the start of [1-13C]-glucose infusion coincided with the onset of ischemia. By initiating the [1-13C]-glucose infusions at varying time points and by alternating infusions with [1-13C]-glucose and [1-12C]-glucose in pulse-chase-type experiments, we expect to gain new insights in the sequence of metabolic changes in both permanent and transient focal cerebral ischemia. Importantly, since hyperglycemia may seriously affect outcome in cerebral ischemia, future studies also should assess the effect of elevated glucose levels on both metabolic impairment and tissue injury.
In conclusion, this study has shown that homonuclear- and heteronuclear-edited MRSI combined with infusion of [1-13C]-glucose may provide a means to distinguish irreversibly damaged and potentially viable ischemic tissue at a metabolic level. These techniques, in combination with other MRI modalities such as perfusion- and diffusion-weighted MRI, thereby would provide a powerful tool to differentiate the penumbra from the core in cerebral ischemia.
Footnotes
Acknowledgments
The authors thank Gerard van Vliet and Yanping Luo for expert technical assistance.
