Abstract
The blood–brain barrier (BBB) closely interacts with the neuronal parenchyma in vivo. To replicate this interdependence in vitro, we established a murine coculture model composed of brain endothelial cell (BEC) monolayers with cortical organotypic slice cultures. The morphology of cell types, expression of tight junctions, formation of reactive oxygen species, caspase-3 activity in BECs, and alterations of electrical resistance under physiologic and pathophysiological conditions were investigated. This new BBB model allows the application of techniques such as laser scanning confocal microscopy, immunohistochemistry, fluorescent live cell imaging, and electrical cell substrate impedance sensing in real time for studying the dynamics of BBB function under defined conditions.
Keywords
Introduction
The blood–brain barrier (BBB) forms a very restrictive and protective barrier between blood and the neuronal parenchyma. It is mainly composed of a tightly sealed monolayer of brain endothelial cells (BECs) that regulate blood flow, nutrient transport, ion homeostasis, clearance of toxic substrates, and various other physiologic processes by the collective action of neurons, astrocytes, pericytes, and microglia (Abbott et al, 2006; Fisher, 2009). This interaction is referred to as a ‘neurovascular unit.’ A dysfunction of the BBB is associated with both acute and chronic neurologic disorders (Sandoval and Witt, 2008; Zlokovic, 2008). For instance, a breakdown of the BBB during an ischemic stroke is associated with the development of a vasogenic edema and represents a life-threatening complication (Rosenberg, 1999). It has been recently shown that besides BEC, other cell types of the neurovascular unit have a significant role in the pathogenesis of BBB breakdown (Abbott et al, 2006; Kuhlmann et al, 2009a). To investigate the pathomechanisms of BBB injury that are relevant in vivo, it is desirable to conduct studies in models that include all components of the neurovascular unit.
In 1998, Duport et al (1998) reported a coculture consisting of cortical organotypic slice cultures (COSCs) and BECs and validated this model using electrophysiological recordings and electron microscopy. However, an extensive study of BBB properties under physiologic and pathophysiological conditions would benefit from the application of additional techniques, such as immunohistochemistry (IHC), fluorescent live cell imaging, and electrical cell substrate impedance sensing (ECIS, which is a validated marker for barrier integrity in real time). Therefore, we established a coculture model consisting of BECs and COSCs that is suitable for the application of these techniques.
Materials and methods
Cortical Organotypic Culture
For detailed information, please see Supplementary Information.
Cell Culture
The murine BEC line (bEnd.3, American Type Culture Collection, Manassas, VA, USA) was cultured as recommended by the manufacturer. Cell cultures were maintained at 37°C and 5% CO2. Media consisted of DMEM Glutamax supplemented with 2% Penicillin/Streptomycin (all purchased from Invitrogen GmbH, Karlsruhe, Germany) and 15% fetal calf serum (Biochrom AG, Berlin, Germany). For coculture procedures, BECs were grown until confluence was reached on glass coverslips.
Dyes, Chemicals, and Antibodies
For detailed information, please see Supplementary Information.
Laser Scanning Confocal Microscopy
For detailed information, please see Supplementary Information.
Electrical Cell Substrate Impedance Sensing in the Coculture
Brain endothelial cells were seeded at a density of ∼50,000 cells per well in an 8-well gold electrode assay (0.8 cm2 growth area, ibidi in cooperation with Applied BioPhysics, Martinsried, Germany). For coculture procedures in ECIS, the BEC media were refreshed after 1 day and COSCs were gently transferred into the wells (Figure 2H, arrows). For monoculture procedures, no COSCs were added and media were only replaced. When impedance measurements reached maximal values, oxygen and glucose deprivation (OGD) was induced to monitor in vitro ischemia-induced BBB breakdown.
Oxygen and Glucose Deprivation
In vitro ischemia was induced by a combined OGD. Coculture coverslips were carefully transferred into 24-well plates. Media were replaced with 500 μL of HBSS (Hank's buffered salt solution) supplemented with 2 mmol/L CaCl2, 1 mmol/L MgCl2, 20 mmol/L 2-deoxy-
Immunohistochemistry
For detailed information, please see Supplementary Information.
Effect of Glutamate Exposure on Tight Junction Distribution
To elucidate whether glutamate triggers tight junction (TJ) redistribution in the novel coculture model, we applied different concentrations of glutamate to the cocultures and monocultures of bEnd.3 for 0.5 h and investigated morphologic changes in the TJ distribution of zonula occludens-1 (ZO-1) and claudin-5 (CL-5) using IHC. Glutamate exposure was performed in HBSS supplemented with 2 mmol/L CaCl2, 1 mmol/L MgCl2, and 20 mmol/L glucose. The following glutamate concentrations were used: 0, 250, 500, and 1,000 μmol/L. After 0.5 h, samples were immediately fixed for immunostaining.
Statistics
Results are expressed as mean values±s.e.m. A value of P<0.05 was considered as significant. Statistically significant effects of OGD on reactive oxygen species (ROS) production were assessed by t-test analyses. Effects of hypoxia on electrical resistance values were analysed with one-way ANOVA (analysis of variance) followed by post hoc Tukey's test.
Results
Preparation and Morphology of the Novel Coculture
Brain endothelial cells were grown on glass coverslips to confluence, mounted onto 0.4 μm Millicell-CM (Millipore, Bedford, MA, USA) culture plates, and put into 6-well plates. Brain endothelial cell media were added beneath the filter inserts and a drop of the BEC media was placed on the coverslip to avoid desiccation of the BEC (Figure 1A). Brain slices should be kept in culture for at least 3 days after preparation. Approximately 400 μL of prewarmed BEC media was added to the culture plates to remove brain slices from their membranes. The slices were gently disconnected from the membrane using a scalpel. A plastic pipette was used to carefully aspirate and transfer the slices onto the grown glass coverslips. The slices were placed in the middle of the BEC coverslip (Figure 1B). Finally, BEC media were added onto the filter so that the coverslips were completely covered with a thin film of the media. After combining COSC and BEC, the plate is moved very gently into the incubation chamber to avoid dislocation of the slice from the coverslip. For the following 2 days, any movement of the coculture should be avoided to let the slice adhere to the BEC. A bEnd.3 medium was used as culture medium. Experiments were carried out after 2 days in coculture.
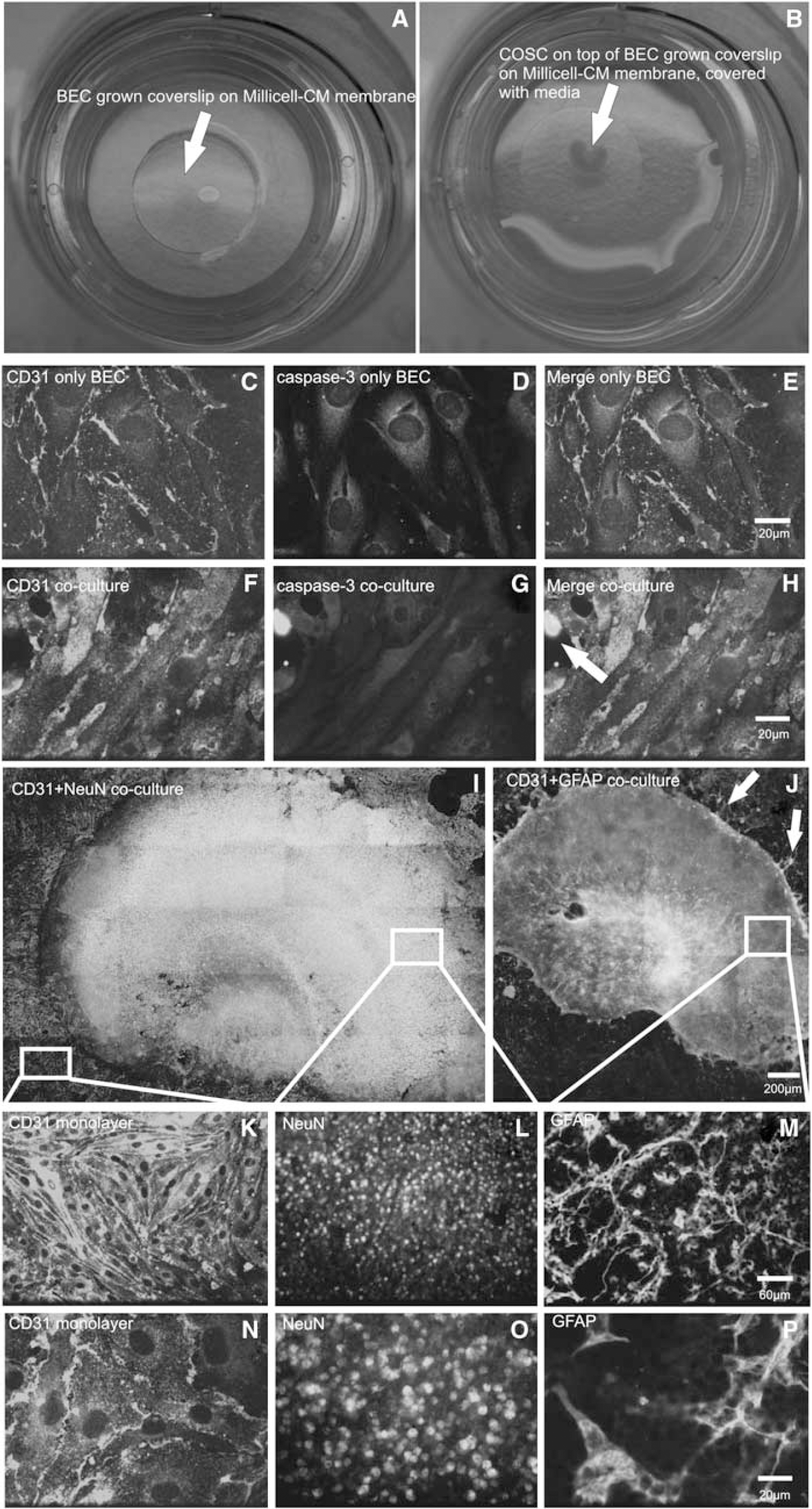
Preparation and morphology of the novel coculture model. (
We observed firm adhesion between BEC and COSC and well-differentiated cell morphologies after 2 days (Figures 1I–1P). To examine the question whether these culture conditions might induce apoptosis in the BEC, we performed IHC costaining of active caspase-3 and CD-31. After 2 days in coculture, the expression of caspase-3 activity was sparse as compared with BECs that were cultured without COSCs (Figures 1C–1H, representative images). Samples were always checked for adherence of the slice on BEC before starting the experiments, because a dislocation of COSC and BEC after 2 days in culture was occasionally observed. Before experimental manipulations, the coculture was also examined for intact cell morphology with light microscopy. We found that adherence after 2 days was crucially dependent on COSC preparation. Only morphologically proper COSC preparations were suitable for further coculture procedures.
Immunohistochemistry of Tight Junction Proteins, Reactive Oxygen Species Fluorescence Live Cell Imaging, and Electrical Substrate Impedance Sensing in the Coculture
We validated the use of our coculture by adopting different methodological approaches, which are commonly used for monitoring BBB function. A hallmark for the integrity of the BBB is the expression of TJ proteins (Krizbai and Deli, 2003), such as (ZO-1) and CL-5 (Wolburg and Lippoldt, 2002). Antibody staining for these proteins revealed a prominent expression of ZO-1 and CL-5 (Figures 2A and 2C). These findings are in good agreement with the data of Brown et al (2007) who previously analyzed TJ expression in a monoculture of bEnd.3. To study the expression of these TJ proteins under pathophysiological conditions, in vitro ischemia was induced by a combined OGD for 10, 45, or 120 mins. Oxygen and glucose deprivation caused a clear redistribution of ZO-1 (Figures 2B and 2D) and CL-5. Excitotoxicity has a key role under pathologic ischemic conditions in the central nervous system and is mainly mediated by glutamate. To further elucidate differences toward glutamate exposure between the novel coculture and monocultures of bEnd.3, we investigated the impact of glutamate exposure on TJ integrity. We found that concentrations of 500 μmol/L (Figures 2J–2M) and 1,000 μmol/L glutamate (not shown) had a significant effect on ZO-1/CL-5 distribution within the coculture but not in the monoculture of bEnd.3. A volume of 250 μmol/L glutamate did not affect TJ integrity (data not shown). Another key factor for BBB breakdown under pathologic conditions is the formation of ROS (Kuhlmann et al, 2008, 2009b). To study the OGD-induced production of ROS in BEC within the coculture, the fluorescent ROS sensor 2′7′-dichlorodihydrofluorescein (DCF) (10 μmmol/L) was added 30 mins before picture acquisition. Coverslips were immediately mounted into a temperature-controlled incubation chamber of a laser scanning microscope set-up (for details, see Supplementary Material) filled with warm HBSS supplemented with 2 mmol/L CaCl2, 1 mmol/L MgCl2, and 20 mmol/L glucose. Confocal pictures were taken in three randomly chosen fields of view per coverslip within the BEC monolayer area (monolayer area: Figures 1K and 1N). The average fluorescent intensity of the entire image was measured. Data were analyzed using Metamorph imaging software (Molecular Devices Corp., Downingtown, PA, USA). Fluorescence of DCF was significantly increased after OGD when compared with the control group (Figures 2E–2G). To analyze the integrity of the BEC monolayer of the coculture in real time, we performed ECIS, which is a very sensitive method to monitor morphologic changes in a monolayer. After 10, 45, and 120 mins of OGD, electrical resistance values decreased significantly (control coculture: 694.5±36.64 Ohm s.e.m.; OGD 10 mins coculture: 420.5±6.5 Ohm; OGD 45 mins coculture: 314.4±8.79 Ohm; OGD 120 mins coculture: 296.8±7.8 Ohm s.e.m., n=4, P<0.01, growth area 0.8 cm2, Figure 2I), which correlates well with the redistribution of TJ proteins as detected with liquid chromatography mass spectrometry after IHC.
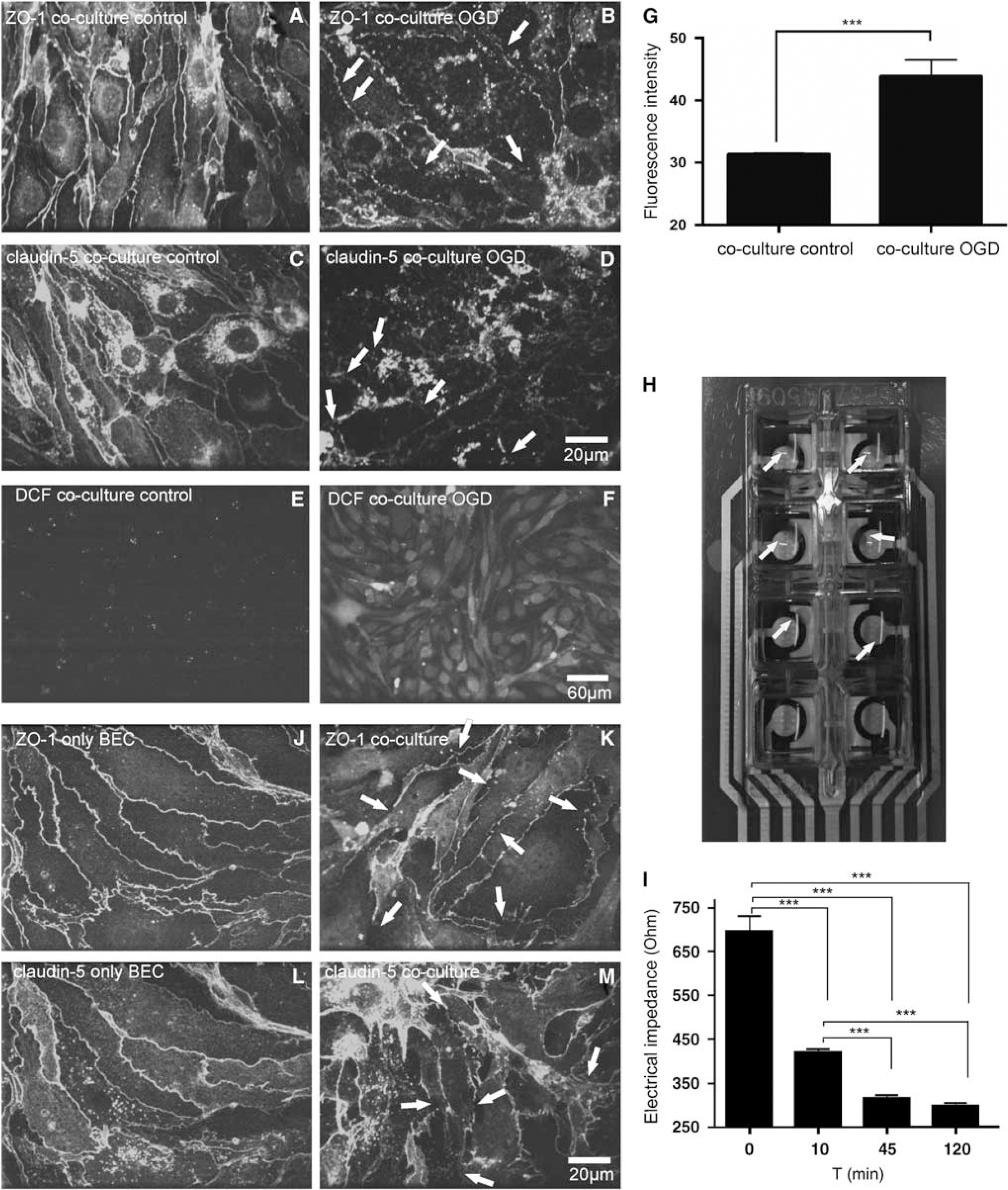
Immunohistochemistry of TJ proteins, ROS fluorescence imaging, and electrical substrate impedance sensing in the coculture. (
Discussion
The following are the main findings of the presented report: (1) The coculture of BEC and COSC shows an intact morphology of neurons, astrocytes, BECs, and TJ protein expression. (2) The pathophysiological conditions in this coculture model can be easily induced, and the underlying mechanisms can be investigated by various established techniques in BBB research including IHC, fluorescent live cell imaging, and ECIS in real time. (3) The preparation of this coculture requires a sufficient control of COSC after explantation before the slices are applied to the BEC monolayers.
A coculture between cortical slices and BEC represents a valuable model that contains all cell components of the neurovascular unit. The commonly used BBB models generally lack some cellular components of the neurovascular unit. As shown by the present glutamate exposure experiments, the contribution of an intact neuronal compartment is essential for the evaluation of changes in BBB integrity. In a recently published study, we were able to show that glutamate at rather high concentrations (1 mmol/L) and a long incubation period of 3 h induces BBB disruption in a BEC/astrocyte coculture model (Kuhlmann et al, 2008). The results of this study indicate that alterations of the BBB occur at already lower glutamate concentrations after shorter incubation periods (500 μmol/L and 30 mins, respectively). This finding clearly underlines the contribution of neurons to BBB damage under excitotoxic conditions. The applications for the BBB model described in this study are not limited to acute BBB injury. It can be a useful model to study changes of BBB function in chronic neurologic disorders, such as inflammatory responses in Alzheimer's disease, multiple sclerosis, and amyotrophic lateral sclerosis (Giri et al, 2000; Schreibelt et al, 2006; Zlokovic et al, 2000; Zlokovic, 2008). Furthermore, the present model can be useful in studying the receptor-mediated transport of pathologic agents (e.g., receptor for advanced glycosylation endproducts (RAGE)-mediated transport of amyloid peptides) across the BBB in Alzheimer's disease.
