Abstract
This study aimed to evaluate by functional near-infrared spectroscopy (fNIRS), the effects induced by an acute exposure (40 mins) to a GSM (Global System for Mobile Communications) signal emitted by a mobile phone (MP) on the oxygenation of the frontal cortex. Eleven healthy volunteers underwent two sessions (Real and Sham exposure) after a crossover, randomized, double-blind paradigm. The whole procedure lasted 60 mins: 10-mins baseline (Bsl), 40-mins (Exposure), and 10-mins recovery (Post-Exp). Together with frontal hemodynamics, heart rate, objective and subjective vigilance, and self-evaluation of subjective symptoms were also assessed. The fNIRS results showed a slight influence of the GSM signal on frontal cortex, with a linear increase in [HHb] as a function of time in the Real exposure condition (F4,40 = 2.67; P = 0.04). No other measure showed any GSM exposure-dependent changes. These results suggest that fNIRS is a convenient tool for safely and noninvasively investigating the cortical activation in MP exposure experimental settings. Given the short-term effects observed in this study, the results should be confirmed on a larger sample size and using a multichannel instrument that allows the investigation of a wider portion of the frontal cortex.
Introduction
Since the exponential increase in mobile phone (MP) usage all over the world, several studies focused on the possible acute (i.e., up to 1h) effects of radiofrequency electromagnetic fields (RF-EMFs) on neurophysiological indexes (Valentini et al, 2007). In recent years, most studies have shown an influence of RF-EMFs on waking electroencephalogram, particularly on alpha rhythm (Croft et al, 2002, 2008; Curcio et al, 2005), as well as on sleep electroencephalogram, in which RF-EMFs influenced the alpha-sigma range (Huber et al, 2002; Regel et al, 2007). Significant effects on cerebral excitability have also been reported, with a localized, transient, and reversible increase of responsiveness of motor cortex as a function of GSM (Global System for Mobile Communications) exposure (Ferreri et al, 2006). Moreover, improvements on attentional performance have been reported as a consequence of GSM irradiation (Koivisto et al, 2000; Curcio et al, 2004).
Furthermore, neuro-imaging studies using PET (positron emission tomography) revealed changes of cerebral blood flow after exposure to RF-EMFs equivalent to those used in MPs. Huber et al (2002) showed that a 30-min Real exposure significantly increases regional cerebral blood flow (rCBF) on the exposed dorsolateral prefrontal cortex. An enhanced regional metabolism was confirmed by a subsequent study (Huber et al, 2005), when the same radiofrequency (RF) signal was compared with a Station Radio Base signal. However, no change in rCBF as a function of the exposure to RF GSM-like signals has been found during the performance on a visual memory task (Haarala et al, 2003), with the exception of a rCBF decrease in the bilateral auditory cortex, possibly because of the noise coming from the working battery. This acoustic confounding effect was ruled out in a methodologically improved protocol (Aalto et al, 2006), in which a decrease in rCBF during the active exposure condition in the fusiform gyrus (directly under the antenna) and a bilateral increase in the medial and superior frontal gyri have been observed. Taken together, these data indicate that exposing the brain to a MP may induce an increase in rCBF in the prefrontal areas.
Functional near-infrared spectroscopy (fNIRS) is a nonharmful, noninvasive, and safe optical imaging technique, which allows the simultaneous acquisition of oxygenated and deoxygenated hemoglobin concentration changes ([O2Hb] and [HHb], respectively) from an array of optical fibers on the scalp (Wolf et al, 2007; Hoshi, 2007). On the basis of the tight coupling of neuronal activity and oxygen delivery, [O2Hb] and [HHb] are considered as indicators of cortical activation (see Hoshi, 2005; Minagawa-Kawai et al, 2008). The fNIRS provides a reliable measure of brain function, as indicated by the linear relationship between hemodynamics measured by fNIRS and neural activity (Villringer et al, 1993), as well as by the correspondence between fNIRS, functional magnetic resonance imaging, and PET measures (Kleinschmidt et al, 1996; Strangman et al, 2002; Toronov et al, 2001; Villringer et al, 1997). These considerations legitimate the use of fNIRS for investigating the effects of the exposure to the RF-EMFs emitted by MPs on cortical oxygenation.
To the best of our knowledge, so far only one study has been carried out for evaluating the oxygenation changes (measured by means of fNIRS) consequent to exposure to GSM signal (Wolf et al, 2006). In that study, because of the technical features of the RF generator, for avoiding electromagnetic interference, the subjects were exposed to the GSM signal for 15 repeated 20-secs cycles (alternating with a 2-sec on and a 2-sec off), with two different power levels: a peak power of 12 and 1.2 W/kg spatial peak specific absorption rate. During exposure to the signal, a close-to-significance short-term decrease of [O2Hb] and an increase of [HHb] were observed. Although these changes were considered much smaller than the regular physiologic ones elicited in [O2Hb] and [HHb] by functional activation of the brain, these effects were interpreted as indicating a general reduction of cerebral blood flow and cerebral blood volume. These results need further clarification, as stated by their authors (Wolf et al, 2006). Moreover, the exposure protocol was very different from the daily exposure that one can experience during a MP call and, finally, the same exposure system was significantly different from a commercial MP, reducing the ecological validity of the study.
The aim of this study was to evaluate, using fNIRS, the effects induced by an acute exposure to a GSM signal emitted by a commercially available MP (with known dosimetric characteristics) on the oxygenation of the frontal areas. To enhance the validity of this study, the protocol will mimic a normal MP daily exposure (a 40-min exposure). Thus, given the well-known responsivity of the frontal-prefrontal cortex to the RF irradiation (Huber et al, 2002; Aalto et al, 2006) and considering that the MP exposure can improve frontal functioning, as suggested by behavioral data (e.g., Koivisto et al, 2000; Curcio et al, 2004), we hypothesized that the MP exposure will induce an increased activity over the frontal areas as measured by fNIRS.
Materials and Methods
Subjects
A group of 31 university students (9 male and 22 female) responded to the recruitment advertisement and agreed to fill in a questionnaire and to undergo a clinical interview to exclude any neurologic disease or psychiatric history, medication, or drug intake. After this selection phase, 15 female healthy volunteers (mean age ± s.d.: 20.9 ± 0.94 years; range: 20 to 23) were enrolled in the study. A written informed consent was obtained before the investigation. All the participants were right-handed and regular MP users. To avoid the potential confounding effect of the cyclical ovarian hormonal impact on the cerebral hemodynamics (Goldstein et al, 2005), each subject was tested in the same menstrual phase (i.e., the first days of follicular phase). During the whole experiment, they were asked to maintain a regular sleep-wake cycle, and to avoid using their MP during 2 to 3 h before the experimental session. As the session started at ~9a.m., all the experimental subjects were free from electromagnetic influences induced by their MPs for at least 7 to 8 h. The whole investigation was approved by the local Institutional Review Board and was conducted at the Department of Health Sciences of the University of L'Aquila, according to the principles established in the Declaration of Helsinki. Owing to data loss because of technical problems, 4 subjects were discarded and all analyses have been performed on 11 subjects.
Protocol
The experiments were carried out in an electromagnetically quiet room of the Department basement. Each subject underwent two sessions (Real and Sham exposure) separated by 2 days, after a crossover, randomized, double-blind paradigm. The randomization generated by a custom software was used to define the order of the sessions (Real followed by Sham exposure or vice versa). During the experiments, the subjects and the researcher who collected the experimental data were masked to both the randomization assignments and to the type of exposure. The whole procedure lasted 60mins: after a 10-min baseline (Bsl), the subjects were exposed to the Real signal for 40mins, and then a 10-min period (Post-Exp) was recorded as recovery from the exposure. The same procedure was repeated in the Sham condition. For data analysis (see below), the period of exposure was divided into four blocks of 10mins each: Exp1, Exp2, Exp3, and Exp4. During the experiment, subjects laid, with their eyes open, on a bed in the supine position. To prevent movement artifacts, the head was restrained by a specially designed head support and the subjects were asked not to move. Heart rate (HR) was monitored by a Nellcor N-200 pulse oximeter (Nellcor Puritan Bennett Inc., CO, USA) using an adhesive finger probe. Moreover, during the experimental procedure, subjects were asked to listen to an audiobook of an Italian literature classic, to objectively control for vigilance levels; at the end of the session (both Real and Sham), participants were asked five multiple-choice questions about the story they had listened to. The number of correct answers has been considered as a control measure of vigilance during the protocol. Finally, the subjects were asked to fill in a visual analog scale of vigilance (Energy, Fatigue) and of subjective symptoms (Tension, Difficulty concentration, Tingling of skin, Dizziness, Redness of ears, Sensations of warmth on skin, Pain, Headache) both before and after each of the two experimental sessions. Each subject was required to assess ‘how do you feel right now’ with regard to the above-reported sensations, making a stroke with a pen on a 100-mm long line, between the extremes of ‘not at all’ and ‘very much’. The dependent variable was the distance from the left end of the line (‘not at all’) and the subject's mark.
Functional Near-Infrared Spectroscopy Measurement
An eight-channel fNIRS system (NIRO-200 with multifiber adapter, Hamamatsu Photonics KK, Japan) was used to investigate the effects of the MP emission on the frontal changes in [O2Hb] and [HHb]. This fNIRS system does not emit RF-EMFs, having passed the RF-EMFs compatibility test for TUV (Technischer Ãœberwachungs-Verein, a German-certifying body involved with product safety for the European community) as medical system. In addition, this fNIRS system, which uses optical fiber bundles for bringing and collecting light to /from tissues, was not sensitive to RF-EMFs, because the light detector unit and the display unit were placed ~2m away from the subject's head. Furthermore, a test performed on a solid plastic optical phantom, simulating the optical properties of human tissues, showed that the fNIRS system was not sensitive to RF-EMFs.
Three laser diodes (775, 810, and 850 nm) were used as light sources (class 1 irradiation level). Two optical fiber bundles (2.5 m length; 3 mm diameter) carried the light to the left and the right frontal cortex; whereas eight optical fiber bundles of the same size (four for each lobe) collected the light emerging from the frontal areas. The two illuminating bundles and the collecting ones were assembled into a specifically designed flexible probe holder (Elastomer LCG20R, Chiorino S.p.A, Italy) ensuring that the position of the 10 optodes, relative to each other, was fixed. The probe holder consisted of two mirror-like units (10 × 8 cm each) held together by a flexible junction. The eight fNIRS measurement points (channels) were defined as the midpoint of the corresponding detector-illuminator pairs (distance set at 3 cm). The optodes were inserted into the elastomer probe holder by fiber optical bundle socket connectors, and then placed over the forehead of both hemispheres. The probe holder was fixed to the head by a velcro brand fastener, adapting them to the individual size and shape of the different heads. This flexible probe holder and its position on the head allowed the creation of a stable optical contact with the forehead's scalp for all optodes. The channels 8, 5, 4, and 1 corresponded to Fp1, AF3, Fp2, and AF4, respectively (Figure 1), according to the extended international 10/20 system of electrode placement (Oostenveld and Praamstra, 2001). The connection between the optodes and the scalp was verified at the end of each measurement.
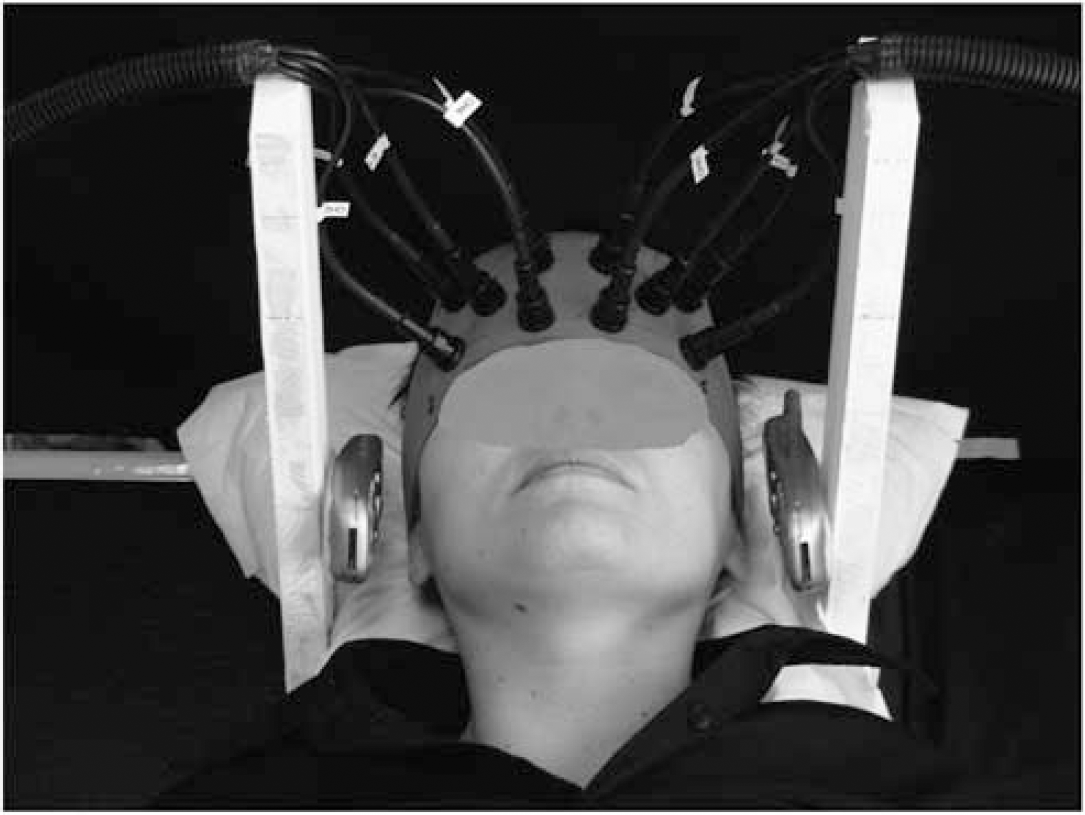
Position of the MPs during the experimental sessions. A distance of −1.5 cm was left between the ear's tragus and the RF source.
The theory of fNIRS and its applications in brain research have been reviewed recently (Wolf et al, 2007; Minagawa-Kawai et al, 2008). Briefly, changes in optical density in the near-infrared region at the different wavelengths were converted into changes in [O2Hb] and [HHb], and consequently in [tHb]. The quantification of the fNIRS parameters has been discussed in detail in earlier reports (Ferrari et al, 2004; Wolf et al, 2007). In this study, the quantification of concentration changes, expressed in Δμmol/L, was obtained by including an age-dependent constant differential path-length factor (5.13 + 0.07 x age0.81) (Duncan et al, 1996). Data were acquired at 0.5 Hz and transferred online from the NIRO-200 monitor to a personal computer. Data were first smoothed with a moving average of five data points, and then averaged over 2 mins. Finally, values of [O2Hb], [HHb], and the derived [tHb] have been averaged over 10-mins blocks (as described in the Protocol section).
Electromagnetic Field Exposure
A Motorola Timeport 260 MP was used, set by a test card for transmitting a typical basic GSM signal with a carrier frequency of 902.40 MHz (modulation components of 8.33 and 217 Hz) at its peak power of 2W (equivalent to an average power of 0.25 W). It was held by means of a wooden structure on the classic calling position of use (antenna oriented to the temporoparietal area, and microphone oriented towards the mouth) at a distance of ~1.5 cm from the left ear's tragus, for eliminating perception of any overheating produced by the phone (see Figure 1). An identical phone, but turned off and without battery, was positioned on the right side of the head to prevent the localization of the side of the GSM exposure by the subjects and to avoid a lateralization of attention to one of the two sides. Following the standards of compliance, the electromagnetic field (EMF) exposure was earlier measured using an anthropomorphic head-shaped homogeneous phantom, and with the phone switched on at its peak power, a maximum value of specific absorption rate equal to 0.5 W/kg was measured (for further technical details see Curcio et al, 2008).
Data Analysis
The amplitudes of [O2Hb] and [HHb] changes in the frontal areas were calculated by subtracting the Baseline period from the Exp1, Exp2, Exp3, and Exp4, and Post-Exp periods. Statistical analyses have been carried out on these differential values. Each dependent variable underwent a three-way model of Analysis of Variance (ANOVA), Condition (Real, Sham) x Time (Exp1, Exp2, Exp3, Exp4, Post-Exp) x Channel (1, 2,…, 8).
Similarly, the HR changes were also calculated and analyzed by subtracting the Baseline period from the Exp1, Exp2, Exp3, and Exp4, and Post-Exp periods, and statistical analyses have been carried out on these differential values. As a consequence, a two-way ANOVA Condition (Real, Sham) x Time (Exp1, Exp2, Exp3, Exp4, Post-Exp) has been carried out. Moreover, we compared the number of correct answers with questions regarding the audiobook story given by the subjects after the two Conditions (Real, Sham) by means of the paired Student's t-test. Finally, self-evaluations of vigilance and subjective symptoms were submitted to a two-way ANOVA Condition (Real, Sham) x Moment (Pre, Post).
Statistical analyses were carried out using the Statistica software version 6.1 (StatSoft, Tulsa, OK, USA). The alpha level was set at P < 0.05.
Results
Functional Near-Infrared Spectroscopy Measurements
Figure 2 shows a single individual subject as a representative of a typical frontal fNIRS response during MP exposure. A linear decrease of [O2Hb] and [tHb] was observed in all measurement points as a function of time, which persisted even after the end of the MP exposure. However, [HHb] showed a nonuniform increase over the eight measurement points.
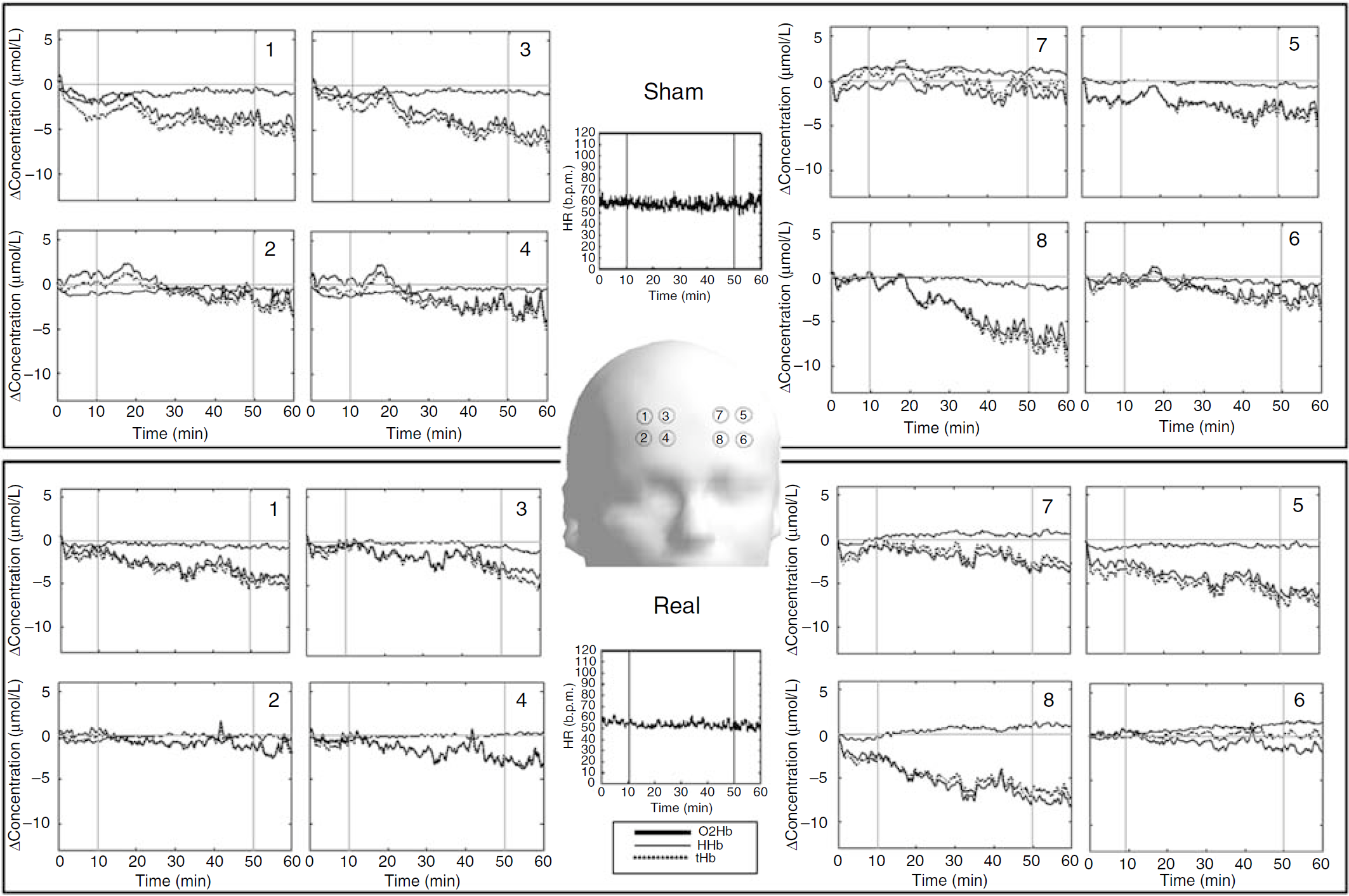
Single subject topographic picture of a representative time course of [HHb], [O2Hb], and [tHb] concentration changes over the eight measurement points on the right (panels 1 to 4) and left (panels 5 to 8) frontal cortex. The arrangement of data resembles the measurement positions. The upper block, including the heart rate (HR) time course, refers to the Sham condition, whereas the lower block refers to the Real exposure condition. Dotted vertical lines indicate exposure switch-on (tenth min) and switch off (fiftieth min). The channels 8, 5, 4, and 1 correspond to Fp1, AF3, Fp2, and AF4, respectively, according to the international 10/20 system of electrode placement. b.p.m, beats per min.
The ANOVA carried out on [O2Hb] showed significant main effects for Time (F4,40 = 16.30; P = 0.00001) and Channel (F7,70 = 2.72; P = 0.01), whereas the factor Condition was not significant (F1,10 = 0.46; P = 0.51; 95% confidence interval, 95% CI: −1.43 to 2.25). Moreover, a significant Time x Channel interaction (F28,280 = 2.11; P = 0.001), indicates a linear decrease of [O2Hb] in all measurement points with the exception of channels 3 and 7, which remained at the baseline level between Exp3 and Post-Exp. No main effect or interaction was significant (Figure 3).
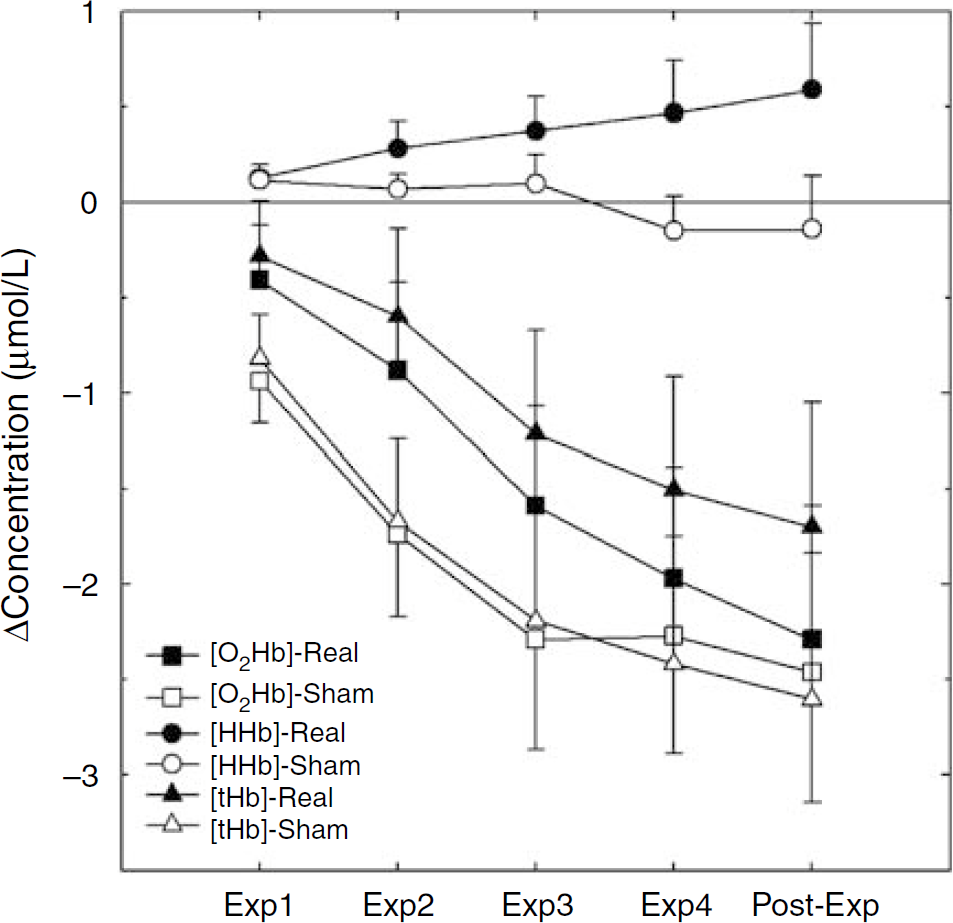
Time course of the [HHb], [O2Hb], and [tHb] changes during the Real and Sham exposure of the whole experimental group. The first four data points (Exp1, Exp2, Exp3, and Exp4) represent the grand average of the four 10-min blocks of exposure subtracted by the 10-min baseline period. The last data point (Post-Exp) represents the grand average of the 10-mins recovery after exposure. During the first 10-mins block of exposure to the Real signal (Exp1), [HHb] remained at the baseline level. The linear increasing trend, indeed, started during the second 10-mins block (Exp2) and did not cease when the field was switched off, persisting also during the last recovery block (Post-Exp). This significant time-dependent increase in [HHb] was not present under the Sham exposure.
The ANOVA on [HHb] showed a main effect for Channel (F7,70 = 2.85; P = 0.01), and an interaction Time x Channel (F28,280 = 2.27; P = 0.0004). Also in this case, the interaction points to a linear decrease of [HHb] in all the scalp channels, except in channels 3 and 7, which instead showed an increase of [HHb]. The crucial Condition x Time interaction was significant (F4,40 = 2.67; P = 0.04), because of a linear increase in [HHb] as a function of time in the Real condition (Figure 3). However, the post hoc comparisons between the Sham and Real values at different time points did not reach statistical significance. The Condition x Channel interaction was not significant, indicating that the exposure of the EMFs did not differently affect the two hemispheres. No other main effect or interaction was significant; in particular, no statistically relevant differences were observed for the factor Condition (F1,10 = 2.78; P = 0.12; 95% CI: 0.10 to 1.34).
The ANOVA on [tHb] again showed significant main effects for Time (F4,40 = 18.84; P = 0.00001) and Channel (F7,70 = 3.20; P = 0.005), whereas no significant effect was observed for the factor Condition (F1,10 = 1.12; P = 0.31; 95% CI: −0.87 to 3.13). Moreover, a statistically significant interaction Time x Channel (F28,280 = 2.54; P = 0.00006) (Figure 3), is explained by the linear decrease of [tHb] as a function of time, in all the scalp channels, except in channels 3 and 7, in which an increase of [HHb] was recorded from the second 10-mins block of exposure until the post-exposure block. No other interaction was significant.
Heart Rate
The ANOVA on HR indicated solely a significant main effect for Time (F4,36 = 4.52; P = 0.004), with a trend to linearly decrease as a function of time, regardless of the exposure condition (Bs1: 72.3 ± 2.6, Exp1:71.3 ± 2.7, Exp2: 70.6 ± 2.4, Exp3: 70.3 ± 2.7, Exp4: 69.9 ± 2.3, Post-Exp: 68.5 ± 2.2).
Control Analysis of Performance During Story Listening
The Real and Sham conditions were characterized by a comparable level of vigilance/attention, because responses to the questions related to the audiobook listening did not show any significant difference (t=−0.38; P > 0.35).
Self-Evaluations
The two-way ANOVA on subjective Energy showed a significant main effect for Moment (F1,10 = 6.73; P = 0.03), indicating a decrease at the end of the sessions.
A complementary main effect for Moment (F1,10 = 9.54; P = 0.01) was observed on Fatigue, which significantly increased at the end of both the sessions.
The significant main effects for Moment have also been observed for Tingling of skin (F1,10 = 7.40; P = 0.02), Dizziness (F1,10 = 13.41; P = 0.004), Sensations of warmth on skin (F1,10 = 10.97; P = 0.008), and Headache (F1,10 = 16.28; P = 0.002), all pointing to an increase of these negative feelings at the end of the sessions.
Moreover, the sensation of Headache also showed a significant main effect for Condition (F1,10 = 5.46; P = 0.04), being greater during the Sham condition with regard to the Real one.
Discussion
To the best of our knowledge, for the first time here, we used fNIRS to investigate the effects of a long MP exposure on frontal oxygenation. We showed that a 40-min continuous exposure to a typical GSM signal emitted by a MP has a significant influence on the cortical [HHb] of the frontal areas. More specifically, [HHb] was increased by the exposure to the GSM signal, and this effect became more evident as a function of the time elapsed under MP exposure. No other measures ([O2Hb], [tHb], HR, objective and subjective vigilance, or self-evaluation of subjective symptoms) showed any GSM exposure-dependent change.
During the first 10-mins block of exposure to the Real signal (Exp1), [HHb] remained at the baseline level (see Figure 3). The linear increasing trend, indeed, started during the second 10-mins block (Exp2) and did not cease when the EMF was switched off, persisting also during the recovery block (Post-Exp). This time-dependent increase in [HHb] was not present under the Sham exposure. Similar results have been recently reported by Wolf et al (2006). Although different interoptode distances and exposure intensities were used in that study, a significant increase in [HHb] paralleled by a more conspicuous decrease in [O2Hb] were found.
These findings support the notion that an acute 40-mins exposure to a GSM signal emitted by a commercially available MP affects the frontal hemodynamics, as indexed by fNIRS. This result extends the PET data showing a metabolic increase over the anterior regions (Aalto et al, 2006; Huber et al, 2002, 2005), and provides a physiologic substratum to the attentional improvements observed under GSM irradiation (e.g., Koivisto et al, 2000; Curcio et al, 2004).
Increases in [HHb] are usually considered as an index of increased oxygen consumption (Wolf et al, 2007). However, the origin of change in [HHb] concentration remains unclear. [HHb], in fact, varies in a complex way with changes in both venous oxygenation (i.e., oxygen consumption) and blood volume (i.e., oxygen supply) (Hoshi et al, 2001; Seiyama et al, 2004). Thus, if [HHb] variations reflect the change in oxygen consumption, it will be considered as a measure of metabolic deactivation. Conversely, if changes in [HHb] concentration mainly reflect variations in oxygen supply, it will be positively correlated with local activation. According to this second hypothesis, the observed increase in [HHb] as a function of MP exposure may be interpreted as a local activation, coherent with the PET studies (Aalto et al, 2006; Huber et al, 2002, 2005).
Another relevant issue relates to the localization of the observed short-term effects induced by MP exposure. Two questions could arise: is it possible to observe the MP-induced effects on brain areas not exposed directly? Furthermore, can these changes in the anterior areas be related to cognitive/behavioral functioning? As far as the first question is concerned, a ‘remote’ effect on regions far from those directly exposed (i.e., the temporal lobe) seems possible. It has been recently shown that even if the temporal lobe ipsilateral to the active MP absorbs at least half of the whole RF energy absorbed in the brain, the frontal one can absorb up to 20% of the total power of the RF-EMFs (Cardis et al, 2008). In this study, the observed increase in [HHb] was not side-specific; even if the exposure side was the left one, the increase in [HHb] was observed bilaterally, over the whole frontal area. Similarly, neuro-imaging studies (Aalto et al, 2006; Huber et al, 2002) reported bilateral changes in cerebral metabolism over the anterior areas during EMF exposure. Regarding the possible relationship between local hemodynamic changes and cognitive/behavioral functioning, these findings indirectly support the hypothesis that attentional performance improvements earlier observed under GSM irradiation (e.g., Koivisto et al, 2000; Curcio et al, 2004) may be associated with the increased activation of the frontal and/or prefrontal areas.
At variance with Wolf et al (2006), the magnitude of [O2Hb] and [HHb] changes observed in this study over the anterior areas seems comparable with that of the other studies of functional activation (Hoshi, 2007), although studies on both fNIRS and EMF reported a high intersubject variability. The lack of statistically significant effects of GSM signal on frontal [tHb] could be attributed to the small contribution of [HHb] changes not accompanied by concomitant statistically relevant [O2Hb] changes. The [HHb] increase could be because of a slight venodilation in vessels with a high oxygen extraction ratio.
The mechanisms underlying the subtle cognitive and neurophysiological effects of MP exposure still remain unclear. Thermal mechanisms are seen as mostly possible, because RF-energy deposition in the tissues would be expected to affect heat balance and may invoke physiologic compensation (Lin and Bernardi, 2007). In this study, the increase in [HHb] could be interpreted as an effect of heat deposition into the brain with a consequent dissipation across cortical areas. This hypothesis has recently been proposed especially for the highest frequencies, in which dielectric and resistive heating could increase the local temperature and increase the entropy of the exposed biologic system (Sheppard et al, 2008). However, a thermal mechanism should be rejected taking into account that: (1) the distance between the antenna and the head drastically reduces the absorbed power (Schönborn et al, 1999); (2) an increase of skin temperature would be easily dissipated through the blood flow (Van Leeuwen et al, 1999); and (3) near-infrared light penetrates through the brain at least 1.5 cm in-depth, in which temperature changes are far below the danger level (Wang and Fujiwara, 1999; Bernardi et al, 2000).
Thus, several nonthermal mechanisms could be involved, such as: (1) proteins conformational variations resulting in proteins functional changes; (2) modifications in the binding of ligands, such as Ca++ to cell receptors, also resulting in a changed receptor function; (3) absorption of RF energy by the vibrational states of biologic components, such as microtubules; (4) enhanced attraction amongst cells (the pearl-chain effect); and (5) demodulation of a modulated RF signal, producing extremely low-frequency electric fields (for a review, see Challis, 2005). Given the short-term effects observed in this study as a consequence of an acute exposure lasting 40mins, conclusions about long-term health consequences of the RF-EMF exposure are premature and need more-in-depth epidemiologic investigations, particularly regarding long-term and repeated exposures over several years.
There are some limitations to the interpretation of our fNIRS findings. Sample size was relatively small and data were characterized by a high interindividual variability. Such variability also depended on the long duration of the whole protocol, as confirmed by the increasing variability across the different time points (see Figure 3). Moreover, taking into consideration the 95% CI of the three dependent variables, only [HHb] data suggest that the observed effect on this variable is independent of the small sample size and reflects a physiologically relevant phenomenon. Although the [tHb] difference between the Real and Sham exposure could represent a potentially relevant physiologic effect, a statistical significance was not found because of a large interindividual variability (much larger than that for [HHb]). Assuming the same means and s.d., such difference would be detected as statistically significant with a larger sample size (at least 31 subjects).
As far as the other measures evaluated in this study are concerned, HR tended to linearly decrease as a function of time, regardless of the exposure condition, probably because of the prolonged supine resting condition. In addition, both objective and subjective levels of vigilance, as measured, respectively by the number of correct responses to the questions about the audiobook story and by the vigilance self-evaluations, were similar under both Real and Sham exposure. The lack of vigilance differences in the two exposure conditions allows us to exclude methodological biases, when assessing the frontal oxygenation measures. In other words, the frontal cortex [HHb] differences between the Real and Sham exposure revealed by fNIRS most likely cannot be accounted for by different levels of vigilance/attention during the two conditions. Finally, self-evaluations of the subjective symptoms (Energy, Fatigue, Tingling of skin, Dizziness, Sensations of warmth on skin, Headache) changed just as a function of elapsed time in the direction of a general worsening of the subjective state. This simple effect of Fatigue, because of the length of the protocol, was not dependent on the exposure condition, confirming that, in healthy subjects, the exposure to a GSM signal does not influence subjective symptoms and self-evaluated vigilance (Cinel et al, 2008).
Conclusions
This study assessed the effects induced by a normal daily exposure (duration: 40mins) to a GSM MP on measures of cortical blood oxygenation noninvasively and safely evaluated by means of fNIRS. Data showed a significant bilateral increase in [HHb] over the frontal areas, indicating a local enhancement of oxygen consumption. Owing to the magnitude of the [HHb] changes and to the high variability of individual data, future fNIRS studies should consider larger sample size, more fNIRS measurement points, and shorter protocols.
Footnotes
Acknowledgements
The authors thank Motorola GmbH, Corporate Technology, Niederkassel-Mondorf (Germany) for supplying the equipment. SAR and field measurements were performed by the Bioelectromagnetic Group at the Department of Electronic Engineering (‘Sapienza’ University of Rome). The authors also thank Patrizio Pasqualetti (AFaR-Associazione Fatebenefratelli per la Ricerca) for his statistical support.
The authors state no conflict of interest.
