Abstract
The near-infrared spectroscopy (NIRS)-derived cerebral oximetry index (COx) has become popularized for non-invasive neuromonitoring of cerebrovascular function in post-cardiac arrest patients with hypoxic-ischemic brain injury (HIBI). We provide commentary on the physiologic underpinnings and assumptions of NIRS and the COx, potential confounds in the context of HIBI, and the implications for the assessment of cerebral autoregulation.
Keywords
Commentary
Near-infrared spectroscopy (NIRS) is utilized in the intensive care and intra-operative settings to continuously and non-invasively monitor regional cerebral oxygen saturation (rSO2). Recently, NIRS has been applied to the assessment of cerebral autoregulation in neurocritically ill patients, including post-cardiac arrest patients with hypoxic-ischemic brain injury (HIBI). 1 With continuous measurements of rSO2 and mean arterial pressure (MAP), autoregulation is ostensibly estimated from the moving Pearson correlation coefficient between rSO2 and MAP – termed the cerebral oximetry index (COx). A COx value <0.3 is considered to reflect intact autoregulation whereas a value ≥0.3 denotes impaired autoregulation.
Recently, Tachino et al. investigated the association between dysfunctional autoregulation (determined by COx) in HIBI patients and all-cause in-hospital mortality. 2 In agreement with prior work,3,4 they concluded that survival was lower in patients with more time spent with impaired autoregulation and monitoring autoregulation may be useful in the early management of HIBI patients to predict outcome. In contrast, prior work has shown that COx does not differ between patients with good and poor outcome 5 and poorly agrees with other autoregulation indices (e.g., pressure reactivity index; PRx).1,6 Therefore, our commentary aims to place the results of Tachino and colleagues into the broader context of monitoring autoregulation as it pertains to 1) physiologic principles of NIRS, and 2) NIRS for monitoring cerebral autoregulation.
Physiologic principles of cerebral oximetry
Several questions regarding the utility of NIRS as a surrogate measure to characterize physiology require consideration. First, what is the variable/function we truly aim to measure? In the study by Tachino et al., and for a broader neuromonitoring context, the true variable of interest is typically cerebral blood flow (CBF). However, NIRS approximates oxygen saturation by estimating mixed arteriovenous hemoglobin saturation within the cerebral vasculature based on the differing absorptions of oxygenated and deoxygenated hemoglobin. Of note, assorted chromophores (e.g., melanin) contribute to the optical properties of the tissue, which impact NIRS measurement beyond just hemoglobin. A second question then becomes, what governs cerebrovascular hemoglobin saturation? Per the Fick equation, cerebrovascular oxygen saturation is determined by cerebral metabolism (CMRO2), CBF, and arterial oxygen saturation (or content).
From this relationship, SvO2 and therefore rSO2 are proportional to CBF by virtue of a higher CBF reducing the oxygen extraction fraction (O2EF). Conversely, SvO2 is inversely proportional to CMRO2, where a greater metabolic demand would increase O2EF and reduce SvO2. Further, rSO2 is influenced by the ratio of arteriole-to-venule blood volume, which is assumed as static when using NIRS. 7 If CMRO2 and the arteriole-to-venule blood volume ratio are constant, rSO2 would be expected to be proportional to SvO2. However, CMRO2 is reduced following cerebral ischemia 7 and the arteriole-to-venule blood volume ratio is not static during alterations in CBF. 8 These physiologic limitations raise the question of, does rSO2 truly reflect CBF? Current evidence suggests that rSO2 does not accurately reflect changes in CBF in healthy humans. 1
Figure 1 highlights the difficulties of interpreting rSO2 values. Under normal physiological conditions, points A and C have identical CMRO2 despite being achieved by different means. This, however, is not reflected in rSO2 where it is lower at point A. Conversely, concurrent reductions in CBF and O2EF (point B) have an equivalent rSO2 with normal physiology (point A). In HIBI this discrepancy could result from increases in cerebrovascular resistance, impaired diffusion of oxygen from the cerebral vasculature into brain tissue or a pathophysiologic depression in CMRO2 following cerebral ischemia, 7 whereby rSO2 would appear normal and be invalid as a CBF surrogate. Therefore, in certain contexts rSO2 is unable to differentiate between normal physiology and pathophysiology. This may in part explain the recurrent finding that rSO2 is not different between favorable and unfavorable outcomes in HIBI.2 –5
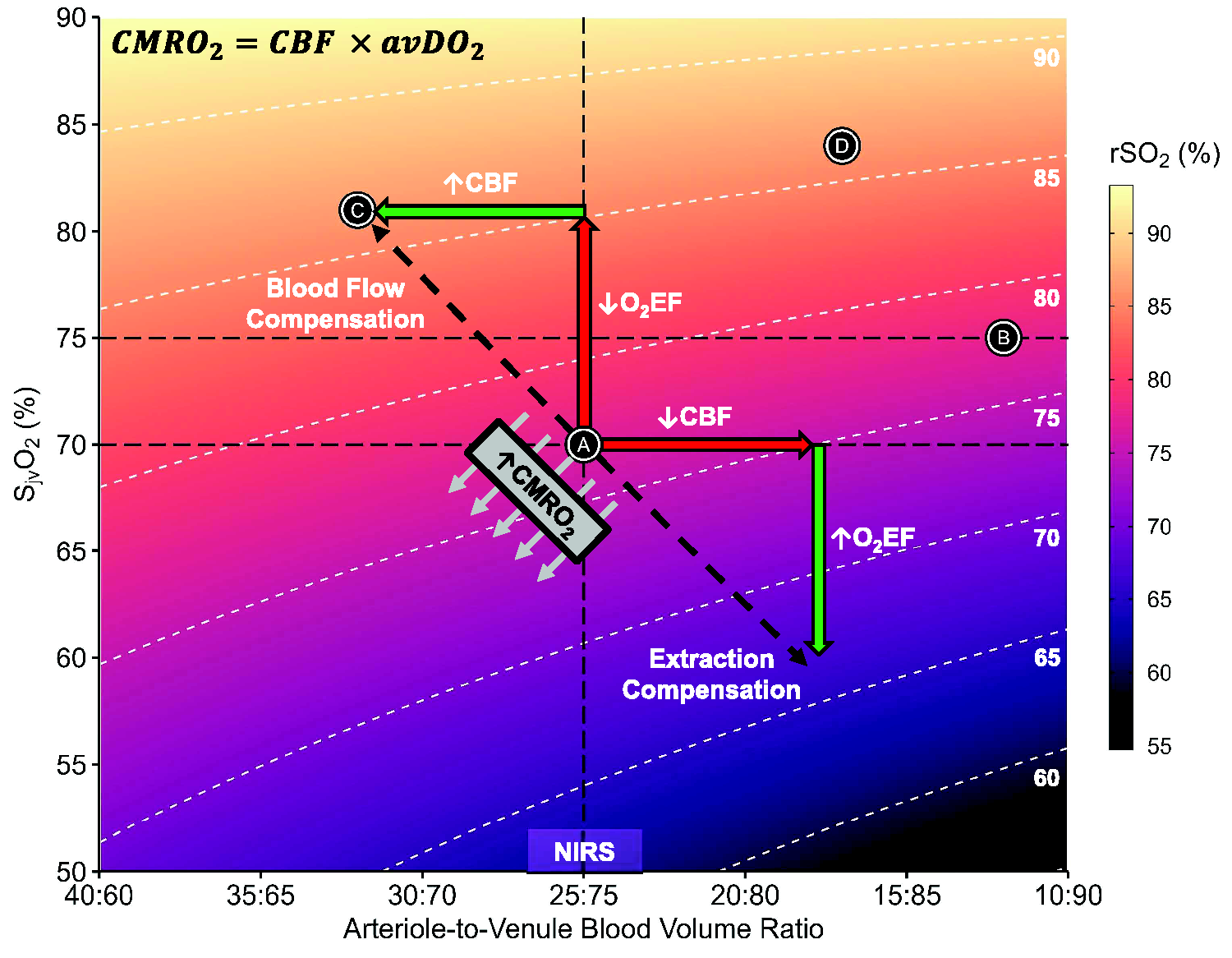
The physiologic underpinnings of cerebral oximetry. This figure depicts the mathematical relationship between SjvO2 (y-axis), the arteriole-to-venule blood volume ratio (x-axis), and near-infrared spectroscopy derived measures of rSO2 (color gradient scale) at a fixed SaO2 of 98%. rSO2 values were computed based on the formula:
NIRS for monitoring cerebral autoregulation
Given the discordance between rSO2 and CBF, the next question that arises is, what are the implications for COx as a metric of autoregulation, particularly in HIBI? First, normal jugular SvO2 is ∼70–75% in healthy subjects 7 but elevated in a subset of HIBI patients as demonstrated by Sekhon et al., 9 and Richter et al., 10 suggesting reduced O2EF. Under these conditions, the arterial-jugular SO2 difference may converge irrespective of CBF (Figure 1), with CBF and oxygen utilization uncoupled. 7 Indeed, raw rSO2 traces show minimal response to changes in MAP in some cases. 1 As such, COx values approaching zero can suggest either (a) functional autoregulation or (b) impaired cerebral O2EF/O2 utilization. In this context COx cannot distinguish between functional and impaired autoregulation in HIBI.
Specific to autoregulation metrics, COx lacks agreement and provides conflicting classifications of whether autoregulation is intact compared with other autoregulation indices.1,6 Further, readily available moving correlation coefficients have encouraged the simplistic notion of autoregulation being dichotomously present or absent. Such classifications are susceptible to mathematical errors that can arise from impaired O2EF 7 or truncated data. For example, raw MAP signals can have minor fluctuations over time, leading to classification of autoregulation based on minimal change (e.g., <2 mmHg). Truncated data confuses steady-state physiology and general signal variability and could explain observations of autoregulation repeatedly appearing then disappearing. 2
Technological advancements have provided many exciting opportunities to interrogate patient physiology. However, as we strive to shine a light on cerebral autoregulatory function in HIBI patients, indices that are built on variable interactions, physiological assumptions, and indirect measures require scrutiny pertaining to edge cases (pathophysiology), limitations (COx not being comparable to other autoregulation indices), and the complexities (mathematical, physiological) that underpin such metrics.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: J.D.B. was funded by a Canadian Institute of Health Research Doctoral Award (#187576). M.S.S. was funded through a Michael Smith Health Research BC Health Professional Award. R.L.H. was funded through the Michael Smith Health Research BC Scholar Program (grant no. SCH-2023-3196).
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J.D.B. is an R&D Consultant for Kent Imaging Inc. D.B.M, D.E.G., M.S.S. and R.L.H. report no conflicts of interest, financial or otherwise.