Abstract
Apolipoprotein E (ApoE), a cholesterol transporter and an immunomodulator, is brain protective after experimental stroke and implicated in brain repair. Here, we study the involvement of ApoE in the restoration of brain function after experimental stroke, by using animal housing conditions that differentially improve recovery after occlusion of the middle cerebral artery occlusion (MCAO). We found that after MCAO the ApoE levels increased in the injured hemisphere over a 30 days recovery period. The exception was a proximal narrow peri-infarct rim, in which ApoE was solely localized in S100
Introduction
Spontanous neurologic recovery after stroke is slow and incomplete because of the growth inhibitory milieu in the poststroke brain (Cramer, 2008a). The observed recovery has been attributed to the activation of remaining, silent and/or newly established neural pathways. In the experimental setting, three types of processes have been discerned that influence the speed and extent of recovery: (1) resolution of inhibitory processes such as edema, diaschisis, inflammation, and axonal growth inhibition; (2) formation and reactivation of synapses; and (3) ‘relearning’ of new neuronal connections and networks (Cramer, 2008a; Wieloch and Nikolich, 2006). The three events are partly overlapping though to large degree consecutive. Recovery can be enhanced by pharmacological treatment, rehabilitative training, by electrical and magnetic stimulation of the ipsilateral or contralateral corticies, or by environmental influences (Cramer, 2008a, 2008b; Wieloch and Nikolich, 2006). An understanding of the complex recovery processes on the cellular and molecular level, particularly those occuring in the peri-infarct area (Carmichael, 2006), may lead to the development of new regenerative poststroke therapies (Wieloch and Nikolich, 2006).
Housing rodents in an enriched environment (EE) mitigates functional deficits caused by brain disease and damage, and when starting days after experimental stroke profoundly improves the neurologic function (Rosenzweig and Bennett, 1996; Ohlsson and Johansson, 1995; Nygren and Wieloch, 2005). This recovery enhancing effect is not because of a decrease in tissue damage, but rather because of brain plasticity (Johansson, 2004). In addition, enriched housing induces dynamic temporal expression of genes encoding synaptic proteins and growth factors (Witte et al, 2000; Carmichael et al, 2005).
We earlier showed that expression of the lipid carrier molecule ApoD is increased in the peri-infarct region after housing rats subjected to middle cerebral artery occlusion (MCAO) in an EE (Rickhag et al, 2008). We proposed that enhancing lipid transfer among cells in the peri-infarct area would facilitate processes of regeneration and wound healing at the infarct core-peri-infarct interface (Rickhag et al, 2008). The 34 kDa glycoprotein apolipoprotein E (ApoE) is another important lipid and cholesterol transport molecule, but also an immunomodulator (Pepe and Curtiss, 1986). It is constitutively expressed in the brain, upregulated after brain injury (Lynch et al, 2002), implicated in Alzheimers disease (Cedazo-Minguez, 2007), and present in neurons, oligodendrocytes, microglial cells, and astrocytes (Aoki et al, 2003; Stoll and Muller, 1986). Apolipoprotein E is involved in mechanisms of neuronal degeneration (Kamada et al, 2003), regeneration (Sun et al, 1998), and glial scar formation (Blain et al, 2006), and is both pro- and antiinflammatory (Barger and Harmon, 1997; Lynch et al, 2001). Apolipoprotein E may therefore be beneficial or detrimental depending on context of brain pathology (Koistinaho and Koistinaho, 2005; Guo et al, 2008).
The aim of the present investigation was to analyze the spatio-temporal ApoE expression in different areas of the hemisphere, ipsilateral to the lesion including the infarct core of rats housed in an EE or standard cages after MCAO, with particular emphasis on a role in tissue regeneration.
Materials and methods
Transient Rat Middle Cerebral Artery Occlusion (tMCAO)
All animal experiments were performed with the approval of the Malmö-Lund ethical committee. Transient MCAO was induced as described earlier (Rickhag et al, 2008). In brief, male Wistar rats (325 to 350 g, HsdBrlHan, Harlan Scandinavia, Denmark) were housed under diurnal light conditions and were fasted for 12 h before surgery. During surgery physiologic parameters (arterial blood pressure and gases after tail artery cannulation and insertion of a catheter (SIMS Portex, UK) for rectal body temperature) were measured and controlled within physiologic limits. Moreover, body temperature was measured after 1 and 2 h of occlusion and after 1 and 2 h of recirculation. Rats were anesthesized (initial 4% fluothane in N2O/O2 (70:30), during surgery 2% fluothane in N2O/O2, Astra Zeneca, Sweden) and the right common carotid artery and external carotid artery were occluded permanently, the internal carotid artery was exposed and ligated. A nylon filament (top diameter 0.3 to 0.4 mm) was introduced into the internal carotid artery through a small incision into the distal end of the common carotid artery and preaded up to occlude the origin of the MCA for 2 h. The filament was withdrawn and awake rats were placed in a cooling box. Two hours after occlusion, a neurologic score was assessed and only rats showing rotational asymmetry and dysfunctional limb placement were included into the study. Rats were killed at different time points after tMCAO (1, 2, 4, 7, 14, and 30d, Figure 1) and fixative-perfused brains were processed for immunohistochemical analyses. The same procedure was performed in sham-operated animals but no filament was introduced into the internal carotid artery.

Experimental design.
Permanent Rat Middle Cerebral Artery Occlusion (pMCAO)
Adult male spontaneously hypertensive rats (17-weeks old) were anesthesized with 60 mg sodium pentobarbital (60 mg/mL/kg BW). Marcain (1.25 mg/kg) was used for local anesthesia. After a dissection of the right temporal muscle, a small hole was drilled into the scull bone. The right MCA was localized and ligated (Ohlsson and Johansson, 1995). After surgery, animals were kept on a warm blanket to maintain body temperature at 37°C. The MCA was exposed but not ligated in sham-operated animals.
Housing Conditions and Behavioral Testing
Two days after surgery, rats were randomly selected for housing conditions, either in standard cages with one cage mate, or multilevel enriched cages with four to seven mates for 3 days (Ohlsson and Johansson, 1995). Multilevel EE cages (size 600 mm × 600 mm × 1,200 mm) were equipped with removable platforms, grids, pipes, and ropes. Sensori-motor function was evaluated using the rotating pole test as described earlier (Rickhag et al, 2008). In brief, rats traverse a rotating wooden pole (length 1,500 mm, diameter 40 mm, and elevation 700 mm) at 0, 3, and 10 r.p.m. Every animal was trained once 12 h before surgery and tested 2 and 5 days after MCAO. Performance was video recorded and evaluated by an experienced person blinded to the study. Sensori-motor dysfunction was assessed by using a 6 to 0 scoring system: 6 denotes the animal traverses pole without any foot slips; 5 denotes the animal traverses the pole with few foot slips; 4 denotes the animal crosses pole with 50% slipping of the foot steps; 3 denotes the animal crosses the pole while jumping with both hindlimbs; 2 denotes the animal falls off during crossing; 1 denotes the animal remains embraced to the pole unable to cross and eventually falls off the pole; 0 denotes the animal falls off immediately. A healthy rat performs with a score of 5 or 6.
Infarct Size Measurements
Fresh frozen sections (thickness 40
Tissue Sample Preparation
Brains were immediately frozen in isopentane (−40°C) and further cooled down to −70°C on dry ice. Tissue samples were collected from 40
Western Blotting
Ten micrograms of protein were separated on a 10% SDS polyacrylamide gel. Blocking was performed onto polyvinyldifluoride membranes using blocking buffer (20 mmol/L Tris, 136 mmol/L NaCl, pH 7,6, 0,1% Tween 20, 5% nonfat dry milk), and detected using primary polyclonal antibody against the ApoE (Santa Cruz Biotechnologies, CA, USA, diluted 1:2,500). After incubation overnight at 4°C, signals were obtained by binding of a secondary anti goat HRP-linked antibody (Santa Cruz Biotechnologies, diluted 1:3,000) and visualized by exposing the membrane to a CCD camera (raytest, Germany) using a chemiluminescence kit (Amersham Biosciences, UK).
Membranes were stripped and reprobed for
Immunohistochemistry
Brain sections (thickness 30
Immunofluorescence
Sections were processed as described for immunohistochemistry. For colocalization of proteins, the following antibodies were used: polyclonal anti-ApoE (1:2,000, Santa Cruz Biotechnology), anti-glutathione-S-transferase-pi (GST-pi) (1:200, BD Tansduction Laboratories, CA, USA), monoclonal mouse anti-NeuN (1:300, Millipore, Hampshire, UK), monoclonal directly Cy3-conjugated anti-GFAP (1:600, Sigma-Aldrich, MO, USA), and monoclonal mouse antibody Ox-42 (1:800, Millipore). After over night incubation at 4°C, cells were incubated with appropiate secondary antibodies (Cy3-conjugated donkey anti mouse antibody, biotinylated horse anti goat antibody, both diluted 1:200, Jackson Laboratories, Suffolk, UK). Sections exposed to the secondary biotinylated horse anti goat antibody before were further incubated with a Alexa 488 streptavidin conjugate (1:200) at room temperature for 60 mins. Fluorescent signals were visualized using a confocal microscopy system (LSM510 Zeiss, Germany). Nuclear stainings were achieved by 4′,6-diamidino-2-phenylindole (DAPI) in a dilution of 1:1,500 for 5 mins and visualized using an appropiate emission filter in a Olympus BX60 microscope (Solna, Sweden).
Interleukin-1β Enzyme-Linked Immunosorbent Assay
Interleukin-1
Cell Counting Analysis
Coronal sections (bregma −1.2 to −0.7) were stained for ApoE from animals housed in standard or EE and only cells showing the typical appearance of ramified astrocytes were counted in the low ApoE expression area around the infarct.
Statistics
Unless otherwise stated all data are presented as means±s.d. Data from the rotating pole test were analyzed by the Mann–Whitney
Results
The Spatio-Temporal Changes in the ApoE Levels in Cells of the Injured Brain After tMCAO
Figure 2 shows ApoE immunoreactivitiy in the core of the infarct and the peri-infarct area at different recovery times after tMCAO in the rat. In noninjured animals ApoE was found in capillaries and astrocytes in the neocortex (Figure 2A). Twenty-four hours after MCAO (Figure 2B), a slight increase in ApoE expression was observed in ramified cells in close vicinity to the infarct border and round cells in the infarct core with further accumulation at 48 h after MCAO (Figure 2C). At days 4 and 7 after MCAO, the ischemic core contained strong ApoE+ cells (Figure 2D-a), whereas in an adjacent approximately 200 to 500
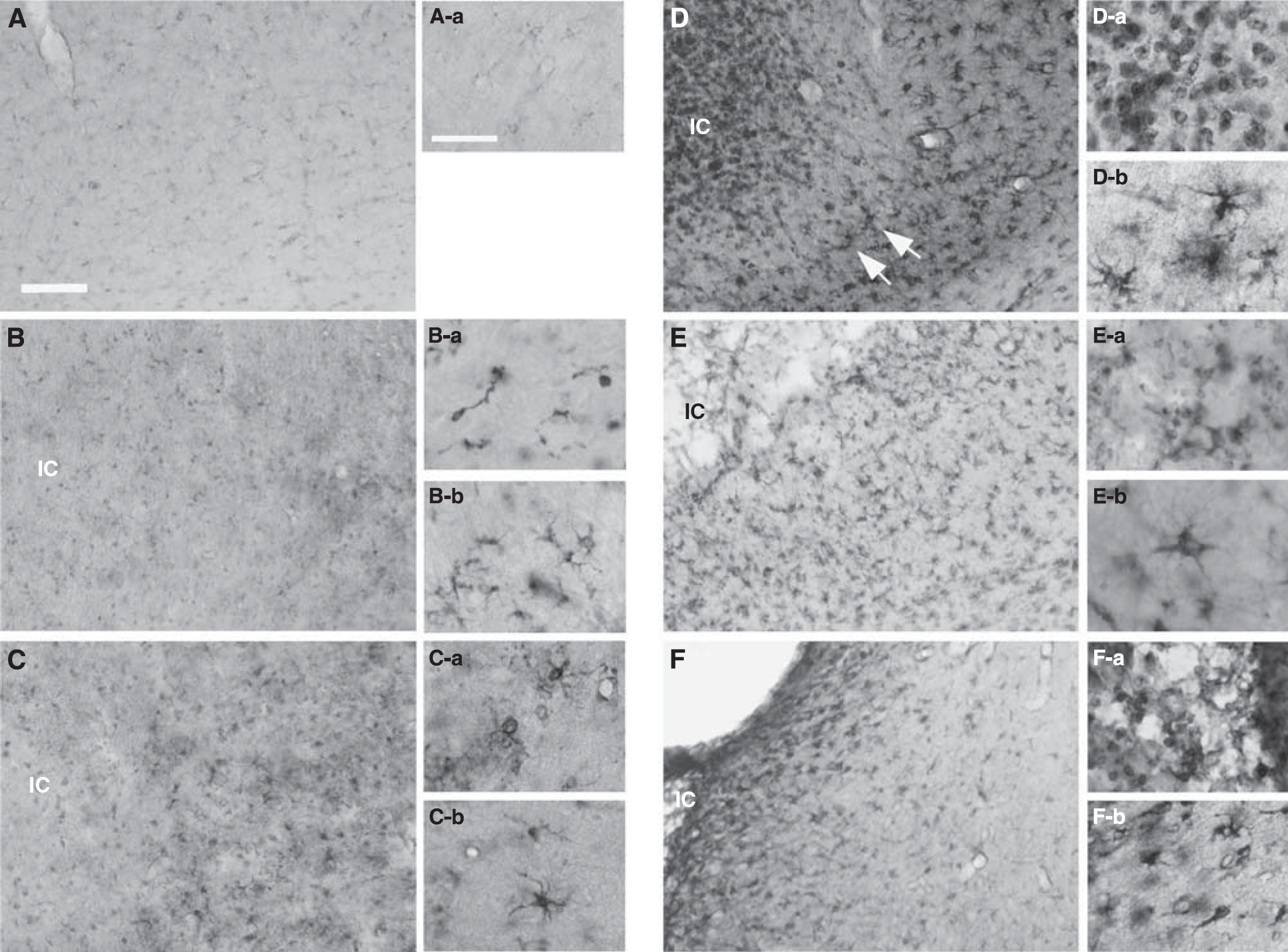
Expression of ApoE after tMCAO. Phase contrast micrographs (
In the Peri-Infarct Rim, ApoE Is Only Present in S100β + Astrocytes
Figure 3 shows a detailed analysis of the low ApoE expression zone. This area had a lower number of DAPI+ cells compared with the core and the distal peri-infarct region (Figure 3A) but still contained unchanged number of NeuN-positive cells (Figure 3B). The ApoE+ cells in the core were mainly CD11b+ microglia cells (Figure 3C) and the ApoE+ cells in distal peri-infarct area were GFAP expressing astrocytes (Figure 3D). Though the infarct rim contained markedly less ApoE+ cells, a population of ramified cloud-like cells showed a strong immunoreactivitiy for ApoE (Figure 3F, magnification in Figure 3G). These cells showed a typical morphology for reactive astrocytes, but weak or no immunoreactivitiy for GFAP (Figure 3D) (Wilhelmsson et al, 2006). In contrast, these ApoE+ cells were strongly immunoreactive for S100
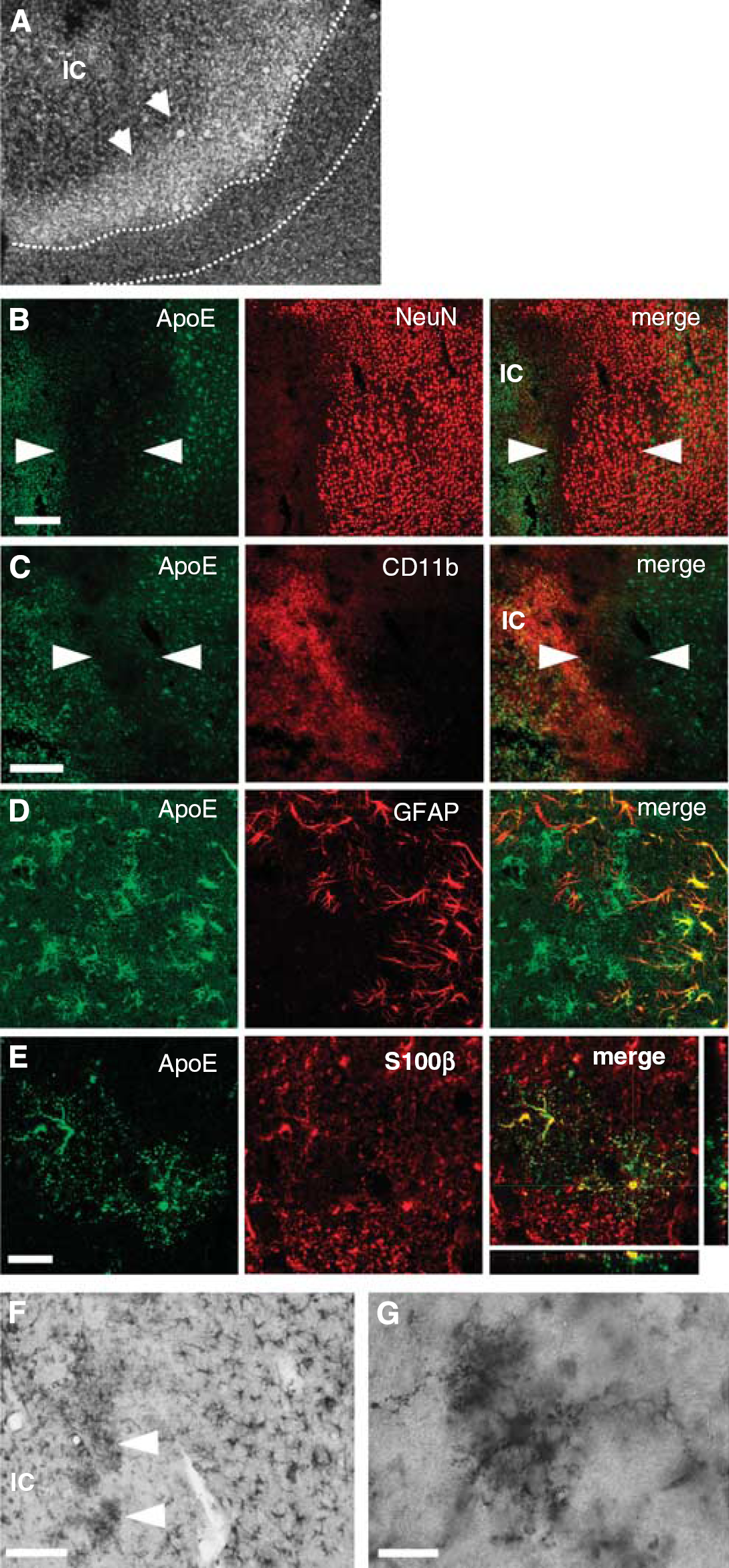
Characterization of a low ApoE expressing zone. (
Housing in an Enriched Environment Decreases ApoE in the Peri-Infarct Region
To assess the significance of the increase in ApoE after MCAO we investigated the effect of housing animals in an EE, which improves motor function without affecting infarct size (Figures 4A and 4B). Using this experimental paradigm we compared the ApoE protein levels in the brains after 5 days of recovery after pMCAO in rats housed in standard conditions with those that have been in enriched housing conditions for the last 3 days. A robust increase in ApoE was seen in GFAP+ astrocytes in the peri-infarct region, with a gradient of increasing ApoE levels toward the peri-infarct rim in rats housed in standard cages (Figure 4A). In contrast, the number of ApoE expressing GFAP+ astrocytes, and also the intensity of ApoE immunoreactivity was lower in rats housed in EE (Figure 4B). In the proximal peri-infarct rim (Figures 3C and 3F), the ApoE+/S100
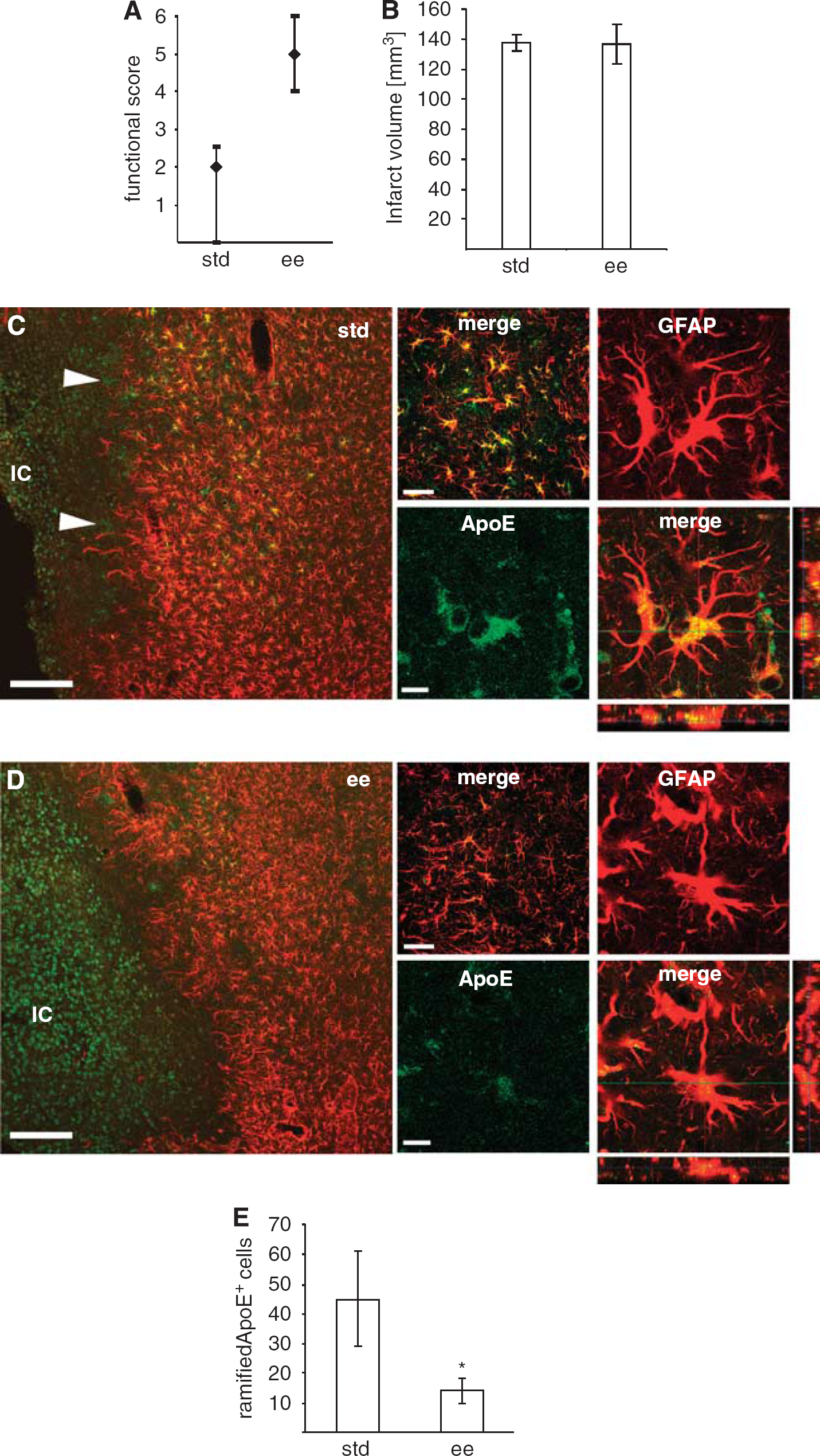
Reduced ApoE expression in reactive astrocytes of the peri-infarct region from rats housed in EE after pMCAO. (
To verify the immunohistochemical findings, the ApoE levels at 5 days after pMCAO in the two housing conditions were analyzed from different brain regions. Samples of brain homogenates were obtained from rats 5 days after MCAO and ApoE levels were analyzed in the core and peri-infarct regions and from the ipsilateral cortex of sham-operated animals (Figure 5A). As shown in Figure 5C, the ApoE levels were markedly higher in the peri-infarct region compared with the infarct core. In addition, the ApoE levels were significantly lower in the infarct core and peri-infarct area, in rats housed in an EE, in the peri-infarct zone by more than 60%. In summary, we show that housing in an EE significantly mitigates the increase in ApoE during recovery after experimental stroke, particular in the peri-infarct zone and in reactive astrocytes of the proximal peri-infarct rim.
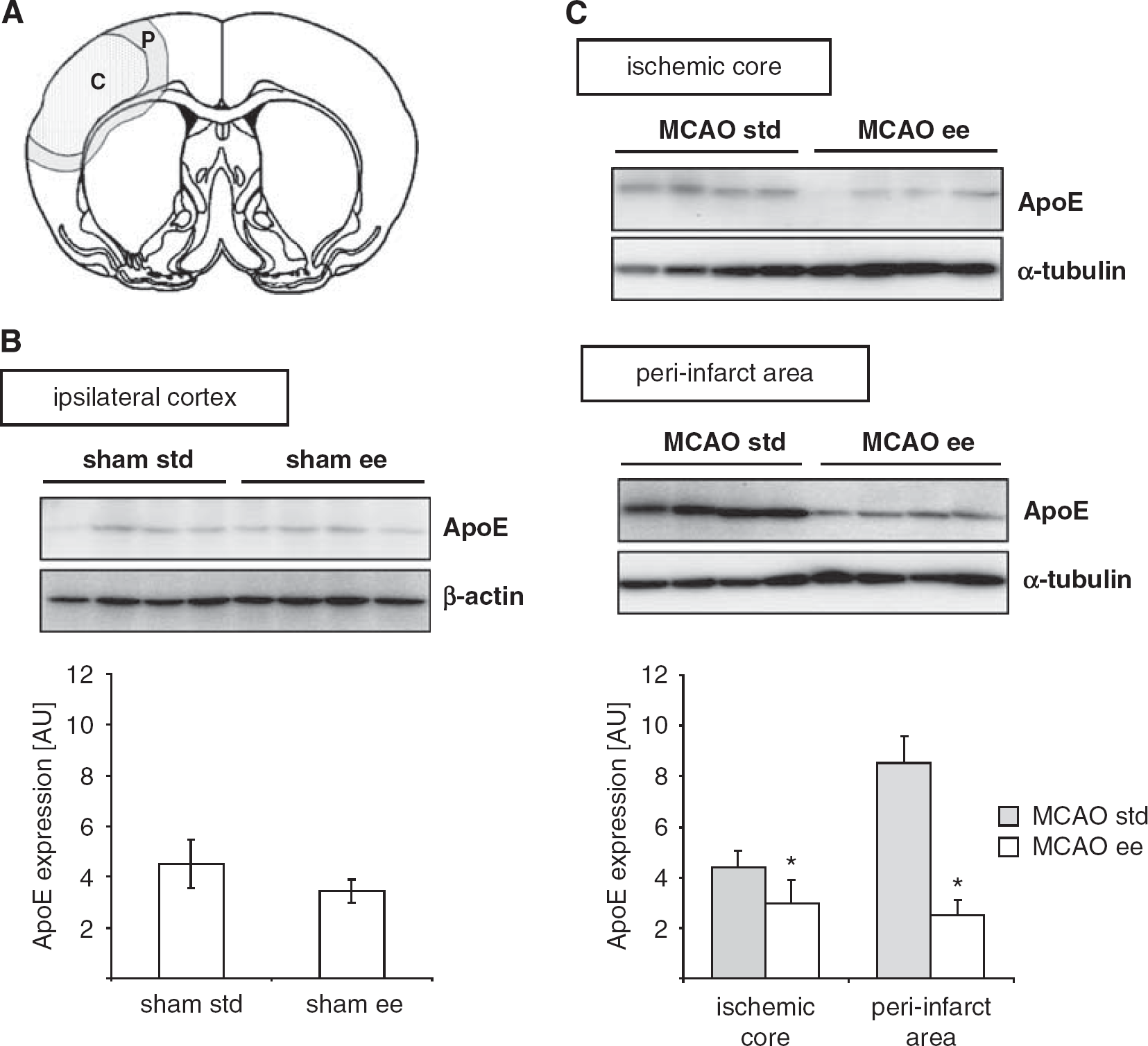
Reduced expression of ApoE in rats housed in EE after pMCAO. (
Interleukin-1β Levels in Rats Housed in Standard and Enriched Environment
ApoE synthesis is strongly affected by cytokines and inflammation (Lynch et al, 2002). As IL-1
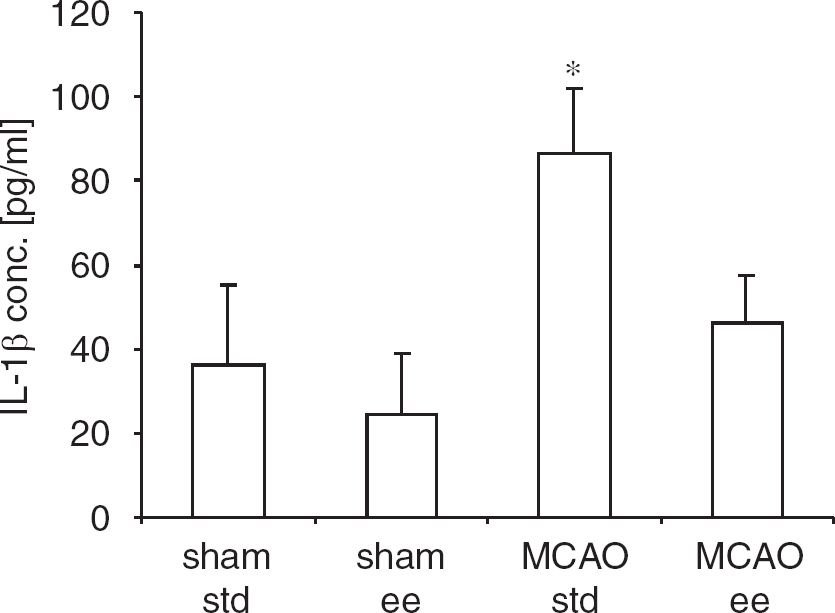
Reduced IL-1
Discussion
We present two important new findings: (1) that a peri-infarct zone devoid of ApoE+/GFAP+ astrocytes is formed at 4 to 7 days of recovery after MCAO, and (2) that the improved recovery of neurologic functions of rats housed in an EE is associated with a marked reduction of ApoE levels, a reduced number of S100
Localization of ApoE
We identified a peri-infarct rim with conspicuously low expression of ApoE at days 4 and 7 after tMCAO (Figure 2). In contrast to the ischemic core, there was no accumulation of CD11b+ microglial cells in this noninjured peri-infarct zone, and there was a normal density of NeuN+ neurons. In this region, we further observed only a limited number of GFAP+ astrocytes suggesting an inhibition of astrocyte proliferation in this zone. Still, in the peri-infarct rim, ramified S100
Our data confirm a delayed induction of ApoE in the infarct core with a peak expression around day 7 after the insult (Kitagawa et al, 2001). Apolipoprotein E accumulated in CD11b+ microglial cells in the infarct core forming a primary scar toward the peri-infarct tissue (Figure 2C). The activated microglial cells have an important function in the reutilization of lipids after peripheral nerve injury (Goodrum et al, 1995). Phospholipids are hydrolyzed into free fatty acids and reutilized in myelin phospholipids synthesis in microglial cells. During nerve regeneration, the cholesterol is transferred into ApoE-containing lipoprotein particles together with esterified free fatty acids to Schwann cells through the low-density lipoprotein receptor to form new myelin sheaths. In fact, microglial cells synthesize and release ApoE. Therefore, it is likely that ApoE released by microglial cells serves as a lipid carrier molecule in mechanisms involved in regeneration of sublethally injured neurons at the border of the infarct core (Carmichael et al, 2005). In addition, the receptor for ApoE, the low-density lipoprotein receptor, was upregulated in microtubule-associated protein 2-positive neurons in peri-infarct areas with a peak expression at 7 days after tMCAO (Kamada et al, 2005).
Decreased Peri-Infarct Levels of ApoE Correlate With Improved Recovery
The robust improvement of neurologic functions in rodents housed in an EE after experimental stroke, has been ascribed to recovery of spine function, that is use-dependent brain plasticity including increased dendritic branching and spinogenesis (Johansson, 2004), also reflected in the changes in gene expression (Keyvani et al, 2004). Although synapses appear to be re-activated after 2 weeks in EE, at least 4 weeks of intermittent enriched housing is required for the recovery effect to become permanent (Nygren and Wieloch, 2005).
Here, we show for the first time that housing rats for only 3 days in an EE after transient MCAO starting at 2 days of recovery is sufficient to improve the lost neurologic function. This improvement is associated with a dramatic decrease in ApoE levels in astrocytes and microglial cells. Apolipoprotein E is neuroprotective and important for brain plasticity. The infarct size is larger in ApoE KO animals subjected to MCAO than in wild-type animals (Laskowitz et al, 1997). In addition, ApoE KO animals and ApoE4 overexpressing animals, the latter carrying a dysfunctional isoform of ApoE, show slower recovery of function after injury and decreased brain plasticity (Sheng et al, 1998). It is not known by which mechanisms ApoE is protective or restorative, but it could be due to its immunomodulatory properties. ApoE suppresses microglial, astrocytic activation
In animals housed in the EE, there were less S100
Our data support the notion that low levels of ApoE and S100
We observed a clear upregulation of parenchymal IL-1
Conclusion
We conclude that the sensori-motor stimulation provided by enriched housing conditions attenuates the inflammatory response after stroke reducing the levels of ApoE, IL-1
Footnotes
Acknowledgements
This study was supported by the Swedish Research Council (KR, No 5909 and TW, No 8466), the EU 7th work program through the European Stroke Network (No 201024), Pia Ståhls Foundation, The Swedish Brain Fund, the Greta and Johan Kocks stiftelse, and the Kungliga Fysiografiska Sällskapet i Lund. We thank Kerstin Beirup and Carin Sjölund for excellent technical assistance.
The authors declare no conflict of interest.
