Abstract
Cerebral venous hypertension (VH) and angiogenesis are implicated in the pathogenesis of brain arteriovenous malformation and dural arteriovenous fistulae. We studied the association of VH and angiogenesis using a mouse brain VH model. Sixty mice underwent external jugular vein and common carotid artery (CCA) anastomosis (VH model), CCA ligation, or sham dissection (n = 20). Hypoxia-inducible factor-1α (HIF-1α), vascular endothelial growth factor (VEGF) and stromal-cell-derived factor-1α (SDF-1α) expression, and matrix metalloproteinase (MMP) activity were analyzed. We found VH animals had higher (P < 0.05) sagittal sinus pressure (8 ± 1 mm Hg) than control groups (1 ± 1 mm Hg). Surface cerebral blood flow and mean arterial pressure did not change. Hypoxia-inducible factor-1α, VEGF, and SDF-1α expression increased (P < 0.05). Neutrophils and MMP-9 activity increased 10-fold 1 day after surgery, gradually decreased afterward, and returned to baseline 2 weeks after surgery. Macrophages began to increase 3 days after surgery (P < 0.05), which coincided with the changes in SDF-1α expression. Capillary density in the parasagittal cortex increased 17% compared with the controls. Our findings suggest that mild nonischemic VH results in a pro-angiogenic stage in the brain by upregulating HIF-1 and its downstream targets, VEGF and SDF-1α, increasing leukocyte infiltration and MMP-9 activity.
Introduction
Angiogenesis is involved in the pathogenesis of brain arteriovenous malformation (BAVM) (Hashimoto et al, 2005) and dural arteriovenous fistulae (DAVF) (Lawton et al, 1997). Clinically, patients with intracranial arteriovenous shunting within BAVM or DAVF usually have complications related to venous hypertension (VH). However, the contribution of these aberrant hemodynamic changes to the lesion formation or progression is poorly understood.
It has been shown that vascular endothelial growth factor (VEGF) increases in both surgically resected dural sinuses harboring DAVF (Uranishi et al, 1999) and brain AVM samples (Hatva et al, 1996; Koizumi et al, 2002). Ng et al (2005) and Sure et al (2004) showed that expression of hypoxia-inducible factor-1 (HIF-1) is significantly correlated to VEGF expression in human AVM, suggesting that HIF-1 plays a role in the induction and maintenance of angiogenesis and vascular remodeling in brain AVM. Increases in VEGF expression and brain microvascular density have also been shown in the brains with VH in animal models (Hai et al, 2003; Lawton et al, 1997; Shin et al, 2003; Zhu et al, 2006).
Inflammatory cells participate in tissue angiogenesis by releasing pro-angiogenic factors and cytokines, including matrix metalloproteinases (MMPs). Inflammatory cell infiltration, as well as their subsequent release of MMPs, might play a role in the angiogenic process in the brain with VH by enhancing the invasion and migration of endothelial cells and macrophages through the surrounding basement membrane and extracellular matrix.
Chemokines are a group of structurally related, small (8 to 14kDa) polypeptide signaling molecules, which can induce the adhesion, migration, and activation of diverse cell types. Recently, stromal-cell-derived factor-1α (SDF-1α or CXCLl2a) has been reported to be a critical mediator in inflammation and angiogenesis. Stromal-cell-derived factor-1α, a strong chemotactic factor, can activate a broad spectrum of leukocytes, including monocytes, neutrophils, and hematopoietic progenitor cells (Aghi et al, 2006; Petit et al, 2007). Du et al (2008) showed that HIF-1, partly through the increases in VEGF and SDF-1α, induces recruitment of CD45+ bone marrow-derived cells to promote angiogenic activity in the brain tumor, and that MMP-9 activity is essential in initiating angiogenesis by increasing VEGF bioavailability. Stromal-cell-derived factor-la can also enhance tumor angiogenesis indirectly, by inducing the secretion of several angiogenic factors including VEGF (Ping et al, 2007).
Several animal models manifesting lesions resembling VH have been well developed and have helped address specific interventional issues, but most of them were created in rats. The relationship of angiogenesis and brain VH has not been clarified using these models. To clarify the causal relationship between VH and angiogenesis, we developed a mouse VH model. We showed that VH in an HIF-1-dependent manner promotes angiogenesis and leukocyte infiltration through increases in VEGF, SDF-1α, and MMP-9 expression under nonischemic conditions, providing a link between VH and angiogenic activities.
Materials and methods
Experimental Design
Animal procedures were approved by the Committee on Animal Research at the University of California, San Francisco. A total of 60 male C57BL/6J mice, weighing 25 and 30 g, were randomly separated into three groups: 20 VH, 20 sham-operated, and 20 with common carotid artery (CCA) ligation. Mice were killed at 1, 3, 7, and 14 days after surgery. Hemodynamic measurements were made pre- and postsurgery, and brains were harvested.
Surgical Procedure
Mice were anesthetized with intraperitoneal injections of ketamine (100 mg/kg) and xylazine (10 mg/kg). Through an anterior midline cervical incision, the right CCA was exposed from its bifurcation down to the clavicle. The external jugular vein (EJV) in the subcutaneous tissue was exposed from the cranial base down to the clavicle. Common carotid artery and EJV were trapped with temporary aneurysm clips and transected. Proximal CCA was connected to the cranial stump of EJV with end-to-end anastomosis performed under an operating microscope with continuous 11-0 monofilament nylon sutures (MONOSOF, Synecture, Norwalk, CT, USA). The distal CCA and caudal stump of EJV were occluded with bipolar cauterization (Figures 1A–1C). All incisions were closed with a 6-0 nylon suture. Forty mice without CCA and EJV anastomosis were used as controls; 20 underwent sham surgery (midline neck exploration) and 20 underwent left carotid artery occlusion.
Hemodynamic Measurement
Mean arterial pressure (MBP), sagittal sinus pressure (SSP), and surface cortex cerebral blood flow (CBF) were measured pre- and postoperation. Mean arterial pressure was measured noninvasively using tail cuff and pulse transducer system (NIBP, AD Instruments, Colorado Spring, CO, USA). Sagittal sinus pressure was documented by cannulating a 30-gauge needle connected to a pressure transducer and signal amplifier (PowerLab, Colorado Spring, CO, USA). Surface CBF was measured using a laser Doppler probe (Laserflo BPM2, Vasomedics, St. Paul, MN, USA) that penetrates brain tissue approximately 1 mm beyond the probe ending. The probe was placed on the surface of the parasagittal parietal cortex. Means of four serial measurements made on each side were documented.
Immunohistochemistry
For single-label immunohistochemistry, 20 μm thick coronal brain sections were fixed in 4% paraformaldehyde for 15 mins and quenched in 0.3% H2O2 with methanol for 30 mins. After blocking in 5% goat serum for 30 mins, sections were incubated with primary antibodies at the following concentrations: rabbit antimouse myeloperoxidase (MPO), 1:200 (Lab Vision, Fremont, CA, USA); rat antimouse CD68, 1:50 (Serotec, Raleigh, NC, USA); rabbit antimouse MMP-9, 1:150 (Chemicon, Billerica, MA, USA); SDF-1 rabbit antimouse, 1:200 (eBioscience, San Diego, CA, USA); VEGF rabbit antimouse, 1:100 (Santa Cruz Biotechnology, Santa Cruz, CA, USA); and HIF-1α antimouse, 1:100 (Novus Biologicals, Littleton, CO, USA). After incubating at 4°C overnight, the sections were incubated with biotinylated secondary antibodies for 1.5 h at room temperature, and were treated with the Avidin-Biotin Complex (ABC) streptavidin detection system (Vector Lab, Burlingame, CA, USA) for 1.5 h. The resulting horseradish peroxidase signal was detected using 3,3'-diaminobenzidene.
For double fluorescent staining, after incubating at 4°C overnight with primary antibodies, sections were incubated with Alexa Fluor 594- or 488-conjugated labeling (Invitrogen, Carlsbad, CA, USA) for 1.5 h at room temperature. They were then mounted and photographed with a fluorescent microscope. Negative controls were performed by omitting the primary antibodies during the immunostaining. The numbers of neutrophils (MPO positive) and macrophage (CD68 positive) was counted blindly in five coronal sections of each animal under a high-power (x 200) Leica microscope (Leica Microsystems, Bannock-bum, IL, USA) with a digital camera. For microvessel counting in the parasagittal cortex region, the sections were stained with fluoresceinlycopersicin esculentum lectin (Vector Lab). Vessel density was counted in five coronal sections per animal using NIH Image 1.63 software (National Institutes of Health, Bethesda, MD, USA).
Gelatin Zymography
The protein isolated from the parasagittal cortex was separated under nonreducing conditions in a 10% zymogram gel (Invitrogen) containing 0.1% gelatin as a substrate. After electrophoresis, gels were washed and incubated overnight in developing buffer at 37°C. Then, the gels were stained with 0.5% Coomassie Blue R-250 (Bio-Rad Laboratories, Hercules, CA, USA) and destained. Matrix metalloproteinase activity could be detected as white bands against the Coomassie blue stained gel. Recombinant MMP-9 and MMP-2 were run in the same gel as references. Gelatinolytic bands were quantified using densitometry and analyzed using NIH Image 1.63 software.
Western Blot Analysis
Aliquots of the proteins for gelatin zymography were used for Western blot analysis. Proteins were separated in 14% of Tris-Glycine gel and electrotransferred onto a nitrocellulose membrane (Bio-Rad Laboratories) in transfer buffer (Invitrogen) by semidry blotting. After blocking in 5% milk, the membrane was incubated with rabbit antimouse VEGF antibody (1:200) overnight at 4°C. After washing, the membrane was incubated with horseradish peroxidase-conjugated antirabbit secondary antibody (Amersham, Buckinghamshire, UK) diluted at 1:10,000, and then reacted with SuperSignal West Femto Maximum Sensitivity Substrate detection reagent (Pierce Biotechnology, Rockford, IL, USA). The membrane was exposed to Hyperfilm ECL (Amersham, Sunnyvale, CA, USA), and developed according to the manufacturer's instructions. Vascular endothelial growth factor expressions were analyzed by optical density using NIH Image 1.63 software.
SDF-1 Enzyme-Linked Immunosorbent Assay (ELISA)
The protein isolated from the parasagittal cortex was collected and stored at −80°C until assayed. The SDF-1α level was measured by ELISA using a commercial kit (Mouse SDF-1α ELISA kit, Minneapolis, MN, USA, MCX120, R&D Systems) according to the manufacturer's instructions.
Statistical Analyses
Data are presented as mean ± s.d. Means were compared using oneway analysis of variance analysis followed by the post hoc t-test. A probability value of less than 0.05 was considered statistically significant.
Results
SSP Increased Dramatically in Mice With CCA and EJV Anastomosis
To analyze whether the CCA and EJV connection caused brain VH and other hemodynamic changes, we measured SSP, CBF, and MBP before and after surgery. We found that the connection resulted in immediate and sustained increase in venous pressure in the mouse brains, with four- to sixfold elevations in SSP. In contrast, sham surgery and CCA ligation did not result in VH in the brains (P < 0.05, Figure 1D). No differences were observed in CBF before and after surgery among the groups, indicating that we created a nonischemic brain VH (Figure 1E). There were also no significant differences in MBP between VH and control mice pre- and postoperation (Figure 1F). Thus, the CCA-EJV connection increased venous pressure in the brains without compromising cerebral and systemic blood flow (no ischemia).
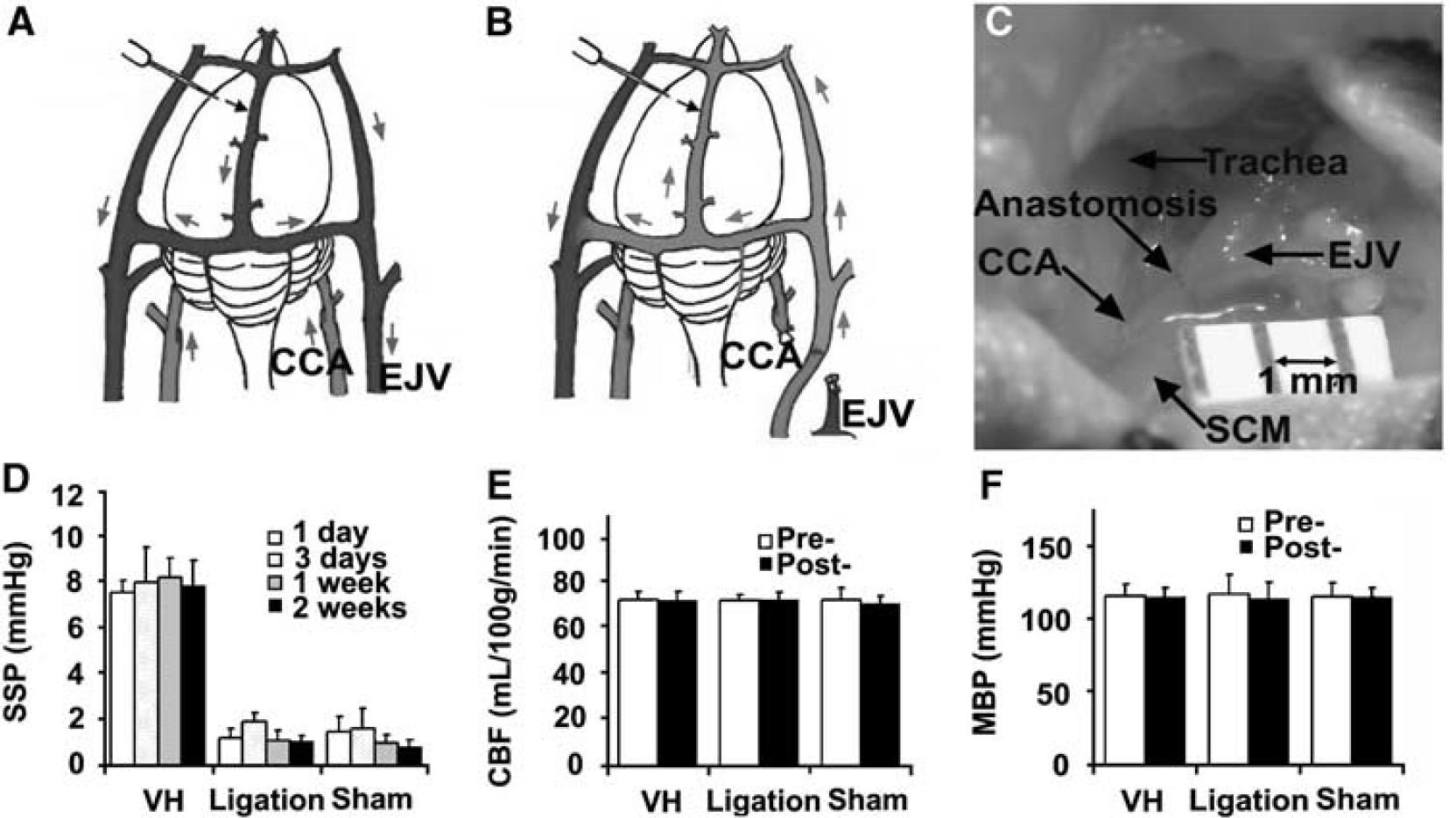
Sagittal sinus pressure increased significantly in the VH brain. (
HIF-1α Expression Increased in Response to VH
To analyze whether nonischemic VH increased HIF-1α expression in the brain, we determined HIF-1α protein in the VH brain using immunohistochemical staining. We found that HIF-1α increased in the endothelial cells at the parasagittal cortex 1 day after the onset of VH (Figure 2A). Hypoxia-inducible factor-la expression was minimal in the brains of mice subjected to sham surgery or CCA ligation. Thus, nonischemic VH increased HIF-1α proteins in the brain.
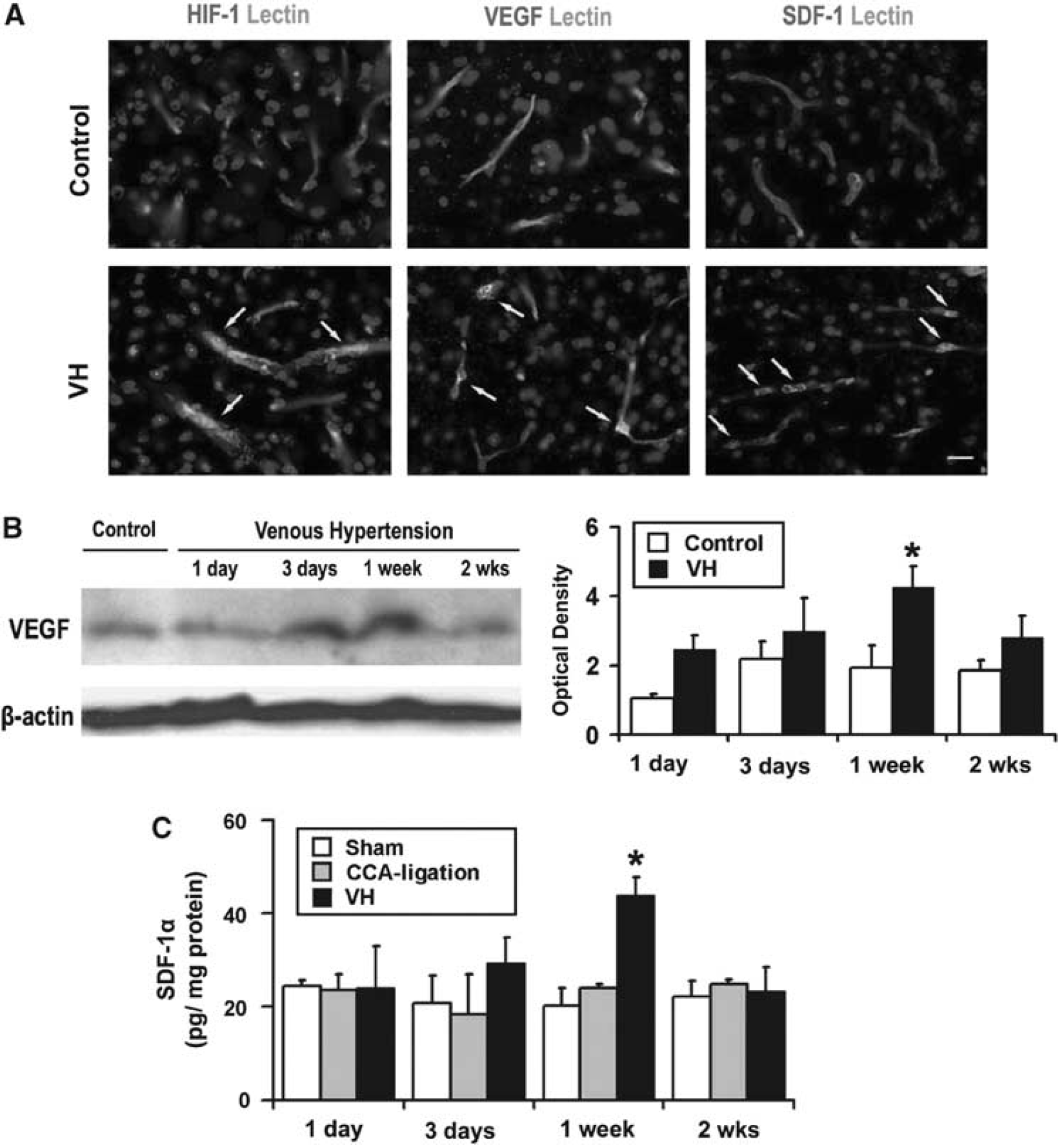
Increased HIF-1, VEGF, and SDF-1 expression in the VH brain. (
HIF-1 Target Genes, VEGF, and SDF-1α Increased in VH Brains
To study whether increased HIF-1 activity in the VH brain upregulated its downstream gene (VEGF), we analyzed VEGF expression using immunostaining and Western blot. The results showed that VEGF expression was higher in the parasagittal cortex region of VH mice than that of control mice (Figure 2A). Double fluorescence staining using lectin and VEGF-specific antibody showed that VEGF was expressed by endothelial cells (Figure 2A). Vascular endothelial growth factor expression began to increase at day 3, peaked at day 7, and decreased 2 weeks after surgery (Figure 2B). There was significantly more VEGF in the brains of VH mice than in the brains of mice without VH (P < 0.05) (Figure 2B). In addition, we observed higher vascular density in the VH brain than in control brains 2 weeks after surgery; however, the differences did not reach a statistically significant level (data not shown).
Stromal-cell-derived factor-la is another HIF-1 downstream gene that promotes angiogenesis by attracting bone marrow-derived mononuclear cells homing to the angiogenic focus (Hiasa et al, 2004), and induces secretion of the angiogenic factor (De Falco et al, 2004; Petit et al, 2007). We found that SDF-1α expression was upregulated in the parasagittal cortex adjacent to the sagittal sinus of HV mice. Minimum SDF-1α-positive signal was detected in control brains (Figure 2A). Double fluorescence staining showed that SDF-1α was mainly expressed by endothelial cells (Figure 2A). ELISA analysis demonstrated that, similar to VEGF, SDF-1α expression also increased 3 days, peaked 1 week, and decreased 2 weeks after surgery (Figure 2C). These findings indicate that SDF-1 was also involved in pro-angiogenic changes in the VH brain.
Neutrophil-Associated MMP-9 Activities Increased in Response to VH
To test whether increased VEGF and SDF-1α expression in VH brains resulted in the recruitment of bone marrow-derived mononuclear cells, we quantified neutrophils in the VH brains by immunostaining using MPO-specific antibody. We found that the number of neutrophils increased significantly in the VH brain 1 day after surgery, whereas minimal neutrophils were found in the control brains (P < 0.05, Figure 3B). The MPO-positive cells were mainly located at the vessel wall in the tissues close to the midline, such as the parasagittal cortex, and to a lesser degree in the basal ganglia (Figure 3A). The number of neutrophils peaked 1 day after surgery, gradually decreased, and returned to the baseline after 2 weeks of surgery (Figure 3B).
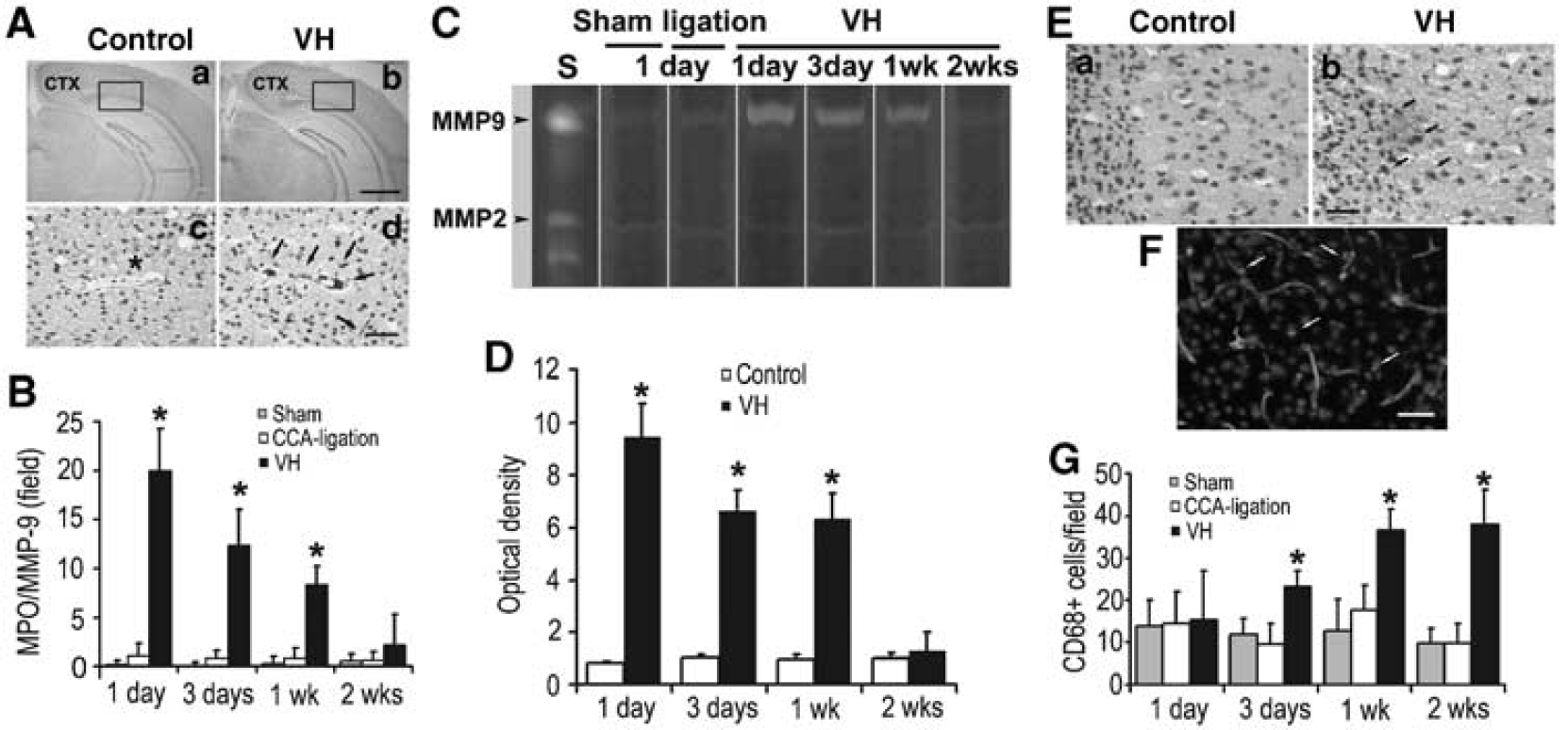
Increased leukocytes in the VH brain. (
As inflammatory cells participate in angiogenesis by producing MMPs(Coussens et al, 2000), we analyzed MMP-2 and MMP-9 activity in the VH brain using zymography. Matrix metalloproteinase-9 activity was most prominent at day 1, remained high for more than 1 week, and then returned to the baseline 2 weeks after surgery (Figures 3C and 3D). In the control groups, minimal MMP-9 activity was observed. Matrix metalloproteinase-2 was equally expressed in all the groups at all time points. The MMP-9 was colocalized with neutrophils (Supplementary Figure 1) and the time course of MMP-9 expression coincided with the neutrophil infiltration, indicating that MMP-9 was neutrophil derived and that it may play a role in early response to shear stress changes, facilitating subsequent inflammatory events such as macrophage infiltration.
Macrophages Increased in Response to VH
Macrophages have also been shown to participate in angiogenesis. To investigate whether the monocyte/macrophages were recruited in the VH brains, we performed immunohistochemical staining using CD68-specific antibody. We found that the number of microphages increased significantly in the VH brain 3 days after surgery (Figures 3E and 3G). The CD68+ cells were found in the parasagittal cortex, including frontal, parietal, and occipital lobes, and, to a lesser degree, in the basal ganglia. The increase in CD68+ cells sustained for more than 2 weeks after surgery (P < 0.05, Figure 3G). The time course of macrophages coincided with SDF-1α expression, suggesting that the microphages were attracted by SDF-1α. Furthermore, immunofluorescence staining showed that CD68+ cells were located on the vessel wall as well as the parenchyma of the VH brains (Figure 3F).
Discussion
This study reports the first cerebral VH murine model. In this model, SSP increased four- to sixfold, whereas CBF and MBP did not change, indicating a nonischemic brain VH. We found that (1) the presence of VH increased HIF-1 and its downstream targets, VEGF and SDF-1α expression, in the parasagittal cortex of the mouse brain; (2) leukocytes, including neutrophils and macrophages, were recruited to the parasagittal cortex; (3) MMP-9 activity increased and MMP-9 protein colocalized with neutrophils—MMP-9 activity peaked with the number of neutrophils in VH brains 1 day after the surgery; (4) the macrophage infiltration began 3 days after the surgery, coinciding with the time course of SDF-1α expression in endothelial cells; and (5) there was a trend of microvessel density increase in the VH brains at 2 weeks after surgery. Our current findings suggest that the pro-angiogenic stage in the VH brains is promoted by HIF-1-upregulated VEGF and SDF-1 expression and leukocyte infiltration. Thus, HIF-1 downstream genes (VEGF and SDF-1) and leukocytes play critical roles in the mechanism that links brain VH and angiogenesis. Furthermore, these factors induced by VH may facilitate vascular remodeling, and participate in the pathogenesis associated with VH in the human brain. Venous hypertension is commonly thought to exert its deleterious effect on the brain by a simple reduction in cerebral perfusion pressure resulting in cerebral ischemia. Our studies point to an additional and underappreciated set of physiologic effects that are caused by nonischemic levels of venous pressure.
Our mouse brain VH model was created by a CCA-EJV fistula, which resulted in a sustained increase in SSP in the brain. None of the mice with cerebral VH showed behavioral changes or neurologic dysfunction associated with ischemia. In this model, the hemodynamics, including surface cortex CBF and systematic MBP, did not significantly change, which suggested that VH did not compromise cerebral perfusion. Thus, the increased HIF-1 expression in our brain VH model was due to mechanisms other than ischemia, such as endothelial share stress.
Hypoxia-inducible factor-1 has been proposed as a key factor that upregulates the expression of SDF-1 and VEGF. These two factors recruit MMP-9-bearing bone marrow-derived cells to participate in tumor angiogenesis of the mouse brain (Aghi et al, 2006; Du et al, 2008; Hao et al, 2008; Lyden et al, 2001). In this study, we demonstrated that focal hemodynamic derangement in the brain resulted in recruitment of MPO-positive leukocytes to endorse angiogenesis. Circulating neutrophils responded to the brain VH earlier than macrophages. The number of neutrophils peaked 1 day after the surgery. Matrix metalloproteinase-9 activity increased in the same region at the same time. Immunostaining showed that MMP-9 was expressed by neutrophils in the vessel wall, which is consistent with findings in human BAVM samples (Chen et al, 2008). Matrix metalloproteinase-9 produced by neutrophils may facilitate the degradation of the extracellular matrix, and thus promotes the invasion and migration of endothelial cells, as well as monocytes through the surrounding basement membrane and extracellular matrix (Philip et al, 2004). In addition, MMP-9 can release VEGF from extracellular matrix, which results in the initiation of angiogenesis (Bergers et al, 2000; Page-McCaw et al, 2007). Macrophage infiltration increased 3 days after the surgery, which is chronologically correlated with the increase in SDF-1 expression. On the basis of immunohistochemistry analysis, macrophages were located both in the vessel wall and brain parenchyma. This finding is consistent with the earlier report (Chen et al, 2008) for human BAVM tissue, and supports the notion that VH induces leukocyte infiltration. Vascular endothelial growth factor and SDF-1α expression peaked 1 week after operation. Both of these factors are expressed by endothelial cells and play a pivotal role in attracting circulating leukocytes homing to angiogenic foci. Stromal-cell-derived factor-1 might mediate the retention of circulating leukocytes in close proximity to angiogenic vessels (De Falco et al, 2004; Petit et al, 2007). Inflammatory cells also enhance local in situ proliferation of endothelial cells through secretion of pro-angiogenic cytokines. Taken together, our findings suggest that nonischemic VH conditions may upregulate the expression of HIF-1 and its downstream genes, VEGF and SDF-1, and facilitate the recruitment of leukocytes.
Brain VH occurs in several human diseases, such as BAVM. The fundamental defect in human BAVM is high-flow arteriovenous shunting, frequently accompanied by VH shown as venous outflow restriction and aberrant venous drainage patterns by angiography. The importance of raised venous pressure in the pathophysiology of AVMs has gained recognition, and the pathogenesis of vascular lesions is closely linked to angiogenesis. In certain cases, active angiogenesis might be a part of the underlying pathophysiology of the growth and regression of BAVM (Du et al, 2007; Nussbaum et al, 1998). In response to VH, endothelial cells are subject to a high intravascular shear stress that is able to convert mechanical stimuli into molecular events, e.g., upregulation of HIF-1α as well as its downstream expressions. It provides a bridge that links HIF-1 and pro-angiogenic stage in response to VH.
In the clinical setting, VH is commonly associated with brain injury because it decreases cerebral perfusion pressure, resulting in ischemic damage. Our results show that even modest levels of VH have a pro-angiogenic effect that can influence the course of diseases in the absence of ischemia. The pro-angiogenic effect of VH has also been observed in the pathogenesis of other diseases. Intracranial hypertension is often accompanied by increased cerebrospinal fluid pressure within the skull. Elevated cerebrospinal fluid pressure compresses the veins, which then results in venous outflow obstruction and VH. Koehne et al (2002) examined 57 cerebrospinal fluid aliquots obtained from patients with hydrocephalus, and found that intracranial hypertension may trigger release of VEGF into cerebrospinal fluid. In addition, VH has been implicated in DAVF (Bederson et al, 1991; Herman et al, 1995; Kusaka et al, 2001; Terada et al, 1994). All of the evidences above highlight the importance of VH in the development and recurrence of vascular lesions.
Pro-angiogenic activities have also been identified in rat VH brain models (Shin et al, 2003). Hai et al (2002, 2003) observed increased capillary density in the rat brains at day 7 after the surgery, and this increase was maintained for up to 3 months. We have found in our earlier study with a rat model that nonischemic brain VH increases HIF-1 and VEGF expression, and proposed a possible role of HIF-1 in the induction of angiogenesis in the VH brains (Zhu et al, 2006). This study provides an important extension of the earlier work by our groups and others, by showing that in addition to increased VEGF, SDF-1α, another HIF-1 downstream gene that participates in tissue angiogenesis, increased in the VH brain. We have also observed leukocyte infiltration and increased MMP-9 activity. The emerging picture that comes into view is that VH exerts its pro-angiogenic effects through VEGF, SDF-1α, and MMP-9 released by leukocytes.
Recently, genetic mutant mice were used in research to generate BAVM that mimics human BAVM, including mice with endoglin (ENG) (Satomi et al, 2003; Xu et al, 2004), activin-like kinase 1 (Park et al, 2008), or Notch (int3) (Carlson et al, 2005; Krebs et al, 2004; Murphy et al, 2008) haploinsufficiency. With brain VH in combination with genetic manipulation, a BAVM model that mimics human BAVM will likely be generated. The model will allow us to better understand the pathogenesis process of the disease and will serve as a testing model for the development of new therapies.
The major limitation of this study is that we did not measure arterial CO2 (Paco2). However, the effect of the anesthetic and surgical preparation was similar among groups and the absolute level of Paco2 or CBF was secondary to the fact that they were equivalent. The VH model has been mostly created on rats. The model by Zhu et al (2006), generated by a cervical fistula combined with the occlusion of transverse and sagittal sinuses, had a higher SSP more than 20 mmHg. In contrast, our model was created by an arteriovenous fistula without sinus occlusion. It may account for the mild increased SSP at 8 mmHg for our mouse model. The fact that the increased microvessels in the mouse VH brain did not reach statistically significant levels showed that the pro-angiogenic activity in the mouse VH brain is lower compared to that in the rat VH brain. We have attempted to reproduce rat surgical procedures in mice; however, the mice could not tolerate the procedures. On the basis of our experiment, we could not identify whether the leukocytes at the angiogenic site were derived from bone marrow. Further studies are needed to address the function of leukocytes in VH-induced angiogenesis. Finally, because neutrophil infiltration occurred earlier than the increase in SDF-1 and VEGF expression, further study is needed to identify the factors that cause the recruitment of neutrophils at the early time point.
In summary, our VH model was based on extradural A-V fistulas, which displayed pro-angiogenic features in the brain, including increased expression of HIF-1α and its downstream genes, VEGF and SDF-1α. Leukocyte infiltration was also observed. All of these results indicate that nonischemic level of VH, without compromising blood flow, can provide a pro-angiogenic environment in the brain that can be a mechanism for vascular remodeling in BAVM and other diseases associated with brain VH.
Footnotes
Acknowledgements
The authors declare no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
