Abstract
We examined the possibility that direct stimulation of the angiotensin II type 2 (AT2) receptor by a newly generated direct AT2 receptor agonist, Compound 21 (C21), enhances cognitive function. Treatment with C21 intraperitoneal injection for 2 weeks significantly enhanced cognitive function evaluated by the Morris water maze test in C57BL6 mice, but this effect was not observed in AT2 receptor-deficient mice. However, C21-induced cognitive enhancement in C57BL6 mice was attenuated by coadministration of icatibant, a bradykinin B2 receptor antagonist. Administration of C21 dose dependently increased cerebral blood flow assessed by laser speckle flowmetry and hippocampal field-excitatory postsynaptic potential (f-EPSP) determined by electrophysiological techniques in C57BL6 mice. Furthermore, activation of the AT2 receptor by C21 promoted neurite outgrowth of cultured hippocampal neurons prepared from fetal transgenic mice expressing green fluorescent protein. Finally, we investigated the pathologic relevance of C21 for spatial learning using an Alzheimer's disease mouse model with intracerebroventricular injection of amyloid-β (1 to 40). We observed that treatment with C21 prevented cognitive decline in this model. These results suggest that a direct AT2 receptor agonist, C21, enhances cognitive function at least owing to an increase in CBF, enhancement of f-EPSP, and neurite outgrowth in hippocampal neurons.
Introduction
The renin—angiotensin system has a role not only in the cardiovascular system but also in the central nervous system. Angiotensin (Ang) II binds two main receptor subtypes, type 1 (AT1) and type 2 (AT2) receptors. In the brain, AT2 receptors are expressed not only in the vascular wall but also in areas related to learning and control of motor activity (Iwai et al, 2004; Mogi et al, 2006). Mice with deletion of the AT2 receptor were reported to exhibit worse cognitive function compared with wild-type mice (Reinecke et al, 2003), whereas AT2 receptor stimulation could contribute to enhancement of neural differentiation and protection after stroke (Li et al, 2005; Reinecke et al, 2003; Wan et al, 2004). Therefore, direct AT2 receptor stimulation has been expected to have a beneficial effect on cognitive function.
Compound 21 (C21), an orally active, nonpeptidergic, selective AT2 receptor agonist (Gelosa et al, 2009), has been generated. Administration of C21 improved systolic and diastolic functions after myocardial infarction in rats, resulting from reduced apoptosis and an antiinflammatory effect (Mertens et al, 2010). Gelosa et al (2009) showed that C21 treatment delayed the occurrence of brain damage and prolonged survival in spontaneously hypertensive stroke-prone rats by preventing renal damage (Wright and Harding, 1995). Local administration of C21 reduced the synthesis of dopamine and tyrosine hydroxylase activity (Zhu et al, 2000).
AT2 receptor activation stimulates the release of nitric oxide (NO)/cyclic guanosine monophosphate (cGMP) and may mediate vascular relaxation and blood flow indirectly by modulation of bradykinin release (Johren et al, 2004). Cerebral blood flow (CBF) functions in concert with neurons and glia (so-called ‘the neurovascular unit’) to maintain homeostasis of the cerebral microenvironment (Iadecola, 2010). Therefore, dysregulation of the neurovascular unit is believed to be a factor contributing to stroke and neurodegenerative disease. Therefore, we speculated that treatment with C21 could enhance cognitive function with improvement of CBF.
Materials and methods
This study was performed in accordance with the National Institutes of Health guidelines for the use of experimental animals. All animal studies were reviewed and approved by the Animal Studies Committee of Ehime University.
Animals
Adult male C57BL/6 mice (Clea Japan Inc., Tokyo, Japan) and AT2 receptor-deficient mice (Agtr2−; based on C57BL/6J strain) (weighing 20 to 25 g; 10 to 12 weeks old) were used. Mice were kept in a room in which lighting was controlled (12 hours on, 12 hours off) and temperature was kept at 25°C. They were given a standard diet (MF, Oriental Yeast, Tokyo, Japan) and water ad libitum. Compound 21 was provided by Vicore Pharma (Gothenburg, Sweden). Mice were subjected to intraperitoneal injection of C21 (1, 3, and 10 μg/kg per day) for 2 weeks before the Morris water maze test. Systolic blood pressure was monitored in conscious mice by the tail-cuff method (MK-1030, Muromachi Co., Tokyo, Japan), as described previously (Krege et al, 1995). After the Morris water maze test, mice were overanesthetized and the brain was removed. The collected cortex and hippocampus were frozen in liquid nitrogen and stored at −80°C until further analysis. A bradykinin B2 receptor antagonist, icatibant (70 μg/kg per day, Sigma-Aldrich, St Louis, MO, USA), was infused through an intraperitoneally implanted osmotic mini-pump (Alzet model 1004, DURECT Corporation, Cupertino, CA, USA). In the vehicle group, phosphate-buffered saline was used instead of C21 and/or icatibant.
Real-Time Reverse Transcriptase-PCR Method
Total mRNA was extracted from brain samples after homogenization in Sepazol (Nacalai Tesque Inc., Kyoto, Japan). Quantitative real-time reverse transcriptase-PCR was performed using a SYBR green kit (MJ Research Inc., Waltham, MA, USA). PCR primers for the AT1 receptor were 5′-GTTCCTGCTCACGTGTCTCA-3′ (forward) and 5′-CATCAGCCAGATGATGATGC-3′ (reverse), and for the AT2 receptor were 5′-CCTGCATGAGTGTCGATAGGT-3′ (forward) and 5′-CCAGCAGACCACTGAGCATA-3′ (reverse).
Morris Water Maze Test
The Morris water maze test was performed in mice after 2 weeks of treatment with or without C21 and icatibant, as described previously (Sakata et al, 2009). In brief, mice were trained 5 times a day at 20-minute intervals for 5 consecutive days. In each trial, mice were given 120 seconds to find the platform. Swimming was video tracked (AnyMaze, Wood Dale, IL, USA), and latency, path length, swim speed, and cumulative distance from the platform were recorded. Mean swim latency for each day was evaluated and compared between groups. All analyses were performed by an investigator blinded to the experimental conditions.
Measurement of Cerebral Blood Flow
After 2 weeks of treatment with C21 and/or icatibant, CBF was determined by laser speckle flowmetry (Omegazone, laser speckle blood flow imager, Omegawave, Tokyo, Japan), which obtains high-resolution two-dimensional images in a matter of seconds as described previously (Tsukuda et al, 2008). Mice were anesthetized with Nembutal in saline, and a midline incision was made in the scalp. Anesthesia did not significantly modify blood pressure. The skull was exposed and wetted with saline. A 780-nm laser semiconductor laser illuminated the whole skull surface. We measured mean CBF in the skull surface. Light intensity was accumulated in a CCD camera and transferred to a computer for analysis. Image pixels were analyzed to produce average perfusion values.
Evaluation of Capillary Density
Capillary density was evaluated in the cerebral cortex using a Blood Vessel Staining kit (Millipore Corp., Billerica, MA, USA), based on immunofluorescence of von Willebrand Factor. Brain sections were embedded in paraffin after perfusion fixation with 10% formalin after infusion of phosphate-buffered saline. The brain was sectioned at 20-μm thickness (5 sections of brain for each sample), and the mean number of vessels in the cortex stained by diaminobenzidine was analyzed using a Leica DMI 6000B microscope (Leica Microsystems, Wetzlar, Bensheim, Germany) in 10 individual fields at × 100 magnification.
Excitatory Postsynaptic Potential
Mice were anesthetized and placed in a stereotaxic adaptor (SA-1, Unique Medical, Tokyo, Japan). A concentric bipolar stimulating electrode (twisted Teflon-insulated stainless steel wires with a diameter of 0.1 mm) was implanted, with bipolar stimulating electrodes placed ipsilaterally in the stratum pyramidale of the CA3 region of the hippocampus (1.70 mm posterior to the bregma, 2.0 mm right lateral to the midline, and ∼2.0 mm below the brain surface), and a recording electrode was placed in the right CA1 stratum radiatum (1.2 mm lateral and 2.2 mm posterior to the bregma and 1 to 1.5 mm below the brain surface). A bare silver wire was fixed to the subcutaneous tissue as ground. The field potential was evoked by repeated stimulation (0.1 ms, 0.3 mA) (UPS-901, Unique Medical). The means of the population spike amplitude and the field-excitatory postsynaptic potential (f-EPSP) slope were calculated (UAS-108S, Unique Medical) from 500 pulses.
Preparation of Hippocampal Neurons
Hippocampal neurons were prepared as described previously (Bock et al, 2004; Li et al, 2007) from the hippocampus of fetal green fluorescent protein mice (provided by Dr Hidemasa Oh, Kyoto University, Kyoto, Japan) (Tateishi et al, 2007). Approximately 95% of cultured cells expressed a neuronal marker, microtubule-associated protein 2 (Supplementary Figure 1). In brief, primary hippocampal neurons were cultured at 60,000 cells per cm2 in 24-well poly-
Assessment of Neurite Length
Measurements of neurite length were taken from all neuritic processes of neurons stained with green fluorescent protein fluorescence. Neurite branches were defined as described previously (Li et al, 2005). Images were randomly selected at × 400 magnification using a Leica DMI6000B microscope (Leica Microsystems) equipped with a computer-based imaging system, FW4000 (Leica Microsystems). For each experimental group, neurite length assessments were performed by analyzing 100 neurons randomly chosen in nine culture wells. Mean neurite length per neuron was calculated using the Densitograph Personal Image Version 3.0 (ATTO Corporation, Tokyo, Japan).
Amyloid-β Intracerebroventricular Injection Model
Amyloid-β (Aβ) 1 to 40 (Peptide Institute, Osaka, Japan) was injected intracerebroventricularly at 200 pmol in 5 μL phosphate-buffered saline simultaneously with drug treatment as described previously (Mogi et al, 2008; Nitta et al, 1994; Tsukuda et al, 2009). For the vehicle control, the same dose of phosphate-buffered saline was injected. To verify that injection was carried out properly, the same volume of Evans blue was injected intracerebroventricularly into another group of mice. We confirmed the distribution of the injected Evans blue throughout both sides of the ventricles. After 4 weeks of Aβ injection, animals were subjected to the Morris water maze test. After the cognitive task, mice were anesthetized and their brains removed. Amyloid-β concentration in the brain was measured by ELISA (enzyme-linked immunosorbent assay) (Human β Amyloid 1 to 40 ELISA Kit Wako II, Wako Chemical Industries, Osaka, Japan) according to the manufacturer's protocol. The amount of Aβ was calculated by comparison with a standard curve of synthetic human Aβ 1 to 40.
Statistical Analysis
All data are expressed as mean±s.e.m. in text and figures. Data were evaluated by analysis of variance, followed by post hoc analysis for multiple comparisons. Differences with P<0.05 were considered statistically significant.
Results
Systolic Blood Pressure and Angiotensin II Receptor mRNA
Treatment with C21 at three different doses had no significant effect on systolic blood pressure over the period of the experiment (Supplementary Figure 2A). mRNA of AT1 and AT2 receptors in the hippocampus was not significantly changed after C21 administration (Supplementary Figures 2B and 2C). Similar results were also obtained in the cortex (data not shown).
Effect of Compound 21 on Cognitive Function
Compound 21-treated mice exhibited significantly shorter swim latency compared with that in control mice over the 5 days of the experiment, and did not show a dose-dependent effect at these doses (Figure 1A). Comparison of swim latency on the final day shown in Figure 1B clearly showed that C21 treatment significantly shortened swim latency. Moreover, we compared the swimming speed during the water maze test (Supplementary Table 1) and observed that administration of C21 did not significantly change the mobility of mice. Conversely, C21 administration had no significant effect on this spatial learning in Agtr2− (Figure 1C), suggesting that the AT2 receptor has a crucial role in spatial learning activity. It was previously reported that Ang II-induced vasodilation through the AT2 receptor is mediated by bradykinin B2 receptor activation and NO production (Yayama and Okamoto, 2008). A B2 receptor antagonist, icatibant, was coadministered with C21 to investigate whether C21-induced enhancement of cognitive function was related to this pathway. We did not find any significant difference in swim latency between control and icatibant-treated mice; however, administration of icatibant attenuated the C21-induced improvement in spatial leaning in the water maze test (Figures 2A and 2B).
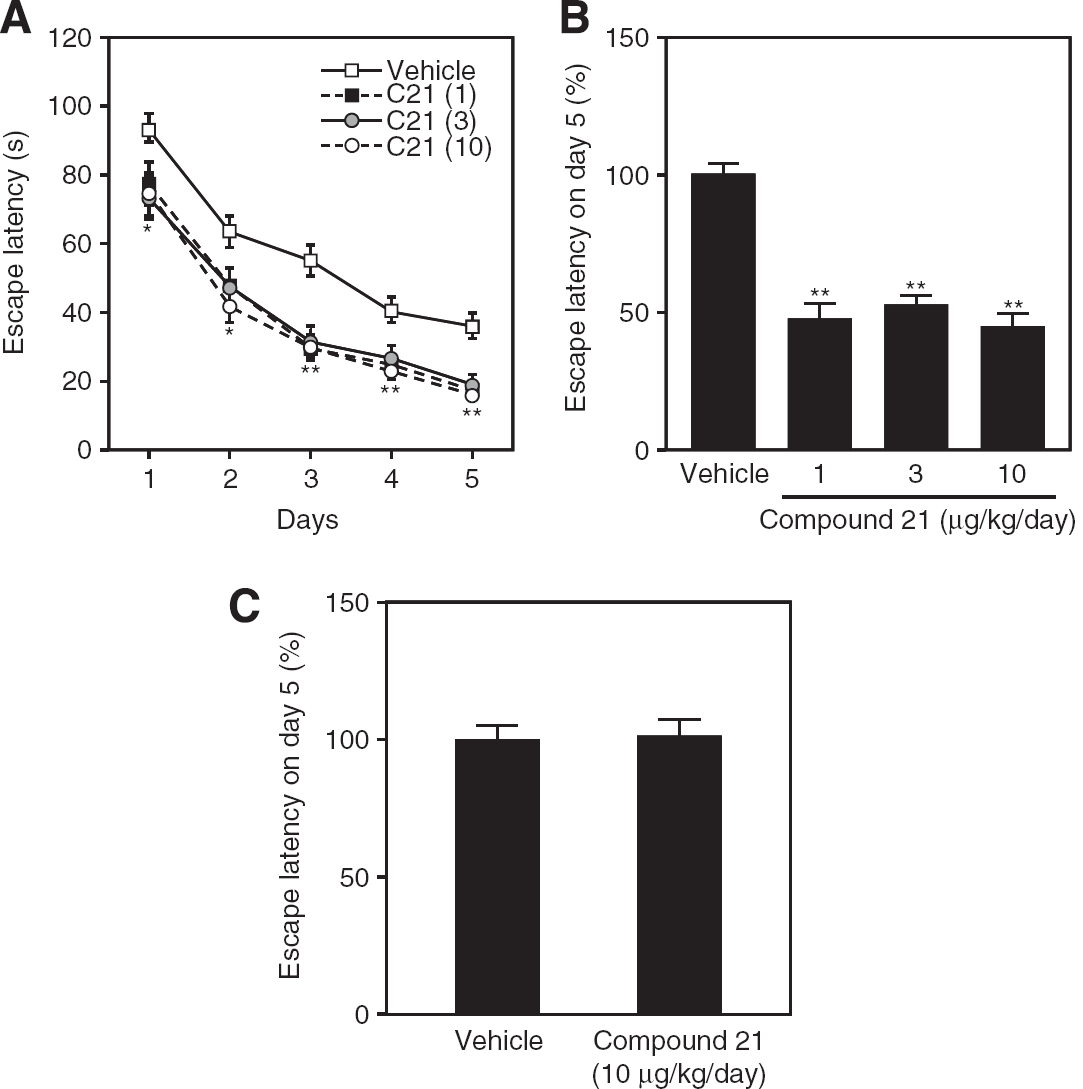
Swim latency determined by the Morris water maze test. (
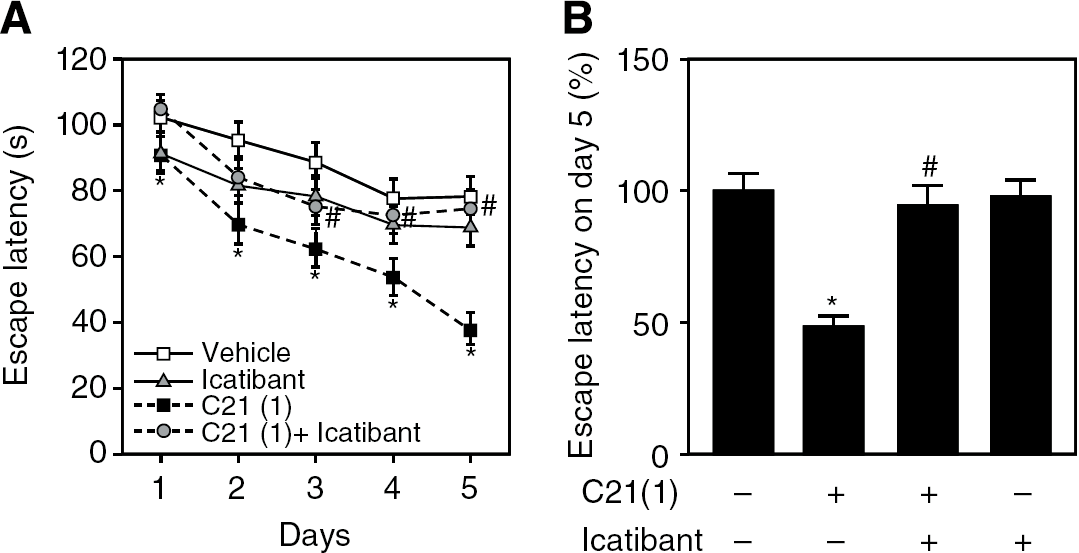
Coadministration of icatibant attenuated C21-induced enhancement of spatial leaning determined by the Morris water maze test. (
Effect on Compound 21 on Capillary Density and Cerebral Blood Flow
After 2 weeks of C21 treatment, capillary density in the brain was not significantly increased before and after the cognitive task compared with that in vehicle-treated mice (Figure 3A). However, mean CBF after the Morris water maze test was significantly increased in Agtr2+ compared with that in vehicle-treated mice, whereas significant difference in mean CBF was not observed before the cognitive task (data not shown). A C21-induced increase in CBF was not observed in the group cotreated with icatibant (Figure 3B; representative photos are shown in Supplementary Figure 3A). In contrast, mean CBF was significantly lower in Agtr2− than in Agtr2+, and C21 administration for 2 weeks had no significant effect on this decrease in Agtr2− (Figure 3C; representative photos are shown in Supplementary Figure 3B).
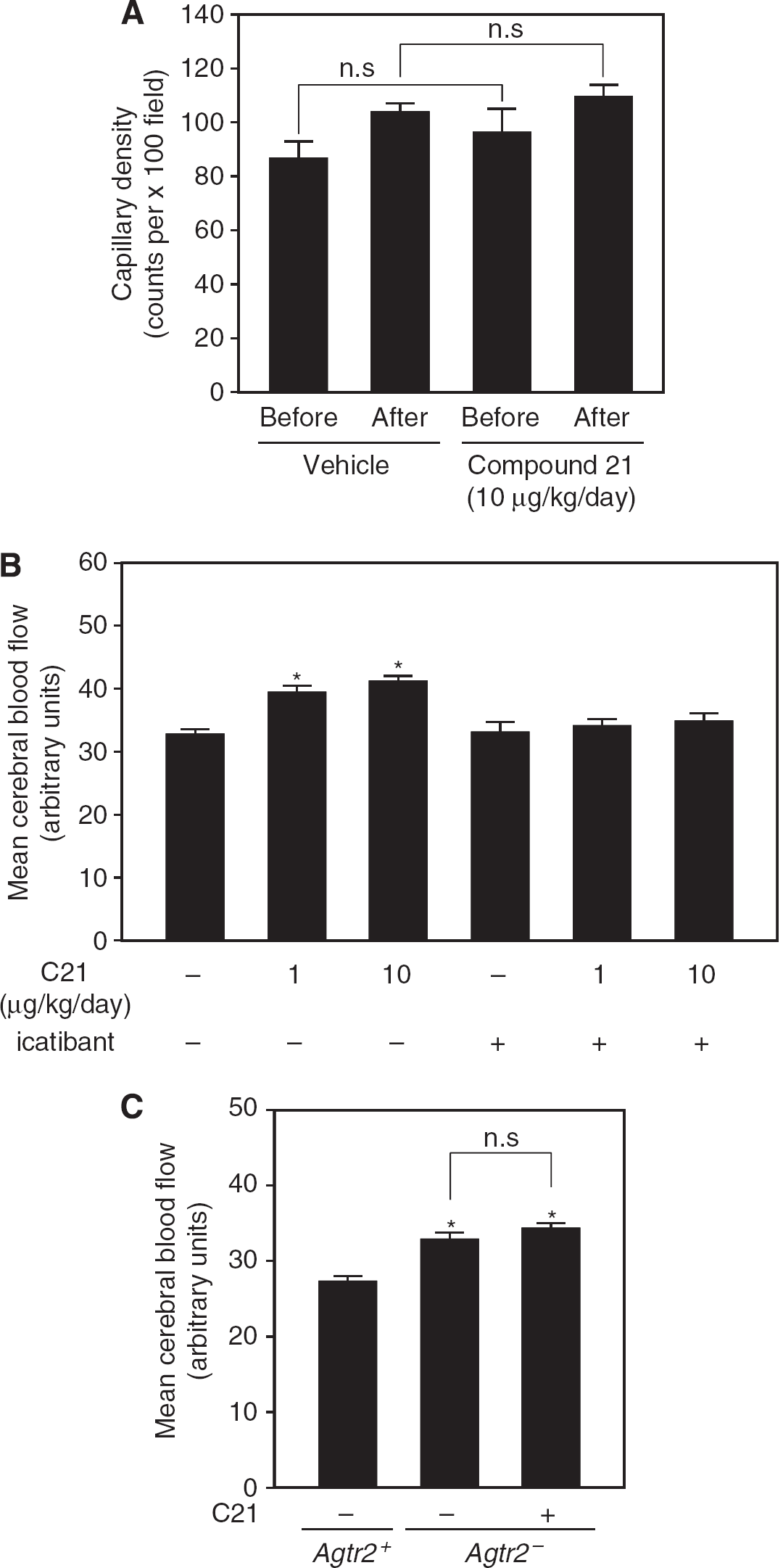
Effect of C21 on capillary density and cerebral blood flow (CBF). (
Effect of Compound 21 on Excitatory Postsynaptic Potential
Compound 21 treatment at two different doses (1, 10 μg/kg per day) significantly increased mean f-EPSP slope and population spike amplitude evaluated by EPSP waveforms elicited by 500 stimulations. The C21-induced enhancement of f-EPSP slope and population spike amplitude was attenuated by icatibant treatment (Figure 4A; representative waveforms are shown in Supplementary Figure 4A), which indicated that the increase in CBF induced by C21 could contribute to neurotransmission in an indirect manner. In contrast, Agtr2− exhibited lower f-EPSP slope and population spike amplitude than those in Agtr2+, which were not affected by C21 treatment (Figure 4B; representative waveforms are shown in Supplementary Figure 4B), suggesting that AT2 receptor activation has a role in synaptic transmission.
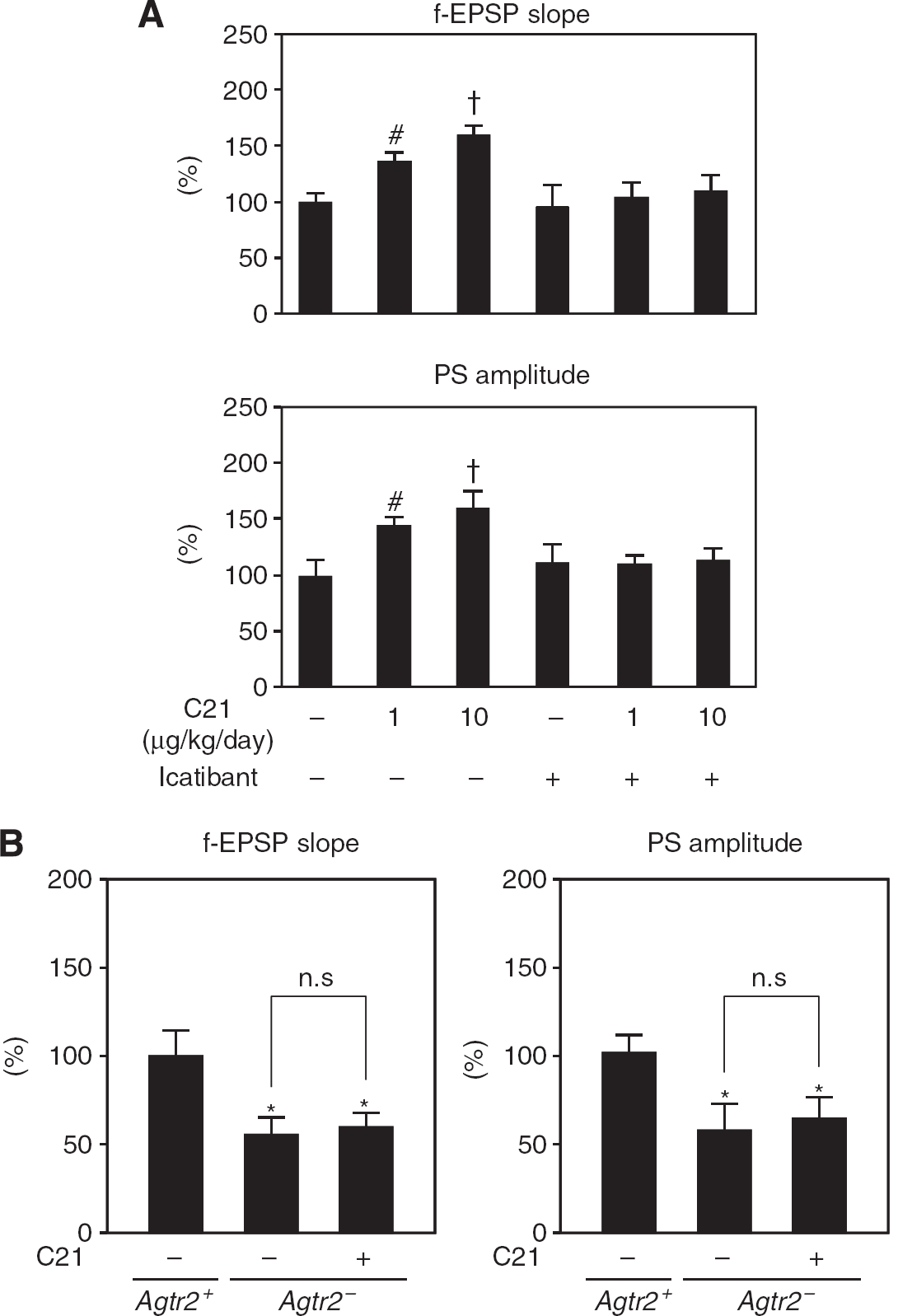
Effect of C21 on baseline of f-EPSP slope and PS amplitude in Schaffer collaterals. (
Effect of AT2 Receptor Stimulation on Neurite Elongation in Hippocampal Neurons
Neurite elongation was assessed using hippocampal neurons prepared from fetal mice. We previously showed that AT1 and AT2 receptors were highly expressed in primary cultured neurons prepared from fetal mice (Li et al, 2007). Treatment with C21 (100 nmol/L) significantly increased mean neurite length per neuron, and an AT2 receptor antagonist, PD123319 (10 μmol/L), decreased this effect (Figures 5A and 5B). Treatment with Ang II (100 nmol/L) enhanced mean neurite length per neuron, which was further increased by an AT1 receptor blocker (ARB), irbesartan (10 μmol/L), and attenuated by PD123319 (Figures 5A and 5B).
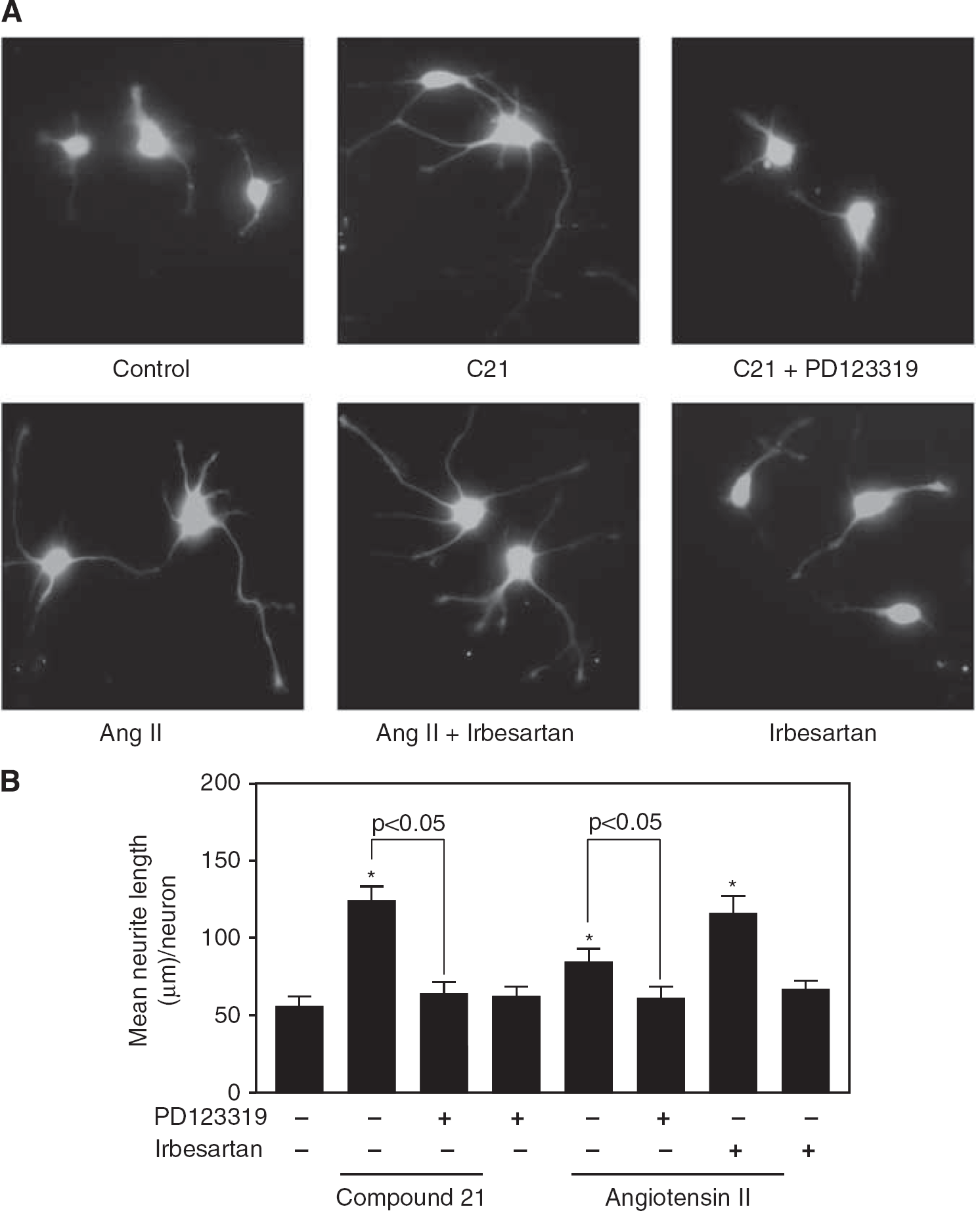
Stimulation of the AT2 receptor promotes neurite elongation in primary cultured hippocampal neurons. (
Effect of Compound 21 on Cognitive Decline an Alzheimer's Disease Mouse Model
We next tried to examine the pathologic relevance of C21-mediated increase in spatial memory. We used an Alzheimer's disease mouse model, produced by intracerebroventricular injection of Aβ 1 to 40. Amyloid-β intracerebroventricularly injected mice showed longer escape latency, as we reported previously (Tsukuda et al, 2009), and administration of C21 attenuated this cognitive decline (Figures 6A and 6B).
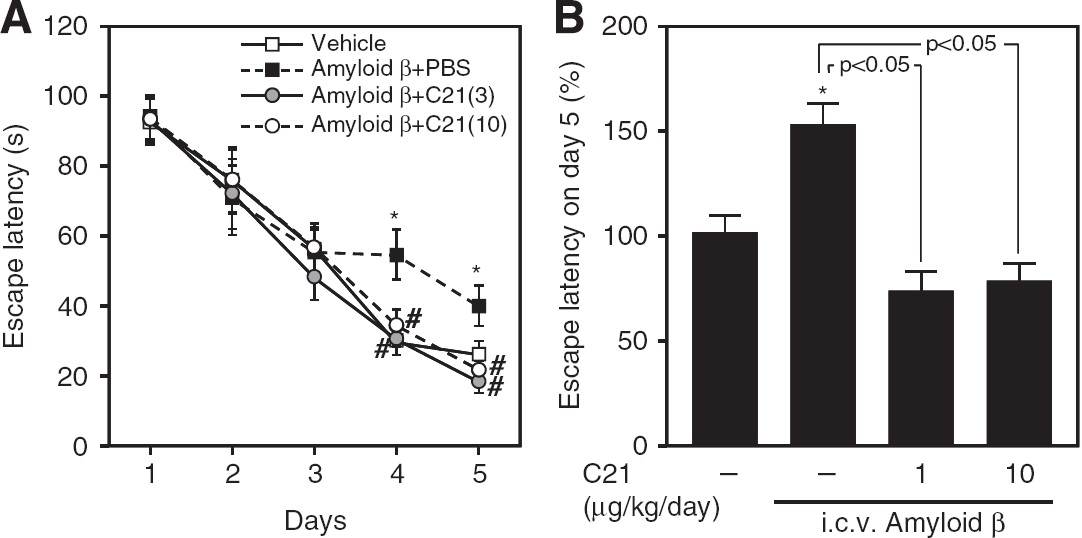
Swim latency of Aβ-injected mice determined by the Morris water maze test. (
Discussion
This study showed that direct AT2 receptor stimulation by C21 enhanced cognitive function possibly through multiple pathways: increase in CBF, enhancement of f-EPSP, and neurite outgrowth in hippocampal neurons. Integration of these C21-mediated mechanisms through further analysis of possible crosstalk would allow us to address the detailed mechanisms of the AT2 receptor stimulation-mediated enhancement of cognitive function.
In recent papers, Rompe et al (2010) showed that C21 has antiinflammatory effects involving inhibition of nuclear factor-κB activity. It was also proved by Matavelli et al (2011) that administration of C21 reduced early renal inflammatory response with production of NO and cGMP. These results indicate that the antiinflammatory action of C21 is mainly focused on the prevention of target-organ damage. Bosnyak et al (2010) showed that administration of C21 evoked a reduction in blood pressure through AT2 receptor stimulation with vasorelaxation in spontaneously hypertensive rats. The detailed mechanism was not investigated in their paper; however, the authors speculated that C21 may elicit classical AT2 receptor-mediated NO signaling in vascular tissue, because C21-induced vasorelaxation was inhibited by N-γ-nitro-arginine methyl ester hydrochloride (
Recent basic studies have suggested the possibility that stimulation of the AT2 receptor could directly promote cell differentiation and regeneration in neuronal tissue (Reinecke et al, 2003). Li et al (2005) reported that AT2 receptor stimulation enhanced neuronal survival and neurite outgrowth in response to ischemia-induced neuronal injury. We previously showed that AT2 receptor stimulation enhanced neural differentiation and the repair of damaged DNA by induction of a neural-differentiating factor, methyl methanesulfonate sensitive-2, which is one of the ubiquitin-conjugating enzyme variants (Mogi et al, 2006). In this study, we observed that direct stimulation of the AT2 receptor enhanced neurite elongation in hippocampal neurons. Moreover, AT2 receptor stimulation directly enhances synaptic plasticity as reported previously (Li et al, 2005, 2007; Reinecke et al, 2003). Therefore, the increase in f-EPSP observed in the hippocampus of mice treated with C21 could be caused by neurite elongation, synaptic augmentation, and increased electrotonic coupling. To investigate the possible direct effect of C21 on cognitive function, we performed intracerebroventricular injection of C21 (Supplementary Table 4). There was no significant difference in swim latency among three groups. However, we are afraid to conclude that C21 does not act directly in the brain or not, because the study design is different in this model compared with systemic treatment model in the original manuscript. First, C21 was continuously administrated by intraperitoneal injection to avoid brain injury with multiple intracerebroventricular injection in the original manuscript. Second, it is not known that the actual dose of C21 was adequate or not in terms of the intracerebroventricular injection in this new set of experiments. Moreover, we could speculate that the blood—brain barrier would be impaired in the diseased states such as stroke, diabetes etc, allowing C21 to penetrate the blood—brain barrier. Therefore, we should further investigate the effects of the systemic treatment with C21 on cognitive function considering the blood—brain barrier penetration, to discuss possible future therapeutic application.
We previously reported that the ARB, telmisartan, could improve cognitive deficit in Aβ-injected mouse model and prevent the deposition of Aβ because of increased CBF (Tsukuda et al, 2009). Kitaguchi et al (2009) also showed that chronic cerebral hypoperfusion increased Aβ fibrils and induced Aβ deposition in the intracellular compartment in amyloid protein precursor transgenic mice. Although the capillary density did not change during the water maze, the CBF increased at least owing to the bradykinin/B2 receptor/NO pathway in our experiment before the water maze. We speculate that increased CBF by C21 prevented Aβ deposition and Aβ-induced neural damage; therefore, cognitive decline was improved by C21 treatment in the Alzheimer's disease mouse model.
In conclusion, administration of an AT2 receptor agonist, C21, enhanced cognitive function, at least owing to an increase in CBF and enhancement of f-EPSP with B2 receptor activation, and increased neurite outgrowth in hippocampal neurons. We also observed that treatment with C21 improved the cognitive decline in an Alzheimer's disease mouse model; however, further investigations are necessary to address the possible direct effects of C21 on cognitive impairment in disease states.
Footnotes
Acknowledgements
The authors thank Takeshi Kiyoi, in Integrated Center for Sciences of Ehime University for technical assistance of immunohistochemical staining for capillary density, Vicore Pharma (Göteborg, Sweden) for providing Compound 21 and Dr Hidemasa Oh, Kyoto University (Kyoto, Japan) for providing green fluorescent protein (GFP) mice.
The authors declare no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
