Abstract
We have developed an experimental model of spontaneous intracranial hemorrhage (ICH) in transgenic mice expressing human renin and human angiotensinogen (R+/A+) treated with high-salt diet and Nω-nitro-
Introduction
Spontaneous intracranial hemorrhage (ICH) is a devastating type of stroke that accounts for 10% to 15% of all strokes, and is associated with high mortality and morbidity (Xi et al, 2006). Hypertension is an important risk factor for ICH. Mechanisms that lead to ICH during hypertension and the pathogenesis of brain injury after ICH, however, remain poorly understood.
Recently, we have developed the first experimental model of spontaneous ICH in hypertensive mice: double transgenic mice with overexpression of human renin and human angiotensinogen (R+/A+) are treated with Nω-nitro-
An important mechanism of vascular injury in hypertension is oxidative stress (Heistad, 2006). Oxidative stress, produced in part by NAD(P)H oxidase, induces dysfunction and cell death of endothelium and vascular muscle (Bhunia et al, 2002; Burlacu et al, 2001; Didion and Faraci, 2003). Angiotensin II activates NAD(P)H oxidase and thereby generates high levels of superoxide in cerebral vessels as well as in the central nervous system (Hanna et al, 2002; Didion and Faraci, 2003; Zimmerman et al, 2004). In addition, several studies suggest that activation of matrix metalloproteinases (MMPs) is redox-sensitive (Gu et al, 2002; Rajagopalan et al, 1996b). Matrix metalloproteinases are a family of zinc endopeptidases involved in the degradation of basal lamina and extracellular matrix (Woessner, 1991). Expression and activity of MMP-9 are increased in cerebral microvessels in rats with chronic hypertension (Liebetrau et al, 2005). Thus, in our model of ICH in double transgenic R+/A+ mice, we hypothesize that oxidative stress in intracranial vessels may predispose to ICH.
Oxidative stress may also contribute to brain injury after ICH. Superoxide, visualized by in situ detection of oxyethidium, is increased in the perihematomal region after ICH (Wang and Tsirka, 2005). Activation of NAD(P)H oxidase contributes to brain injury after ICH (Tang et al, 2005). Previous studies were performed after injection of bacterial collagenase into the brain to produce ICH (Tang et al, 2005; Wang and Tsirka, 2005). However, no studies have examined the role of oxidative stress in spontaneous ICH.
The major goal of this study was to determine whether generation of superoxide and activation of NAD(P)H oxidase are associated with spontaneous ICH during chronic hypertension in R+/A+ mice.
Materials and methods
Experimental Animals
Studies were conducted in female double transgenic mice (R+/A+), which were generated by cross-breeding human renin (R+) transgenic mice with human angiotensinogen (A+) transgenic mice (Merrill et al, 1996). Nontransgenic (R−/A−) and single transgenic littermate mice (R+/A− or R−/A+) were used as controls for R+/A+ mice, because strict specificity in the enzymatic reaction exists between the mouse and human renin—angiotensinogen system (Merrill et al, 1996). Breeding and genotyping of R+/A+ mice and their control mice were performed in the transgenic animal facility located in a virus- and pathogen-free animal-care facility.
We also studied male wild-type (C57BL/6) mice and gp91phox-deficient mice (C57BL/6 background) that lack the membrane gp91phox (Nox2) subunit of NAD(P)H oxidase. The gp91phox-deficient and C57BL/6 mice were purchased from Jackson Laboratories (Bar Harbor, ME, USA) and were housed in the Animal Care Facility at the University of Iowa.
All mice were housed in individual cages and kept on a 12-h light/dark cycle (light from 0600 to 1800 hours). All experimental protocols were approved by the Institutional Animal Care and Use Committee at the University of Iowa.
Spontaneous Intracranial Hemorrhage
Mice developing spontaneous ICH were generated as described previously (Iida et al, 2005). Briefly, female R+/A+ and control mice (4 months old) were given an 8% high-salt diet (TD-92012; Harlan Teklad, Madison, WI, USA) and 100 to 120 mg/kg
Systolic blood pressure (SBP) was measured using a tail cuff (Vistech System BP-2000, Apex, NC, USA). R+/A+ and control mice on treatment were trained for 3 consecutive days in the prewarmed tail-cuff device, and then SBP was measured for 2 days. On each day, 30 measurements were obtained and averaged for each mouse. Averaged values of SBP were measured at baseline and once every month for 24 weeks.
Clinical signs of stroke were assessed by daily neurologic examinations, including contralateral forelimb extension, circling behavior, or other motor dysfunction (Garcia et al, 1995). When an R+/A+ mouse developed these signs, the mouse was euthanized with Nembutal (150 mg/kg intraperitoneally), and an age-matched R+/A+ mouse without signs of stroke and an age-matched control mouse were euthanized. The mice were perfused in situ for 10 mins through a cannula in the left ventricle with phosphate-buffered 0.9% saline (PBS) at about 70 mm Hg. Brains were cut sagittally into half and analyses were conducted according to several procedures described below.
Half of the brain of each mouse was immersion-fixed in 7% formaldehyde for histometric analysis. Paraffin-embedded tissue was serially sectioned at 5 μm and stained with hematoxylin and eosin (H&E). To facilitate precise identification of ICH, one section of every five serial sections was stained with DAB (diaminobenzidine), which reacts with peroxidases in red blood cells. Diaminobenzidine highlights hemorrhages, leaving nonhemorrhagic areas unstained. We have found that this method is much more sensitive in detecting ICH, and thus allows far more accurate quantification of ICH, than examining sections stained with H&E. All H&E- and DAB-stained sections were screened using a light microscope, and each section was scanned in toto using an Aperio Scanscope System (Model T3; Aperio, Vista, CA, USA) to create a digital image with a final resolution of 0.5 μm/pixel (equivalent to a final magnification × 200) of the entire section. Digital images were analyzed with Image J software (NIH) to identify the location and quantify the number of hemorrhages.
Fresh, unfixed half brains were used to evaluate oxidative stress by lucigenin-enhanced chemiluminescence and to estimate mRNA levels by real-time reverse-transcription PCR. These unfixed half brains were cut and divided into four parts at 3 mm intervals from anterior pole to cerebellum. The most anterior quarter of brain contained cerebral cortex and anterior part of striatum. The second anterior quarter of brain contained cerebral cortex, posterior part of striatum, and anterior part of thalamus and hippocampus. The third anterior quarter of brain contained posterior part of thalamus and hippocampus, and upper part of brainstem. The most posterior quarter of brain contained cerebellum and middle and lower part of brainstem (Franklin and Paxinos, 1997).
Striatal Injection of Blood
To determine whether ICH itself produces oxidative stress in the brain, we examined the levels of superoxide and the activity of NAD(P)H oxide after injection of blood in the striatum of male gp91phox-deficient mice and C57BL/6 mice (4 months old). The gp91phox-deficient mice were used to investigate whether Nox2 is associated with the oxidative stress after ICH. Mice were placed in a stereotactic frame, and a 27-gauge stainless-steel cannula was introduced through a burr hole into each striatum (2 mm lateral to midline, 1 mm anterior to bregma, and depth 4 mm below the surface of the skull). Each mouse received a 4-μL injection over 5 mins of autologous whole-blood into the left striatum and artificial cerebrospinal fluid (aCSF) (temperature 37°C; ionic composition (in mmol/L): 132 NaCl, 2.95 KCl, 1.71 CaCl2, 0.65 MgCl2, 24.6 NaHCO3, 3.69
The injection cannula was slowly withdrawn 10 mins after the injection of blood or aCSF. The wound was sutured, and the mouse was returned to its cage with free access to normal food and water. Twenty-four hours later, mice were euthanized by an injection of overdose of Nembutal (150 mg/kg intraperitoneally) and perfused transcardially with PBS as described above. The brain was immediately removed and cut coronally into a section 2 mm anterior and posterior to the injection site. The brain sections with injected blood and aCSF were used for the measurements described below.
Evaluation of Oxidative Stress
Superoxide levels were quantified with lucigenin-enhanced chemiluminescence. Fresh, unfixed brains were homogenized and sonicated in PBS containing protease inhibitors (protease inhibitor cocktail, Complete Mini; Roche Diagnostic, Mannheim, Germany) at 0°C to 5°C. Brain homogenates were placed in 0.5 mL PBS and 5 μmol/L lucigenin, preincubated for 60 mins at room temperature with or without 1 mmol/L tiron (a superoxide dismutase mimetic) or 100 μmol/L diphenyliodinium (DPI; an inhibitor of flavoprotein-containing enzymes, including NAD(P)H oxidases). Relative light units (RLUs) were measured for 10 mins. Background counts were determined and subtracted, and values were normalized per 1 mg brain tissue. NAD(P)H oxidase activity was estimated by adding NADPH (1, 10, or 100 μmol/L) to brain homogenates with or without addition of tiron (1 mmol/L). Under all conditions, the tiron-inhibitable chemiluminescence value (RLU per sec per mg brain tissue) was used as a measure of superoxide levels.
Dihydroethidium (DHE), an oxidative fluorescent dye, was used to localize superoxide in brain, in situ, after intrastitial injection of blood. Some mice were treated with DHE (300 μL; stock solution of DHE: 20 mmol/L in dimethylsulfoxide, diluted to 3.3 mmol/L in PBS just before use). Dihydroethidium solution was injected intraperitoneally 3 h before the mice were euthanized. In these mice treated with DHE, we did not use brain tissue for estimating superoxide levels using lucigenin-enhanced chemiluminescence because DHE was reported to be neuroprotective and reduce superoxide in mice after stroke (Yu et al, 2003). Fresh, unfixed brains were frozen in TBS compound (Triangle Biomedical Sciences, Durham, NC, USA). Coronal sections (20-μm thick) were cut in a cryostat and placed on glass slides, and images were obtained using the Bio-Rad MRC-1024 laser (krypton/argon)-scanning confocal microscope. The fluorescence excitation/emission spectra for ethidium bromide used during the imaging process were 488 and 610 nm, respectively. Fluorescence was detected with a 585-nm long-pass filter.
Statistics
Results are expressed as mean±s.e.m. Repeated-measures ANOVA followed by Scheffé test was used for comparison of multiple groups. Mann—Whitney's U test was used for comparison of two groups. A probability value of P<0.05 was considered significant.
Results
Spontaneous Intracranial Hemorrhage
R+/A+ mice presented neurologic signs 57±13 (mean±s.e.m.) days after the start of feeding high-salt diet and
Systolic blood pressure
In R+/A+ mice with ICH, SBP was higher at baseline (4 months of age) than in control mice. In R+/A+ mice without ICH, SBP also tended to be higher at baseline than in control mice (P=0.07) (Table 1). R+/A+ mice developed a progressive increase in SBP compared with control mice within 4 to 8 weeks of treatment with high-salt diet and
Systolic blood pressure in control mice, R+/A+ mice without ICH, and R+/A+ mice with ICH

ICH, intracranial hemorrhage; R+/A+, human renin and human angiotensinogen expressing mice.
Values are mean±s.e.m.; number of mice in parentheses.
R+/A+ ICH(−), R+/A+ mice without ICH; R+/A+ IVH(+), R+/A+ mice with ICH.
∗P<0.05, ∗∗P<0.01 compared with control mice at each time point.
†P<0.01 compared with R+/A+ ICH(−) at each time point.
Histometric analysis of the brain
All R+/A+ mice with neurologic signs had multiple ICH, shown in H&E- and DAB-stained sections (Figures 1A to 1H). Hemorrhagic legions were mainly located in the brain stem, although ICHs were distributed widely in the brain including the cerebral cortex, basal ganglia, and cerebellum (Figure 1K). R+/A+ mice with neurologic signs also had a small number of ischemic infarcts in the brain (Figure 1L). Most ischemic infarcts were separated from hemorrhagic lesions (Figure 1I), both longitudinally and horizontally, but a few ischemic infarcts colocalized with hemorrhagic lesions (Figure 1J). None of the control mice or R+/A+ mice without signs of stroke had ICH or ischemic infarcts.
Superoxide levels
In R+/A+ mice with ICH, lucigenin-enhanced chemiluminescence signals in each quarter of brain were significantly attenuated by preincubation with tiron (Figure 2). In R+/A+ mice without ICH, tiron reduced chemiluminescence signals in three of four quarter-parts of the brain (Figure 2). In control mice, chemiluminescence signals were reduced only in the posterior quarter of the brain (Figure 2). Diphenyliodinium significantly reduced chemiluminescence signals in each quarter of the brain in R+/A+ mice with and without ICH. Chemiluminescence signals were decreased in posterior half of the brain in control mice (Figure 2).
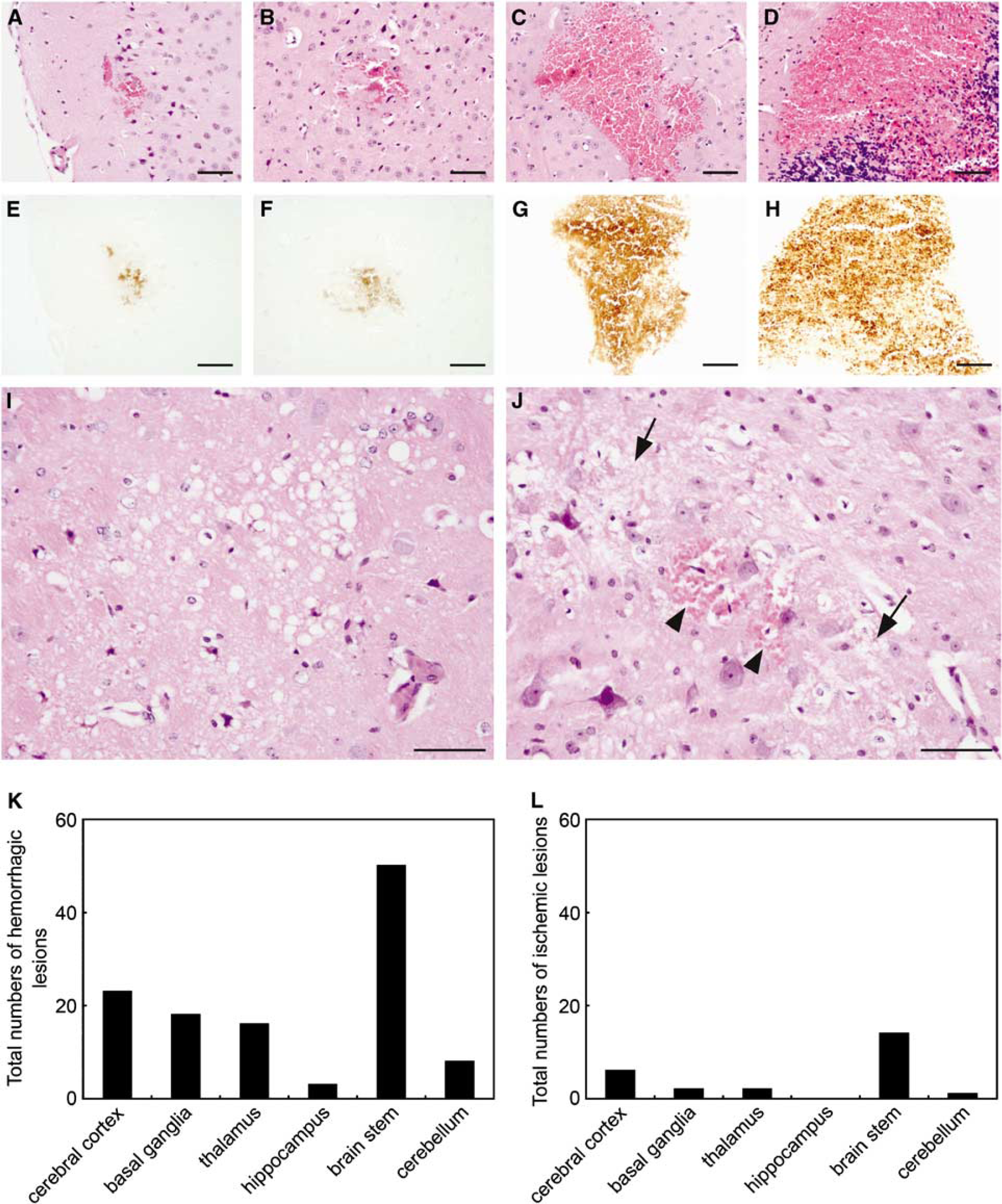
Histologic changes in brains of R+/A+ mice with signs of stroke. (
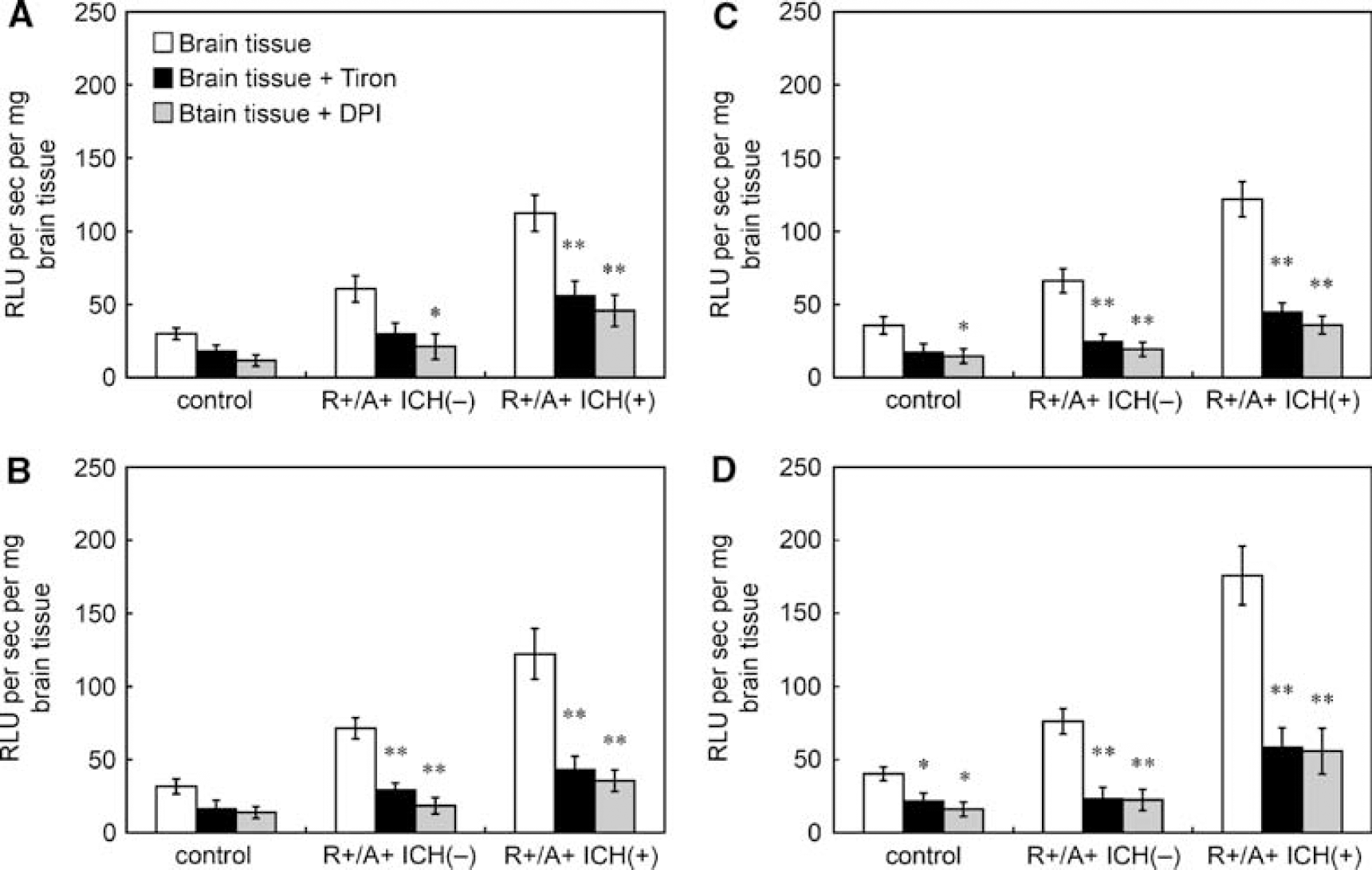
Effects of tiron and DPI on lucigenin-enhanced chemiluminescence signals in brain tissue of control mice, R+/A+ mice without ICH (R+/A+ ICH(−)), and R+/A+ mice with ICH (R+/A+ ICH(+)). (
Levels of superoxide under basal conditions, as measured using tiron-inhibitable chemiluminescence, were significantly higher in R+/A+ mice with ICH than in control mice and R+/A+ mice without ICH (Figure 3). The increase in superoxide was observed in each quarter of the brain in R+/A+ mice with ICH. In the most posterior quarter of the brain of R+/A+ mice with ICH, where spontaneous ICH was frequently observed, superoxide levels were greater than in the other parts of the brain (P<0.01). In R+/A+ mice without ICH, superoxide levels were also significantly higher than in control mice in each quarter of the brain (Figure 3).
NAD(P)H oxidase activity
NAD(P)H oxidase activity, estimated as levels of superoxide using tiron-inhibitable chemiluminescence after adding NADPH, was higher in R+/A+ mice with ICH than in control mice and in R+/A+ mice without ICH (Figure 4). In R+/A+ mice with ICH, NAD(P)H oxidase activity of the most posterior quarter of brain was significantly higher than in the anterior and second quarter-parts of the brain (P<0.05). NAD(P)H oxidase activity was also higher in R+/A+ mice without ICH than in control mice in all the quarters of the brain.
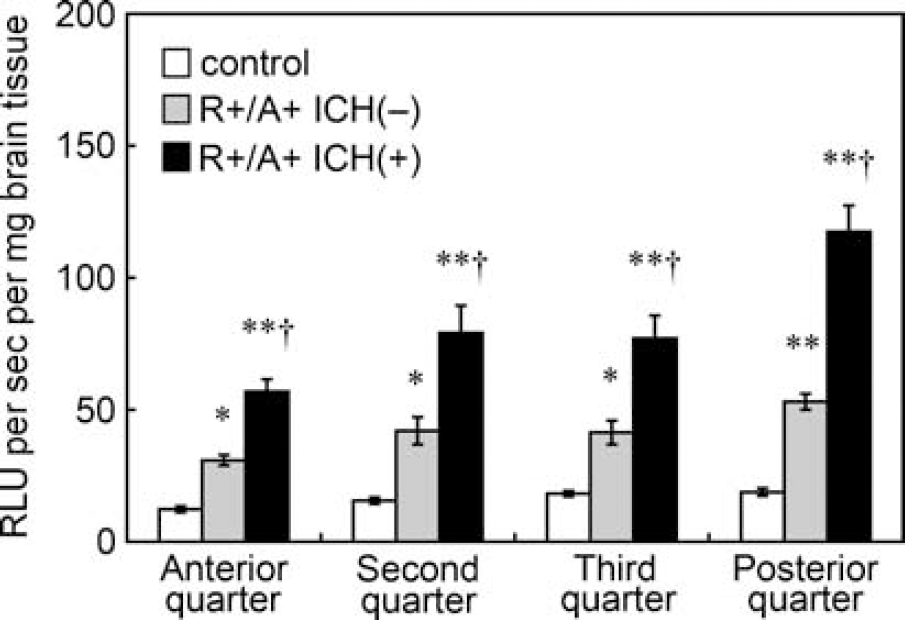
Basal levels of superoxide, calculated from Figure 2 as tiron-inhibitable signal, in brain tissue of control mice, R+/A+ mice without ICH (R+/A+ ICH(−)), and R+/A+ mice with ICH (R+/A+ ICH(+)). Values are means±s.e.m. (n=8 in each group); RLU, relative light units. ∗P<0.05, ∗∗P<0.01 versus respective control mice. †P<0.01 versus respective R+/A+ mice without ICH.
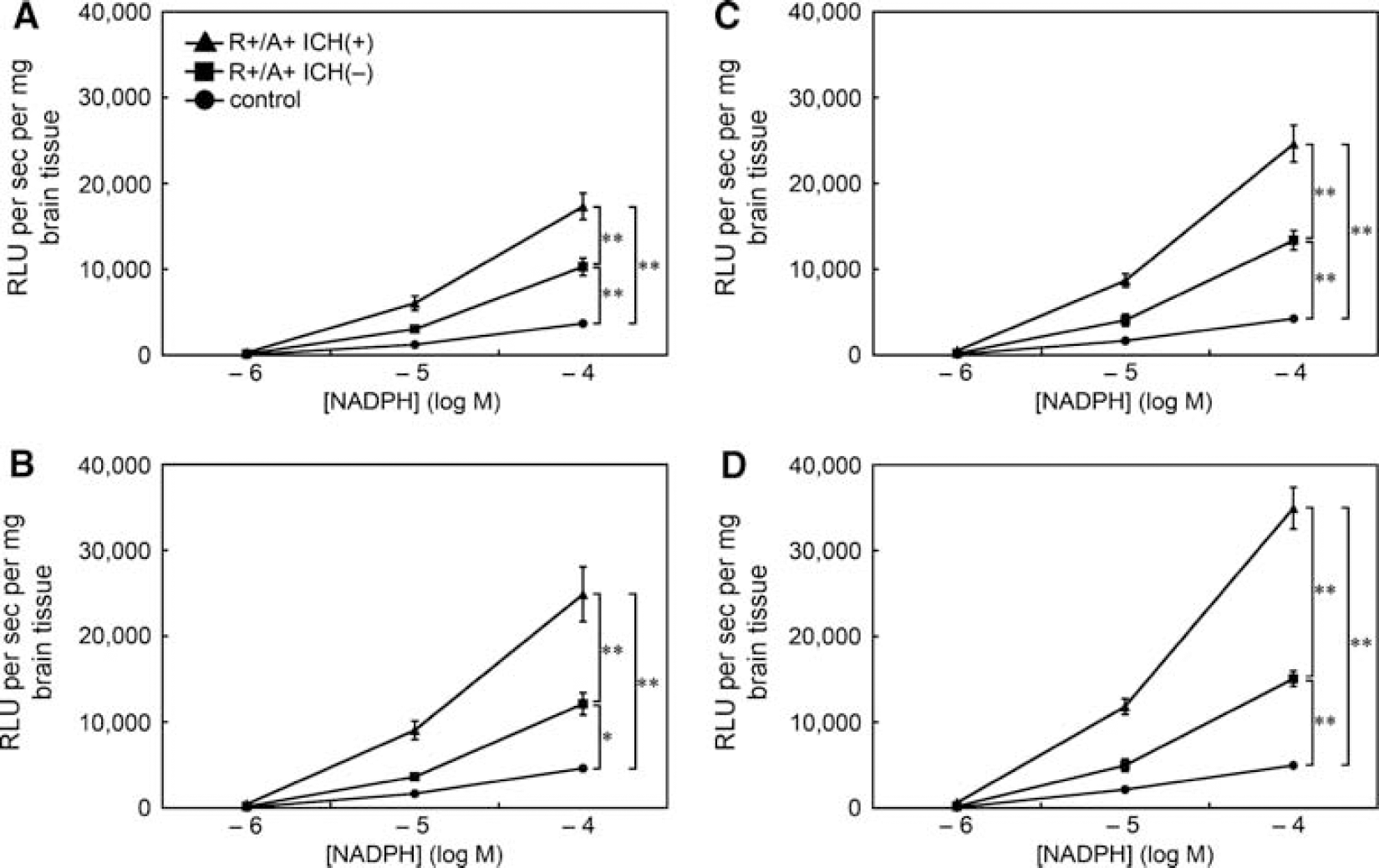
NAD(P)H oxidase activity in brain tissue of control mice, R+/A+ mice without ICH (R+/A+ ICH(−)), and R+/A+ mice with ICH (R+/A+ ICH(+)). (
To examine the mechanisms that contributed to increased basal levels of superoxide in R+/A+ mice with and without ICH, we measured the expression of the catalytic subunits of NAD(P)H oxidase (Nox2 and Nox4). Measurements were made in the posterior quarter of the brain in which basal levels of superoxide were greater than in other parts of the brain in R+/A+ mice with ICH. In contrast to evidence for increased activity of NAD(P)H oxidase in R+/A+ mice with and without ICH, quantitative real-time reverse-transcription PCR showed no significant difference in the expression of Nox2 and Nox4 among the three groups (data not shown).
Striatal Blood Injection
Superoxide levels
We used two methods to detect superoxide after injection of blood in the striatum: tiron-inhibitable chemiluminescence and DHE staining. In wild-type mice and in wild-type mice treated with apocynin, lucigenin-enhanced chemiluminescence was greater in slices of brain after the injection of blood than in slices after the injection of aCSF (Figure 5A). In gp91phox-deficient mice, there was no significant difference in lucigenin-enhanced chemiluminescence between brain sections after injection of blood and after injection of aCSF (P=0.09). The increase in chemiluminescence after injection of blood was, however, significantly reduced by tiron or DPI in all groups (Figure 5A).
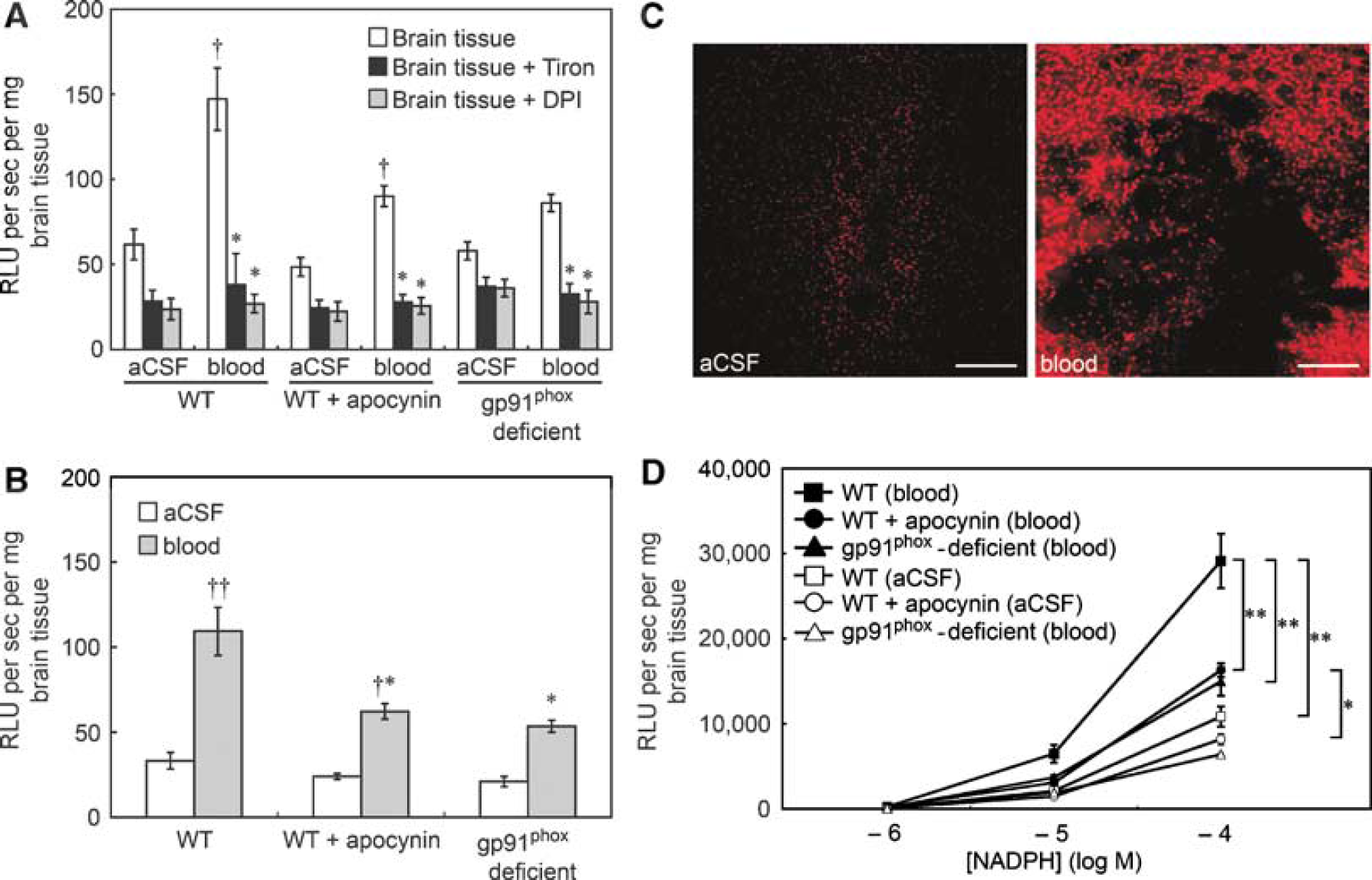
(
In wild-type mice, basal levels of superoxide, as measured using tiron-inhibitable chemiluminescence, were significantly higher in the blood-injected slices of brain than in aCSF-injected slices (Figure 5B). There was no significant difference in basal levels of superoxide in blood-injected brain between male and female wild-type mice in a study of eight male and four female mice (data not shown). In wild-type mice treated with apocynin, basal superoxide levels were also higher in blood-injected brain than in aCSF-injected brain, but the increase in superoxide was significantly attenuated compared with the wild-type mice that did not receive apocynin (Figure 5B). The increase in levels of superoxide after injection of blood was less in gp91phox-deficient mice than in wild-type mice, and there was no significant difference in levels of superoxide between brain sections after injection of blood and after injection of aCSF (P=0.20) (Figure 5B).
Consistent with results obtained with lucigenin-enhanced chemiluminescence, DHE staining showed higher levels of superoxide in blood-injected brain than in aCSF-injected brain in wild-type mice. Increases in DHE fluorescence were restricted to the area of needle insertion in aCSF-injected brain, whereas enhanced DHE fluorescence was widely distributed around the intraparenchymal hematoma after injection of blood (Figure 5C).
NAD(P)H oxidase activity
NAD(P)H oxidase activity was higher after injection of blood than after aCSF in both apocynin-treated wild-type mice and in mice that did not receive apocynin (Figure 5D). The increase in NAD(P)H oxidase activity after injection of blood was largely prevented by apocynin (Figure 5D). In gp91phox-deficient mice, the increase in NAD(P)H oxidase activity after injection of blood was less than in wild-type mice, and there was no significant difference in NAD(P)H oxidase activity between brain sections after injection of blood and after injection of aCSF (P=0.11) (Figure 5D).
In contrast to the evidence for increased activity of NAD(P)H oxidase in brain sections after injection of blood, expressions of Nox2 mRNA and Nox4 mRNA were not different between brain sections after injection of blood and after injection of aCSF in wild-type mice (data not shown).
Discussion
There are several major findings in this study. First, basal superoxide levels were significantly higher in brains from R+/A+ mice treated with high-salt diet and
Experimental Model of Spontaneous Intracranial Hemorrhage
We have previously observed spontaneous ICH in R+/A+ mice treated with high-salt diet and
Superoxide and Hypertension
Treatment of R+/A+ mice with high-salt diet and
NAD(P)H oxidases are a major enzymatic source of superoxide in the systemic vasculature, and activity of NAD(P)H oxidase is increased in several pathophysiological states, including hypertension (Didion et al, 2002; Griendling et al, 2000; Rajagopalan et al, 1996a). NAD(P)H oxidase is also expressed and activated in cerebral blood vessels during chronic hypertension (Didion and Faraci, 2003; Girouard et al, 2007; Kazama et al, 2004; Paravicini et al, 2004).
We observed in this study that superoxide is markedly reduced by preincubation of brain tissue with DPI, an inhibitor of NAD(P)H oxidase. We are aware of limitations of use of DPI as an NAD(P)H oxidase inhibitor, and the suggestion that DPI also inhibits electron flow through other enzymes, including mitochondrial respiratory chain complex I, NADH reductase, and NOS (Li et al, 2003). We cannot exclude the possibility that mitochondrial enzyme is a major source of increased oxidative stress observed in our study. The possibility that NOS is the source of increased oxidative stress is unlikely in our study, because R+/A+ mice received
Role of Superoxide in Spontaneous Intracranial Hemorrhage
A major finding in this study was that basal levels of superoxide and NAD(P)H oxidase activity in brain tissue were greater in R+/A+ with ICH than in R+/A+ mice without ICH. Although there is no direct evidence that superoxide contributes to spontaneous ICH, accumulating evidence suggests that oxidative stress may be involved in spontaneous ICH.
Chronic oxidative stress, especially from activation of NAD(P)H oxidase, produces dysfunction of endothelium and smooth muscle in blood vessels and induces cell death (Bhunia et al, 2002; Burlacu et al, 2001; Didion and Faraci, 2003). Furthermore, activity of MMPs is upregulated by oxidative stress, including superoxide (Gu et al, 2002; Rajagopalan et al, 1996b). The MMPs are a family of zinc endopeptidases involved in the degradation of basal lamina and extracellular matrix (Woessner, 1991). They are also reported to disrupt the blood—brain barrier in experimental ischemia—reperfusion model with transient middle cerebral artery occlusion (Yang et al, 2006). Expression and activity of MMP-9 are increased in cerebral microvessels in rats with chronic hypertension (Liebetrau et al, 2005). In addition, permeability of the blood—brain barrier was increased when blood pressure was greatly, but not moderately, elevated (Tuor et al, 1986; Nukhet Turkel and Ziya Ziylan, 2004). In this study, we observed that ICH occurred in R+/A+ mice with signs of stroke, in which there were greater increases in blood pressure in most mice (Table 1) and in basal levels of superoxide in all cases (Figure 3), than in other R+/A+ mice without hemorrhages, in which there were only moderate increases in blood pressure and basal level of superoxide. Thus, it seems reasonable to suggest that increased MMP activity, through oxidative stress associated with severe hypertension perhaps beyond a threshold level of blood pressure, may facilitate proteolysis of extracellular matrix components of the basement membrane, contributing to the pathogenesis of ICH. Therefore, we speculate that increases in superoxide in R+/A+ mice treated with high-salt diet and
Oxidative Stress after Intracranial Hemorrhage
The increase in superoxide levels in R+/A+ mice with ICH may also reflect brain injury after ICH. Enhanced superoxide levels resulting from activation of NAD(P)H oxidase have been reported to be associated with brain injury after ICH in experimental studies (Tang et al, 2005; Wang and Tsirka, 2005). Increased superoxide levels were shown in the perihematoma region after injection of bacterial collagenase, and the authors suggested that superoxide may mediate ICH injury (Wang and Tsirka, 2005). Expression of the gp91phox subunit of NAD(P)H oxidase was upregulated in brain after injection of collagenase in wild-type mice (Tang et al, 2005). In addition, brain edema, neurologic deficit, and mortality rate were reduced after injection of collagenase in gp91phox-deficient mice, which suggested that oxidative stress from activation of NAD(P)H oxidase contributed to brain injury (Tang et al, 2005). In those studies, bacterial collagenase was injected in the brain. Collagenase dissolves extracellular matrix and blood vessel lamina, and produces ICH (Brown et al, 1995). Collagenase also produces inflammation, which may contribute to oxidative stress, because inflammatory cells can generate superoxide (Green et al, 2001; Wang and Tsirka, 2005).
To determine whether ICH itself, in the absence of bacterial collagenase, produces oxidative stress in the brain, we examined superoxide and NAD(P)H oxidase activity after injection of blood in the striatum. Basal superoxide levels and NAD(P)H oxidase activity were increased after injection of blood in wild-type mice. The increase in superoxide and NAD(P)H oxidase activity after injection of blood was significantly, but not completely, reduced in wild-type mice treated with apocynin and in gp91phox-deficient mice. For several reasons, we are aware of the possibility that an NAD(P)H oxidase containing a Nox subunit other than Nox2, as well as other sources beside NAD(P)H oxidase, may contribute to increases in chemiluminescence signals in brain sections after injection of blood. First, DPI significantly reduced lucigenin-enhanced chemiluminescence signals in blood-injected brain tissue of wild-type mice treated with apocynin and in brain tissue from gp91phox-deficient mice, as well as in blood-injected brain tissue of wild-type mice. Second, apocynin does not inhibit activation of Nox4, as Nox4 does not require the cytosolic subunit p47phox for its activation (Infanger et al, 2006). Third, endothelial NOS (eNOS) could also produce superoxide (Vásquez-Vivar et al, 1998), and DPI also inhibits eNOS-mediated generation of superoxide (Li et al, 2003). Nevertheless, these findings suggest that intraparenchymal blood per se produces an increase in superoxide due, at least in part, to activation of the gp91phox subunit of NAD(P)H oxidase.
We cannot exclude the possibility that brain infarction may affect basal levels of superoxide in R+/A+ mice with ICH. The levels of superoxide were reported to increase in cerebral ischemic lesions produced by permanent as well as transient middle cerebral artery occlusion (Fujimura et al, 1999; Noshita et al, 2003). The ischemic lesions in R+/A+ mice with ICH were, however, far smaller than the ischemic lesions produced by middle cerebral artery occlusion. In addition, the number of ischemic lesions was much less than hemorrhagic lesions in R+/A+ mice with ICH. Thus, it is likely that ischemic lesions have much less effect than hemorrhagic lesions on basal levels of superoxide in R+/A+ mice with ICH.
Limitations of Study
There are methodological limitations of the present study. First, angiotensin II without hypertension generates superoxide in vascular cells in vitro (Berry et al, 2000; Zhang et al, 1999). In addition, because long-term administration of
Footnotes
Acknowledgements
We thank the University of Iowa Transgenic Facility for genotyping R+/A+ mice used in this study. We also thank Arlinda A LaRose for typing the manuscript and Robert M Brooks II for technical assistance.
