Abstract
Multiple sclerosis (MS) is primarily an autoimmune disorder of unknown origin. This review focuses iron overload and oxidative stress as surrounding cause that leads to immunomodulation in chronic MS. Iron overload has been demonstrated in MS lesions, as a feature common with other neurodegenerative disorders. However, the recent description of chronic cerebrospinal venous insufficiency (CCSVI) associated to MS, with significant anomalies in cerebral venous outflow hemodynamics, permit to propose a parallel with chronic venous disorders (CVDs) in the mechanism of iron deposition. Abnormal cerebral venous reflux is peculiar to MS, and was not found in a miscellaneous of patients affected by other neurodegenerative disorders characterized by iron stores, such as Parkinson's, Alzheimer's, amyotrophic lateral sclerosis. Several recently published studies support the hypothesis that MS progresses along the venous vasculature. The peculiarity of CCSVI-related cerebral venous blood flow disturbances, together with the histology of the perivenous spaces and recent findings from advanced magnetic resonance imaging techniques, support the hypothesis that iron deposits in MS are a consequence of altered cerebral venous return and chronic insufficient venous drainage.
Keywords
Cerebral veins and iron stores in multiple sclerosis
Multiple sclerosis (MS) is an inflammatory, demyelinating disease of the central nervous system (CNS) of elusive origin, which is widely considered to have autoimmune determinants. The multistep mechanism of the disease involves inflammation, demyelination, and neurodegeneration (Compston and Coles, 2002; Barnett and Sutton, 2006; Frohman et al, 2006). Interestingly, from the time of the first histologic description of Charcot, MS plaques is known to be venocentric (Charcot, 1868; Barnett and Sutton, 2006). Both magnetic resonance imaging (MRI) venography and postmortem studies show a central vein oriented on the long axis of the inflammatory lesion (Kermode et al, 1990; Kidd et al, 1999; Tan et al, 2000; Fog, 1964, 1965). In addition, common to several neurodegenerative disorders, the brain and spinal cord of MS-affected patients contain abnormally high levels of redox-active metals, particularly iron (Sayre et al, 2005), documented by advanced MRI (Haacke et al, 2005; Tjoa et al, 2005) and enhanced histochemical methods (Sayre et al, 2005; LeVine and chakrabarty, 2004). Histologic studies show the peculiar disposition of the iron stores in MS constantly encircling the venous wall, as shown in Figure 1 (Adams, 1988, 1989; Adams et al, 1989). Iron stores are arranged in hemosiderin deposits as well as in ferritin-like structures inside the macrophage, curiously resembling perivenous iron stores commonly observed in peripheral venous disease. Starting from histology, it has been delineated an impressive parallel between the inflammatory process activated in the course of chronic venous disorders (CVDs) (Zamboni, 2006; Zamboni et al, 2008), and that profoundly studied in MS (Minagar et al, 2006; Frohman et al, 2006), although a parallel evidence of venous hemodynamic (VH) impairment in MS was still lacking (Zamboni, 2006). However, two important occurrences have taken place in the past 2 years: (1) a demonstration of altered venous flow in the cerebral venous system in the course of MS and (2) the development of advanced MRI techniques that have brought about an extraordinary improvement in the capacity to assess iron stores and cerebral veins.
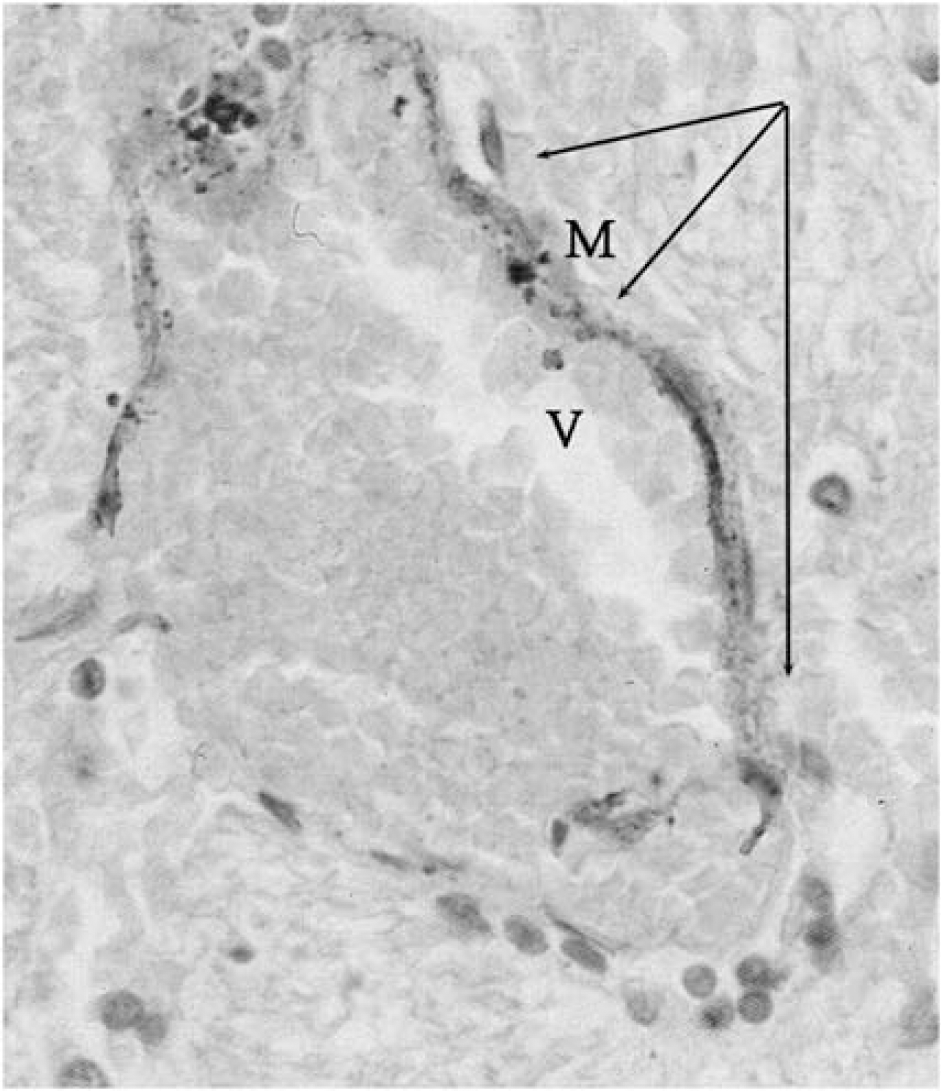
Histology of a brain MS plaque (hematoxylin-eosin × 50). The figure depicts a streak of blood (arrows) encircling the wall of a vein (V) at the center of an MS brain plaque. Heme iron triggers macrophages infiltration, demonstrated by the presence of iron-laden phagocyte (M).
This review delineates the role of iron in the complex pathogenesis of MS and corroborates the hypothesis that iron overload in MS is secondary to disturbed venous flow in the cerebral veins.
Metallobiology of central nervous system iron and putative role in pathogenesis of multiple sclerosis
Physiologic Role and Regulation of Iron in the Central Nervous System
Iron is important for CNS physiology as it acts as cofactor for enzyme cascade of neural metabolism and ATP production. Further, it is required by all living cells, whether neuronal or other cells, taking part in tissue formation, angiogenesis, oxygen transport, electron transfer, and neurotransmitter synthesis (Chua et al, 2007). Most importantly, iron is instrumental in myelination and oligodendrocyte development as it is required by most of the growth factors involved in these processes. This fact is supported by such evidence as low iron content in hypomyelinated brains and deeply stained iron-enriched oligodendrocytes in the white matter of normal and diseased brains (Todrich et al, 2009). Moreover, neuronal cells from the brain and spinal cord use iron for routine functions and maintenance of cellular homeostasis that can be reflected by the presence of abundant transferrin receptors on brain capillary endothelial cells (Sipe et al, 2002; Ke and Qian, 2003).
Transferrin is a glycoprotein that is responsible for iron homeostasis in vivo; it maintains the iron pool in the systemic circulation by binding with iron tightly but reversibly. Oxygen is the elixir of life, and glial cells in the brain require more oxygen and glucose consumption to generate a continuous ATP pool in vivo than do other organs. A continuous ATP pool is essential to normal functioning of the brain, which is one of busiest organs and keeps all other organs active and under control. Neurons are highly polarized, and proteins of neuronal homeostasis and CNS physiology are synthesized in cell bodies and then transported to functional sites at synapses. In cooperation with iron regulatory proteins, ferritin, and ferroportin, transferrin maintains the iron requirement under control across neuron transport in the CNS by regulating iron absorption, transport, storage, and utilization by brain cells (Rouault, 2001; Yang et al, 1984; Chua et al, 2007).
Role of Iron in Aging
With advancing age, iron accumulates in the brain and has been associated with senile dementia, many cognitive dysfunctions, and neurodegenerative disorders (Altamura and Muckenthaler, 2009). Although it is unclear how iron accumulates as age progresses, in a recent report Jeong and coworkers showed that multiple mechanisms may lead to iron accumulation in neurons and glia. These mechanisms include dysregulation of the proteins involved in iron influx and sensing of intracellular iron; iron accumulation in ventral motor neurons secondary to blockage of anterograde axonal transport; and increased mitochondrial iron load (Jeong et al, 2009). This finding may pave the way to a better understanding of iron accumulation in the aged brain.
Moreover, anatomic and metabolic features of the aged brain make it more susceptible to iron accumulation and subsequent reactive oxygen species-mediated toxicity. Glia, the principal cells in the brain that provide metabolic and anatomic support to neurons and other CNS cells, have a high lipid content and a greater requirement for glucose and oxygen for the ATP pool in the brain (Giaume et al, 2007). A higher lipid and oxygen content, in the presence of iron in glial cells and associated neurons, creates an environment of peroxidation of lipids, proteins and DNA in the aged brain through the Fenton reaction, described in later section (Zaleska and Floyd, 1985; Van Rensburg et al, 2004). In addition, low antioxidant activity and low regenerative capacity of brain cells after injury, render the aged brain weak in combating iron-mediated toxicity in older people (Van Rensburg et al, 2004).
Role of Iron in Neurodegenerative Disorders
The role of metallobiology in neurodegenerative disorders has long been implicated, with particular attention given to iron as it is one of the most important redox metals, which have been largely linked to senile toxicity and neurodegenerative disorders such as Alzheimer's and Parkinson's diseases and aging patients (Bush, 2000; Sullivan, 2004; Altamura and Muckenthaler, 2009; Stankiewicz et al, 2007; Stankiewicz and Brass, 2009). The redox switching capability of iron from ferrous to ferric state, and vice versa, makes it one of the most dangerous catalytic elements responsible for the neurodegenerative process (Levenson and Tassabehji, 2004). The role of iron in the neurodegenerative process can be best described in three distinct phases: accumulation, invasion, and catalytic activity. Iron accumulation in the brain follows a nonuniform distribution, both regionally and cellularly, that reflects the differential requirements of iron and its regulation in proportion to regional brain activity. Regions of the brain that are associated with motor functions (extrapyramidal regions) tend to have more iron than nonmotor-related regions (Koeppen, 1995). Similarly, oligodendrocytes, microglia, and neurons undergo deep immunostaining in the aged brains of patients with Alzheimer's and Parkinson's diseases, indicating a higher than usual cellular iron content (Levenson and Tassabehji, 2004; Zecca et al, 2004). Particularly in microglia, a higher iron content clearly depicts the role of iron in activating the immunoinflammatory cascade in neurodegenerative disorders (Zecca et al, 2004). During invasion and increased activity, stored iron generates free radicals and reactive oxygen species in the aged brain, as evidenced by higher heme oxygenase-I, which contributes to increased susceptibility to oxidative stress in older people (Hirose et al, 2003). Biochemical events surrounding iron-mediated catalytic events, which give rise to oxidative stress and free radical generation can be given as
Fe3++•O2− → Fe2++O2 (Step I)
Fe2++H2O2 → Fe3++OH−+•OH (Step II) ∗
Combining Step I & II
•O2−+H2O2 → •OH+HO−+O2∗Known as Fenton reaction (Hunt, 2009).
Iron Overload in Multiple Sclerosis
Although investigations into the role of iron in MS are still few, many high-resolution MR techniques have shown stored iron regions inside the brain and spinal cord. Moreover, in experimental autoimmune encephalomyelitis (EAE), an animal model of MS, dietary modifications have revealed an incidence of EAE in ∼70% of mice with a normal iron level or iron overload, but 0% in iron-deficient mice. This is clear evidence that iron deficiency protects against the progress of MS in mice with induced EAE, with obvious clinical implications (Grant et al, 2003). The authors speculated that the failure of iron-deficient mice to develop EAE is impressive, but controversy remains as iron deficiency may lead to much more serious health hazards. However, they conclude that any of the pharmaceutical approaches to inhibiting EAE are less effective than iron deficiency. Another study that correlated iron overload and MS revealed variations in the level of protein expression involved in iron homeostasis. Sfagos et al investigated the serum concentration of soluble transferrin receptor in a group of MS patients. The levels were found to be significantly higher in patients with active MS, in both progressive and relapsing-remitting clinical forms, than in controls. Serum ferritin levels were also significantly elevated in patients affected by the active and progressive form of MS. These findings reinforce the above argument, which proposes local iron overload as a pivotal signal of the inflammatory chain in MS (Sfagos et al, 2005). In addition, higher levels of soluble transferrin receptors and increased transferrin receptor in the serum of nonanemic MS patients with active disease, which reflect increased iron turnover, may indicate active inflammation with ongoing oxidative damage that is not detectable by patient history or examination (Abo-Krysha and Rashed, 2008). In a recent review, Trap and Stys hypothesized in MS that imbalance in the increased energy demand of impulse conduction along excitable demyelinated axons, together with reduced axonal ATP production, induce a hypoxia-like state in chronically demyelinated axons, caused by mitochondria and ion-gated channel dysfunction (Trapp and Stys, 2009).
Biological Links of Iron Overload to Autoimmune Inflammation in Multiple Sclerosis
Multiple sclerosis is believed to be primarily an autoimmune disorder with an initial trigger suspected among environmental factors (including viral infection, bacterial lipopolysaccharides, and superantigens) impacting a genetic predisposition. The initial trigger facilitates the movement of autoreactive T-helper (Th) cells and demyelinating antibodies from the systemic circulation into the CNS through disruption of the blood–brain barrier (Noseworthy et al, 2000).
Autoimmune cells mainly destroy oligodendrocytes, the cells responsible for creating and maintaining a fatty layer known as the myelin sheath, which helps the neurons to carry action potentials across axonal pole. In MS, this myelin sheath becomes thin or completely disappears. Most importantly, due to the repeated attacks of autoimmune cells, the remyelination capacity of the insulating sheath is lost, leading to scar-like plaques or lesions situated around the damaged axons (Chari, 2007). However, in such a scenario, iron overload and oxidative stress may be the underlying cause that leads to immunomodulation in chronic MS. Perivascular extravasation of erythrocytes has been demonstrated in plaques of patients with a recent relapse of MS (Adams, 1988). Biologically, the erythrocyte is degraded in the tissues by heme oxygenase. This enzyme, together with carbon monoxide, which is one of the products of the reaction, is a major regulator of the T-lymphocytes CD4+ (Brusko et al, 2005). In addition, the iron deposits represent a powerful chemotactic stimulus that attracts macrophages. This is confirmed by the presence of iron-laden macrophages in the MS plaques, as shown in Figure 1 (Adams, 1988, 1989; Zamboni, 2006; Zamboni et al, 2008).
Furthermore, it has been observed that the cells involved in iron overload having the greatest effect on immunity are the macrophages, and that there is a close relationship between iron and the major cells of adaptive immunity, the T-lymphocytes, as they have a major function in recycling the iron from hemoglobin (Porto et al, 2007).
Figure 2 describes the role of major cells from immune cascade those attack and damage to the myelin sheath across cerebrospinal region triggered by iron overload and oxidative stress during the course of autoimmune inflammation.
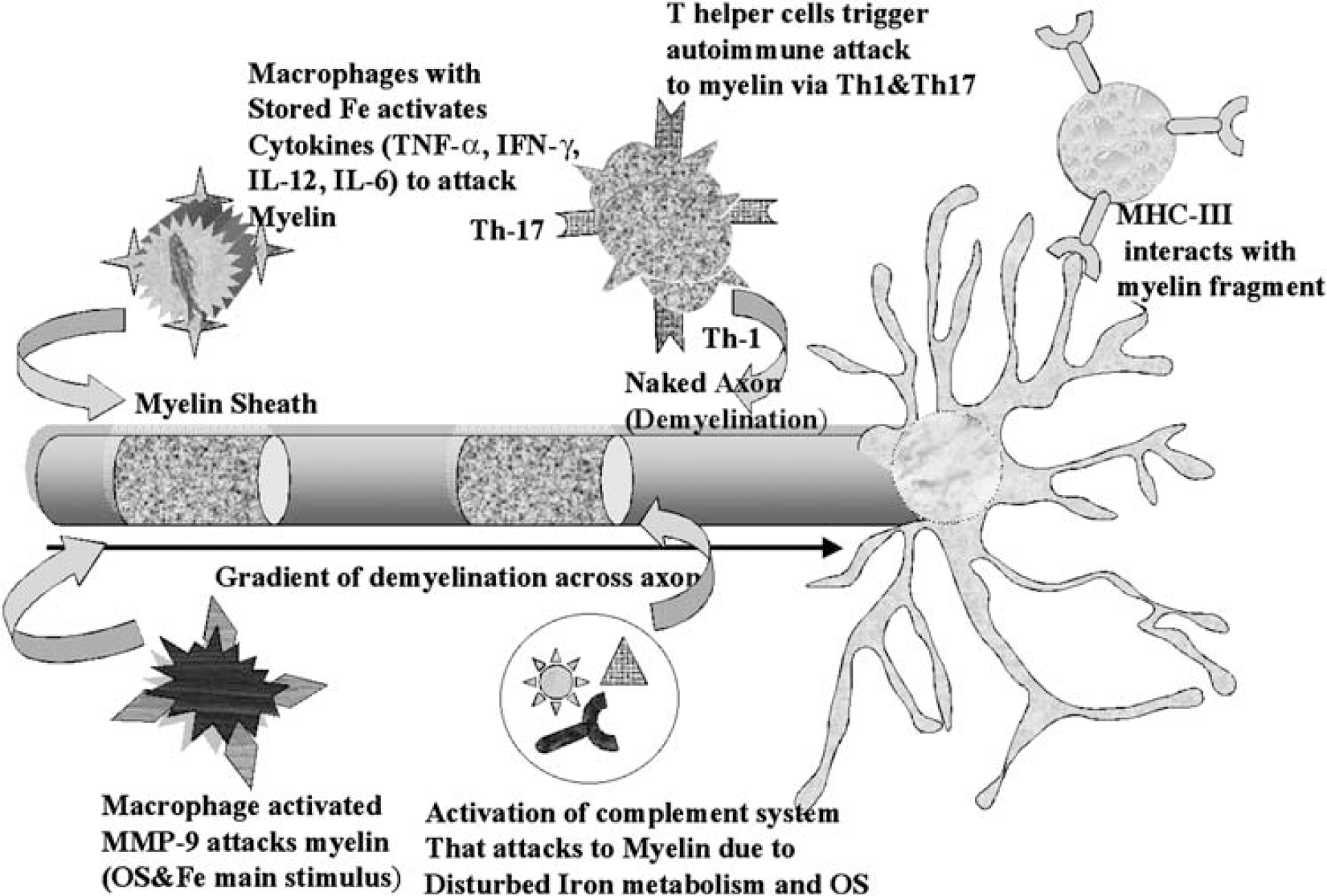
Autoimmunitary attack to myelin. The role of major cells from immune cascade those attack and damage to the myelin sheath under autoimmune modulation across cerebrospinal region, triggered by iron overload and oxidative stress (IL, interleukin; Th, T-helper cells; MMP, matrix metallo proteinase; MHC, major histocompatibility complex; TNF-α, tumor necrosis factor-α; IFN-γ, interferon-γ).
Moreover, INF-γ is produced by T-lymphocytes and seems to be the main mediator of neurodegeneration (Brusko et al, 2005; Frohman et al, 2006).
However, INF-γ is simultaneously responsible for T-lymphocyte apoptosis. Therefore, under normal conditions, a positive feedback loop resulting in a high local concentration of INF-γ cannot develop. Several studies have shown the role of iron in T-lymphocyte regulation, and iron induces their refractoriness to INF-γ/STAT1 signaling. Thus, iron can determine a longer survival of T-lymphocytes exposed to INF-γ, increasing the potentiality of INF-γ-mediated neuronal damage (Regis et al, 2005).
Physiology of cerebral venous return and venous blood flow alterations discovered in multiple sclerosis
The blood leaves the brain by using the back propulsion of the residual arterial pressure (vis a tergo), complemented by antegrade postural and respiratory mechanisms (vis a fronte) as shown in Figure 3 (Ursino and Lodi, 1997; Schaller, 2004; Menegatti and Zamboni, 2008).
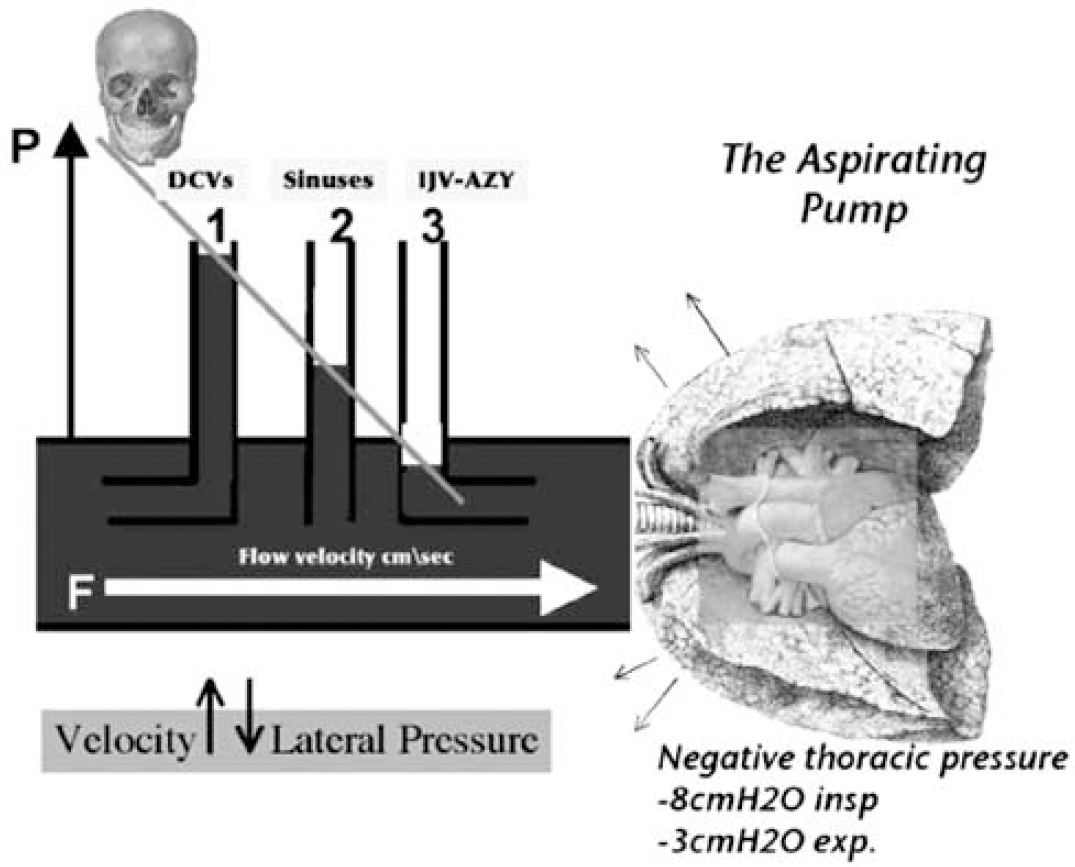
Physiology of cerebral venous return. The blood leaves the brain by using the back propulsion of the residual arterial pressure (vis a tergo), much more important in the cerebral veins, complemented by antegrade postural and respiratory mechanisms (vis a fronte), which has a major function in the dural sinuses and in the jugular and azygous vein (IJV-AZ). In fact, the blood flow velocity is higher close to the chest, due to the negative thoracic pressure gradient, increased by inspiration. According to the Bernoulli law, by increasing the blood flow velocity there is a corresponding reduction of the lateral pressure resulting in a natural aspiration of the deep cerebral veins (DCVs) into the sinuses, and finally into the main outflow extracranial pathways, IJV-AZ.
The latter consists of increased venous outflow during inspiration, thanks to increased thoracic negative pressure, which improves the aspiration of blood toward the right atrium. Moreover, the supine posture favors cerebral venous outflow through the internal jugular veins (IJVs); on the contrary, in the upright position, blood is redirected through the vertebral veins and the azygous vein (AZ), which become the predominant pathways in that position (Ursino and Lodi, 1997; Schaller, 2004; Menegatti and Zamboni, 2008).
It has been recently shown that MS is significantly associated with a condition defined as chronic cerebrospinal venous insufficiency (CCSVI) (Zamboni et al, 2009a, 2009b, 2009c). The CCSVI is characterized by multiple stenoses of the principal pathways of extracranial venous drainage, the IJVs and the AZ, with opening of collaterals, clearly demonstrated by means of selective venography, as depicted in Figure 4. The presence of stenotic lesions that cannot be crossed by postural and respiratory mechanisms explains the higher rate of reverse flow recorded in the main extracranial venous outflow route.
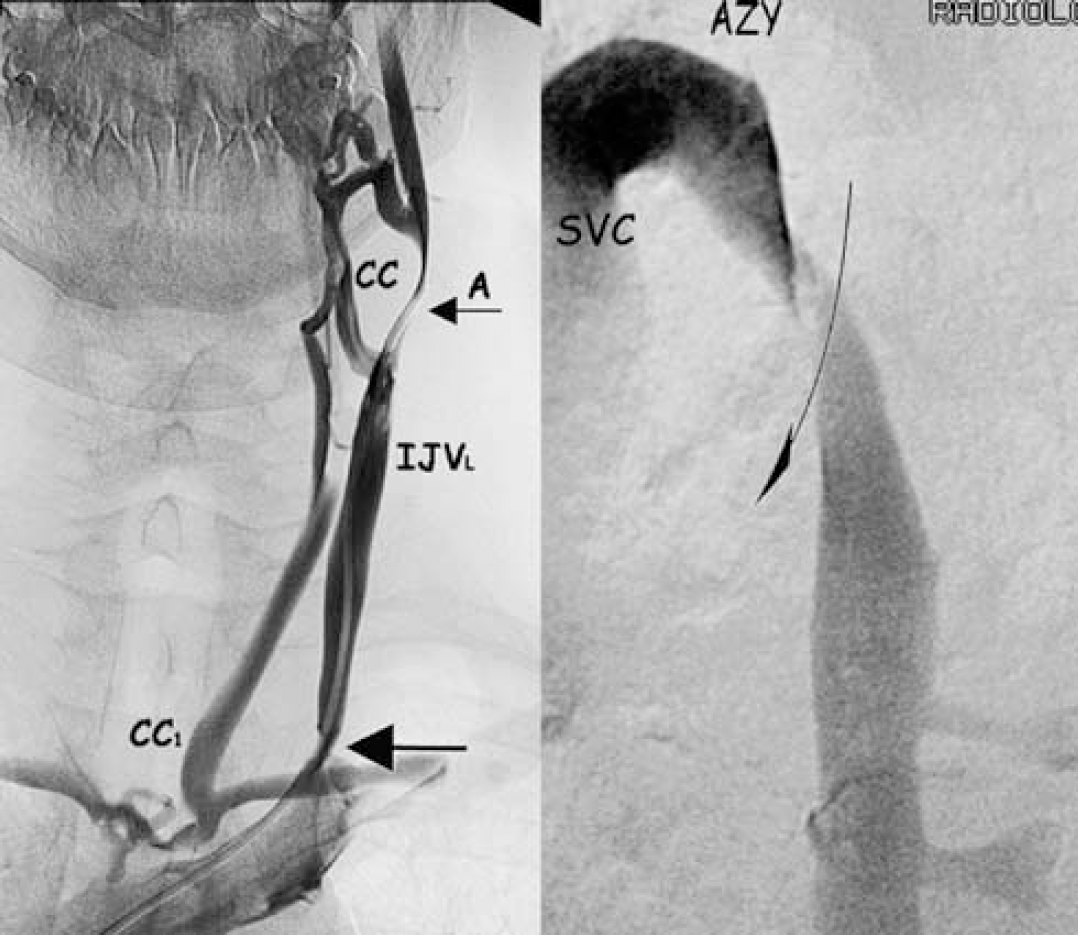
Extracranial venous stenosis associated to MS (CCSVI). Selective venography in the course of CCSVI associated to MS. Left: proximal stenosis of the left IJV (arrow) with agenesia more distally (A). Intra–extra cranial collateral circle (CC1) represented by condylar veins shunts the double block, whereas the agenesia is bypassed by an extracranial collateral (CC). Right: Twisting of the descending segment of the AZ just below the arch communicating with the superior vena cava (SVC).
In MS patients, venous stenoses in the main cerebrospinal outflow pathways were never found to be isolated. Rather, they were combined in the IJVs, AZ, and lumbar systems, defining four main patterns of distribution (Zamboni et al, 2009b):
Type A pattern (30%) is characterized by a stenoobstruction of the proximal AZ, associated with a closed stenosis of one of the two IJVs, with a compensatory controlateral IJV that appears with an ample cross-sectional area; Type B pattern (38%) is characterized by significant stenoses of both IJVs and the proximal AZ; Type C pattern (14%) is characterized by bilateral stenosis in both IJVs, with a normal AZ system; Type D pattern (18%) is characterized by the multilevel involvement of the AZ and lumbar systems. Association with the IJVs was observed in approximately 50% of cases, causing an additional obstruction in these patients. Type D is significantly associated with the PP course of MS, characterized by a plaque topography in the spinal cord.
The CCSVI brings about an overturning of the above-described postural and respiratory mechanisms, rendering abnormal the physiology of cerebral venous return (Zamboni et al, 2007, 2009b, 2009c). Combined transcranial and extracranial echo-color-Doppler allows for measurement of VH parameters indicative of CCSVI (Zamboni et al, 2009c). The CCSVI diagnosis needs to fulfill at least 2 out of 5 VH parameters (Zamboni et al, 2009b, 2009c). The detection of ≥2 parameters in the same subject was never observed in controls (Zamboni et al, 2009c), but perfectly overlapped with the diagnosis of clinically definite MS (sensitivity 100%, specificity 100%, positive predictive value 100%, and negative predictive value 100%). Despite the strong association found between CCSVI and MS, it has not yet been clarified whether such extracranial venous obstructions can be considered a cause or an effect of MS.
Role of Venous Collateral Circulation and the Concept of Vicarious Venous Shunt
Significant obstacles to the main venous outflow collectors, with overload of collaterals and associated refluxes, have been described at the level of the iliac veins in obstructive CVD, and also in the inferior vena cava in primary Budd-Chiari syndrome (Bergan et al, 2006; Raju et al, 2006; Lee et al, 2006). Similarly, in CCSVI, the main extracranial cerebrospinal veins are blocked, with opening of substitute circles. When venous flow is deviated into collaterals to bypass an obstacle, there exists an anatomic bypass called a vicarious shunt (Franceschi, 2009; Zamboni et al, 2009a).
This type of shunt is desirable because it bypasses blocked veins and thereby reduces resistance to drainage. Blood flows regularly in the substitute circle under the effects of distal cardiac residual pressure and proximal thoracoabdominal aspiration. Opening of extraintracranial draining anastomosis is depicted by vascular imaging in CCSVI (Zamboni et al, 2009b) as shown in Figure 4. However, the time of venous outflow is longer than normal, leading to insufficient venous drainage, as confirmed by the mean transit time measured in MR perfusion study, even in normal-appearing white matter (Law et al, 2004). The main collateral pathways activated in the course of CCSVI are depicted in Figure 5: the condylar venous system, the pterygoid plexus, the thyroid veins. Additionally, the suboccipital cavernous sinus and the hemiazygous-lumbar venous anastomosis with the left renal vein may also become prominent substitute circles. Collateral circulation prevents brain edema and intracranial hypertension (Schaller, 2004; Zamboni et al, 2009a), and ensures a correct but slower, and thus insufficient, venous drainage.
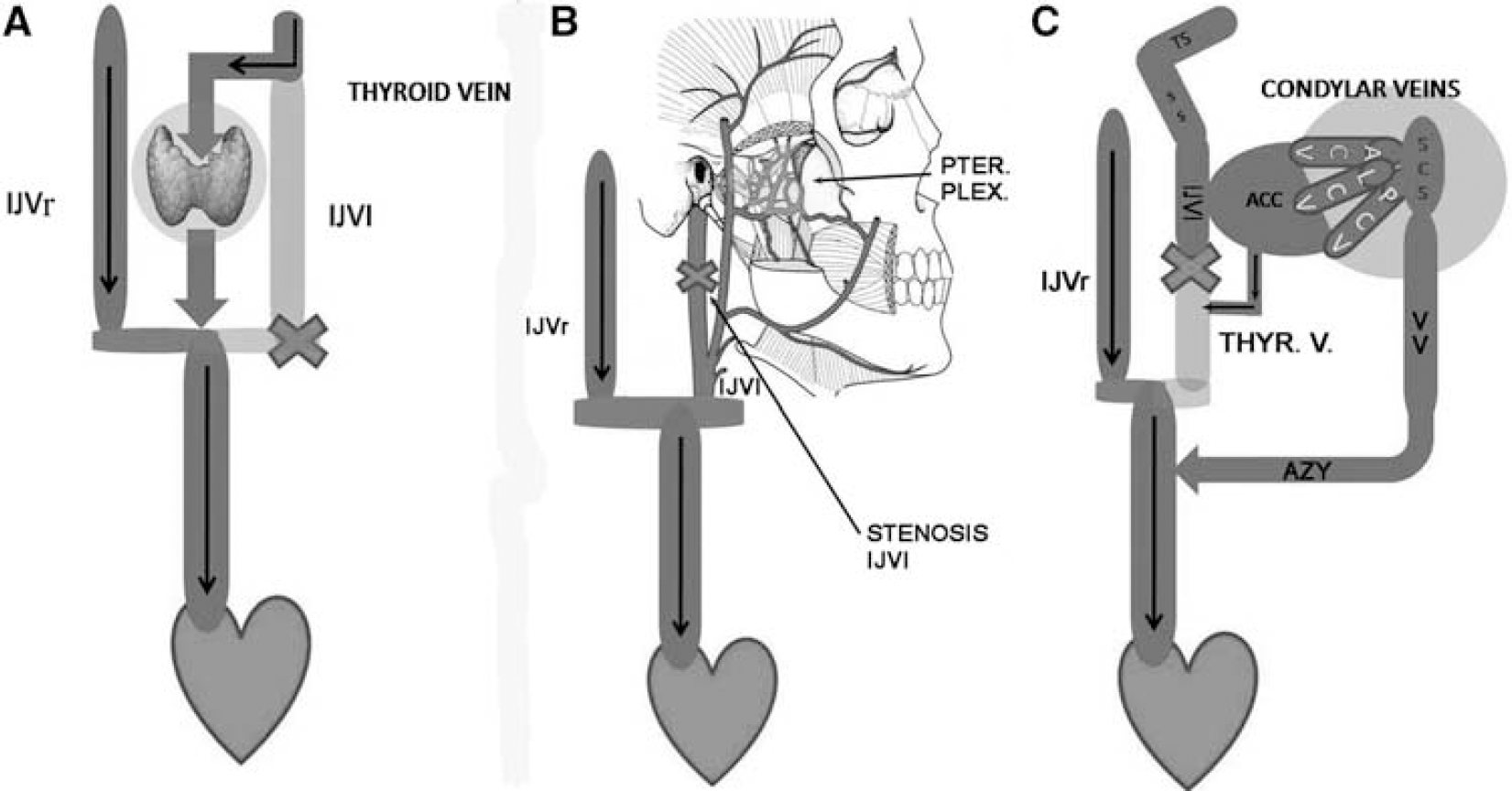
Main collateral circles activated in the course of CCSVI-MS. Substitute circles frequently activated in the course of chronic obstruction of the IJVs. (
Peculiarity of Anomalous Cerebral Venous Blood Flow in Multiple Sclerosis and Possible Contribution to the Inflammatory Chain
Blocked extracranial venous blood outflow causes a high rate of cerebral venous reflux in MS patients, as shown in earlier studies (Zamboni et al, 2007, 2009a, 2009b, 2009c). This detected reflux, propagated from the chest and neck veins into the parenchymal veins of the brain, may have an important function in explaining the mechanism of iron overload in MS. By contrast, venous reflux and oscillatory flow in the periventricular veins have not been found in patients affected by other neurodegenerative diseases with increased iron stores, such as Alzheimer's and Parkinson's diseases, and amyotrophic lateral sclerosis, as shown in Table 1. The VH parameters detected in these and other miscellaneous neurologic disorders closely resembled those found in healthy controls and healthy elderly subjects (Zamboni et al, 2009b, 2009c). More specifically, the rate of venous reflux flow detected in the main extracranial cervical veins of MS patients was 70% with respect to 0% detected in the three control populations. It has been shown that extracranial reflux was also transmitted up to the deep cerebral venous system in 50% of MS cases, as shown in Figure 6, but was detected neither in healthy controls nor in patients with other neurologic diseases (Table 1) (Zamboni et al, 2009b, 2009c).
Cerebral venous Doppler haemodynamic parameters of CCSVI
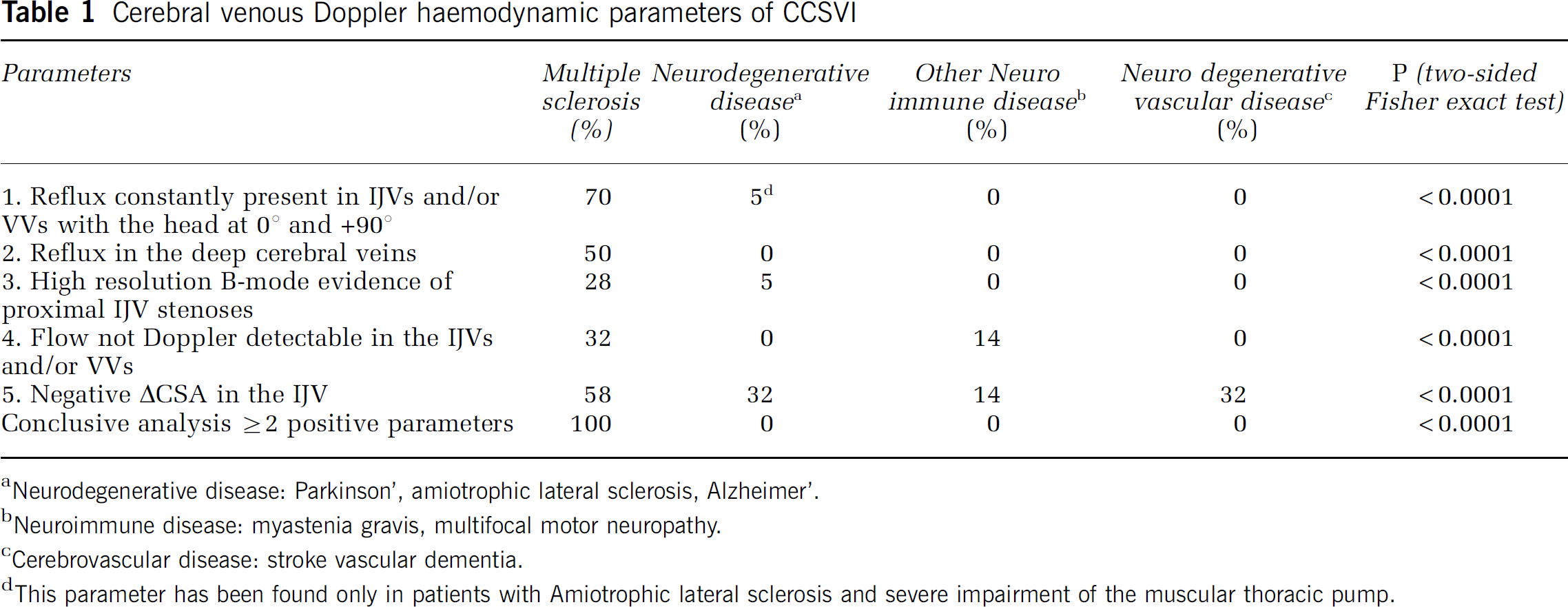
Neurodegenerative disease: Parkinson’, amiotrophic lateral sclerosis, Alzheimer’.
Neuroimmune disease: myastenia gravis, multifocal motor neuropathy.
Cerebrovascular disease: stroke vascular dementia.
This parameter has been found only in patients with Amiotrophic lateral sclerosis and severe impairment of the muscular thoracic pump.
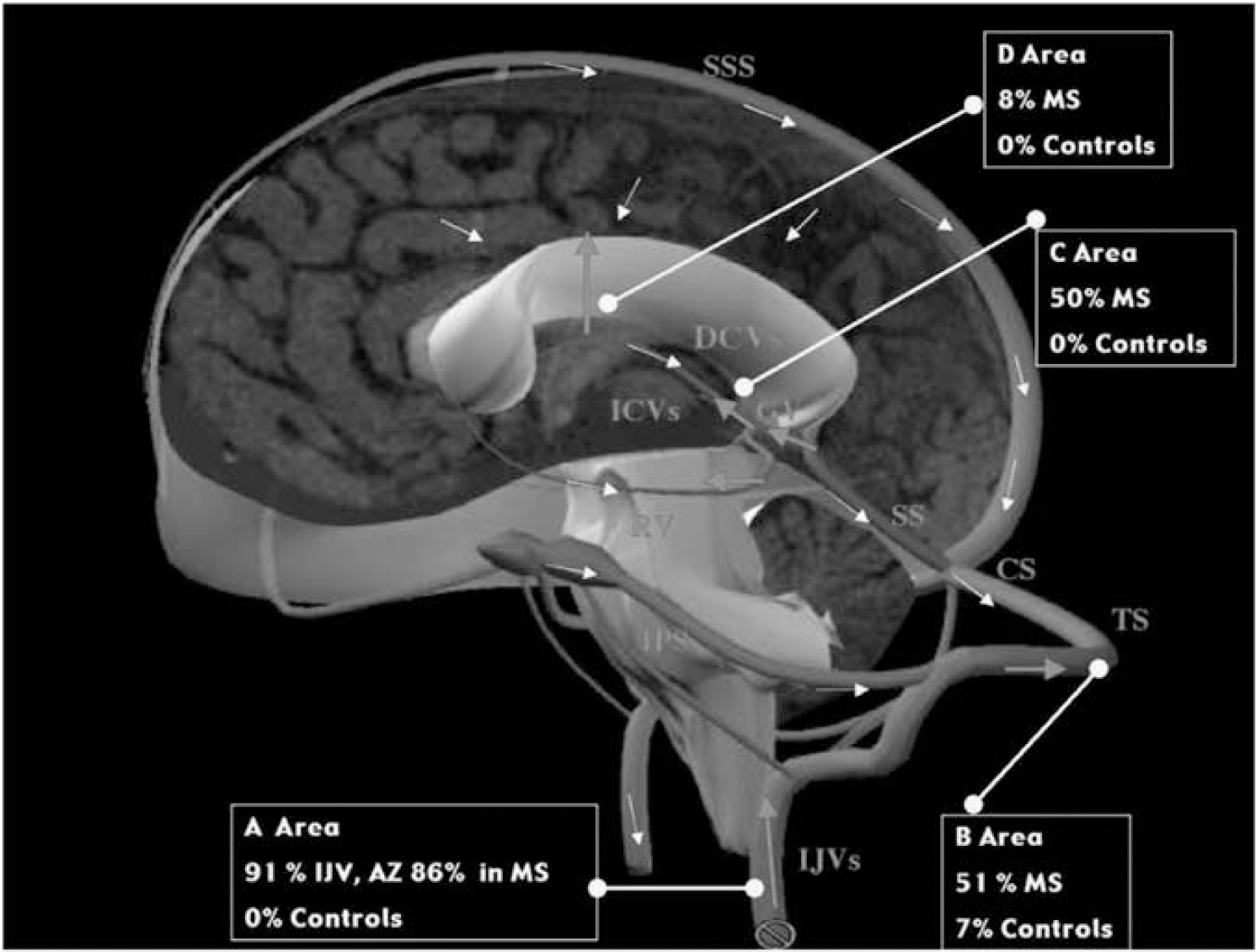
Reflux flow mapping in the cerebral venous system found in CCSVI-MS. Map of venous reflux flow in MS and in healthy controls. In the diagram, areas investigated by means of high resolutions vascular ultrasounds. Legends: Yellow arrows normal flow direction, red arrows reflux flow; SSS (superior saggital sinus), CS (confluence sinus), TS (transverse sinus), SS (straight sinus), DCVs (deep cerebral veins including Galen vein (GV), internal cerebral veins (ICVs), Rosenthal vein (RV)), IPS (inferior petrosus sinus), IJVs (internal jugular veins). A Area: reflux rate in the extracranial main pathways, IJVs and azygous vein; B Area: reflux rate in the TS; C Area: reflux rate in the DCVs; D Area: reflux rate in the veins connecting the subcortical gray matter with the DCVs. The color reproduction of this figure is available on the html full text version of the manuscript.
Altered hemodynamics have been documented by transcranial echo-color-Doppler in veins anatomically related to MS lesions. These alterations cause a high rate of reverse flow with a chaotic displacement of blood at the activation of the thoracic pump, a phenomenon never observed in controls (Zamboni et al, 2007, 2009b, 2009c). By contrast, cerebral VHs in healthy subjects are characterized by a laminar, monodirectional flow with low velocity (Valdueza et al, 1996; Stolz et al, 1999; Baumgartner et al, 1997; Zamboni et al, 2007).
It has been established that steady laminar shear stress promotes a release of factors from endothelial cells that inhibit coagulation and migration of leukocytes, while simultaneously promoting normal function in endothelial cells (Bergan et al, 2006; Sorescu et al, 2004). Conversely, where there occurs a disturbance or, especially, a reversal of flow direction, one or both factors may promote an inflammatory reaction, and particularly the expression of surface adhesion molecules (Bergan et al, 2006; Sorescu et al, 2004). Therefore, the oscillatory flow assessed in the cerebral venous system can be considered a proinflammatory stimulus, potentially contributing to MS multifactorial etiopathogenesis (Zamboni et al, 2007). The expression of adhesion molecules on the endothelial part of the blood/brain barrier facilitates macrophage and T-cell adhesion, migration, and infiltration, and is considered to be a crucial vascular factor in the development of MS (Frohman et al, 2006; Minagar et al, 2006; Geppert and Losy, 1999).
Molecular and Pathophysiologic Parallelism between Chronic Venous Disorder and Multiple Sclerosis: The Hypothetical Mechanism of Iron Deposition in Multiple Sclerosis
Chronic venous diseases and CCSVI associated with MS have many similarities in the molecular and physiologic mechanisms involved in iron-mediated disease development. The basic molecular mechanisms involved in iron overload and oxidative stress, leading to inflammation in CCSVI and CVD, follow similar routes at the molecular level (Simka and Rybak, 2008). In CVD, the chain is triggered by altered VHs with consequent disturbed microcirculation, bringing about erythrocyte extravasation as a primary source of iron stores (Bergan et al, 2006; Zamboni et al, 2006, 2008). We hypothesize that the same mechanism could also be responsible for iron deposition in CCSVI associated with MS (Zamboni, 2006; Zamboni et al, 2008). Histology confirms erythrocyte extravasation in brain plaques of MS just at the perivenular level (Adams, 1988, 1989; Zamboni, 2006). Figure 1 depicts a streak of blood encircling the venous wall in an MS plaque. The mechanisms involved in MS and CVD have many common features that exhibit intriguing similarities in the iron-mediated pathophysiology of the disease mechanisms (Zamboni, 2006). The recent description of altered VHs, together with immune cascade infiltration, systemic cellular pool extravasations, altered physiology of nephron metallobiology and classical theories explaining fibrin cuff and T-cell iron-laden macrophages indicate many similarities between MS and CVD, and are summarized in Table 2.
Molecular and physiological parallelism in CVD and CCSVI-MS
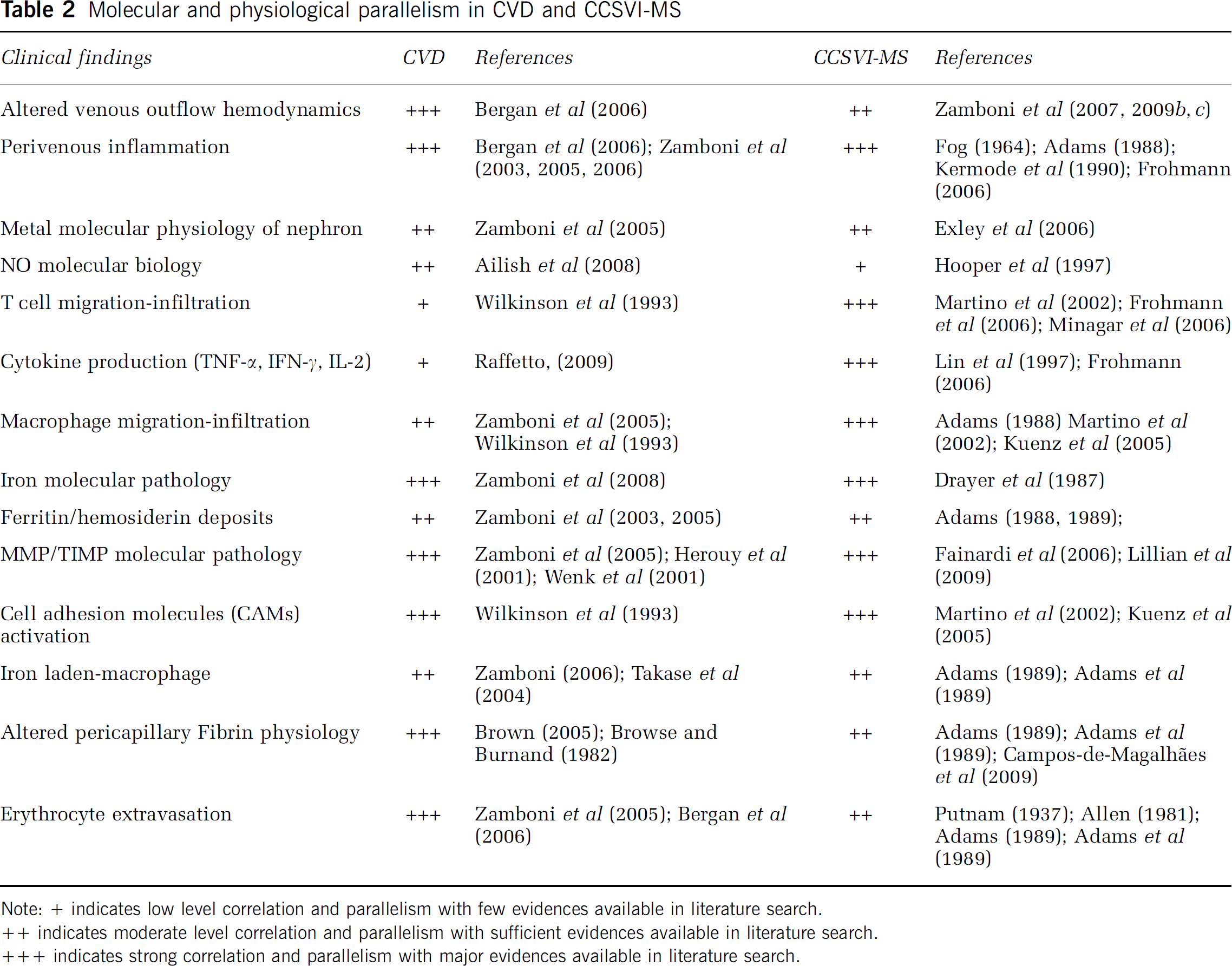
Note: + indicates low level correlation and parallelism with few evidences available in literature search.
++ indicates moderate level correlation and parallelism with sufficient evidences available in literature search.
+++ indicates strong correlation and parallelism with major evidences available in literature search.
In CVD, it is well established that the same altered VH conditions negatively affect tissue drainage, with development of chronic inflammation, iron deposition, and various degrees of tissue injury. In CVD at the microcirculatory level, erythrocyte sludge facilitates the transmigration of these cells and subsequent extravascular hemolysis, leading to increased pericapillary iron deposition (Bergan et al, 2006; Zamboni et al, 2005; Zamboni, 2006). Interestingly, several peculiar histologic aspects of insufficient venous drainage, that is, erythrocyte extravasation, iron-laden macrophages with extracellular hemosiderin deposits, and pericapillary fibrin cuffs, have also been demonstrated in CCSVI associated with MS (Adams, 1988, 1989; Zamboni et al, 2008).
Perspectives in advanced magnetic resonance imaging techniques
Recent advances in imaging techniques for iron detection may contribute substantially to understanding the role of iron in MS pathology and to developing iron-based biomarkers for disease progression (Ceccarelli et al, 2009), which might correlate better to the CCSVI condition. Recently published studies support the hypothesis that MS progresses along the venous vasculature, exactly as the reflux pathways described above (Hammond et al, 2008; Haacke et al, 2009a, 2009b). However, at present, despite the intriguing parallel in the mechanisms of iron deposition between CCSVI and CVD, we cannot definitely prove this hypothesis. Advanced MRI techniques will be probably needed for deeper insights. Absolutely promising from this point of view is the susceptibility weighted neuroimaging technique (SWI), which uses the magnetic susceptibility differences of tissues to generate a unique contrast (Haacke et al, 2005), technically different from that of spin density, T1-weighted, T2-weighted, which can detect iron deposits (Tjoia et al, 2005; Zivadinov and Bakshi, 2004; Zivadinov et al, 2008). Additionally, SWI is capable of extracting and impressively depicting the morphology of cerebral veins, possibly measuring the cerebral blood volume (Haacke et al, 2009a, 2009b). In perspective, the ability to unravel cerebral blood volume changes in MS and oxygen saturation changes is another key element that may be possible combining SWI with single vessel imaging (Haacke et al, 1997; Trapp and Stys, 2009) or susceptibility mapping (Haacke et al, 2005), all of which may help paint a more consistent picture of what is happening on the venous side in the course of CCSVI.
Moreover, recently researchers have shown that ultra-small superparamagnetic particles of iron oxide (USPIO) can visualize cellular infiltration and pluriformity of inflammation in MS more accurately than do traditional techniques.
On systemic application, USPIO is preferentially phagocytosed by monocytes before clearance within the reticuloendothelial system of the liver, spleen, and lymph nodes. On acute migration into the diseased nervous system, these iron oxide-laden macrophages become visible on MRI by the superparamagnetic effects of iron oxide resulting in a signal loss on T2-weighted and/or bright contrast on T1-weighted MRI. Iron oxide-contrast-enhanced MRI allowed in vivo visualization of cellular inflammation in patients with MS (Stoll and Bendszus, 2009). Importantly, cellular MRI provides additional information to gadolinium-DTPA-enhanced MRI as cellular infiltration and breakdown of the blood–brain barrier are not closely linked (Dousset et al, 2006).
In addition, USPIO-enhanced MRI seems to be a new marker for diffuse inflammation in MS normal-appearing white matter (Vellinga et al, 2009). Interestingly, three patterns of USPIO-enhancement have been observed, contrary to routine Gd-DTPA, showing that sensitivity and specificity of MRI in MS can be improved using USPIO. These patterns are (1) focal enhancement, (2) ring-like enhancement, and (3) return to isointensity of an earlier hypointense lesion (Vellinga et al, 2008). Both USPIO and SWI detect ring-like enhancement in MS lesions. Perhaps these two approaches could be combined, thereby contributing to a better understanding of the anatomical and functional relationships between cerebral veins and plaques in MS.
Finally, the latest and most promising technique is 4D MRI angiography, which may make it possible to measure blood flow in cerebral veins, helping to confirm and/or correlate the transcranial Doppler hemodynamic parameters described above (Eddleman et al, 2009; Spuentrup et al, 2009; Willinek et al, 2008).
Footnotes
Acknowledgements
Hilarescere Foundation supports a research program on Venous Function and Multiple Sclerosis at the Vascular Diseases Center, University of Ferrara, Italy. AVS thanks to European School of Molecular Medicine (SEMM) for supporting his own research grant. We thank Uttara Bayani, MS, for invaluable assistance at various stages of manuscript preparation. We thank Mrs Patricia Jo Ennis for her revision of the English language.
The authors declare no conflict of interest.
