Abstract
Moyamoya disease (MMD) is an unusual form of chronic cerebrovascular occlusive disease that involves the formation of characteristically abnormal vessels. Recent studies have reported that colony-forming unit (CFU) and outgrowth cells represent a subpopulation of endothelial progenitor cells (EPCs). Here, we attempted to determine the significance of CFU number and outgrowth cell yield in MMD. Endothelial progenitor cells were isolated from the blood of 24 adult MMD patients and from 48 age- and risk factor-matched control subjects. After 7 days of culture, CFUs were determined, and yields of outgrowth cells were measured during 2 months of culture. The EPC function was also evaluated using matrigel plate assays. It was found that CFU numbers were significantly lower in MMD patients than in controls. Moreover, during long-term culture, outgrowth cells were isolated from only 10% of control subjects but from 33% of MMD patients, and CFU numbers and tube formation were found to be lower in advanced MMD cases than in those with early stage disease, whereas outgrowth cells were more frequently detected in those with early MMD and moyamoya vessels than in those with advanced disease. These characteristics of circulating EPCs reflect mixed conditions of vascular occlusion and abnormal vasculogenesis during the pathogenesis of MMD.
Keywords
Introduction
Moyamoya disease (MMD) shows progressive and bilateral steno-occlusive changes around the terminal portions of the internal carotid artery, and the formation of extensive collateral vessels at the base of the brain (Suzuki and Takaku, 1969). Neurologic manifestations in MMD result from the occlusions of specific arteries, hemodynamic instability, or the rupture of leaking collateral vessels (Suzuki and Kodama, 1983). Furthermore, these occlusions of major intracranial arteries are the results of eccentric fibrocellular thickening of intima because of smooth muscle cell proliferation and fibrosis (Yamashita et al, 1983; Masuda et al, 1993). In addition, robust neovascularization develops from various collateral pathways and may form by indirect revascularization from surgically placed vascularized tissues (Hoshimaru et al, 1991; Yoshimoto et al, 1996). It also should be noted that these features are not generally observed in other cerebrovascular occlusive diseases.
Reduced levels of circulating endothelial progenitor cells (EPCs) have been shown to be related to endothelial dysfunction, cerebral infarction, and coronary artery disease, which suggests that EPCs play an important role in vascular homeostasis (Vasa et al, 2001; Tepper et al, 2002; Hill et al, 2003; Schmidt-Lucke et al, 2005). Furthermore, it was recently suggested that reduced numbers and impaired functions of circulating EPCs are related to the pathogenesis of stroke (Ghani et al, 2005; Chu et al, 2008). Thus, given that MMD involves dynamic interplay between arterial occlusion and neovascularization, the involvement of EPCs in MMD may be more complex than in other cerebrovascular occlusive diseases.
Some investigators have employed flow cytometry as a means of estimating EPC levels (Vasa et al, 2001; Tepper et al, 2002; Schmidt-Lucke et al, 2005), whereas others have used colony-forming units (CFUs) (Hill et al, 2003; Ghani et al, 2005; Chu et al, 2008). Matters are further complicated by the fact that a subset of outgrowth cells with the endothelial phenotype develops during long-term cultures of peripheral blood mononuclear cells (PB-MNCs) (Hur et al, 2004; Yoon et al, 2005; Jung et al, 2008). Outgrowth cells represent an EPC population with vasculogenic potential. Current evidence indicates that EPC-CFU numbers represent an accumulated EPC dysfunctional status, whereas outgrowth cell appearance reflects the resilience of the systemic circulation to ongoing vascular stress (Chu et al, 2008; Jung et al, 2008).
A recent study reported a positive correlation between moyamoya-like neovascularization of the cerebral arterial circulation and increased levels of circulating CD34+ cells (Yoshihara et al, 2008). However, the issues about EPC number and function, and outgrowth cell generation, are much more complicated during the dynamic courses of MMD. Therefore, in this study, we aimed to characterize EPCs at various stages of adult MMD in terms of EPC-CFU number, function, and outgrowth cell yield, and elucidate the mechanisms that contribute to the pathogenesis of MMD.
Materials and methods
Study Population
Twenty-four consecutive patients with an angiographic diagnosis of MMD registered at the Neurology clinic at Seoul National University Hospital were enrolled. Study subjects were selected from adult patients aged more than 20 years at disease onset who had undergone cerebral angiography at the most 2 years before enrollment. As EPC might represent a dynamic response to an acute stroke event (Chu et al, 2008), MMD patients with no new stroke or symptom exacerbation ≤ 3 months were recruited for blood tests. Given that vascular risk factors could have influenced EPC populations, we also enrolled control subjects who had (a) no history of stroke and (b) one or more of the following vascular risk factors: hypertension, diabetes mellitus, dyslipidemia, or a history of smoking (RF control group). The RF controls were recruited by 1:2 case-to-control matching (age ≤ ± 1, same gender, Framingham risk scores ≤ ± 2) of individuals who visited the neurology clinic at Seoul National University Hospital for a medical examination and a routine neurologic checkup. All eligible patients provided written informed consent before enrollment. The study was conducted according to the Declaration of Helsinki and its subsequent amendments, and approved by the Institutional Review Board of Seoul National University Hospital. Patient demographic and medical data were collected as described in the Supplementary Methods.
Angiographic Classification
Moyamoya disease was diagnosed by digital subtraction angiography, and a diagnosis of MMD was verified when digital subtraction angiography showed stenosis or occlusion of the terminal internal carotid or proximal basal arteries and the presence of moyamoya vessels (Suzuki and Takaku, 1969). Digital subtraction angiographic findings of each patient were independently reviewed by a neuroradiologist and a neurologist who were unaware of the patients' identities. Angiographic findings were evaluated in both hemispheres of all 24 patients using the 4-stage angiographic classification scale proposed by Suzuki and Takaku (1969). Patients were classified according to the stage of the disease in more advanced sides.
Isolation and Culture of Peripheral Blood-Derived Mononuclear Cells
After obtaining informed consent, PB samples were collected from the 24 stroke patients and the 48 age- and sex-matched control subjects. Total human PB-MNCs were isolated from 50 mL of blood from each of the 72 enrolled subjects by density gradient centrifugation using Histopaque-1077 (Sigma, St Louis, MO, USA), as described previously (Chu et al, 2008; Jung et al, 2008) and in Supplementary Methods.
Colony-Forming Unit Counts
A CFU was defined as a central core of rounded cells surrounded by elongated spindled-shaped cells, and CFUs were counted after culturing MNCs for 7 days. Cell clusters without emerging spindle cells were not counted. Colonies were counted manually in a minimum of 3 wells in 12-well plates by two independent observers who were unaware of clinical profiles; results are expressed as average number of CFUs per well. In selected samples, the endothelial phenotype was confirmed using specific indicators, that is, according to the uptake of 1,1ʹ-dioctadecyl-3,3,3ʹ,3ʹ-tetramethylindocarbocyanine-labeled acetylated low-density lipoprotein (Biomedical Technologies Inc., Stoughton, MA, USA), and by immunostaining using FITC-labeled Ulex europaeus agglutinin-1 (Sigma) and anti-CD31 (1:20; DAKO, Carpinteria, CA, USA), antivascular endothelium cadherin (1:50; Chemicon, Billerica, MA, USA), anti-CD34 (1:20; DAKO), and anti-kinase domain receptor (1:50; Sigma) antibodies, as previously described (Hur et al, 2004; Yoon et al, 2005; Chu et al, 2008; Jung et al, 2008).
Matrigel Plate Assay
In selected subjects (n = 4 per disease stage), the angiogenic activities of conditioned media were assessed using Matrigel plates (BD Biosciences, Bedford, MA, USA) and human umbilical vein endothelial cells (HUVECs). Conditioned media were obtained by replacing media in 4-day MNC cultures with serum-free endothelial basal media (EBM; Clonetics, Baltimore, MD, USA) supplemented with single aliquots of EGM-2 (growth factors like vascular endothelial growth factor and fibroblast growth factor-2 were omitted), and by culturing for an additional 72 h. The wells of 96-well tissue culture plates were then coated with Matrigel (0.04 mL/well), which was left for 1 h at 37°C to solidify; HUVECs (15,000 in 0.15 mL EBM) were then added to each well. Conditioned medium (200 μL) was then added to these HUVECs (in sextuplicate) and 16 h later the media were removed. The cells were then fixed and the lengths of structures resembling capillaries were measured over whole-well areas using Image-Pro Plus imaging software (Media Cybernetics, Silver Spring, MD, USA). Total lengths of tube networks are expressed as mm/mm2, and the total areas of the complete tubes formed are expressed as mm2/mm2.
Statistical Analysis
Results are expressed as means ± s.d. The two study populations were compared in terms of the following variables: background characteristics (age, gender), risk factors (hypertension, diabetes mellitus, dyslipidemia, smoking, family history of coronary artery disease, and Framingham risk scores), and culture parameters (CFU number, outgrowth cell yield). Continuous variables were tested for normal distribution using the Kolomogorov-Smirnov test. Comparisons between groups were analyzed by using the two-tailed unpaired student's
Results
Baseline Characteristics of the Study Subjects
We enrolled 24 MMD patients and 48 RF controls. The clinical characteristics of the study subjects are summarized in Table 1. Sixteen MMD patients were women and eight were men. The MMD patients had a mean age of 43.7 ± 13.4 years, and a mean age at disease onset of 36.2 ± 12.3 years. Nineteen of the 24 MMD patients were diagnosed as having typical ʻbilateralʼ MMD, and the other 5 patients as having ‘unilateral’ MMD. Ten (41.7%) patients were classified as stage-2; 10 (41.7%) as stage-3; and 4 (16.6%) as stage-4. Initial symptoms were transient ischemic attacks in 7 (29.2%), cerebral infarction in (16.7%), and intracerebral hemorrhage in 10 (41.7%). Bypass surgery, including superficial temporal artery to middle cerebral artery anastomosis, in one or both hemispheres was eventually performed in 3 of the 24 MMD patients. The remaining 21 patients were treated medically with anticonvulsants, antiplatelet agents, or other pharmacological agents. The control subjects comprised 32 women and 16 men (mean age 43.8 ± 13.6 years). Primary diseases in the control group were as follows: hypertension in 4 (16.7%), diabetes mellitus in 1 (4.2%), and dyslipidemia in 4 (16.7%). As shown in Table 1, the patients and controls did not differ with respect to age, gender, hypertension, diabetes mellitus, dyslipidemia, smoking, family history of CVD, Framingham risk scores, or drug medication.
Clinical characteristics of the study population
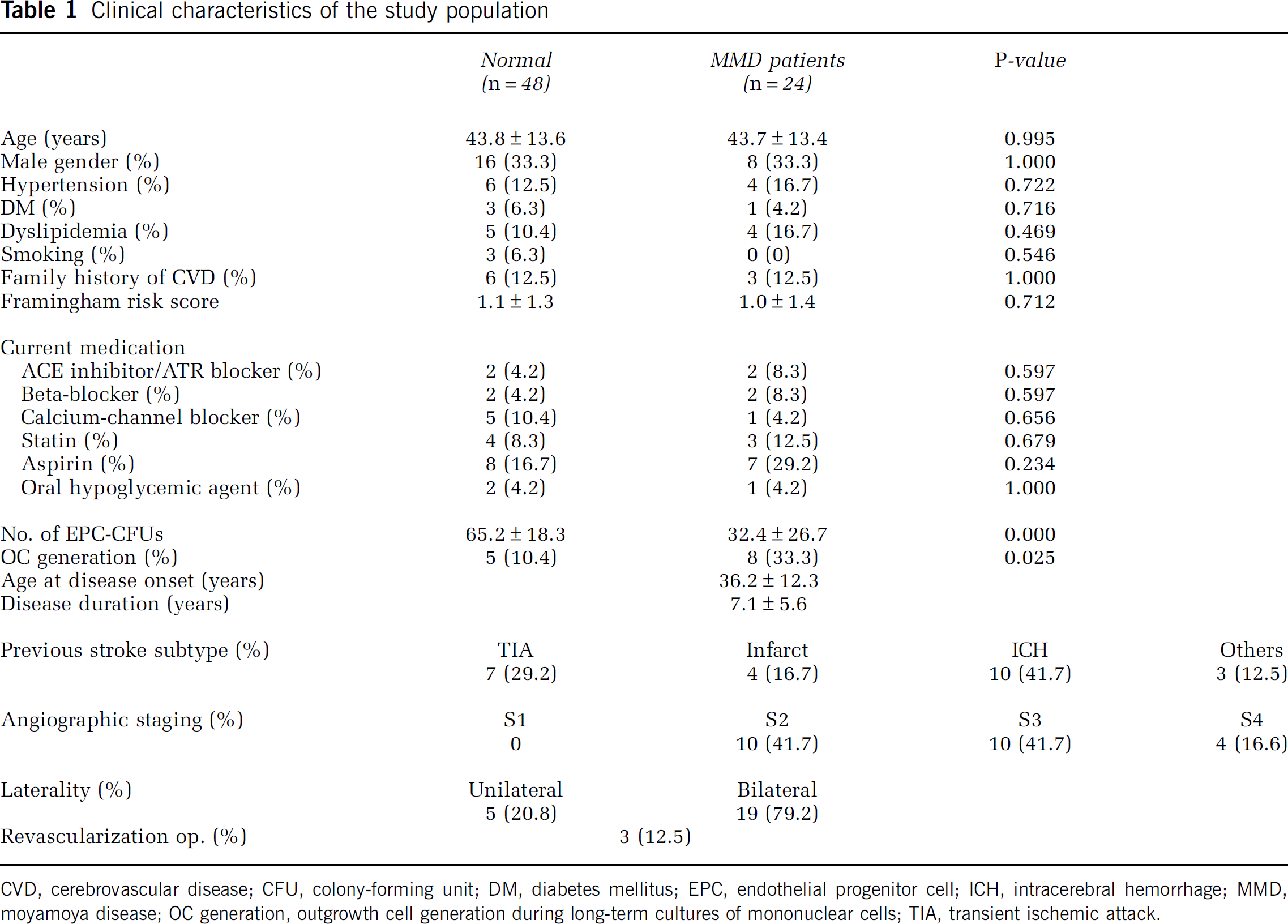
CVD, cerebrovascular disease; CFU, colony-forming unit; DM, diabetes mellitus; EPC, endothelial progenitor cell; ICH, intracerebral hemorrhage; MMD, moyamoya disease; OC generation, outgrowth cell generation during long-term cultures of mononuclear cells; TIA, transient ischemic attack.
Culture Outcomes in the Study Populations
Culturing PB-MNCs resulted in the formation of CFUs or outgrowth cells, as described previously (Hur et al, 2004; Yoon et al, 2005; Chu et al, 2008; Jung et al, 2008). The interval between blood sampling and cerebral angiography was 15 ± 7 months (range: 3 to 23 months). In all patients, the clinical status was stable throughout the period between cerebral angiography and blood sampling, and no new stroke or exacerbation of transient ischemic attack occurred during this period. We examined culture outcomes in the two study populations with respect to EPC-CFU numbers and outgrowth cell isolation frequencies. The EPC-CFU numbers were significantly lower in the MMD patients than in the controls (32.4 ± 26.7 versus 65.2 ± 18.3, respectively,
Factors Associated with Low EPC-CFU Numbers in MMD Patients
To identify the factors that affect CFU number in MMD patients, clinical and radiologic parameters were compared in low EPC-CFU (≤ 30 CFUs,
Factors associated with CFU numbers from adult MMD patients
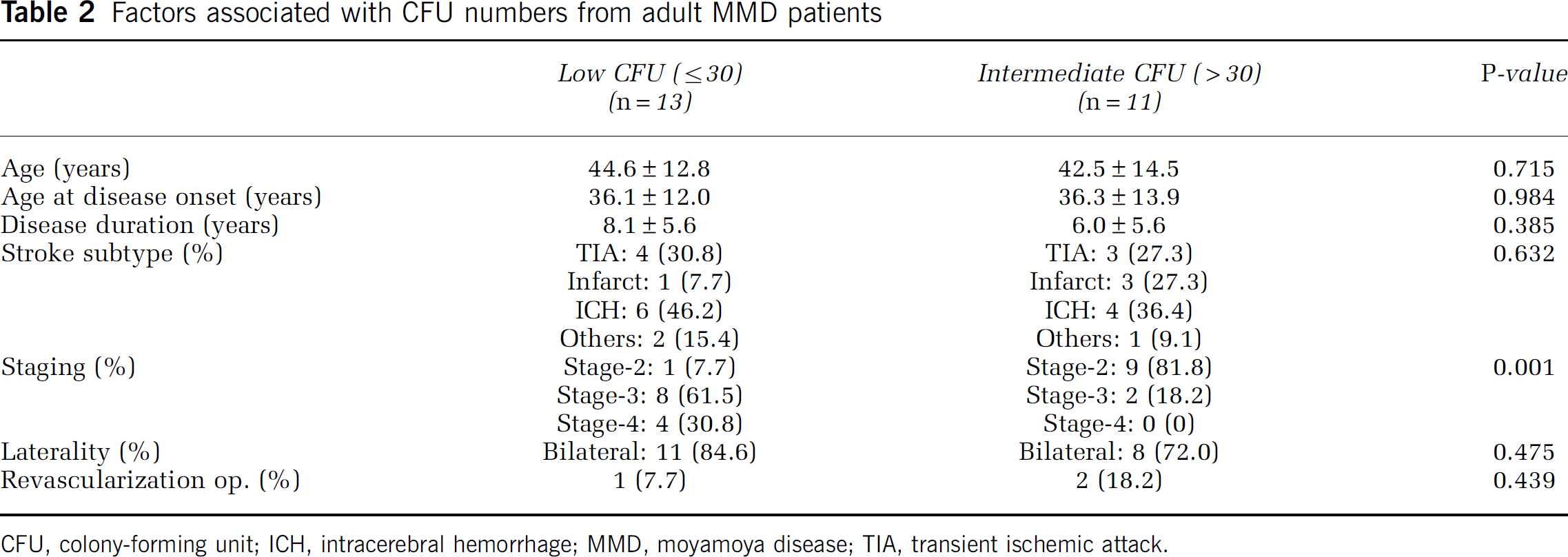
CFU, colony-forming unit; ICH, intracerebral hemorrhage; MMD, moyamoya disease; TIA, transient ischemic attack.
In addition, we also compared the numbers of EPC-CFUs isolated from MMD patients by disease stage. Advanced MMD patients (stages 3 and 4) were found to have lower numbers of EPC-CFUs than stage-2 or control subjects (Figure 1D;
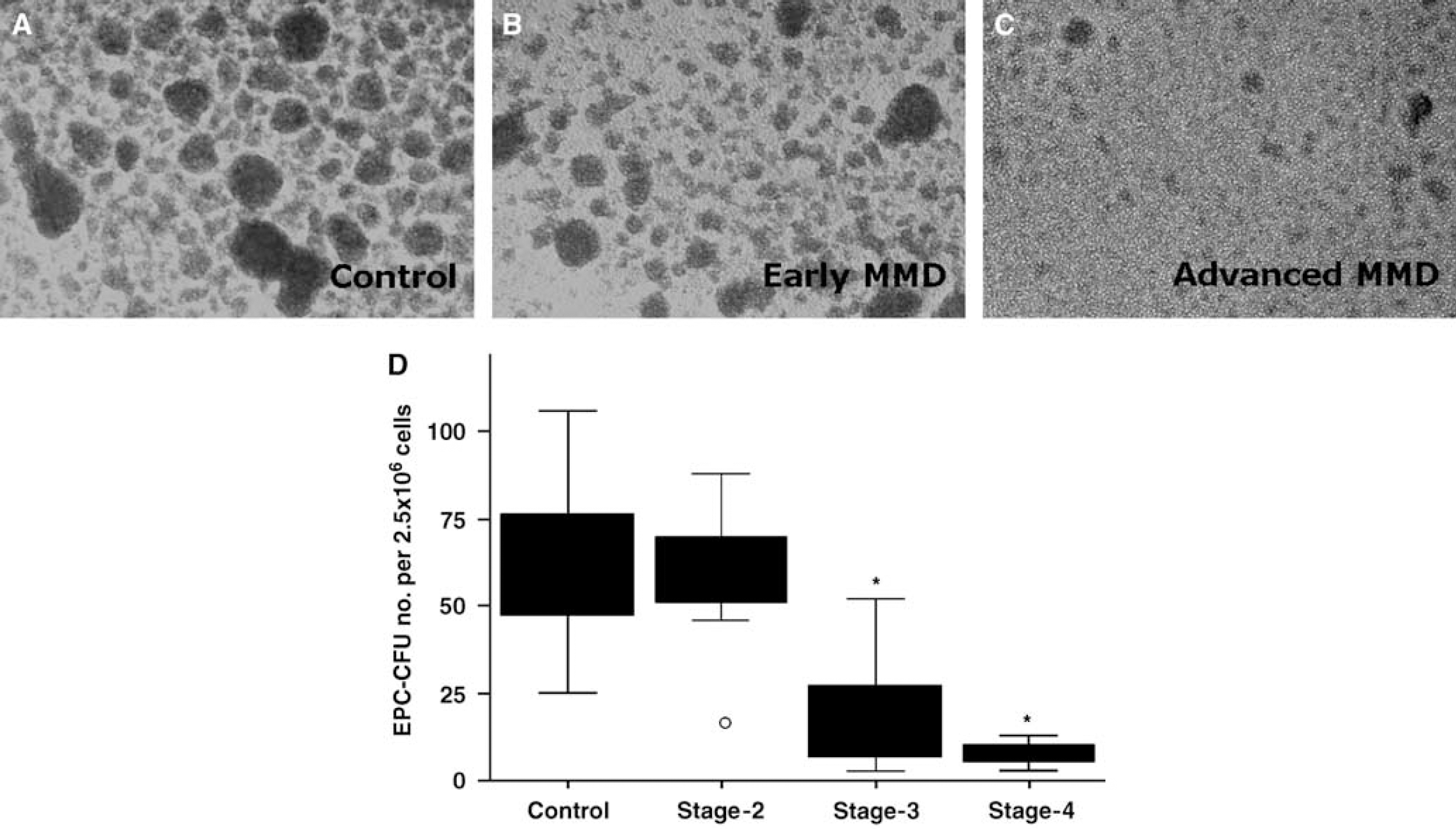
The EPC-CFU numbers from MMD patients. Phase contrast images of PB-MNC cultures show the formation of EPC-CFUs at 7 days after plating the MNCs of MMD patients at different disease stages (
Matrigel Tube Assays in Moyamoya Disease Subpopulations
To investigate whether the paracrine functions of EPCs are differentially affected by MMD stage, we examined the angiogenic potentials of conditioned media using Matrigel assays. Conditioned media of control subjects led to the formation of an extensive tubule network when compared with nonconditioned media (EBM), thereby showing that EPCs can facilitate angiogenesis in a paracrine fashion (Figure 2A). In contrast, the conditioned media from the EPCs of MMD patients induced significantly less tube formation, suggesting that cultured EPCs from MMD patients can impair angiogenesis
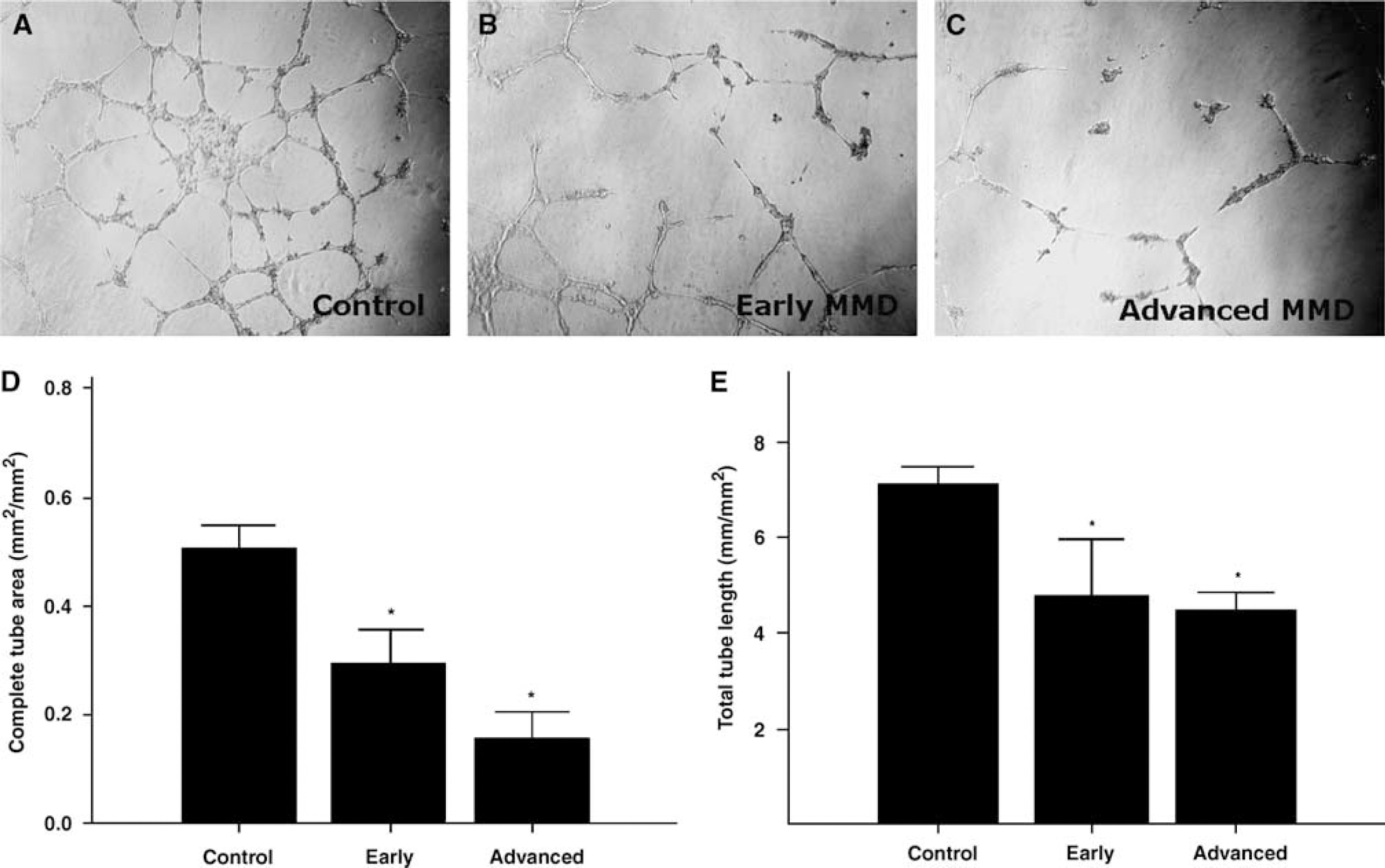
Matrigel assays. Three-day cultures of PB-MNCs were used to produce conditioned media, which were added to HUVECs on Matrigel-coated 96-well plates. Representative photomicrographs show the formation of tube-like structures in controls (
Factors Associated with Outgrowth Cell Appearance in Moyamoya Disease Patients
Outgrowth cells were isolated from 8 (33.3%) of the 24 MMD patients. To identify factors that predict outgrowth cell generation in MMD, we allocated patients to two groups, the outgrowth cell-negative and outgrowth cell-positive groups. The two groups were similar in terms of patient age, age at disease onset, disease duration, stroke subtype, and laterality. However, outgrowth cell yield tended to be greater for those with an earlier stage disease or a history of revascularization surgery (Table 3).
Factors associated with outgrowth cell appearance in adult MMD patients
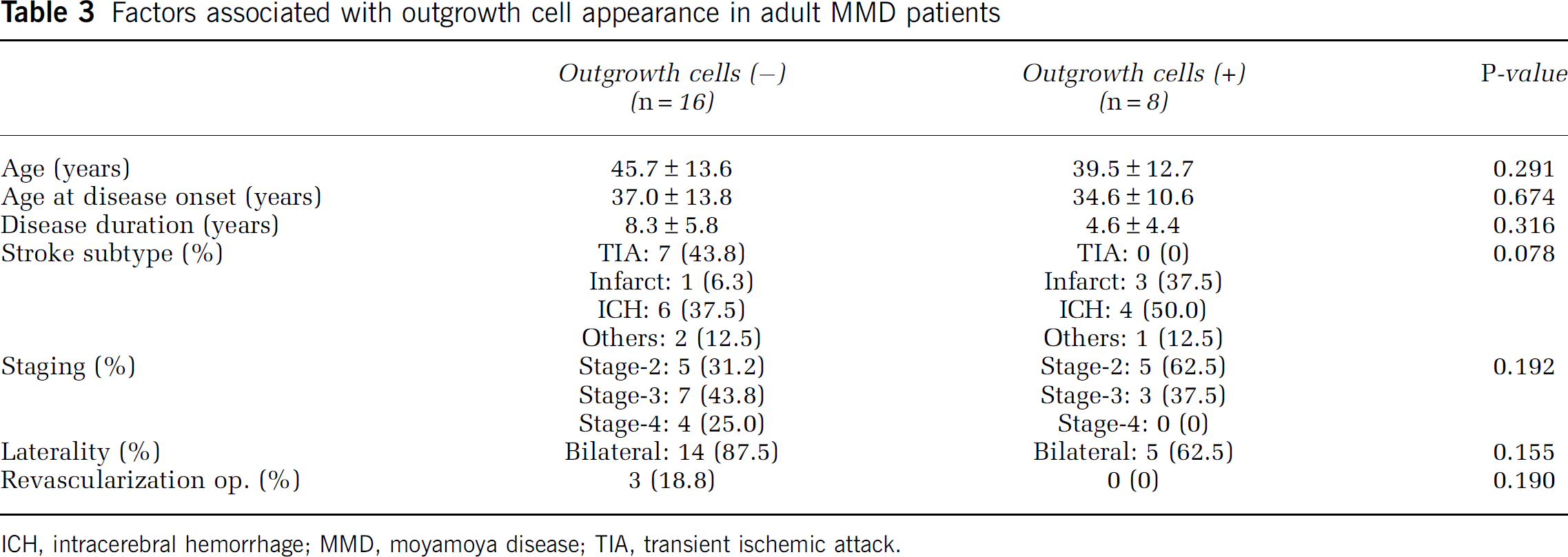
ICH, intracerebral hemorrhage; MMD, moyamoya disease; TIA, transient ischemic attack.
Outgrowth cells were more efficiently isolated from stage-2 MMD patients (5 out of 10, 50%) than from patients with more advanced disease (stage-3: 3 out of 10, 30%; stage-4: 0 out of 4, 0%), and were also isolated more frequently from patients who had undergone revascularization surgery than from patients treated conservatively (100% versus 0%; Supplementary Figure 1).
Discussion
In the present study, we compared EPC numbers and functions in MMD patients, and in control subjects without a history of stroke, and found that MMD is associated with a reduced number of EPC-CFUs, impairment in EPC functional activity, and a higher yield of outgrowth cells. These observations suggest that circulating EPCs are a pathogenetic marker of MMD and that they induce vaso-occlusive changes and/or recruit the compensatory vascular network.
The pathophysiology of MMD has not been elucidated, but major intracranial arteries are known to be occluded by eccentric fibrocellular intimal thickening because of smooth muscle cell proliferation and fibrosis (Yamashita et al, 1983; Masuda et al, 1993). Moreover, collateral pathways, that is, fine-vessel networks (moyamoya vessels) and transdural anastomoses develop because of intimal proliferation (angiogenesis) or recruitment of EPCs (vasculogenesis) (Hoshimaru et al, 1991; Nanba et al, 2004; Yoshimoto et al, 1996). In the present study, CFU numbers and outgrowth cell yields were used to identify EPCs in blood. Moyamoya disease patients have lower EPC-CFU numbers than control subjects, and although it is unclear whether this effect is causative or simply associated with pathologic arterial changes, the EPC-CFU number appears to indicate a cumulative vascular risk in MMD. The MMD patients were also found to have higher frequencies of outgrowth cells, which have recently been reported to represent a regenerative response to ongoing tissue damage (Chu et al, 2008; Jung et al, 2008) and, thus, they may be associated with the formation of an abnormal vascular network. Our results are in line with a recent report that found that a prominent feature of moyamoya-like vessels in MMD is correlated with a robust increase in the level of circulating CD34+ cell levels (Yoshihara et al, 2008).
In adult patients, the incidence of disease progression in both unilateral and bilateral MMD is about 20%, and occurs in both anterior and posterior circulations in symptomatic and asymptomatic patients (Kuroda et al, 2005). Moyamoya staging is based mainly on angiographically observed changes in the severities of steno-occlusive lesions (Suzuki and Takaku, 1969; Mugikura et al, 2002). However, until now, no study has addressed the blood markers associated with MMD stage in adult patients. Our results clearly show that patients with advanced stage disease have substantially lower EPC-CFU numbers than patients with early stage disease. In addition,
Moyamoya disease staging is based on angiographic changes in the intensification or decrease of moyamoya vessels (Suzuki and Takaku, 1969; Mugikura et al, 2002), that is, moyamoya vessels initially develop as occlusive changes progress and then reduce in number and size when occlusive changes become severe (Suzuki and Takaku, 1969). The abnormal vascular networks formed act as collateral vessels and compensate for arterial stenosis (Takekawa et al, 2004). ‘Angiogenesis,‘ the paradigm of postnatal neovascularization, is a process whereby new vessels and endothelial cells are derived by sprouting from preexisting differentiated endothelial cells (Folkman and Shing, 1992). However, some of these new vessels and endothelial cells in MMD may be derived from EPCs of bone marrow origin, a process referred to as postnatal vasculogenesis (Isner and Asahara, 1999). Further-more, outgrowth cells that arise from an unidentified population during the long-term culture of MNCs were reported to have EPC characteristics (Gulati et al, 2003). In the present study, outgrowth cells were generated from majority of the early moyamoya patients, but rarely from control subjects or from advanced stage moyamoya patients. Although the exact cellular and/or paracrine mechanisms involved in EPC-mobilization, recruitment, and homing are unclear, this regenerative process appears to be regulated by a variety of chemokines and cytokines (Hristov et al, 2003). In terms of growth factors and cytokines in MMD, the vascular endothelial growth factor, fibroblast growth factor, hepatocyte growth factor, and platelet-derived growth factor have been reported to increase in the cerebrospinal fluid and dural tissue in MMD patients (Hoshimaru et al, 1991; Aoyagi et al, 1996; Yoshimoto et al, 1996; Soriano et al, 2002; Kim et al, 2003; Nanba et al, 2004; Sakamoto et al, 2008). Another analysis of the cerebrospinal fluid from MMD patients revealed high levels of cellular retinoic acid-binding protein-I expression, which is known to enhance the expressions of growth factors (Kim et al, 2003). Therefore, outgrowth cells may be an important population that is involved in the developmental and/or compensatory processes of MMD.
In the present study, EPC-CFU numbers and the output of outgrowth cells were somewhat higher in patients who had undergone revascularization surgery, and although the significance of this finding was borderline, we attribute this lack of significance to the limited sample size. Collateral circulation is frequently well developed in MMD patients, and revascularization surgery is known to improve cerebral hemodynamics and outcome in MMD (Matsushima et al, 1992; Golby et al, 1999). More-over, as EPC levels were found to be related to the effect of revascularization surgery in the present study, it is possible that they reflect the status of vascular reserve in MMD patients. However, whether the restoration of EPC status after bypass surgery is a reliable indicator of revascularization remains to be determined.
Although it is appreciated that circulating EPCs in MMD patients may be critical during the development of stenosis of intracranial major arteries and in the vascularization of a collateral circulation, several clinically related questions need to be fully addressed. First, basal EPC profiles may vary among individuals, and these may be related to individual disease course variations. Second, we enrolled MMD patients with no new stroke or symptom exacerbation during the 3-month period before the recruitment, because acute stroke might influence circulating EPC number and function. However, stroke, especially in watershed areas in MMD patients, could be silent. Third, MMD is known to be a progressive disease and it has been shown that unilateral MMD is likely to progress as well. The disease could have progressed between the time when the angiogram was performed and the time of inclusion into the study, especially in 20% of unilateral MMD patients in our series.
We speculate that our findings may shed light on the pathogenesis of MMD, and that they represent the basis of new treatment strategy aimed at preventing disease progression. Further prospective studies are required to determine whether alterations in the number and function of circulating EPCs may be used as diagnostic or prognostic markers in MMD.
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
