Abstract
The inflammatory response that exacerbates cerebral injury after ischemia is an attractive therapeutic target: it progresses over days and strongly contributes to worsening of the neurologic outcome. The authors show that, after transient ischemic injury to the rat brain, systemic application of interferon-beta (IFN-β), a cytokine with antiinflammatory properties, attenuated the development of brain infarction. Serial magnetic resonance imaging (MRI) showed that IFN-β treatment reduced lesion volume on diffusion-weighted MRI by 70% (P < 0.01) at 1 day after stroke. IFN-β attenuated the leakage of contrast agent through the blood–brain barrier (P < 0.005), indicating a better-preserved blood–brain barrier integrity. Both control and IFN-β-treated animals showed a similar degree of relative hyperperfusion of the lesioned hemisphere, indicating that neuroprotection by IFN-β was not mediated by improved cerebral perfusion as assessed 24 hours after stroke onset. IFN-β treatment resulted in an 85% reduction (P < 0.0001) in infarct volume 3 weeks later, as determined from T2-weighted MRI and confirmed by histology. This effect was achieved even when treatment was started 6 hours after stroke onset. Quantitative immunohistochemistry at 24 hours after stroke onset showed that IFN-β almost completely prevented the infiltration of neutrophils and monocytes into the brain. Gelatinase zymography showed that this effect was associated with a decrease in matrix metalloproteinase-9 expression. In conclusion, treatment with the antiinflammatory cytokine IFN-β affords significant neuroprotection against ischemia/reperfusion injury, and within a relatively long treatment window. Because IFN-β has been approved for clinical use, it may be rapidly tested in a clinical trial for its efficacy against human stroke.
Keywords
To date, experimental stroke research has produced only one FDA-approved drug, tissue plasminogen activator (tPA), to combat the consequences of ischemic stroke in the clinic. In practice, however, only very few patients meet the criteria for tPA treatment. The protracted evolution of the inflammatory response to ischemia makes its attenuation an attractive target in human stroke because there is a potentially broad therapeutic window. The events triggered by an inadequate blood supply to the brain transform the vascular endothelium from a quiescent to a proinflammatory and prothrombotic state. Cytokines induce the upregulation of cell adhesion molecules (CAMs) on the vascular endothelium, leading to leukocyte recruitment. This causes a further augmentation of the inflammatory reaction, accompanied by generation of reactive oxygen species, release of cytotoxic enzymes and a compromised blood–brain barrier (BBB) status (Barone and Feuerstein, 1999; DeGraba, 1998; Iadecola and Alexander, 2001). Interferon-beta (IFN-β) is a regulatory cytokine with antiinflammatory properties that has been approved for the treatment of multiple sclerosis (MS). Previously, we have shown that IFN-β strongly inhibits disease activity in rats afflicted with experimental autoimmune encephalomyelitis, a model for the human disease MS. Our data showed that the beneficial effects are associated with inhibition of the extravasation of blood-derived monocytes into the CNS (Ruuls et al., 1996; van der Meide et al., 1998). More recently, we have demonstrated that IFN-β dose-dependently prevents the upregulation of vascular CAM-1 and intercellular CAM-1 (ICAM-1) expression, both in vitro and in vivo (Floris et al., 2002). In MS patients, the effect of IFN-β on monocyte and T-cell infiltration has been shown to be associated with a reduction in matrix metalloproteinase (MMP)-9 expression (Galboiz et al., 2001; Stuve et al., 1997; Trojano et al., 1999; Waubant et al., 2001). After stroke, upregulation of MMP-9 contributes to injury and correlates to an increased risk of hemorrhagic transformation (Asahi et al., 2000; Montaner et al., 2001).
For the past decade the therapeutic properties of IFN-β have been the subject of extensive study, but mainly in relation to MS. The above observations led to the hypothesis that IFN-β could attenuate the inflammatory response after cerebral ischemia, and consequently, have therapeutic value in stroke. To that aim, using magnetic resonance imaging (MRI) we assessed the effect of IFN-β treatment on tissue damage, perfusion status, and BBB integrity after transient middle cerebral artery occlusion in the rat. MRI was chosen as the primary readout modality, because it allows for the longitudinal follow-up of the same animals over time, and provides both dynamic anatomical information on the evolution of brain injury and informs on functional parameters such as cerebral perfusion and BBB leakage. We used both diffusion-weighted and T2-weighted MRI, as these techniques are applied worldwide for the clinical diagnosis of stroke in humans. A decrease in the apparent diffusion coefficient (ADC) of water, as measured using diffusion-weighted MRI, is thought to correlate to acute, excitotoxic tissue changes and is a sensitive indicator of early ischemic injury (Mintorovitch et al., 1994; Moseley et al., 1990; van der Toorn et al., 1996). In the more chronic stages of the disease, T2-weighted MRI data correlate well with infarct size and tissue damage as assessed histologically (Palmer et al., 2001).
To study the effect of IFN-β treatment on tissue damage after stroke, animals were treated with IFN-β, either prophylactically or at various time points after stroke onset, and were compared with animals that received treatment with saline. To assess the evolution of ischemic brain injury, diffusion-weighted and T2-weighted MRI data sets were acquired at 1, 7, and 21 days after stroke. Furthermore, at the 24-hour time point, cerebral blood flow and BBB leakage were also determined. To correlate the putative antiinflammatory properties of IFN-β to its effect on lesion size and BBB integrity, we used immunofluorescence microscopy and gelatinase zymography to quantify the infiltration of inflammatory cells and determine MMP expression at the 24-hour time point.
MATERIALS AND METHODS
Animal model
The Utrecht University Animal Experiment Ethical Committee approved all animal experiment protocols. Male Fischer rats (F344/Ico, Iffa-Credo Broekman, Someren, The Netherlands) aged 8 to 12 weeks were used. Animals had free access to standard laboratory chow and water. Anesthesia was induced by injection of a mixture of 0.5-mL/kg fentanyl citrate (0.315 mg/mL, intraperitoneally) and fluanisone (10.0 mg/mL, intraperitoneally), and 0.5 mL/kg midazolam (5.0 mg/mL, intraperitoneally), followed by 0.1-mL/kg atropine sulfate (0.5 mg/mL, subcutaneously) and injection of 0.5-mL/kg gentamicin sulfate (10 mg/mL, i.m.). The animals were endotracheally intubated and mechanically ventilated with O2/N2O (30/70 volume/volume). Body temperature was maintained at 37.0° ± 0.5°C by means of a feedback-controlled heating pad. During surgery and MRI measurements, anesthesia was continued by adding 1% to 1.5% halothane to the O2/N2O mixture. After surgery, the animals received 1-mL/kg buprenorfine (0.3 mg/mL, subcutaneously).
Focal ischemia was induced by unilateral permanent occlusion of the right common carotid artery and transient occlusion of the middle cerebral artery (MCA) via a modification (Veldhuis et al., 2001) of the procedure as described elsewhere (Brint et al., 1988). Briefly, while keeping the zygoma intact, a 2-mm-diameter hole was drilled just rostral to the oval foramen, exposing the right MCA. After incising the dura and arachnoidea, the MCA was transiently occluded using a vascular microclip (Codman, Raynham, MA, U.S.A.). Perfusion was reinstated after 1 hour under visual inspection by removing the clip.
Animals were treated with subcutaneous injections of 500,000 U (8 μg) recombinant rat IFN-β in 1 mL phosphate-buffered saline once daily until 7 days after reperfusion. The dose of IFN-β was chosen on the basis of doses earlier reported to inhibit experimental autoimmune encephalomyelitis disease activity in rats (Floris et al., 2002; van der Meide et al., 1998). Control animals (n = 10) received subcutaneous injections of saline, from 2 days before surgery up to 7 days after reperfusion. Treated animals were divided into four groups based on whether treatment started 2 days before surgery (n = 3), on reperfusion (n = 5), 4 hours after stroke onset (n = 4), or 6 hours after stroke onset (n = 4). Seven additional animals were treated with saline (n = 3) or IFN-β (n = 3) on reperfusion or were sham operated (n = 1), and were killed and processed for immunohistology after the MRI measurement at 24 hours.
MRI experiments
All MRI experiments were performed on a 4.7-T Varian (Palo Alto, CA, U.S.A.) horizontal bore spectrometer. Radiofrequency excitation and signal detection were accomplished by means of a Helmholtz volume coil (9-cm diameter) and an inductively coupled surface coil (2-cm diameter), respectively. Animals were positioned in an animal cradle and immobilized with ear bars. During the experiments, exhaled CO2 and rectal temperature were continuously monitored. In five control animals and five treated animals, a tail vein was cannulated at day 1 for injection of the MRI contrast agent dimegluminegadopentate (Gd-DTPA) (Runge et al., 1994). A single-shot diffusion-trace MRI sequence (4 b values: 100–1,780 s/mm2, repetition time [TR] = 2 seconds, echo time [TE] = 100 milliseconds, number of excitations [NEX] = 2, 8 consecutive 1.7-mm-thick slices, field of view [FOV] = 3.2 × 3.2 cm2, matrix [M] = 128 × 64, zero-filled to 256 × 256) was used to generate quantified images of tissue water trace ADC. T2-weighted images were acquired using a multiecho sequence (8 TEs: 17.5 milliseconds + 7 × 17.5 milliseconds, TR = 2 seconds, NEX = 2, eight 1.7-mm-thick slices, FOV = 3.2×3.2 cm2, M = 128 × 64, zero-filled to 256 × 256). A T1-weighted dataset (TR = 0.65 seconds, TE = 0.0125 seconds, NEX = 2, FOV = 3.2 × 3.2 cm2, M = 256 × 128, zero-filled to 512 × 512) was acquired before and 15 minutes after injection of the contrast agent Gd-DTPA (0.5 mmol/kg). Perfusion-weighted imaging was performed using the flow-sensitive alternating inversion recovery (FAIR) technique (Kim, 1995). A slice-selective and a nonselective inversion recovery image (Mss and Mns) were acquired with Turbo-fast low angle shot (FLASH) acquisition and a sufficient inversion time (TI) to allow inflow of labeled spins into brain tissue. Acquisition was preceded by flow-crushing to suppress intravascular signal (Ye et al., 1997). Magnetic resonance parameters were as follows: flip angle (α) = 22°, TE/TR/TI = 3 ms/6 ms/2 s, predelay = 2 seconds (total TR = 4.75 seconds), 128 × 128 matrix, 3.2 × 3.2 cm2 FOV, slice thickness = 2 mm, and NEX = 96. For normalization of FAIR signal an equilibrium magnetization (M0) image was acquired with the same parameters, but without inversion. For quantification of FAIR signal, a slice-selective T1-map was acquired by Turbo-FLASH look-locker acquisition (α/TE/TR = 5°/4.5 ms/11 ms, TIs = 0.4 + 9 × 1.4 seconds, and NEX = 8).
Histology
Gelatinase zymography
Recombinant rat IFN-β
The cloning and expression of the chromosomal gene encoding rat IFN-β in Chinese hamster ovary cells has been described elsewhere (Ruuls et al., 1996). The glycoprotein was purified to homogeneity by dye-ligand affinity chromatography and analyzed for endotoxin contamination. The biologic activity of each batch was determined in an antiviral bioassay (Olsson et al., 1994) and compared with the bioactivity of a laboratory standard preparation. Purified recombinant rat IFN-β was found to exert a bioactivity of approximately 6 × 107 U/mg of protein. IFN-β-treated rats received dosages on the basis of equal bioactivity.
Data analysis
Apparent diffusion coefficient and T2 maps were generated by monoexponential fitting using IDL (Research Systems, Boulder, CO, U.S.A.). Parametric images were analyzed in anatomic regions of interest using in-house software. Calculations of volumes of affected tissue were based on ipsilateral ADC or T2 differing more than 20% from the mean value in the contralateral hemisphere. This threshold corresponds to a
The ICAM-1–stained histologic sections were analyzed with a NIKON Eclipse E800 microscope and a digital NIKON DXM1200 camera. Within each section, standardized recordings were made from the cortical and striatal area with at ×10 objective. For each acquisition session, pictures were captured using a standardized procedure. The ICAM-1–stained area was calculated using the digital image analysis program AnalySIS (Soft Imaging System GmbH, Münster, Germany). Each pixel in the photographs was divided into three color components (hue, saturation, and intensity). The threshold for these three color components was defined in such a manner that only immunohistochemical-positive areas were selected for analysis. The threshold was kept constant throughout the analysis of all animals. For analysis, the number of pixels in the immunopositive area was used and expressed in arbitrary units. Statistical analysis was done using a linear mixed effects model (in S-PLUS 2000 Professional Edition Release 3, Mathsoft Inc., Cambridge, MA, U.S.A.), with Rat as random effect and Day and Treatment as fixed effects. After showing significance of all treatment effects, further statistical analysis was performed using SPSS 10.0 (SPSS Inc., Chicago, IL, U.S.A.). All data are presented with standard deviation as the measure of dispersion around the mean. Reported P values correspond to two-tailed Student's t-tests for the dichotomized comparisons (treatment start 2 days prior versus treatment start after onset) or one-way analysis of variance where appropriate, with correction for multiple comparisons. Pearson's correlation coefficient was calculated using SPSS 10.0. Reported P values for the correlations correspond to two-tailed tests of significance.
RESULTS
IFN-β reduces ischemic lesion size
A more restricted diffusion of water is one of the earliest manifestations of ischemic brain injury. ADC maps, calculated from diffusion-weighted datasets acquired at 24 hours after stroke, showed ischemic lesions, as evidenced by decreased ADC values (Fig. 1). In control animals, both the cortex and a large part of the caudate putamen of the ipsilateral hemisphere were affected, whereas in IFN-β–treated animals the lesion was restricted to a smaller area of the caudate putamen and to only a small rim of cortex, if at all (Fig. 1). At this time point, the volume of the lesion on ADC maps was 70% smaller (P < 0.01) in IFN-β–treated animals than in controls (Fig. 2A). Delaying IFN-β treatment to even up to 6 hours after stroke onset did not result in a significantly different lesion size on ADC maps compared with prophylactic treatment.
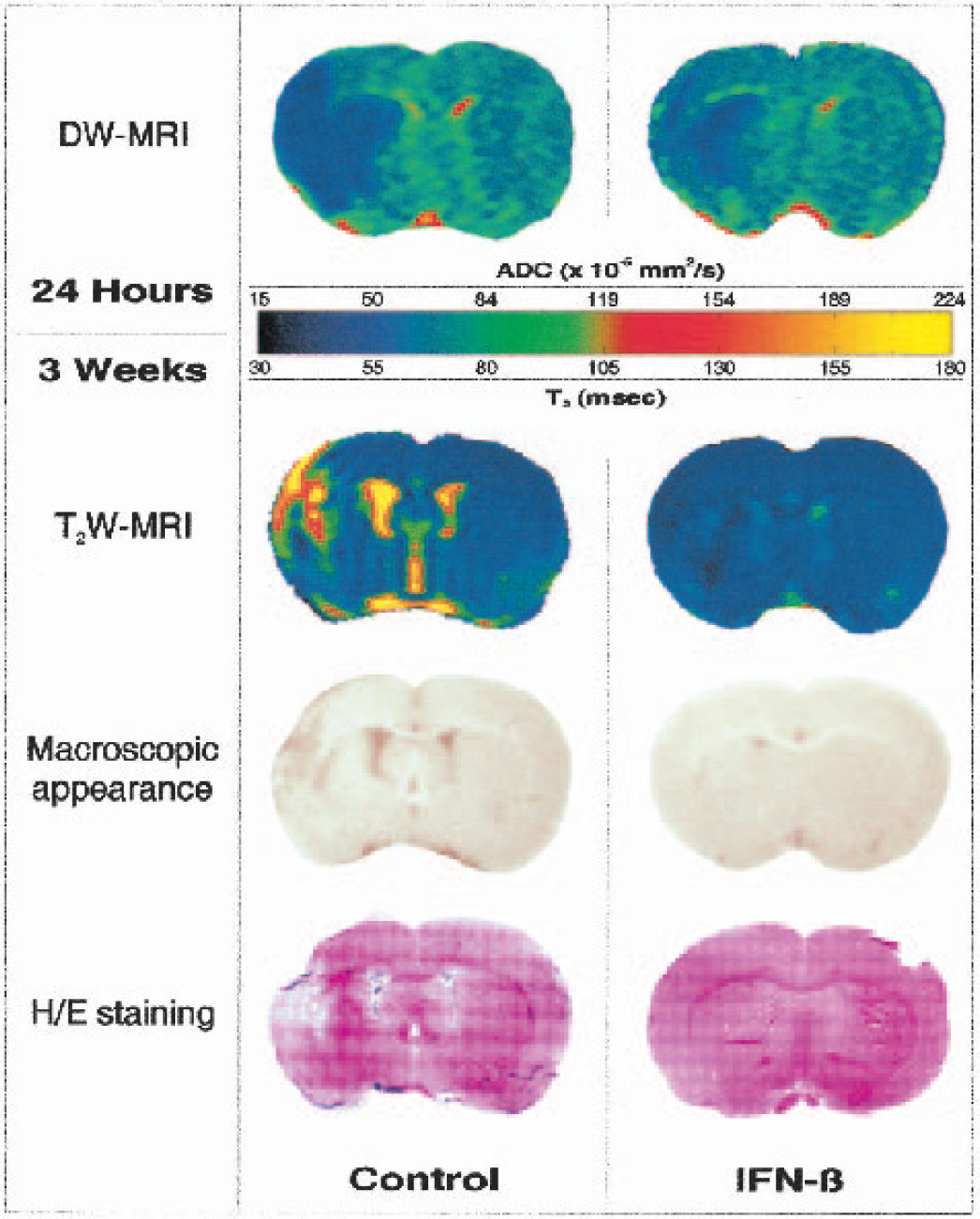
Parametric maps of MRI data of a control rat and a rat that received 500,000 U IFN-β subcutaneously, starting 6 hours after stroke onset. Shown are ADC maps of a 1.7-mm-thick slice through the center of the lesion, calculated from diffusion-weighted MRI data acquired at 24 hours after stroke. The ADC data showed ischemic lesions at risk of undergoing infarction, as evidenced by reduced ADC values. IFN-β reduced both the volume of tissue at risk and the severity of tissue damage, as determined from the extent of ADC reduction. Shown below the diffusion-weighted MRI data are T2 maps calculated from T2-weighted images acquired in the same animals 3 weeks later. The corresponding macroscopic appearance before H/E staining and H/E stained histologic sections are also shown.
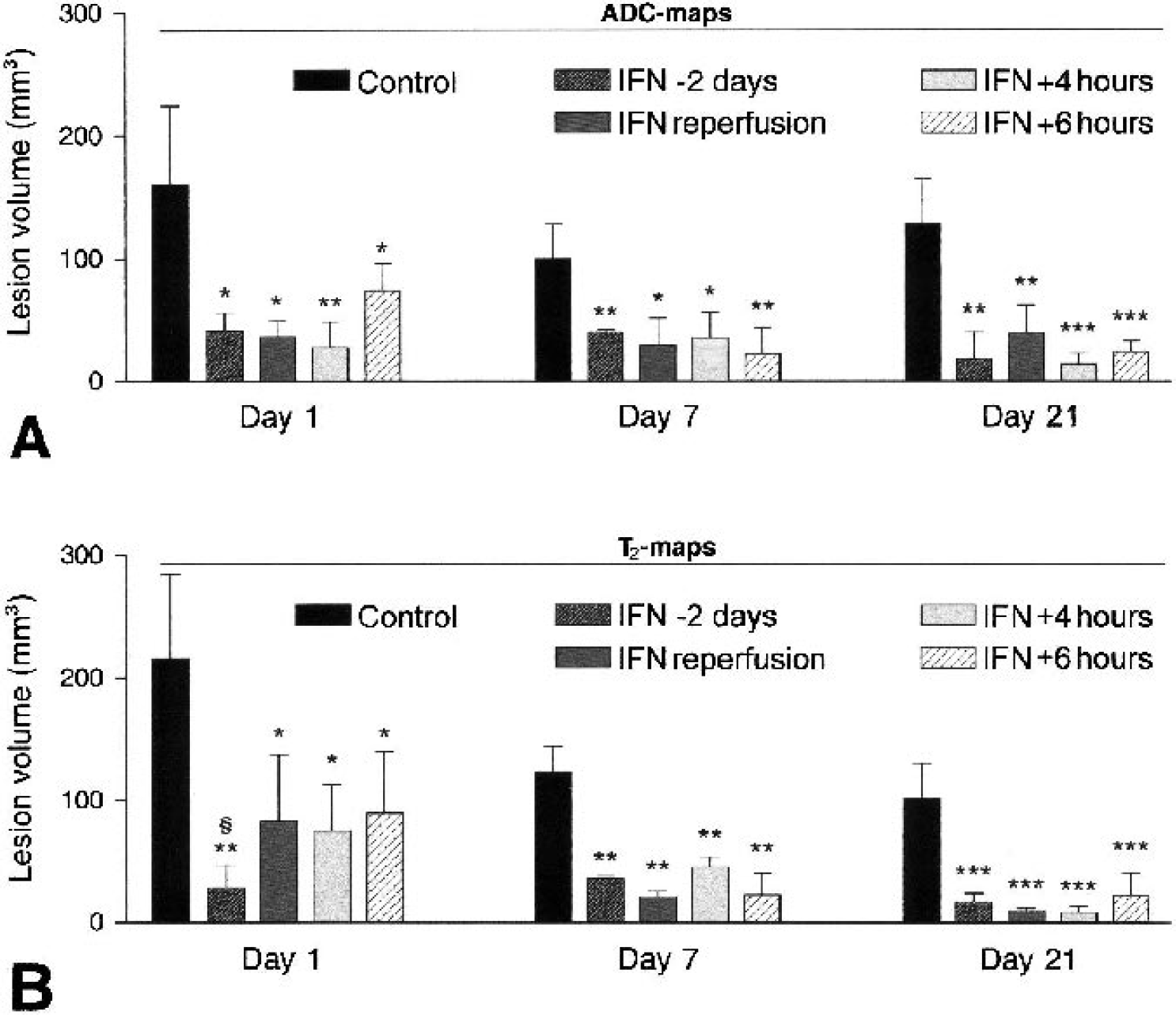
Lesion volumes of all treatment groups on all days, calculated from ADC data (
Edema formation is a common complication after brain injury. We therefore examined the effect of IFN-β treatment on vasogenic edema, using T2-weighted MRI at 24 hours, the time of maximal edema (Jiang et al., 1998). T2 maps, calculated from T2-weighted images, showed edematous tissue, as evidenced by increased T2 values (Fig. 1). At 24 hours after stroke the lesion on T2 maps was 67% smaller (P < 0.05) in IFN-β–treated animals compared with controls (Fig. 2B). This difference was also demonstrated by the midline shift caused by the swelling of brain tissue, which was absent or much less pronounced in IFN-β–treated animals than in controls (data not shown). We subsequently analyzed the effect of different onset of treatment on T2 lesion size at 24 hours. In terms of the ADC data, there was no difference between prophylactic treatment and post hoc treatment. In terms of the T2 data, prophylactically treated animals had 65% smaller lesion volumes on day 1 (P < 0.05, Fig. 2B) than animals treated after stroke onset. From day 7 onwards, the effect of starting treatment up to 6 hours after stroke was similar to that of starting treatment 2 days before surgery.
On day 21, ADC values had increased to supranormal values, which are indicative of vasogenic edema and tissue loss. The infarct size on ADC maps was 80% smaller (P < 0.0001) in IFN-β–treated animals than in controls (Fig. 2A). T2 data acquired on day 21 confirmed the presence of vasogenic edema and tissue loss, and correlated well with the macroscopic appearance and with the corresponding H/E stained histologic sections (Fig 1). Infarct size as determined from T2 maps was 85% smaller (P < 0.0001) in IFN-β–treated animals than in controls (Fig. 2B).
IFN-β treatment is associated with a preservation of blood–brain barrier integrity
Interferon-beta has been reported to prevent opening of the BBB (Stone et al., 1995). The intravascular contrast agent Gd-DTPA cannot pass the intact BBB. To study the effect of IFN-β on BBB integrity, the leakage of Gd-DTPA into the brain parenchyma was determined from postcontrast T1-weighted images (Neumann-Haefelin, et al. 2002). Detection of BBB disruption by determination of Gd-DTPA leakage (Jiang et al., 1997) correlates very well to Evans Blue uptake (Prabhu et al., 2000) and is more sensitive than using horseradish peroxidase effusion (Blamire et al., 2000). Gd-DTPA–induced signal enhancement in the brain parenchyma of IFN-β–treated animals was significantly lower than in control animals (Fig. 3A). This was true for both the cortex (P < 0.005) and the striatum (P < 0.01), indicating that IFN-β treatment was associated with a sparing of BBB integrity in both regions (Fig. 3B).
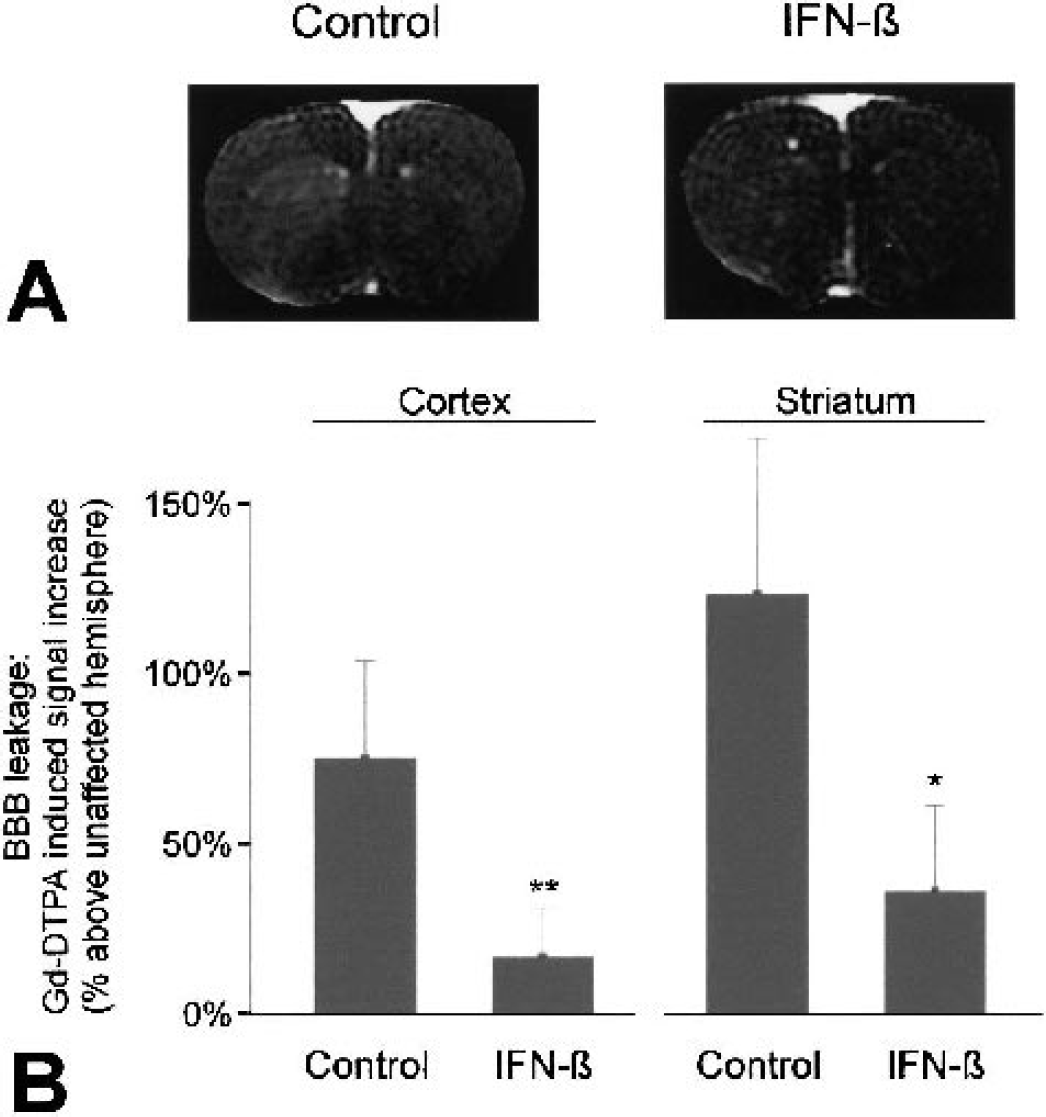
(
To correlate BBB damage to the severity of parenchymal injury, the mean ADC values of those regions of interest in the cortex and striatum were determined. The ADC values in each region were compared with the corresponding contralateral region and analyzed for differences between control and IFN-β–treated animals. The ADC values in the ipsilateral cortex of IFN-β–treated animals were not significantly different from the ADC values in the contralateral cortex (72.9 × 10−5 mm2 /s versus 75.0 × 10−5 mm2 /s, P > 0.05). In control animals, the ADC values in the cortex were significantly lower than the contralateral ADC values (58.8 × 10−5 mm2 /s versus 77.9 × 10−5 mm2 /s, P < 0.01). The difference with IFN-β–treated animals was significant (P < 0.05), indicating that IFN-β attenuated the ADC decrease in the cortex. In the striatum, in the center of the lesion, the ADC was significantly reduced compared to contralateral values, in both control and IFN-β–treated animals (controls: 57.5 × 10−5 mm2/s versus 75.2 × 10−5 mm2/s, P < 0.001; IFN-β–treated: 61.0 × 10−5 mm2/s versus 72.7 × 10−5 mm2/s, P < 0.001). Thus, in the striatum of IFN-β–treated animals, BBB leakage was attenuated despite the similar degree of tissue injury in that region compared with control animals.
Neuroprotection by IFN-β is not mediated by improved cerebral perfusion
Clogging of the microvasculature, initiated by neutrophils adhering to the vascular endothelium, has been described to interfere with reperfusion of ischemic tissue (Ames et al., 1968; del Zoppo et al., 1991; Mori et al., 1992). To study the occurrence of this so-called no-reflow phenomenon and to evaluate its attenuation as a possible mechanism of IFN-β–mediated neuroprotection, cerebral perfusion was assessed at 24 hours using perfusion-weighted MRI. In control animals, CBF after reperfusion was higher (albeit nonsignificantly) in the previously ischemic hemisphere than in the contralateral hemisphere. This was the case in both the cortex and the striatum (Fig. 4). Identical results, relative hyperperfusion of the previously ischemic hemisphere, were found in the brains of IFN-β–treated animals (Fig. 4) excluding attenuation of “no-reflow” as a likely mechanism for IFN-β–mediated neuroprotection.
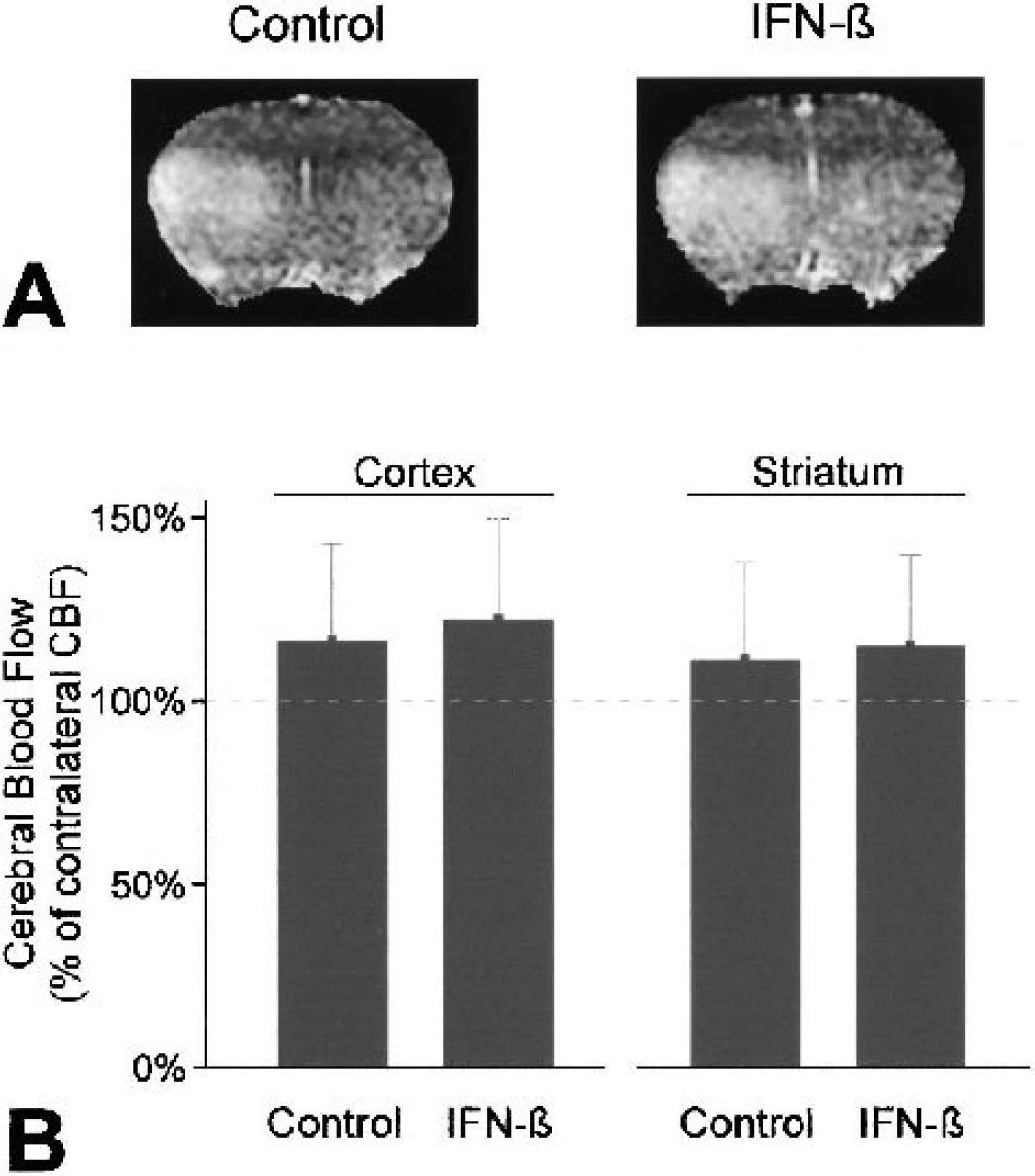
(
IFN-β blocks the infiltration of inflammatory cells after ischemia
To correlate the putative antiinflammatory properties of IFN-β to its effect on lesion size and BBB integrity, we assessed the infiltration of inflammatory cells into brain at 24 hours after stroke. Sections were analyzed from the center of the MCA territory where ischemia was most severe, and from the caudal part of the MCA territory, which suffered less severe injury. To study the effect of IFN-β treatment on blood-derived inflammatory infiltrate in general, histologic sections were stained for CD45, expressed on all leukocytes. No cellular infiltrates were present in the contralateral hemispheres of both control and IFN-β–treated animals. At 24 hours after stroke, massive infiltration of leukocytes was observed in the cortex and striatum of the ischemic territory (Figs. 5A and 5B). IFN-β treatment significantly reduced cellular infiltration in the cortex, both in the center (Fig. 5A) and in the caudal part of the MCA territory (Fig. 5B). We subsequently assessed the relative contribution of neutrophils and monocytes to the infiltrate. At this time point, some monocytic infiltrate was already present in the brains of control animals (Figs. 5C and 5D). Little or no monocyte infiltrate was present in the brains of IFN-β–treated animals, although the absolute difference was too small to become significant at the current time point. As expected, at this acute time point, the majority of the infiltrate consisted of neutrophilic granulocytes as identified by mAb HIS48 (Fig. 6). Treatment with IFN-β almost completely prevented the influx of neutrophils into the ischemic area (Figs. 6A–6C).
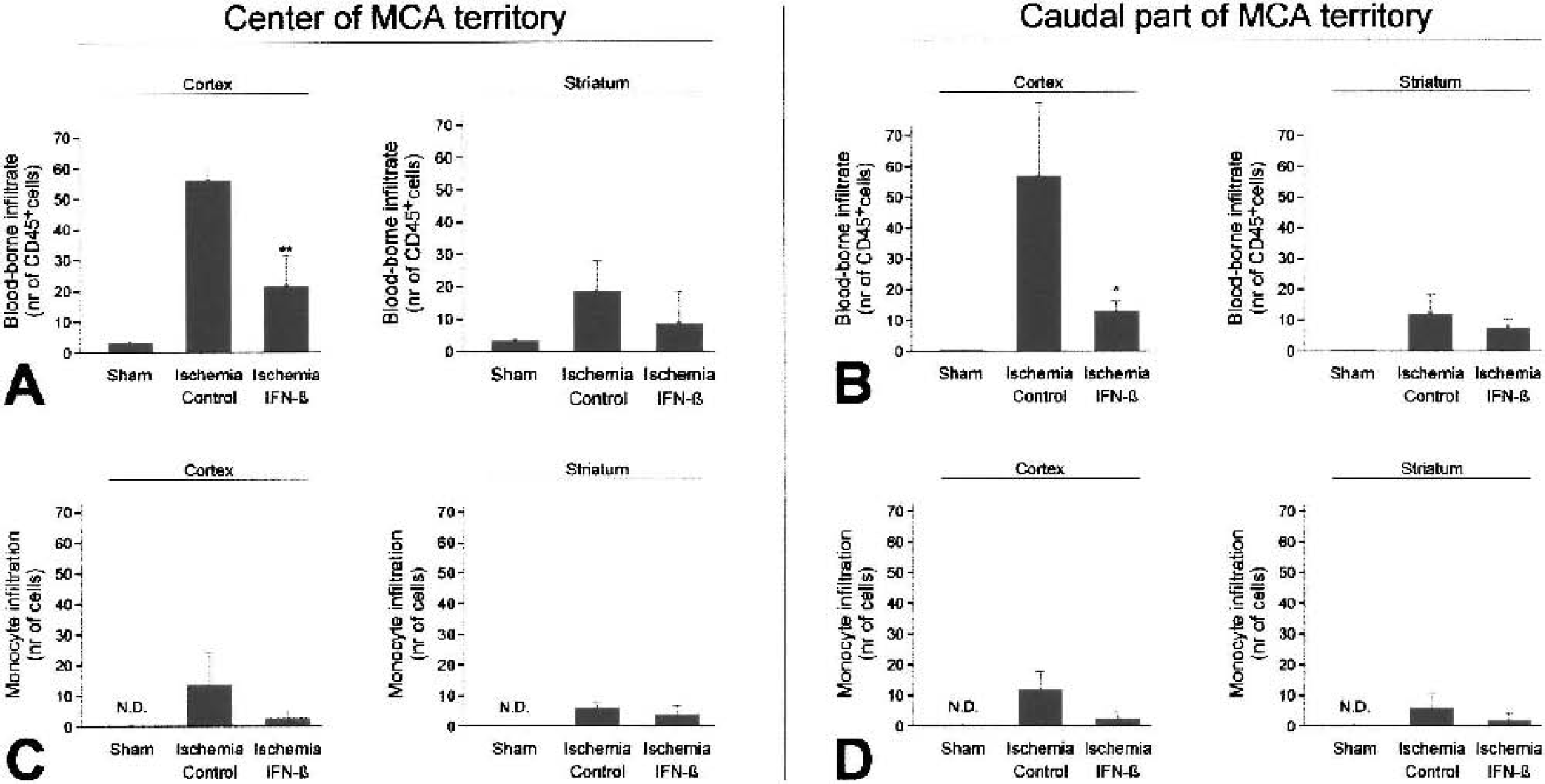
Inflammatory cell infiltration at 24 hours after stroke. (
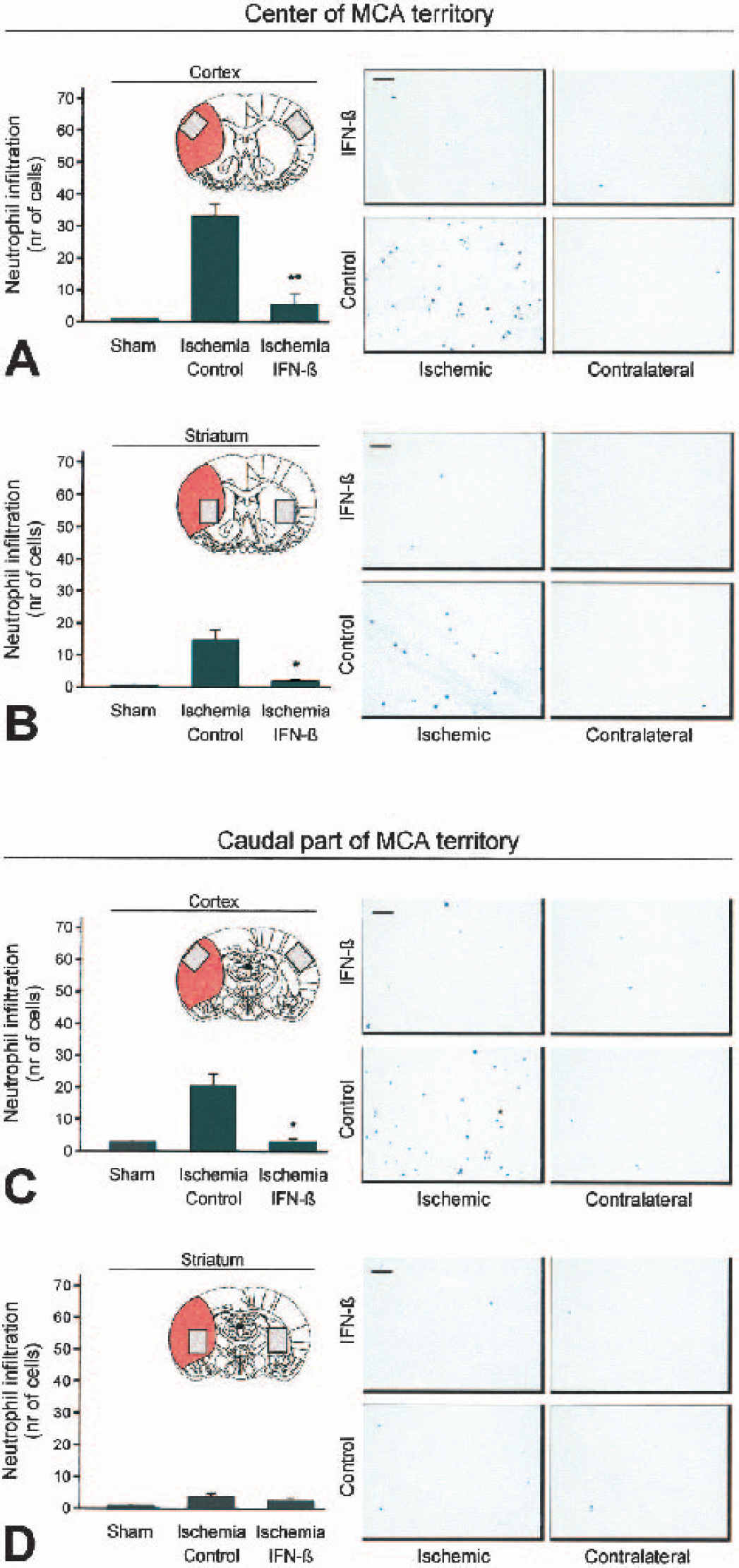
Neutrophil infiltration at 24 hours after stroke. Shown are representative cortical and striatal sections from the center (
Firm adhesion of neutrophils to ICAM-1 on microvascular endothelium is essential to infiltration. In light of earlier observations in experimental MS that IFN-β may block the infiltration of leukocytes into the brain by downregulating adhesion molecule expression (Floris et al., 2002; Ruuls et al., 1996; van der Meide et al., 1998), we investigated the effect of IFN-β on the expression of ICAM-1 after stroke. ICAM-1 expression was upregulated in the cortex and striatum of the ischemic hemisphere at 24 hours after stroke (Fig. 7). However, there was no difference between control and IFN-β–treated animals, indicating the effect of IFN-β on neutrophil influx was not mediated by a reduction in ICAM-1 expression (Fig. 7).
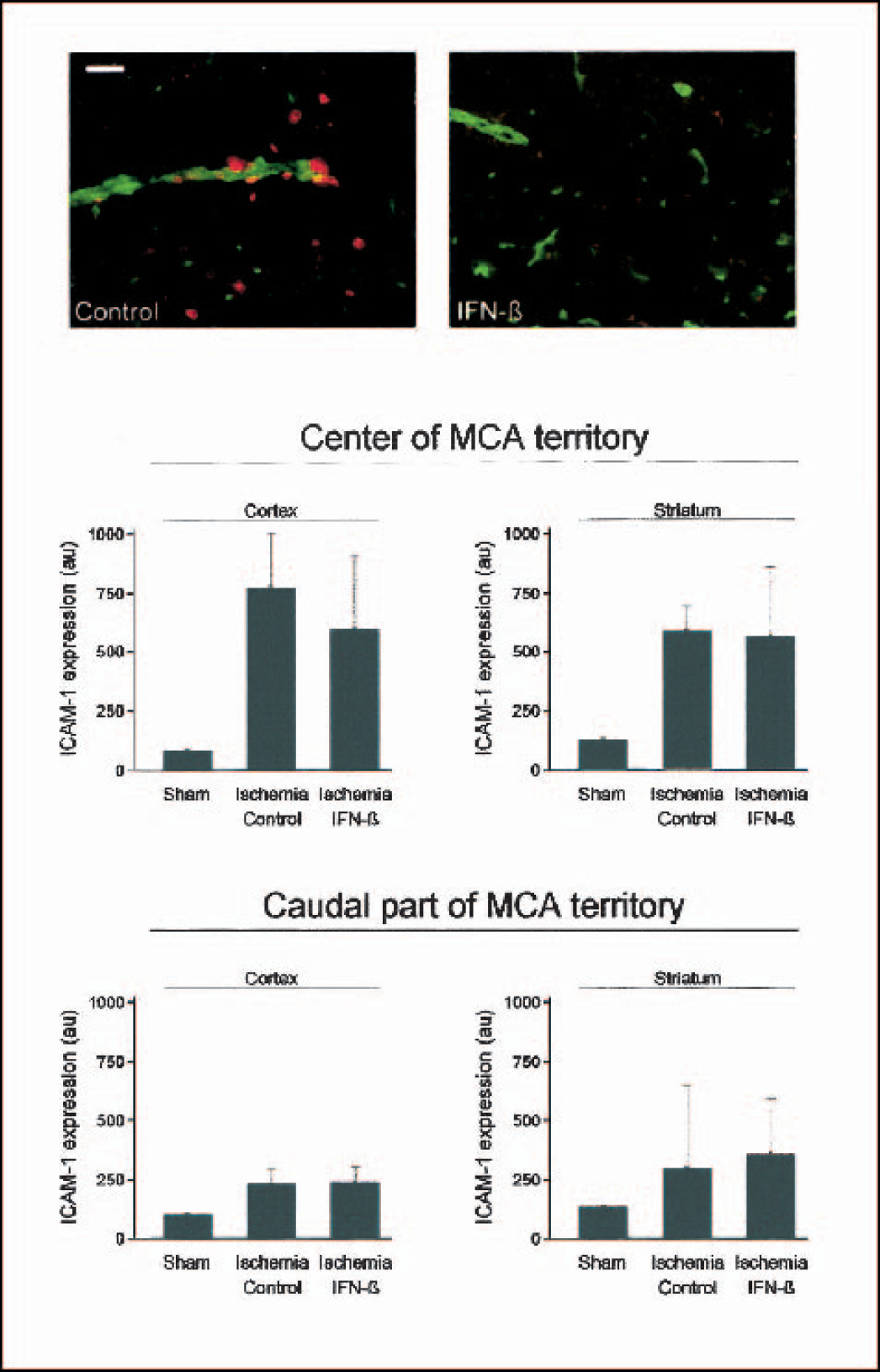
Intercellular adhesion molecule-1 expression at 24 hours after stroke. ICAM-1 expression (in green) was upregulated to an equal extent in the ischemic hemisphere of both control and IFN-β–treated animals. Neutrophils, detected by mAb HIS48 (in red), were present in the vascular lumen and in the brain parenchyma of control animals. Scale bar = 250 μm.
Finally, the number of infiltrated neutrophils in the cortical and striatal regions of interest (nr_pmns) was related to the extent of BBB damage in those regions as determined from the amount of Gd-DTPA leakage (gd_leak) on the postcontrast T1-weighted magnetic resonance images. The Pearson correlation coefficient for nr_pmns versus gd_leak was 0.833 (P < 0.05, two-tailed) and 0.815 (P < 0.05, two-tailed) for the cortex and striatum, respectively, indicating the extent of neutrophilic infiltrate positively correlated with the extent of BBB disruption.
IFN-β treatment is associated with a reduction in MMP-9 expression
In MS patients, the effect of IFN-β on monocyte and T-cell infiltration has been attributed to downregulation of MMP-9. Therefore, we performed gelatinase zymography, after gelatin-Sepharose purification, to assess MMP-2 and MMP-9 expression at 24 hours after stroke in relation to the neutrophil influx and BBB breakdown detected at that time point. In the lesion homogenates of all animals, a clear band of pro–MMP-2 was observed at 72 kd. The effect IFN-β of treatment was not associated with a decrease in MMP-2 expression (Fig. 8). The assay further showed a clear band corresponding to pro–MMP-9 at 92 kd in the control animals after ischemia. Although in one animal, after IFN-β treatment, subtle gelatinolytic activity was still detected, overall MMP-9 expression was strongly reduced compared with control animals (Fig 8.)
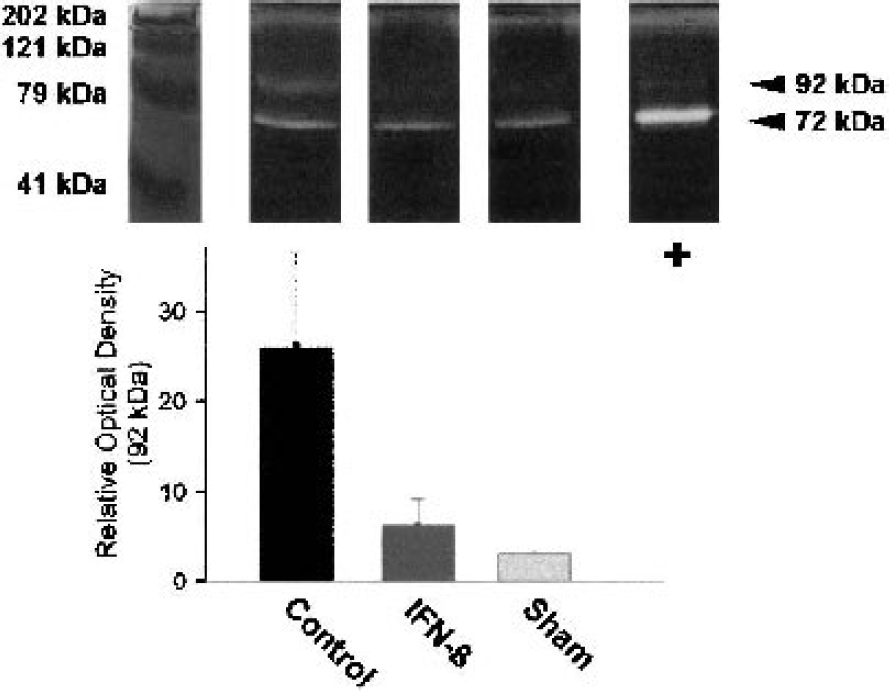
Gelatinase zymography for MMP-2 and MMP-9 at 24 hours after stroke. There was no upregulation of pro–MMP-2 (72 kd) after ischemia compared with sham operation. Quantification by optical densitometry showed that IFN-β prevented the upregulation of pro–MMP-9 (92 kd) witnessed in control animals. Zymography was performed in duplicate. The lane labeled ‘+’ contains pro–MMP-2 from the SK-N-SH human neuroblastoma cell line as a positive control.
DISCUSSION
There is an urgent need for acute stroke therapies that can be given over a wide time window, preferably in addition to thrombolytic therapy. Attenuation of inflammation may provide a target for such a therapy. In this study we applied MRI to a rat stroke model to longitudinally assess the effects of the antiinflammatory cytokine IFN-β on tissue damage, BBB disruption, and cerebral perfusion status after transient ischemic stroke. The data show that treatment with IFN-β affords significant protection against ischemia/reperfusion injury, even when treatment is started 6 hours after stroke onset. At 24 hours after stroke, IFN-β treatment was associated with a preservation of BBB integrity and with a 70% reduction in lesion volume. Postischemic CBF levels were not significantly different between control and IFN-β treated animals, which excludes improved reperfusion (e.g., by attenuating “no-reflow”) as the main neuroprotective mechanism. Reduced brain injury was sustained over the following days, and an 85% reduction in infarct size was measured 3 weeks after stroke. This observation lends support to the suggestion that IFN-β prevents aggravation of brain damage after transient ischemia by attenuating deleterious processes that progress over days, such as inflammation. We have previously shown that IFN-β inhibits the extravasation of monocytes into the CNS in an experimental model of the chronic inflammatory disease MS (Floris et al., 2002; Ruuls et al., 1996; van der Meide et al., 1998). A similar mechanism, reducing acute inflammatory infiltrate, appears to be active after ischemic brain injury. IFN-β treatment strongly reduced the number of neutrophils and monocytes that infiltrated the ischemic region. The powerful salutary effect of attenuating inflammation after stroke was recently clearly established in studies using the tetracycline derivative, minocycline, which strongly reduced brain injury, even after a 4-hour treatment delay (Yrjanheikki et al., 1999). We demonstrated that ICAM-1 was upregulated after stroke, but the effect of IFN-β was not mediated by a reduction in ICAM-1 expression, as has been reported for IFN-β treatment after MS in humans or experimental autoimmune encephalomyelitis in rats (Defazio et al., 1998; Floris et al., 2002; Muraro et al., 2000; Soilu-Hanninen et al., 1995; Trojano et al., 2000). At present, it is not known how IFN-β shields the brain from immune activity. IFN-β may have attenuated inflammation by stimulating the production of the classic antiinflammatory cytokine IL-10 (Chabot and Yong 2000; Liu et al., 2001; Tuohy et al., 2000), or by inhibiting the production of IL-8 (Oliveira et al., 1992), thereby directly reducing neutrophil recruitment to the ischemic area. Both downregulation of IL-8 and upregulation of IL-10 have been shown to protect the brain against ischemia/reperfusion injury (Mukaida et al., 1998; Ooboshi et al., 2002; Spera et al., 1998; van Exel et al., 2002). Noteworthy, the effect of IFN-β was shown to be associated with a reduced expression of MMPs. Because expression of adhesion molecules and proteolysis of the BBB are both essential to infiltration, the effect on MMP-9 expression may, at least in part, explain the effect on monocyte and neutrophil infiltration. The effect on MMP-9 may, however, also be secondary to the reduction in neutrophil infiltration. Nevertheless, IFN-β treatment resulted in diminished proteolytic activity, which correlates well with the observation that the protection afforded by IFN-β at 24 hours after stroke was accompanied by a preservation of BBB integrity. This effect cannot simply be explained by the reduction in lesion size because, as evidenced by the ADC data, the striatum in the core of the lesion suffered comparable injury in both control and IFN-β–treated animals, while leakage of contrast agent through the striatal BBB was nevertheless diminished in IFN-β animals.
Currently, the only available treatment for acute ischemic stroke patients is thrombolysis, which is severely limited in its application by the risk of hemorrhagic transformation. Matrix metalloproteinases are directly linked to this risk of hemorrhagic transformation (Montaner et al., 2001; Montaner et al., 2003; Sumii and Lo 2002), and their expression has been shown to be upregulated not only by ischemia but also by tPA treatment itself (Sumii and Lo, 2002). The present data show that IFN-β treatment was associated with a downregulation of MMP-9, which is in agreement with previous studies (Galboiz et al., 2001, Stuve, 1997; Trojano et al., 1999; Waubant et al., 2001) IFN-β may, in addition to its neuroprotective potential, lower the risk of hemorrhagic transformation and thereby widen the time window during which thrombolytic therapy can be given.
In conclusion, this study shows that treatment with the antiinflammatory cytokine IFN-β may afford significant neuroprotection against ischemia/reperfusion injury, and within a relatively long treatment window. IFN-β has shown effectiveness against MS both in humans and in rats indicating a conserved mode of action in these species. Because IFN-β has been approved for clinical use, it could be rapidly tested in a clinical trial for its efficacy against human stroke.
Footnotes
Acknowledgments:
Johnson & Johnson Medical supplied the applier for the vascular microclips used in this study. The authors thank Henk Veldman and Gerard van Vliet for their technical expertise.
