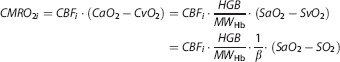Abstract
Pathophysiologic mechanisms involved in neonatal hypoxic ischemic encephalopathy (HIE) are associated with complex changes of blood flow and metabolism. Therapeutic hypothermia (TH) is effective in reducing the extent of brain injury, but it remains uncertain how TH affects cerebral blood flow (CBF) and metabolism. Ten neonates undergoing TH for HIE and seventeen healthy controls were recruited from the NICU and the well baby nursery, respectively. A combination of frequency domain near infrared spectroscopy (FDNIRS) and diffuse correlation spectroscopy (DCS) systems was used to non-invasively measure cerebral hemodynamic and metabolic variables at the bedside. Results showed that cerebral oxygen metabolism (CMRO 2i ) and CBF indices (CBF i ) in neonates with HIE during TH were significantly lower than post-TH and age-matched control values. Also, cerebral blood volume (CBV) and hemoglobin oxygen saturation (SO 2 ) were significantly higher in neonates with HIE during TH compared with age-matched control neonates. Post-TH CBV was significantly decreased compared with values during TH whereas SO 2 remained unchanged after the therapy. Thus, FDNIRS–DCS can provide information complimentary to SO 2 and can assess individual cerebral metabolic responses to TH. Combined FDNIRS–DCS parameters improve the understanding of the underlying physiology and have the potential to serve as bedside biomarkers of treatment response and optimization.
Keywords
INTRODUCTION
Neonatal hypoxic ischemic encephalopathy (HIE) occurs in ∼2 to 3 per 1,000 live births in the USA, with devastating consequences including neurodevelopmental disabilities such as cerebral palsy, life-long cognitive impairment, and epilepsy. 1 In HIE, there is a period of reduced blood flow (ischemia) and oxygen delivery (hypoxia) followed by reperfusion with transient energy recovery, and then a secondary energy failure. 2 Treatment is aimed at preventing the cascade of events that begin with reperfusion and lead to secondary energy failure and inevitable cell death; this is the ‘window of opportunity.’ 3 Theories for injury mechanisms in this window suggest a central role for excitotoxicity and oxidative stress with the injury evolving over hours to weeks. 2 Therapeutic hypothermia (TH) is the treatment of choice for HIE, as it is the only treatment with proven efficacy and acts through a variety of mechanisms, including those associated with decreased neuronal energy metabolism. 4 Although early studies showed that TH decreased rates of both mortality and disability, recently published long-term outcomes have demonstrated significance only for decreased mortality rates. 5 In addition, a recent meta-analysis advocated refinement of patient selection and TH duration whereas others have suggested that currently excluded neonates be considered in future trials.6, 7 Thus a need for further optimization of TH remains.
Magnetic resonance imaging (MRI) is the gold standard for detecting acute brain injury, but is impractical as an early screening tool or as a tool to optimize therapy of HIE due to its non-portability and high cost. Electroencephalography and amplitude-integrated electroencephalography can provide essential measures of neuronal activity but would benefit by additional direct measures of cerebral hemodynamics and cerebral metabolic rate of oxygen consumption (CMRO 2 ) changes that precede or accompany secondary energy failure. Commercial continuous wave near infrared spectroscopy systems have been used in HIE to monitor relative cerebral hemoglobin oxygen saturation (rSO 2 ) and fractional tissue oxygen extraction (FTOE=(SaO 2 ) – rSO 2 )/SaO 2 ) where SaO 2 is the arterial oxygen saturation. 8 However, neither rSO 2 nor fractional tissue oxygen extraction accurately reflect CMRO 2 , as CMRO 2 also depends on oxygen delivery (via CBF).
In contrast, frequency domain NIRS (FDNIRS) allows the absolute quantification of hemoglobin oxygen saturation (SO 2 ) and cerebral blood volume (CBV). 9 Frequency domain near infrared spectroscopy has been used to demonstrate that CBV is significantly elevated in acute HIE brain injury, whereas SO 2 shows no significant difference from neonates without suspicion of brain injury. 10
Diffuse correlation spectroscopy (DCS) was developed for in vivo applications to measure microvascular blood flow non-invasively in deep biologic tissue without the injection of external contrasts. In previous work, we used DCS to measure an index of cerebral blood flow (CBF i ) in the preterm brain and combined this measurement with FDNIRS measures of SO 2 to provide an index of CMRO 2 (CMRO 2i ). 11 More recently, we described the relationship between CMRO 2i and gestational age in the developing premature brain by using concurrent FDNIRS and DCS measurements. 12 FDNIRS–DCS CMRO 2i , unlike SO 2 , is sensitive to early brain development, increasing with gestational age and throughout the first year of life 12 and might facilitate early detection of abnormalities and delays in maturation of specific brain regions. 13 Using our FDNIRS–DCS bedside instrumentation, the aim of this current study was to determine cerebral hemodynamics and oxygen metabolism in neonatal HIE during TH and compare the parameters to values measured after TH and measured in age-matched control.
MATERIALS AND METHODS
Patients
Twelve term neonates diagnosed with moderate to severe HIE were enrolled in a prospective observational study at Boston Children's Hospital from July 2009 to January 2012 with institutional review board approval from Partners Healthcare, which includes both Massachusetts General Hospital and Brigham and Women's Hospital and an inter-institutional agreement granted by Boston Children's Hospital. Each neonate that underwent TH met inclusion criteria based on published studies: 14 (1) gestational age36 weeks and birth weight2 kg, (2) evidence of fetal distress including history of acute perinatal event, and (3) evidence of neonatal distress as evidenced by at least one of the following: apgar score5 at 10 minutes, pH7.0 within 1 hour of birth or base deficit16 mEq/L or need for ventilation for at least 10 minutes after birth, (4) evidence of neonatal encephalopathy by physical exam from the neurologist, and (5) abnormal cerebral function monitor evidenced for example by the presence of seizures identified by amplitude-integrated electroencephalography. Two of the twelve neonates recruited were excluded from analysis due to a change in diagnosis from HIE to a rare metabolic disorder with encephalopathy and myopathy in one neonate and inability to obtain FDNIRS–DCS frontal measurements in the other. All neonates had light skin.
Twenty-two control neonates were recruited from the well baby nursery at Brigham and Women's Hospital between 2009 and 2011 under the same institutional review board. No neonate had signs or symptoms of fetal distress and only routine resuscitation was required after delivery. To further homogenize the healthy population and make it more comparable to the HIE population, five neonates with darker skin color were excluded from the analysis resulting in a total of 17 control neonates. Demographics are detailed in Supplementary Table 1.
Neurodevelopmental Outcomes
Gross motor function was assessed using either the gross motor function classification system for cerebral palsy (GMFCS) score 15 or the Bayley psychomotor developmental index score whereas cognitive function was assessed using the Bayley mental development index (MDI) score. 16 Motor and cognitive scores were then combined yielding a single score that was classified into three categories: normal (Bayley MDI or PDI85 and GMFCS=1), mildly abnormal (70Bayley MDI or PDI<85 or GMFCS=2) or abnormal (Bayley MDI or PDI<70 or GMFCS>2, or death). Neurodevelopmental outcomes were assessed in eight infants at 18 months (median), patient #7 was lost to follow-up after NICU discharge and patient #10 died a few days after birth. Thus the outcome score was normal in five infants (5/9, 55.6%), mildly abnormal in three infants (3/9, 33.3%), and the newborn who died was scored abnormal (1/9, 11.1%). Details are provided in Supplementary Table 1.
Therapeutic Hypothermia
Standard TH procedures 17 were initiated within 6 hours after birth by passive cooling at the referral hospital or on transport, and by active whole-body cooling upon admission to Boston Children's Hospital. Body temperature was targeted to 33.5°C for 72 hours of TH. Rewarming procedures were then applied for 12 hours, raising the temperature by 0.5°C every 2 hours. For all measurements acquired within 90 hours after birth, whole-body temperature was<36°C. Both the cooling and rewarming periods are grouped together as a single ‘TH’ period as the patient is technically considered hypothermic (temperature<36°C). Post-TH measurements were all acquired at normothermia. Medications used during TH period are listed in Supplementary Table 2 and include sedatives, analgesics, and neurotransmitters.
Optical Measurements
Non-invasive FDNIRS–DCS measurements were performed at the bedside during TH (6–78 hours), the rewarming period (78–90 hours), and post-TH (106–305 hours) in neonates with HIE and after birth in healthy neonates (13–77 hours). The hybrid instrumentation has been described previously in literature. 11 The power emitted by FDNIRS–DCS was well below the American National Standards Institute light levels and the combined instrumentation was compact enough to be moved to the neonate's bedside on a small cart.
The probe was designed such that the light would penetrate into the neonatal cerebral cortex and subcortical white matter 9 while minimizing the error due to extra-cerebral contamination. 18 Measurements were obtained from up to three locations on the head including left, middle, and right frontal regions. The probe was positioned on the scalp for 10–16 seconds up to three times per location. Repeating the measurement by slightly moving the probe helped to ensure reproducibility of the data. This procedure was also used to minimize data contamination from local heterogeneities such as hair and extra-cerebral veins. 18 Data quality and rejection criteria were based on a published study 11 and used to discard spurious source of noises such as motion artifacts and ambient light contamination, resulting in only high-quality data.
In neonates with HIE, arterial oxygen saturation (SaO 2 ) was recorded from routine monitors whereas hemoglobin concentration in the blood (HGB) [g/dl] was determined by complete blood count analysis at the time of the measurement and extracted from medical records. In healthy neonates, SaO 2 was assumed to be 98% 19 and HGB values were derived from clinical reference tables. 20
Cerebral blood volume [ml/100 g] and SO 2 [%] were computed from the data acquired with the FDNIRS system using a published approach. 9 Diffuse correlation spectroscopy data was modeled as a diffusion process and included a set of temporal auto-correlation functions acquired sequentially every second. The diffusion equation was used to derive CBF i [mm2/s] by fitting the normalized intensity temporal auto-correlation function, to the measured signal of multiply scattered light.21, 22
Cerebral oxygen metabolism was computed via the calculation of an index of CMRO 2 (CMRO2i, [Mol/dl·mm2/second]) using Fick's principle 22 such that
where CaO2 and CvO2 [Mol/dl] are the arteriolar and venous O2 concentrations, MWHb [g/Mol] is the molecular weight and, SaO2 and SvO2 are the arteriolar and venous O2 saturations. As in our previous studies, we assumed SO2=αSaO2+βSvO2 with α+β=1 where the arterial:venous (α:β) contribution ratio is 1/4:3/4. 23
Statistical Analysis
Non-parametric Wilcoxon signed rank tests were performed to compare hemodynamic and metabolic variables in neonates with HIE during TH (HIE–TH) and after TH (post-TH), and in control neonates (Control) at a significance level of 0.05. Age-matched comparisons were also performed by defining two control age sub-groups with respect to hours of age at the time of measurement: 0<age≤48 hours and 48<age96 hours. A paired Wilcoxon signed rank test was performed at a significance level of 0.05 to compare each neonate with HIE with its corresponding age-matched control sub-group. Number of measurements for each variable in subsequent comparisons are not identical for all variables because of their independent data quality criteria. 11
RESULTS
Figure 1 depicts temporal distribution of frontal (A) CMRO 2i , (B) CBF i , (C) CBV, and (D) SO 2 values in neonates with HIE treated with TH (colored bullets) and control neonates (black squares) with respect to age at the time of measurement. Standard errors of the mean are shown when more than one frontal location was averaged. Therapeutic hypothermia period (light gray background) corresponds to the period between 6 and 90 hours and includes both cooling and rewarming phases whereas normothermic post-TH period (light blue background) starts at 91 hours and ends at the last measurement (at 305 hours). Note that the x-axis is broken twice in post-TH phase, as no measurements were taken during those particular periods. Cerebral oxygen metabolism in HIE–TH was almost always lower than in healthy controls (Figure 1A). Most of the infants had CBF i ranging between 1.5 and 4.5 × 10−6 mm2/second during TH (Figure 1B). Infant #9 had the highest value (7.1 × 10−6 mm2/second at 65 hours of age), which coincided with periods of desaturation corresponding to abnormally low partial pressure of oxygen in the arterial blood, per the medical record. The CBV value was low for infant #8 at 20 hours of age (Figure 1C). Infants #6, #7, and #9 had the highest CBV values during TH all around 72 hours of age. Infant #6 had right temporal lobe and intraventricular hemorrhages. Subpial and intraventricular hemorrhages were reported in infant #7 as well as a subdural hemorrhage within the posterior regions in infant #9. However, none of the infants had intracranial blood products in the frontal cerebral regions where FDNIRS–DCS measurements were acquired. Infant #8 also exhibited the lowest SO 2 value during TH (54.5% at 20 hours, Figure 1D) when compared with all other treated or control newborns.
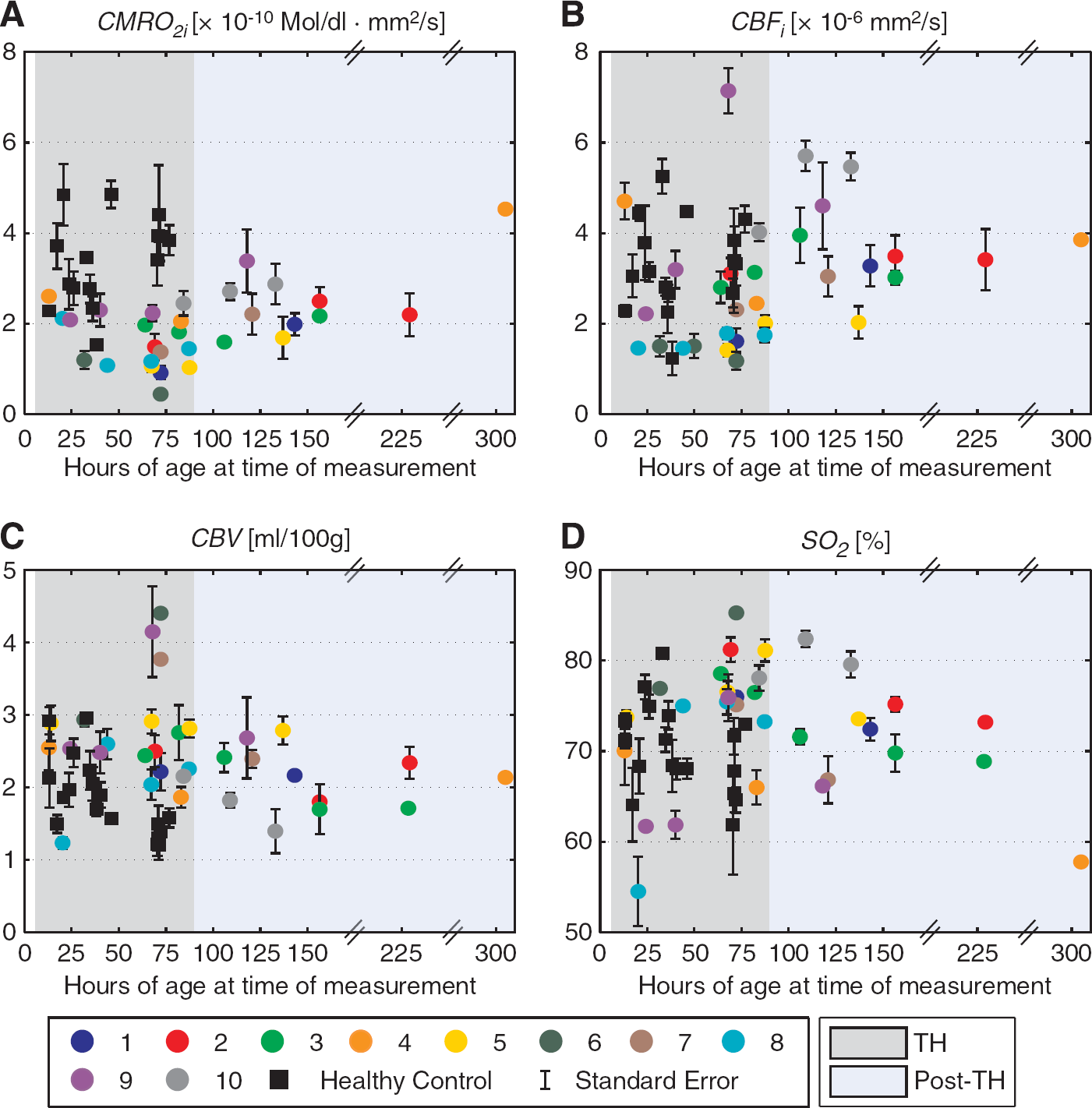
Frontal averages of (
Corresponding temporal distribution of the mean frontal oxy- (HbO), deoxy- (HbR), and total hemoglobin (HbT) used to derive CMRO 2i , CBV and SO 2 are shown in Supplementary Figure 1. In addition, individual raw values of regional (middle, left, and right frontal) HbO, HbR, HbT, and CBF i measured in neonates with HIE and healthy neonates are shown in Supplementary Figure 2 and Supplementary Figure 3, respectively.
Clinically indicated MRI was performed within 6 days of life in all 10 patients who underwent TH in this study. Two small posterior punctate foci with decreased apparent diffusion coefficient around 3 cm deep from the scalp were reported in infant #3, as well as few scattered foci of decreased apparent diffusion coefficient deep in the cortex and white matter in infant #7. All other neonates had apparent diffusion coefficient maps that were clinically interpreted as normal.
Recall that CBF i is calculated using the DCS system whereas CBV and SO 2 are calculated using the FDNIRS system. Thus, there are neonates whose data met inclusion criteria for one instrument and not the other and therefore not all subjects have all the same number of CBF i measurements as CBV and SO 2 measurements at every time point. Figure 2 shows boxplots of (A) CMRO 2i , (B) CBF i , (C) CBV, and (D) SO 2 values between HIE–TH and post-TH conditions (left box), and between HIE–TH and Control groups (right box) with corresponding number of measurements. On each box, the central mark is the median, the black square is the mean, the edges of the box are the 25th and 75th percentiles, and the whiskers show the 95% confidence interval. Outliers are depicted by empty circles whereas P value is provided in the box when the comparison is significant. Neonates with HIE had significantly lower CMRO 2i during TH than both post-TH (P<0.01) and Control conditions (P<10−5; Figure 2A). The low CMRO 2i in HIE–TH compared with Control remained significant when age-matched (P<10−4). Consistent with the low CMRO 2i , CBF i in HIE–TH was also significantly lower than post-TH values (P<0.01) and Control neonates (P<0.01; Figure 2B). The difference between HIE–TH and Control conditions remained significant when age-matched (P<0.01). Surprisingly, CBV was significantly elevated despite the low CBF i in the HIE–TH condition compared with both post-TH (P<0.01) and Control conditions (P<10−3; Figure 2C). Age-matched comparison also demonstrated significantly higher CBV in HIE–TH than Control groups (P<10−3). Although SO 2 is the parameter monitored most frequently with commercial systems, differences in SO 2 were the least discriminating and were only significantly elevated in the comparison between HIE–TH and Control (P=0.014; Figure 2D), with this significance remaining when age-matched (P=0.019). A similar analysis was also performed with HbO, HbR, and HbT measurements. Oxyhemoglobin and HbT in HIE–TH were significantly higher than in post-TH condition (P<0.01 and P=0.01, respectively) whereas HbR was not significantly different. None of the comparisons between HbO, HbR, and HbT in HIE–TH versus Control was significantly different.
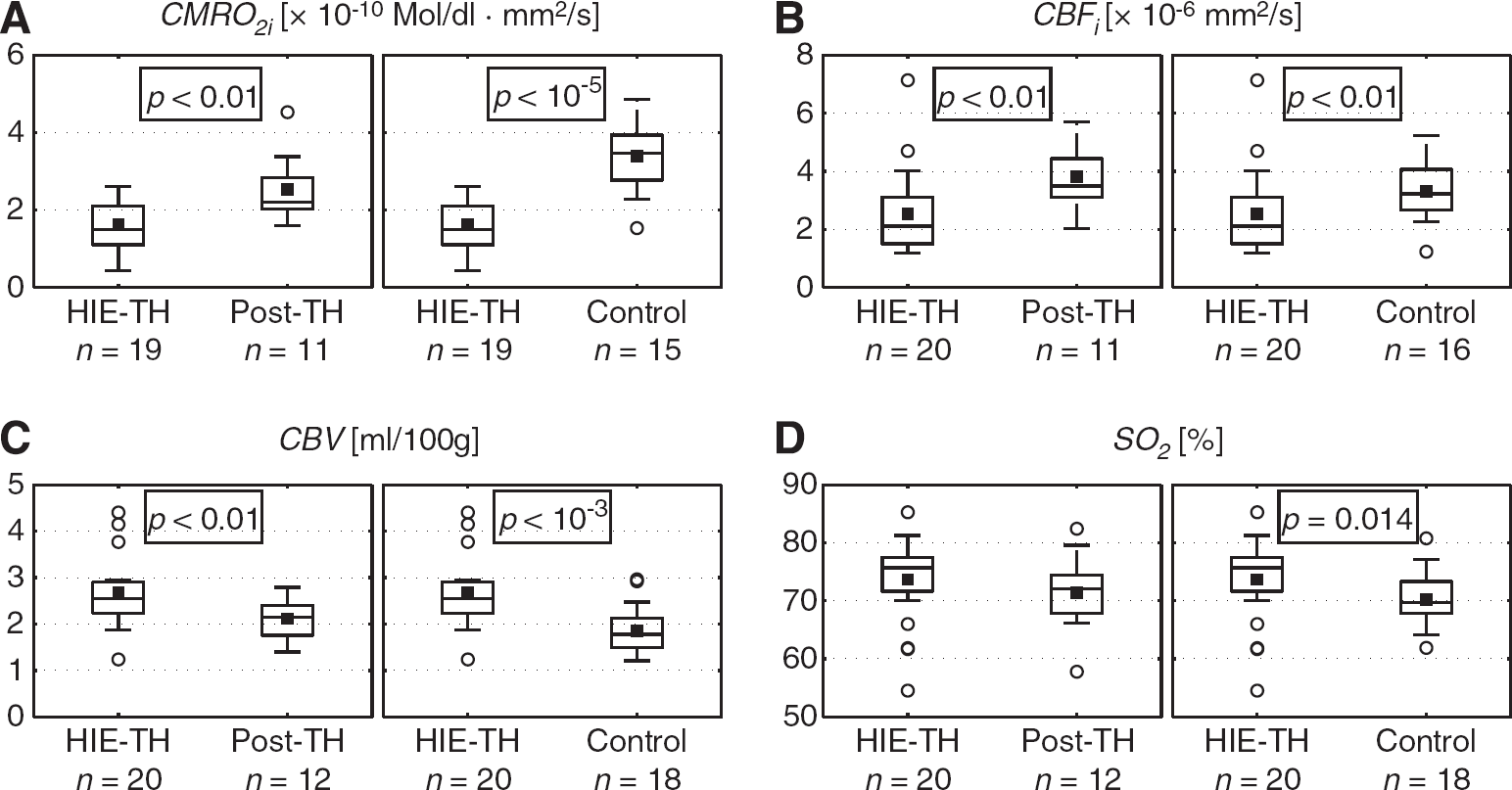
Boxplots of (
DISCUSSION
Indices of Cerebral Oxygen Metabolism and Cerebral Blood Flow
In this prospective pilot study of 10 term neonates undergoing TH for HIE and 17 healthy control neonates, bedside FDNIRS–DCS measurements of CMRO 2i were significantly lower in neonates with HIE during hypothermia (HIE–TH) compared with post-hypothermia (post-TH) and age-matched controls (Control). In fact, of all hemodynamic parameters studied, CMRO 2i was the parameters that best discriminated among the different conditions and groups. To our knowledge, this is the first report of bedside human neonatal CMRO 2i measures that demonstrates the expected lower CMRO 2i during TH compared with post-TH measures and age-matched healthy controls. These data suggest that FDNIRS–DCS instrumentation has the potential to be a good bedside monitor of newborns with HIE undergoing TH or other therapies.
This low CMRO 2i we observed in neonates with HIE during TH contrasts sharply with our prior study of untreated neonates with HIE. In particular, we found a higher relative CMRO 2 (rCMRO 2 ) in acute neonatal HIE without TH compared with neonates without suspicion of brain injury. 10 Although data were acquired between 1 and 12 days of birth all neonates were within 5 days of symptom onset and all had acute injury documented by diffusion-weighted imaging near the time of the FDNIRS measures. In addition, rCMRO 2 was estimated using the Grubb's relationship, 24 as the DCS system was not available at the time of the study. However, in a previous MRI study, we have shown increased cerebral perfusion in untreated neonatal HIE. 25 Thus with prior studies showing higher rCMRO 2 and increased cerebral perfusion in neonatal HIE, it was important to discover that both CBF i and CMRO 2i were decreased during TH. This observation was also consistent with a previous MRI study that showed decreased cerebral perfusion in acute neonatal HIE with hypothermia. 26
Although MRI methods for estimating CMRO 2 have been published in the adult literature,27, 28, 29, 30, 31 there is currently little data on the MRI measurement of CMRO 2 in neonates. One recent abstract reported results for 6 normothermic preterm infants without acute brain injury. 32 These MRI methods are based on a relationship between venous T2 imaging and venous oxygenation that is described through a calibration process determined in vitro. As NIRS, this calibration method depends on hemoglobin (hematocrit).33, 34 These methods provide whole brain estimates of CMRO 2 . Although a regional variant of this method is being developed it has not been applied to neonates. 35 In addition, it is currently not practical to screen for abnormal CMRO 2 with MRI before the necessary rapid initiation of TH, nor is MRI a practical tool to monitor response to TH or other future therapies because of high cost, difficulty in scheduling rapid studies and need to transport unstable infants outside the NICU.
Few reports of neonatal CMRO 2 using gold standard techniques such as positron emission tomography 36 or Xenon clearance 37 exist and as a result of increasing concerns for radiation exposure in neonates, no new data has been published since 1993. Thus although other, arguably more accurate, approaches exist, these approaches are currently not acceptable for research purposes in neonates.
The calculation of CMRO 2 is strongly dependent on the technique utilized to measure CBF. Here the subscript ‘i’ in CMRO 2i is used to acknowledge that CMRO 2 is derived using a CBF index (CBF i ), which although a flux (in mm2/second), correlates well with other measures of cerebral perfusion (in ml/100 g per minute). In fact, DCS CBF i correlates well with MR arterial spin labeling 38 and with phase-encoded velocity mapping (VENC) MRI measures of relative blood flow in the jugular veins and superior vena cava in neonates/children with congenital heart defect. 39 Further support for the use of DCS CBF i measures was provided by the high correlation (r2=0.73, P<0.01) between relative CBF i and Xenon CT cerebral blood perfusion measures in critically brain-injured adults. 40 These comparisons show that CBF i measured by DCS offers reasonable agreement with other modalities, in particular those providing perfusion measurements, and therefore provides a reliable index of CMRO 2 .
Cerebral Blood Volume
Our current observation of elevated CBV in HIE during TH has also been observed in one term newborn with moderate HIE between 2 and 8 hours of age during TH compared with pre-treatment value using commercial continuous wave near infrared spectroscopy. 41 This increase was followed by a decrease in CBV between 68 and 74 hours of age during TH, and another increase during the rewarming phase. However, the study did not specify if patient or table values for HGB were used to derive CBV. In a previous study, van Bel et al, 42 found CBV lowered during cooling and increased during rewarming in 12 neonates and infants (age 0 to 11 months) undergoing open-heart surgery using commercial continuous wave near infrared spectroscopy. 42 However, none of these neonates had documented brain injury, the degree of hypothermia was inconsistent among the patient population and there was no detail on the duration of cooling. An increase in CBV during rewarming was also noted using commercial continuous wave near infrared spectroscopy in seven out of eight neonates. However these neonates were preterm infants (<31-week gestational age) with very low birth weight presenting with hypothermia at admission and none had documented brain injury. 43
Elevated CBV with TH is interesting as CBF i is decreased rather than increased. Possible reasons for increased CBV include mediations such as dopamine, which has a similar effect as cocaine, 44 increased carbon dioxide partial pressure and increased venous pressure. In addition, there is a variable relationship between CBV and CBF depending on cerebral perfusion pressure and the effectiveness of autoregulation. If CBF decreases sufficiently to begin impacting oxygen supply, CBV may increase to compensate. A similar phenomenon has been seen in other mechanisms of brain injury.45, 46 Thus there are many factors that must be considered and explored in future work.
Cerebral blood volume is determined using HbT measured with our FDNIRS system and HGB. To determine if HGB changes may be driving the increase in CBV, we first provided values of HbO, HbR, and HbT in Supplementary Figure 1. We confirmed HbT to be significantly higher in neonates with HIE during TH (P=0.01) compared with post-TH values. However, HbT in HIE–TH was not significantly higher than in Control group. We further discuss the accuracy of HGB measurements in the next section presenting the modeling assumptions.
The clinical significance and etiology of increased CBV remain unclear. The role of medications and the potential of inadequate cerebral perfusion leading to vasodilatation needs to be explored in neonates with HIE undergoing TH. This observation raises questions about the optimal CBF and CBV during therapy. 47
Cerebral Hemoglobin Oxygen Saturation
Near infrared spectroscopy-measured rSO 2 have been used to monitor neonates with HIE before the clinical standardization of TH. Results among the literature are variable. In a previous study, we measured higher SO 2 values in neonates with HIE within 5 days of symptom onset compared with neonates without suspicion of brain injury; however, the difference was not significant. 10 In a time window between 24 and 48 hours of age in neonates with HIE, rSO 2 was significantly higher in neonates with adverse outcomes as compared with neonates with good outcomes. 8 However, this difference was not significant between 6 and 18 hours of age. In a recent study of neonates with HIE who underwent TH, rSO 2 was reported as slightly higher in HIE with adverse outcomes at 6, 12, and 24 hours of life compared with neonates with good outcomes; however the difference was only significant at 12 hours. 48 In the current study, SO 2 in neonates with HIE was higher than post-TH, but the difference was not significant. With an elevation of SO 2 occurring both with and without TH, SO 2 alone appears to be less sensitive to the therapy and metabolic disturbances occurring in the first few days after birth. Therefore, FDNIRS–DCS parameters presented in this study have potential to compliment SO 2 measures to provide a more complete picture of the hemodynamic and metabolic changes in neonates with HIE treated with TH.
Medication Effects
The primary role of analgesics and sedatives is to prevent secondary brain injury during the treatment by maintaining an adequate mean arterial pressure, which in turn will influence CBF and volume to lower cerebral metabolic demand. 49 For example, barbiturates (Phenobarbital) decrease CMRO 2i and benzodiazepines (midazolam or lorazepam) are commonly used as a sedative in ventilated pediatric patients. They have minimal adverse cardiovascular effects. Opioids (fentanyl, morphine, or methadone) produce analgesia and comfort. 50 In rhesus monkeys, propofol anesthesia concurrent with TH did not impair the coupling between cerebral metabolism and blood flow. 51 Neurotransmitters are also commonly used in neonatal population for their action on the cardiovascular system. For example, Dopamine is known to increase systemic and regional CBF as well as blood pressure. 52 Cardiovascular effects of Dopamine in piglets were maintained during mild hypothermia and were similar to responses in normothermia. 53 Given the wide range and inconsistent use of medications in the neonates presented here (see Supplementary Table 2), we believe that medication is less likely the dominant factor causing the low oxygen metabolism we observed. However, future studies are necessary to further assess the effects of medications on FDNIRS–DCS measures and to better understand flow–volume–metabolism coupling.
Modeling Assumptions and Statistical Analysis
Factors that could contribute to CMRO 2i errors include shifts in arterial:venous volume ratios during the therapy. For example, as CBF i decreases and CBV increases during TH, there could be a relative increase in the venous compartment from 75% to 85%. Recalculating CMRO 2i during TH with 85% venous content further reduces CMRO 2i by 12% and therefore CMRO 2i remains significantly lower than controls (P<10−8). However, the 1/4:3/4 (arterial:venous) ratio has not been validated for in vivo circulation in neonates and TH could also lead to a decrease in the venous content. Assuming a fixed ratio of 1/3:2/3 (arterial:venous) leads to an increase of 12% in CMRO 2i but CMRO 2i remains significantly lower compared with controls (P<10−5). Thus our major findings remain unchanged.
Errors in CMRO 2i propagate with respect to all physiologic variables present in its modeling (equation (1)). Assuming a coefficient of variation of 13%, 2.3%, 5.5%, and 4% for CBF i , 11 HGB, 54 SaO 2 , 55 and SO 2 ,11, 18 respectively, variation in CMRO 2i was estimated to be 19.9%. Error of 12% in rCMRO 2 has been calculated in rats during focal ischemia using a similar instrumentation and was particularly sensitive to the error in relative oxygen extraction fraction. 22 The major source of error in our method is likely due to the variability of CBF i . Furthermore, the fitting process used to derive CBF i requires the knowledge of the average reduced scattering coefficient of the cerebral tissue, which is quantified with the FDNIRS system. We verified that the reduced scattering coefficient was not statistically different between neonates with HIE treated with TH and healthy controls, thus differences in CBF i between the groups are due to differences in CBF and not confounded by differences in scattering. In this study, we used the average scattering coefficient for each population as the input for the fitting process. As a result, future research is aimed at improving the accuracy of CBF i measurements.
The calculation of both CBV and CMRO 2i necessitates knowledge of HGB values. We found consistently lower HGB in neonates with HIE than normal clinical table values and believe this may be due to the high number of blood draws and intravenous fluids these neonates receive, as well as pre-existing anemia in some HIE newborns. To determine if elevated CBV and decreased CMRO 2i in HIE–TH compared with healthy controls were solely due to HGB values in Control we performed an additional analysis using HGB lower (13.5 g/dl) and upper (21.5 g/dl) limits from the reference table values. 20 We confirmed the persistence of a statistically significant difference between HIE–TH and Control groups for CMRO 2i (see Supplementary Figure 4(a)). It is only when HGB in Control is dropped below the 5th percentile that the increase in CBV in HIE–TH compared with Control increase is not significant (see Supplementary Figure 4(b)). Our results may lose significance only if our normal neonates have very low HGB (below the 5th percentile). As blood is not drawn on healthy neonates, in future a direct measurement of HGB using non-invasive techniques would help improve the accuracy of CBV and CMRO 2i measures.
In the statistical analysis, the selection of the two sub-groups about the age at measurement is justified by prior studies showing significant decreases in cerebral rSO 2 in 26 healthy control neonates during the first 5 days of age. 56 This result is consistent with our data: SO 2 in control neonates from birth to 2 days old is significantly higher than SO 2 from 48 to 96 hours of age (P<0.05). We identified a similar relationship with CBV in controls (P<10−3). However, control CBF i and CMRO 2i data did not show a significant difference between the two age sub-groups (P>0.1 and P=0.06, respectively).
Future Studies
HIE is associated with a complex cascade of events including vascular and metabolic changes. 2 This evolution depends on the severity and timing of the hypoxic ischemic insult. In this pilot study, the aim was to compare hemodynamic and metabolic variables in neonatal HIE during and following the therapy. The addition of time-points during the first week after birth would better characterize individual injuries and their evolution. For example, continuous monitoring of FDNIRS–DCS parameters during the therapy would provide more details of the time course of changes. 8 Furthermore, with the primary goal of TH to reduce the extent of brain injury, 17 bedside measurement of hemodynamic and metabolic variables might be useful concurrent measures with electroencephalography or amplitude-integrated electroencephalography prior the therapy to aid in the decision to initiate TH in neonates with HIE. However, this clinical translation would be facilitated with a simplified device and standardization of the measured parameters. The role of animal studies in providing pre-, intra- and postmeasurements with and without TH might also help to refine and optimize inclusion criteria and TH parameters. 57
The rate of normal outcome (56.6%) from the current study was similar to the normal rate reported in literature. 17 However, the rate of mildly abnormal outcome (33.3%) was higher compared with literature and the rate of abnormal outcome (1 death, 11.1%) was lower than reported trials.5, 17 This discrepancy is most likely due to the small sample size included in this particular study, as the overall outcome of neonatal HIE treated with TH at our institution is similar to published data. Due to the low number of patients, scores can only be used to describe individuals but predictive modeling will be implemented for a planned larger cohort.
CONCLUSION
In summary, we have demonstrated that human term neonates with HIE who meet inclusion criteria for TH have low CMRO 2i and CBF i while undergoing TH when compared with post-TH and healthy age-matched controls. In contrast, CBV in neonates with HIE treated with TH was significantly higher than post-TH and Control values whereas SO 2 was only significantly higher in HIE–TH compared with Control group. We believe that these hemodynamic and metabolic changes observed during TH are of multi-factorial nature including factors related to mechanisms associated with TH, hypoxia-ischemia, timing of measurements and potentially, medications administered. In conclusion, FDNIRS–DCS parameters have the potential to serve as bedside biomarkers for monitoring individual response and optimization of TH but further studies are needed to explore the effects of medications on individual responses as well as long-term outcome.
Footnotes
Maria Angela Franceschini hold patents on the technology developed in this article. The other authors declare no conflict of interest.
ACKNOWLEDGMENTS
The authors thank all the families and the nurses, physicians, and staff in the NICU, the Special Care Nursery, Pediatric Neurology, and the maternity units at Brigham and Women's Hospital and Boston Children's Hospital. In particular, the authors thank Breda Hayes, Deirdre O’Reilly, Marcia Kocienski-Filip, Maddy Artunduaga, Linda J Van Marter, Robert M Insoft, Jonathan H Cronin, Julianne Mazzawi, and Steven A Ringer. The authors would also like to thank Barry D Kussman, David A Boas, Stefan A Carp, Erin M Buckley and Bruce R Rosen for their discussion and support, and Marie Drottar for reading through the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.