Abstract
Diffusion tensor imaging (DTI) is a powerful method to visualize white matter, but its use in patients with acute stroke remains limited because of the lack of corresponding histologic information. In this study, we addressed this issue using a hypoxia–ischemia (HI)-induced thrombotic model of stroke in adult mice. At 6, 15, and 24 hours after injury, animals were divided into three groups for (1)
Keywords
Introduction
White matter (WM), occupying 50% of total brain volume in humans, has a metabolic rate similar to that of gray matter (GM) (Zhang and Sejnowski, 2000; Goldberg and Ransom, 2003). Yet, WM receives a disproportionately small blood supply and little collateral circulation, making it particularly susceptible to hypoxic-ischemic insults (Dewar et al, 1999; Arai and Lo, 2009). However, although the importance of WM injury in cerebrovascular diseases is recognized, it is difficult to distinguish it from GM damage and determine the point of irreversible WM injury in acute stroke using traditional imaging methods.
Diffusion tensor imaging (DTI) is a relatively new magnetic resonance imaging (MRI) technique to visualize axonal organization and to measure directional water diffusion in the nervous system (Basser et al, 1994). In DTI, measurements of water diffusion along multiple axes are fitted into a three-dimensional (3D) model to determine three eigenvalues in the principal (
In trauma- or axotomy-induced Wallerian degeneration, axial diffusivity steadily declines and radial diffusivity gradually increases, resulting in a progressive reduction of FA (MacDonald et al, 2007; Zhang et al, 2009). In contrast, patients with acute ischemic stroke often exhibit a rapid reduction of both axial and radial diffusivities, accompanied by little to no change of initial FA (Yang et al, 1999; Bhagat et al, 2008; Sakai et al, 2009). A sizable reduction of FA only occurred ∼24 hours after stroke onset, which was postulated to indicate irreversible axonal injury (Bhagat et al, 2008). However, this hypothesis of important clinical application is yet to be validated, because to date, there are no DTI–neuropathology correlation data in patients with acute ischemic stroke (<24 hours).
In this study, we describe DTI measurements and corresponding histologic changes in a rodent model of thrombotic stroke induced by cerebral hypoxia–ischemia (HI) (Adhami et al, 2006). We show that a combined hypoxic-ischemic insult triggers a rapid reduction of both axial and radial diffusivities in multiple axonal tracts, similar to the pattern observed in acute stroke patients. In contrast, a large reduction of FA only occurs slowly and is restricted to the nerve tracts with severe axonal destruction. Furthermore, the rapid reduction of radial diffusivity correlates with oligodendrocyte swelling and compression of the axoplasma. Finally, the reduction of anisotropy contrast inside the GM of the hippocampus signifies dendritic degeneration. Taken together, these results suggest that DTI can be used to detect and distinguish multiple aspects of axonal and dendritic injuries in acute ischemic stroke.
Materials and methods
Animal Surgery
Male CD-1 mice (Charles River, Wilmington, MA, USA) and Thy1-YFP mice (Jackson Laboratories, Bar Harbor, ME, USA; stock number 003782; Feng et al (2000)), aged 8 to 12 weeks, were used in this study. Details of the HI-induced thrombotic model of stroke have been described previously (Adhami et al, 2006). In brief, animals were anesthetized using 1% to 2% isoflurane while maintaining respiration at 80 to 120 breaths per minute. Partial cerebral ischemia was established by permanent unilateral (right) common carotid artery occlusion. After carotid ligation, hypoxia was initiated by administering 7.5% O2 balanced by nitrogen through a gas mask for 50 minutes under anesthesia. The body temperature of mice was maintained at 37°C±0.5°C using a thermo-controller connected to a rectal probe and heating light. The animal procedures were approved by the Institutional Animal Care and Use Committee and conform to the NIH
At 2 hours after hypoxia, mice were evaluated for neurologic symptoms, and those walking in circles towards the ipsilateral hemisphere were used for analysis in this study. Approximately 80% of HI-challenged animals fulfilled this criterion, and the mortality rate was <10% in CD-1 and Thy1-YFP mice by 24 hours recovery. In total, the following six groups of CD-1 mice were used for sequential MRI histology examinations.
Six hours after HI:
Fifteen hours after HI:
Twenty-four hours after HI:
Six hours after HI:
Fifteen hours after HI:
Twenty-four hours after HI:
Additional CD-1 and Thy1-YFP mice were killed at 1, 6, 15, or 24 hours after HI for various biochemical, histochemical, and immunocytochemical evaluations without MRI or EM (the number of animals used for each assay is indicated in text).
In Vivo Magnetic Resonance Imaging
All data were collected on a Bruker BioSpec 7-T system (Bruker BioSpec 70/30, Karlsruhe, Germany) equipped with 400 mT/m actively shielded gradients. Animals were anesthetized by 1% to 2% isoflurane delivered by oxygen, and the respiration rate was maintained at 60 to 100 breaths per minute. The core temperature was maintained at 37°C by warm air circulated through the magnet bore. All animals were scanned with a custom-built radio frequency coil. Anatomic data were acquired with a 3D, fast spin-echo sequence and the following parameters: echo train length=16, TEeff (echo timeeff)/TR (repetition time)=70.56/1,000 msec, FOV (field of view)=32 × 19.2 × 19.2 mm3, matrix size=256 × 96 × 96, and 1 average, resulting in a resolution of 125 × 200 × 200
Ex Vivo Magnetic Resonance Imaging
Biochemistry
Brain samples for lipid peroxidation were taken from the cerebral cortex on either side of the brain, and homogenized in the phosphate buffer. The amount of malondialdehyde was quantified using a commercial kit following the manufacturer's instructions (Oxis Research, Portland, OR, USA).
Histology
All immunohistochemistry procedures were performed on frozen brain sections following transcardial perfusion of 4% paraformaldehyde and sucrose cryoprotection. The following antibodies were used: a rabbit polyclonal antibody against fibrinogen (a gift from Dr J Degen), MBP (Chemicon, Temecula, CA, USA), Olig2 (Chemicon), MAP2 (Sigma, St Louis, MO, USA), and NeuN (Chemicon). Biotinylated secondary antibodies and streptavidin conjugated to Alexa Fluor 488 or Alexa Fluor 594 (Molecular Probes, Carlsbad, CA, USA) were used to amplify immunosignals. The TUNEL (terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling) was performed as described previously (Yang et al, 2009). Cerebral perfusion analysis was performed by injecting fluorescein isothiocyanate-dextran (5
Electron Microscopy
At indicated times after HI, animals were perfused transcardially with 4% paraformaldehyde and 1% glutaraldehyde in 0.1 mol/L phosphate buffer. The brains were removed and horizontal 100-
Statistical Analyses
All data were analyzed using SPSS for Windows version 12.0 (SPSS, Chicago, IL, USA). Data were first analyzed using a general linear model analysis with time, side (contralateral or ipsilateral), and location as factors. Side and time were significant factors for all locations, but there was no significant side × location effect. There was a significant time × side effect for axial diffusivity and a significant location × time effect for FA. To determine the specific interactions, all data were analyzed for each side using one-way ANOVA (analysis of variance) with time as the factor. Contrasts were defined in this analysis comparing the 6- and 15-hour time points and the 6- and 24-hour time points. After that analysis, each time point was analyzed comparing the contralateral and ipsilateral sides by Student's
Results
Cerebral Hypoxia–Ischemia Produces Progressive Gray Matter Damage and Occasional Axonal T2-Hyperintensity
To induce stroke, mice were treated with unilateral ligation of the common carotid artery, followed by 50 minutes systemic hypoxia (7.5% O2) with the core temperature kept at 37.5°C±0.5°C. This model produced thrombosis and infarction on the HI-challenged side of the brain, while causing no discernible damage to the contralateral side of the brain (Adhami et al, 2006). The undamaged contralateral side of the brain thus served as the internal control for MRI and additional analyses. In Supplementary Figure 1, we showed that at 4 hours after injury, there were still many punctate fibrin clots and pockets of perfusion deficits on the HI-challenged side of the brain but not on the contralateral hemisphere, suggesting an extended period of thrombosis in small arteries.
For
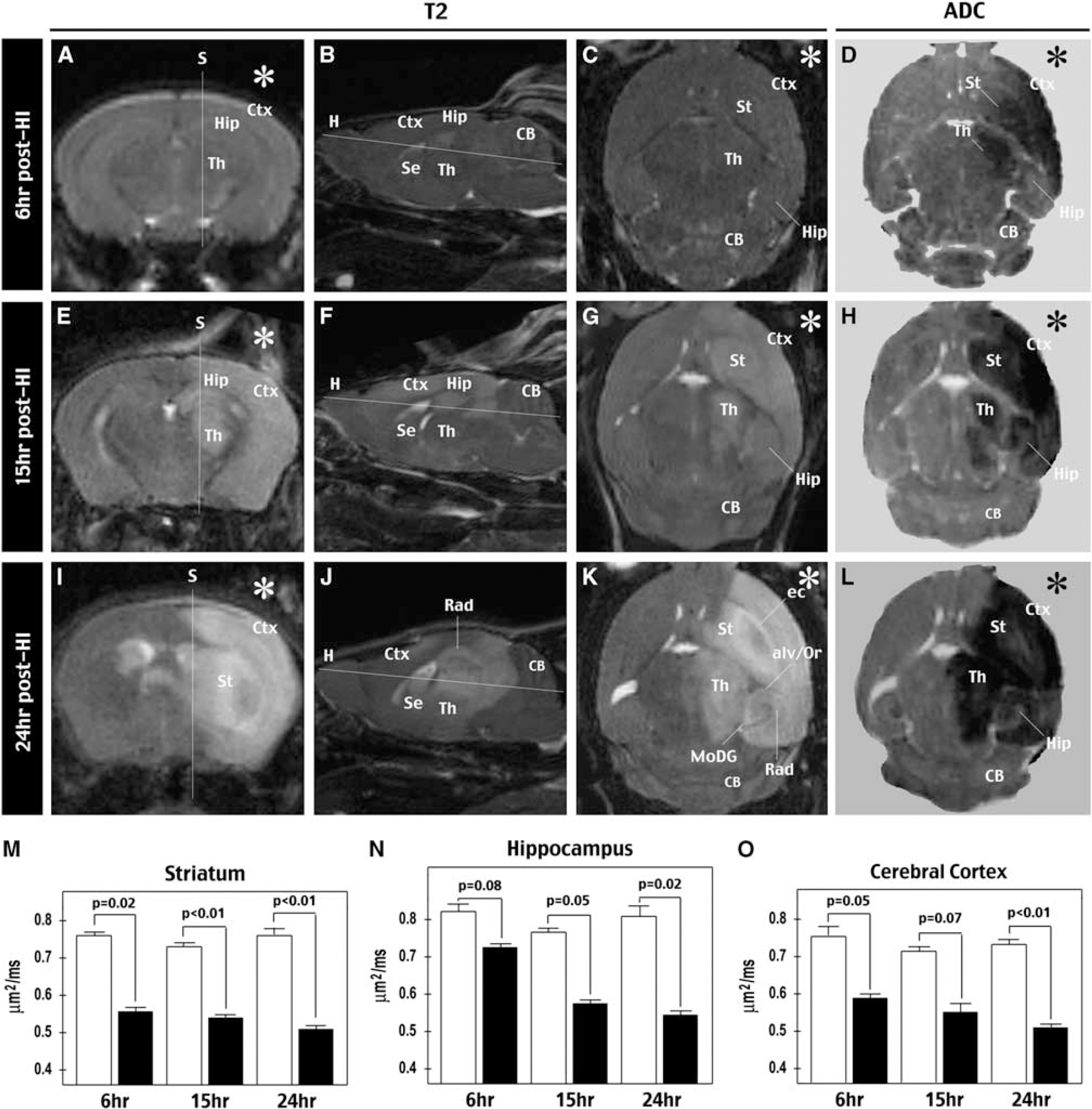
Evaluation of HI-induced brain damage by
The ADC map showed a rapid reduction of isotropic diffusion signals (caused by water accumulation intracellularly or in a tortuous extracellular space) in the striatum and cerebral cortex at all time points (
Visualization of White Matter and Comparison of λ II, λ ⊥, and Fractional Anisotropy Alterations after Cerebral Hypoxia–Ischemia
Next, we used
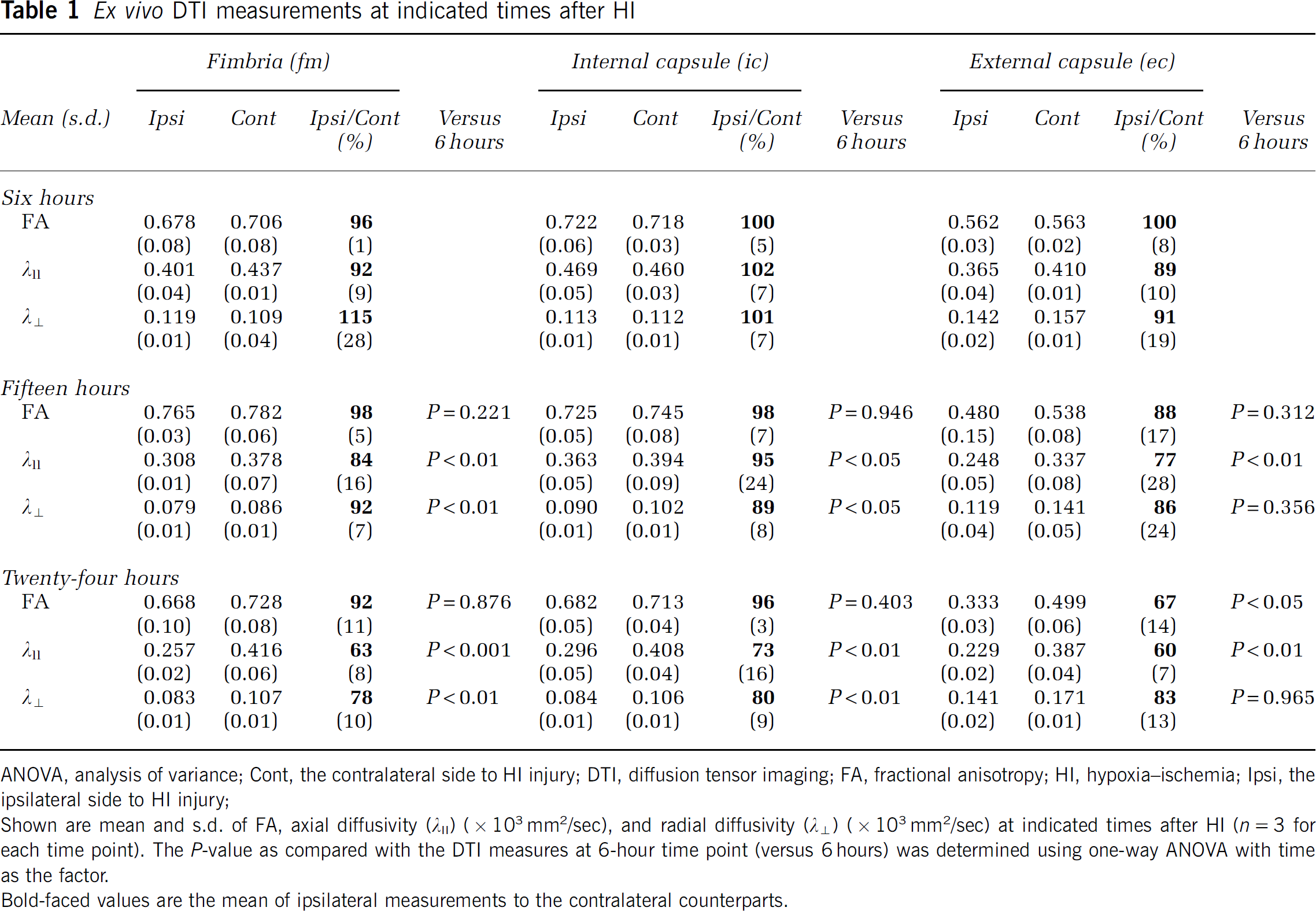
ANOVA, analysis of variance; Cont, the contralateral side to HI injury; DTI, diffusion tensor imaging; FA, fractional anisotropy; HI, hypoxia–ischemia; Ipsi, the ipsilateral side to HI injury;
Shown are mean and s.d. of FA, axial diffusivity (
Bold-faced values are the mean of ipsilateral measurements to the contralateral counterparts.
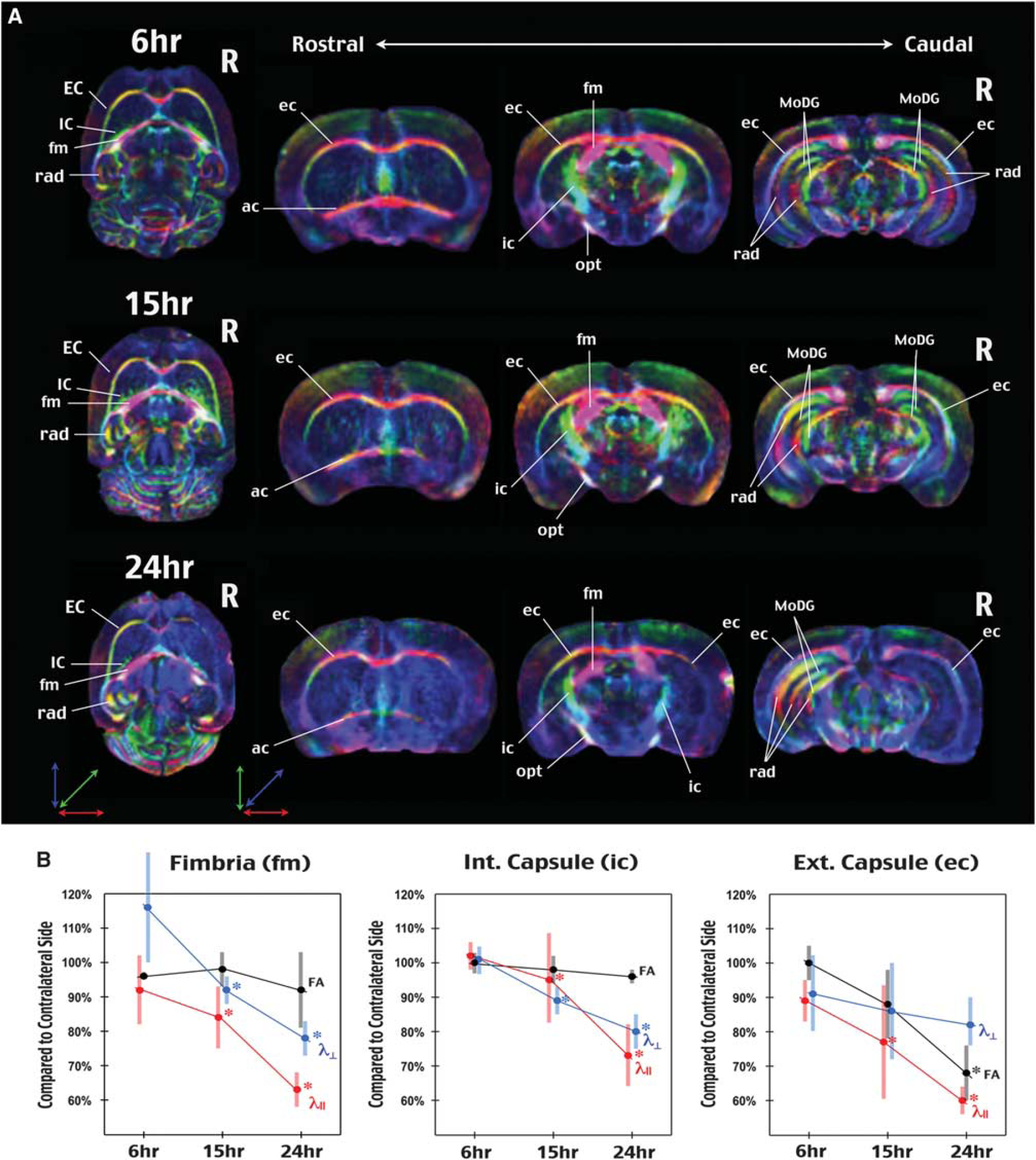
Assessment of HI-induced WM injury by
This analysis showed that axial diffusivity exhibited a steady decline in all three axonal tracts after cerebral HI, decreasing to 63%±8% (mean±s.d., relative to the contralateral side) in the fimbria, to 73%±16% in the internal capsule, and to 60%±7% in the external capsule at 24 hours recovery. These values were also significantly smaller than those at 6 hours on the ipsilateral hemisphere (
In contrast to the trend of gradual decrease of axial and radial diffusivities in all three axonal tracts, a significant reduction of FA after cerebral HI was only detected in the external capsule, in which FA decreased to 67%±14% at 24 h recovery (
Substantial Reduction of Fractional Anisotropy Correlates with Severe Axonal Destruction
To examine this possibility, we used EM and Thy1-YFP mice to assess the extent of axonal injury in this model of thrombotic stroke. Thy1-YFP is a line of transgenic mice that expresses the yellow fluorescent protein in a soluble form to fill the entire cellular architecture of most cortical layer V and hippocampal neurons, thus offering a global survey of axonal injury after cerebral HI (Feng et al, 2000). The analysis using Thy1-YFP mice showed severe breakdown of axonal fascicles in the external capsule (
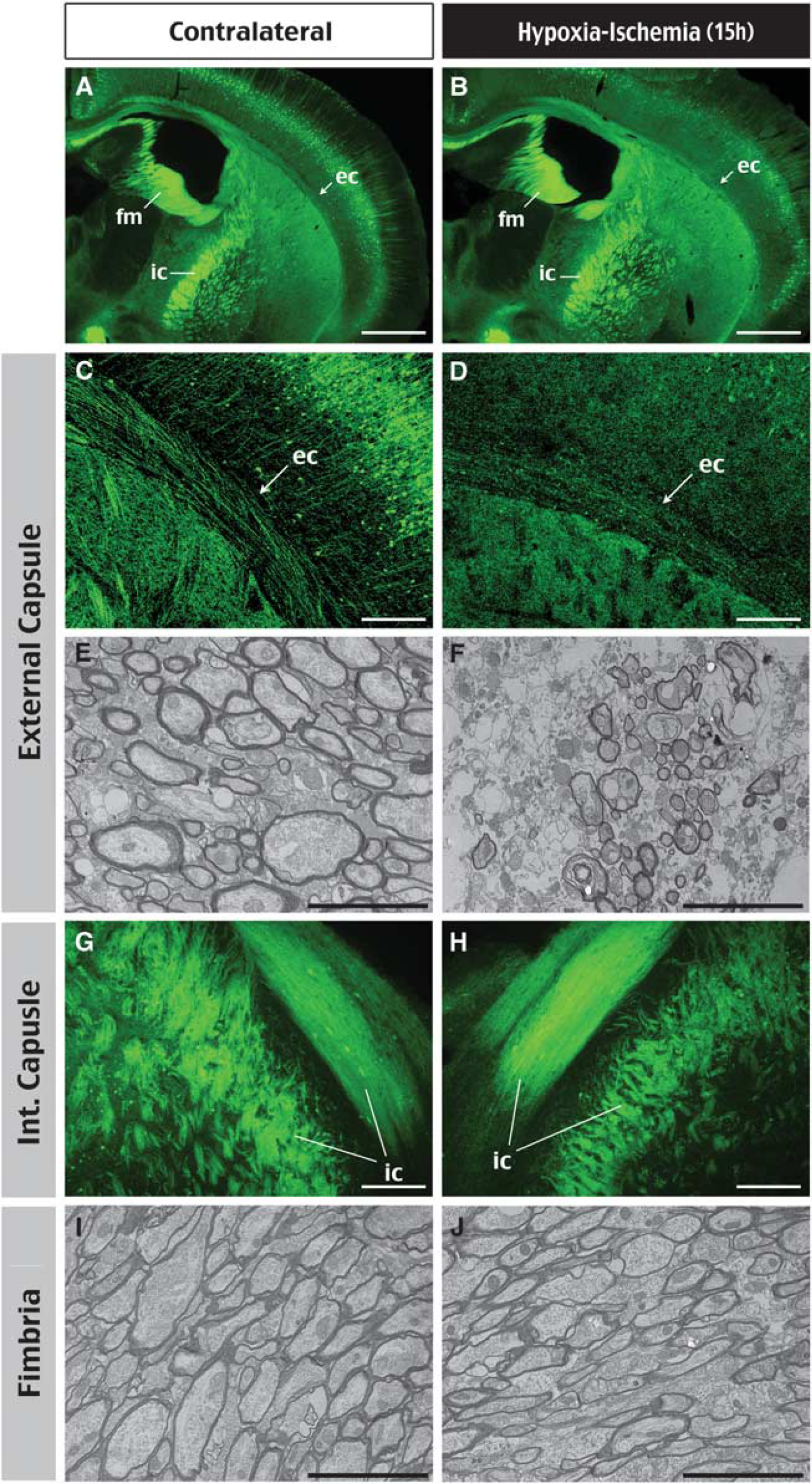
Histologic correlates of HI-induced changes in fractional anisotropy. (
Reduction of Radial Diffusivity after Hypoxia–Ischemia Signifies Acute Oligodendrocyte Damage
The observed early reduction of radial diffusivity in patients and our animal model of stroke stands in contrast to the slow increase in radial diffusivity in trauma- or axotomy-induced Wallerian degeneration (MacDonald et al, 2007; Zhang et al, 2009). Chronic demyelination often causes an increase in radial diffusivity (Song et al, 2002). Thus, the rapid reduction of radial diffusivity after cerebral HI may be caused by retention of myelin sheaths but expansion of the less-anisotropic extraaxonal components in WM (Beaulieu, 2002). To test this hypothesis, we used immunohistochemistry and EM to examine the relationship between myelin and axoplasma after cerebral HI.
We found that HI caused greater reduction of the immunoreactivity against MBP inside the cerebral cortex than in the external capsule or the fimbria at 15 hours recovery (Figures 4A and 4D;
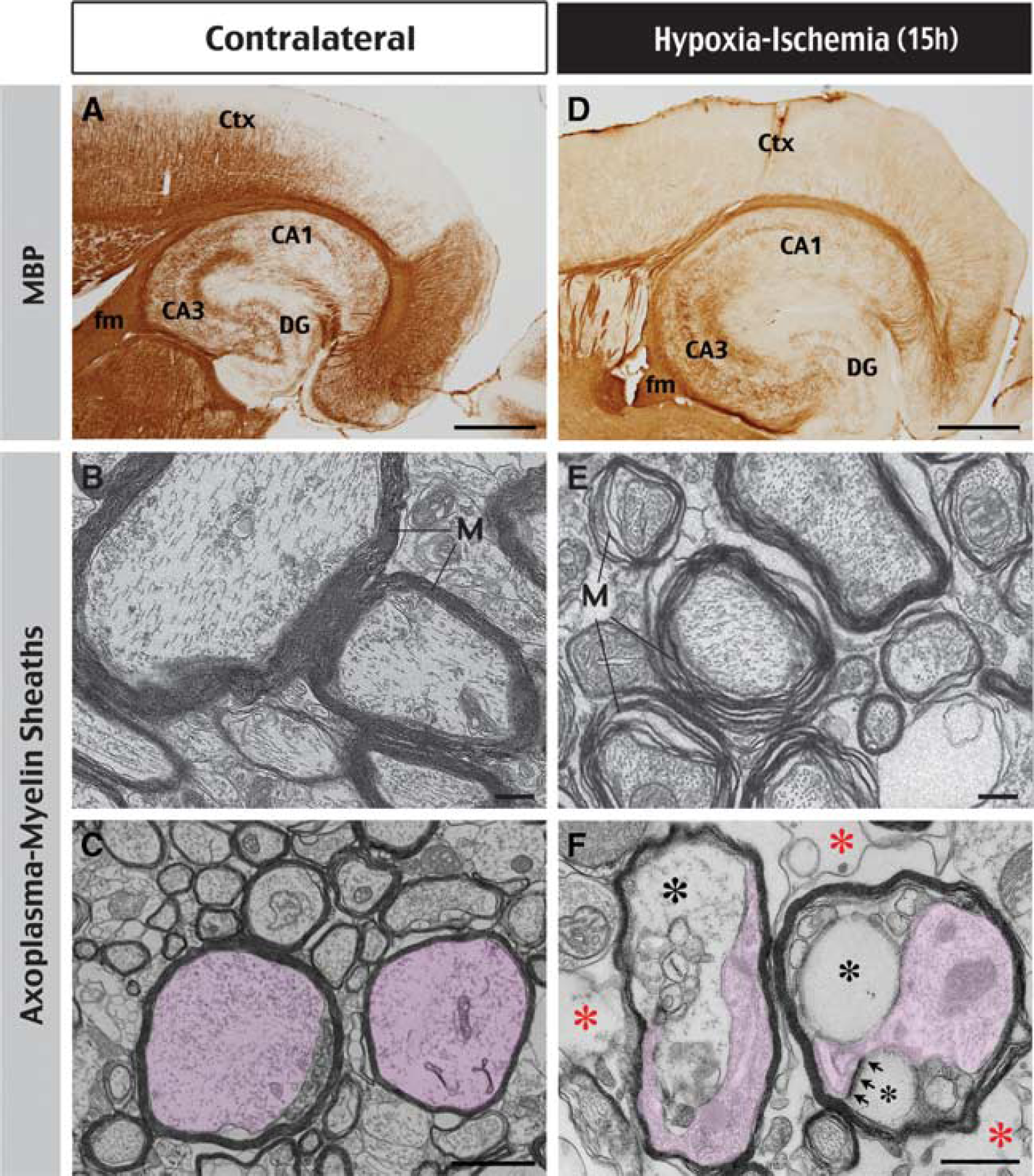
Histologic correlates of HI-induced changes in radial diffusivity. (
With regard to the mechanisms of HI-induced oligodendrocyte injury, previous studies have suggested that oligodendrocytes are very susceptible to oxidative stress owing to an insufficient antioxidant system and a high content of cellular iron (Thorburne and Juurlink, 1996; Irving et al, 1997; McCracken et al, 2000). To test this possibility, we used DHE stain to detect superoxide and found intense signals along the blood vessels on the HI-challenged hemisphere at as early as 1 hour recovery (Figures 5A;
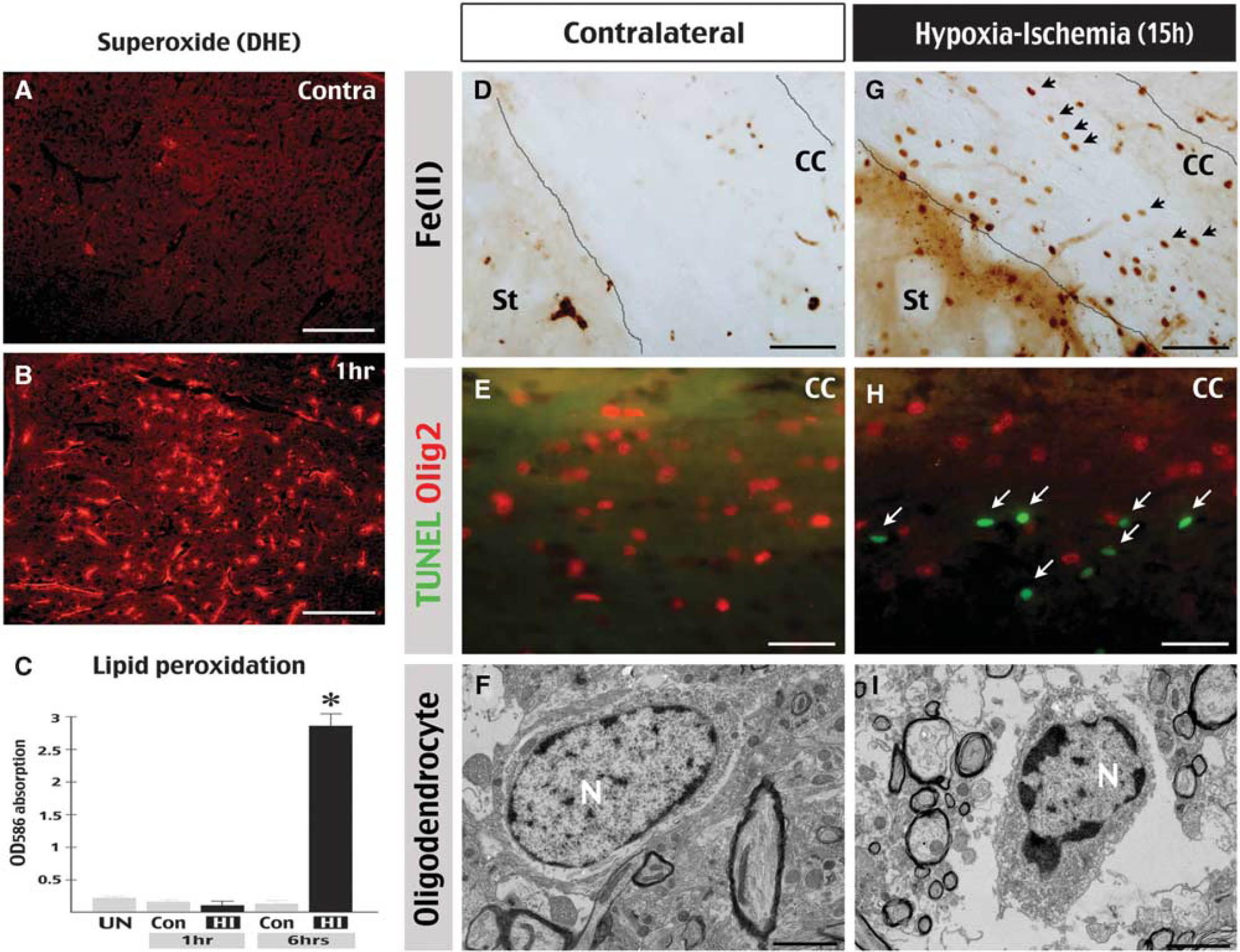
Cerebral HI induces oxidative stress and oligodendrocyte injury. (
Diffusion Tensor Imaging Captures Hypoxia–Ischemia-Induced Dendritic Degeneration in the Hippocampus
Although axons have the highest FA signals in the nervous system, in theory, any tissue with a polarized and orderly microstructure could display diffusion anisotropy (Basser et al, 1994). For example, a previous study using
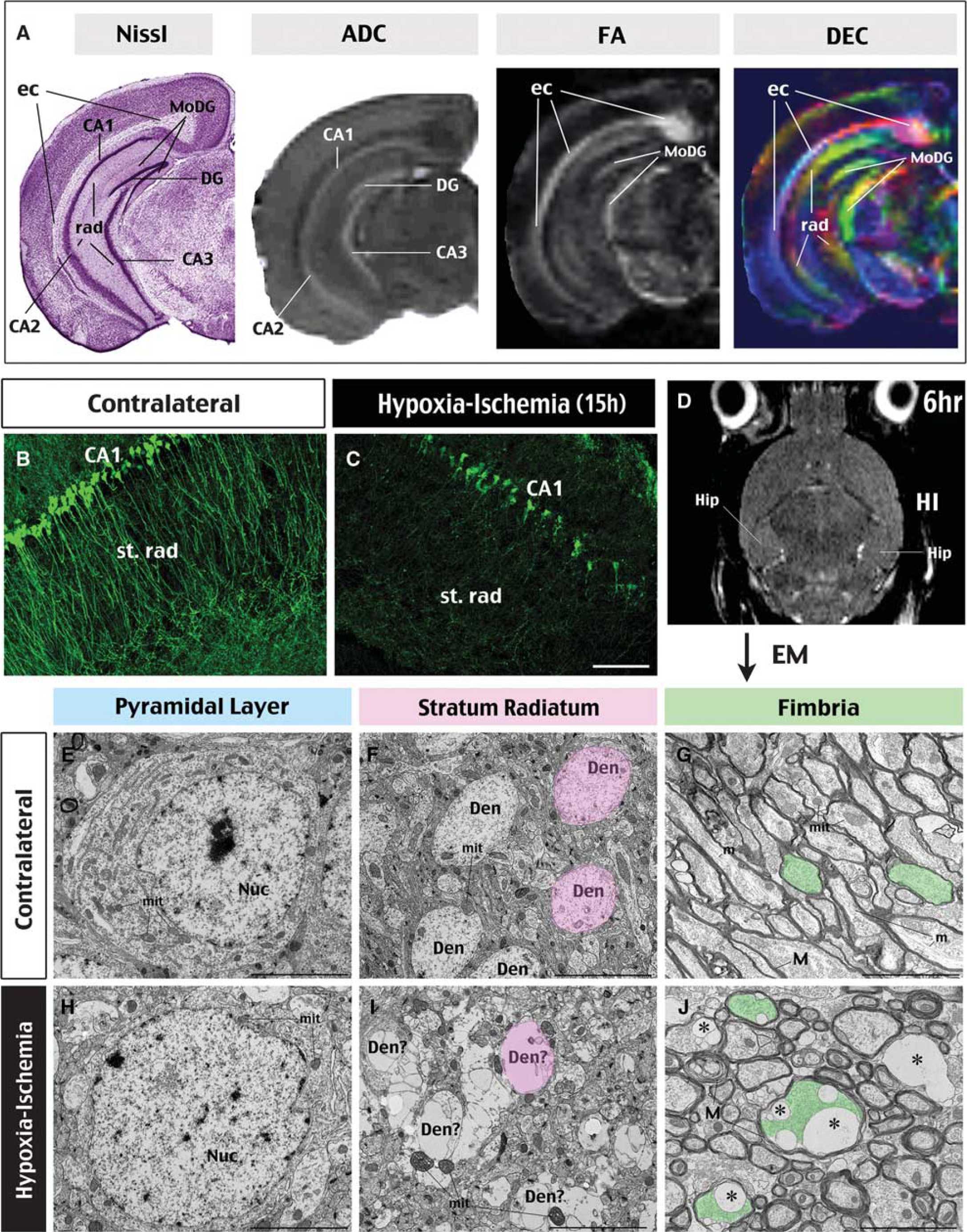
DTI detects HI-induced dendritic degeneration in hippocampal neurons. (
Interestingly, close inspection of the post-HI DEC maps indicated a rapid reduction of diffusion anisotropy contrast in the stratum radiatum on the ipsilateral hemisphere (Figure 2A). By 15 hours after HI, DEC maps showed that the ipsilateral stratum radiatum was almost completely black, coinciding with severe dendritic degeneration shown in Thy1-YFP mice (Figures 6B and 6C). This finding suggests that dendrites of hippocampal neurons are highly sensitive to HI injury. To further examine this possibility, in Supplementary Figure 2, we used TUNEL, Golgi, anti-MAP2, and NeuN immunestains to compare the timing of somal-versus-dendritic injury in the hippocampus. This analysis indicated that dendritic destruction precedes the apoptosis of hippocampal neurons after cerebral HI.
Finally, to evaluate how early HI-induced dendritic degeneration occurs, we used T2-weighted MRI and EM to examine mouse brains collected at 6 hours after HI (
Discussion
Histopathology is the gold standard for establishing the diagnostic utility of any imaging method; however, the systematic imaging–histology correlation is not always feasible in patients. Hence, studies of animal models of the disease may illuminate the neuropathological basis of imaging findings in similar clinical settings. Previous studies have characterized the temporal evolution of diffusion tensor MRI in rodent or nonhuman primate models of cerebral ischemia at 3 and 24 hours and beyond (Carano et al, 2000; Liu et al, 2007). This study uses
Reduction of Fractional Anisotropy after Ischemic Stroke Signals the Structural Breakdown of Axons
Previous studies in patients of ischemic stroke showed a rapid reduction of eigenvalues in all diffusion axes within 24 hours of symptom onset (Yang et al, 1999; Bhagat et al, 2008; Sakai et al, 2009). As FA is a measurement of the relative difference of the three eigenvalues, there was little-to-no decrease in the FA value in the hyperacute (2.5 to 7 hours) phase of ischemic stroke. Only at ∼24 hours after stroke onset did patients show reduction of FA in subcortical WM. This secondary reduction of FA has been suggested to indicate severe axonal damage, but this hypothesis of important clinical application has not been validated by histology (Bhagat et al, 2008). If validated, changes of FA values in individual axonal tracts can be used to monitor WM injury following ischemic stroke.
In this study using a murine model of HI-induced thrombotic stroke, we have reproduced the unique pattern of DTI alterations in ischemic stroke patients. This pattern includes acute reduction of eigenvalues in all three axes (thus, the concomitant reduction of axial and radial diffusivities), as well as a smaller and more variable change of FA in different axonal tracts. We found that the fimbria, internal capsule, and external capsule all had 25% to 40% reduction of axial diffusivity at 24 hours after HI, but showed a great variation in the degree of FA reduction. In the fimbria and internal capsule, there was <10% decrease in FA at 24 hours after HI, whereas the external capsule showed >30% reduction at 24 hours recovery. Importantly, a severe structural breakdown of the axons was only detected in the external capsule, but not in the fimbria or internal capsule. These results support the hypothesis that a large reduction of FA is an imaging marker of severe WM injury in acute ischemic stroke (Bhagat et al, 2008). However, more studies of the DTI–histopathology correlation are necessary to determine the threshold of FA reduction for irreversible axonal injury in acute stroke.
Oligodendrocyte Injury Likely Contributes to the Acute Reduction of Radial Diffusivity
The cellular basis of alterations of radial and axial diffusivities remains incompletely understood (Beaulieu, 2002). Axial diffusivity is believed to represent water diffusion in the lengths of axons, and its reduction could be caused by increased viscosity in the intraaxonal space (owing to cytoskeletal breakdown) or focal enlargement constriction of neuritis (‘beading’) (Budde and Frank, 2010). Microtubules and myelin sheaths are generally agreed to be principally responsible for radial diffusivity, because they constrain transverse water diffusion across the axons. In trauma- or axotomy-induced Wallerian degeneration of axons, radial diffusivity slowly increases as a consequence of demyelination (MacDonald et al, 2007; Zhang et al, 2009). In contrast, clinical studies of stoke patients and our data in animal models all demonstrated an acute, concomitant reduction of axial and radial diffusivities. This disparity of DTI alterations suggested unique pathologic changes in ischemic stroke. We found that edema, separation of myelin sheaths, and protrusion of oligodendrocyte-derived vesicles compressing the axoplasma are typical WM pathologies in acute ischemic stroke. Our data of large, intramyelinic vesicles are strikingly similar to those previously described in isolated optic nerves after anoxic insults (Waxman et al, 1992), and consistent with the notion that oligodendrocyte swelling is one of the earliest morphologic changes in cerebral ischemia (Pantoni et al, 1996). It is conceivable that an increase in extraaxonal water content and compression of the axoplasma by swollen myelin sheaths may lead to reduction of radial diffusivity and even axial diffusivity in acute cerebral ischemia.
Unlike Wallerian degeneration in which the injury of oligodendrocytes is a slow process (Vargas and Barres, 2007), our data suggested that oligodendrocytes undergo rapid degeneration after hypoxic-ischemic insults. Although the exact causes of this hypersensitivity remain to be determined, our results support the notion that reactive oxidative stress and a high iron content in oligodendrocytes contribute to their increased sensitivity to hypoxic-ischemic insults (Thorburne and Juurlink, 1996; Irving et al, 1997; McCracken et al, 2000; Dewar et al, 2003). Importantly, recent studies have suggested that oligodendrocytes have a critical role in maintaining axonal function and survival (Kassmann and Nave, 2008). Hence, supply of antioxidants to oligodendrocytes may be a useful strategy for WM protection in acute ischemic stroke.
Diffusion Tensor Imaging may Detect Hypoxia–Ischemia-Induced Dendritic Degeneration in Laminated Brain Structures
This study also suggests the potential of DTI for detecting dendritic degeneration in hippocampal neurons, which may have implications beyond acute ischemic stroke. Diffusion anisotropy results from directional water flow in any tissue of a polarized intracellular structure and an orderly architecture (Basser et al, 1994). Although neural fibers are the prime example for structures with diffusion anisotropy, they are not the only such structures in the body. For example, a previous study using
Moreover, using T2-weighted MRI followed by EM analysis, we have shown that the degeneration of hippocampal dendrites is a very early event in ischemic stroke. This conclusion is consistent with a recent
Microscopic dendritic degeneration (only detectable by EM). Swelling of oligodendrocytes (reduction of radial and axial diffusivities). Massive degeneration of hippocampal dendrites (absence of diffusion contrast in DTI). Structural breakdown of axons (at least >10% reduction of FA in DTI). Massive accumulation of water in axon tracts (hyperintensity on T2-weighted MRI).
It is interesting to note that WM-associated T2-hyperintensity in our study resembles the hallmark of leukoaraiosis, an incident MRI finding in the elderly and an important contributor of vascular dementia (Hachinski et al, 1986; Schmidt et al, 2007). Moreover, a recent study suggested that leukoaraiosis-like WM lesions are caused by chronic hypoxia hypoperfusion (Fernando et al, 2006), which could also injure dendrites. Taken together, our results and the relevant literature raise an intriguing possibility that the decline of cognitive functions in leukoaraiosis-like vascular dementia may be in part caused by the ‘invisible’ dendritic degeneration (and the consequential loss of synaptic inputs) instead of strict WM injury. If this hypothesis is correct, DTI may be a useful tool for early diagnosis of dendritic degeneration and vascular dementia in the future.
In conclusion, we have used an animal model to study the utility of anisotropic diffusion imaging in acute ischemic stroke. Although our findings with
Footnotes
Acknowledgements
The authors thank Drs Ton Degrauw, Joseph Broderick, Lawrence Wechsler, and Charles Dumoulin, the anonymous reviewers for critical reading of this manuscript, and Dr Steve Danzer for providing Thy1-YFP-H mice.
The authors declare no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
