Abstract
Netrin-1 is a critical molecule for axonal pathfinding during embryo development, and because of its structural homology to the endothelial mitogens, it may share its effects on vascular network formation. Using an adeno-associated viral netrin-1 vector (AAV-NT-1) gene transfer, we demonstrated that netrin-1 was able to stimulate the proliferation and migration of human cerebral endothelial cells (HCECs) and human aortic smooth muscle cells (HASMCs) compared with the control (P < 0.05), and could also promote HCEC tube formation on matrigel (P < 0.05) in vitro. Moreover, netrin-1 hyperstimulation could promote focal neovascularization (P < 0.05) in the adult brain in vivo. Unlike VEGF-induced microvessel increase, netrin-1-induced newly formed vessels showed an artery-like phenotype, with an intact endothelial cell monolayer surrounded by multiple cell layers, including smooth muscle cells and an astrocyte-connected outer layer. Our findings suggest that netrin-1 plays an important role in promoting blood vessel formation in the adult rodent central nervous system, and could have broad implication in cerebrovascular development and remodeling.
Keywords
Introduction
The nervous and vascular systems are organized in a strikingly parallel way, consisting of highly branched, ramified networks (Martin and Lewis, 1989). Accumulated evidence indicates that crosstalk between these two systems occurs at the molecular level in both development and response to injury (Weinstein, 2005). For example, vascular endothelial growth factor (VEGF), a potent endothelial cell mitogen and morphogen, drives angiogenesis in a wide variety of tissues and lesions (Cebe-Suarez et al, 2006) and, in addition, plays an important role in neural development, axonal outgrowth, cell survival, and neural progenitor attraction (Jin et al, 2006; Rosenstein et al, 2003). Conversely, neuroregulatory signals and neuronal guidance factors also influence vascular development. For example, the neural regulator notch is involved in both nervous system development and vascular morphogenesis and differentiation (Louvi and Artavanis-Tsakonas, 2006). The Robo receptor and Slit ligand families interact to initiate signaling cascades that repel growing axons and also provide a repulsive cue to migrating endothelial cells (Park et al, 2003).
Netrin-1 (NT-1), one of the major neuronal guidance cues, was first isolated from chick brain (Kennedy et al, 1994). It belongs to a family of homologs that includes netrin-1, −2, −G1, −G2, and −4/b. Netrin-1 receptors include deleted in colorectal carcinoma (DCC), uncoordinated 5H (UNC5H) family members, and other molecules (Cirulli and Yebra, 2007; Leonardo et al, 1997; Stein et al, 2001). By binding to these receptors, NT-1 functions as a chemotropic or repulsive factor that mediates axon outgrowth, axon orientation, and neuronal migration during development (Forcet et al, 2002; Kennedy, 2000). It is also crucial to the survival of UNC5H-and DCC-expressing neurons (Llambi et al, 2001; Mehlen and Llambi, 2005). Netrin-1 and its homologs have been extensively studied in neuronal development, but their effects on the vascular system are still controversial (Lu et al, 2004; Park et al, 2004). Identifying the effect of NT-1 on angiogenesis or vascular remodeling in the adult brain is a crucial first step for its future application. If exogenous NT-1 hyperstimulation promotes angiogenesis or neovascularization in the adult brain, it can provide a unique therapeutic approach to chronic cerebral ischemia or brain injury-caused neurovascular remodeling, where angiogenesis, arteriogenesis, and even neurogenesis are required.
On the basis of this view, we have constructed a novel adeno-associated viral netrin-1 vector (AAV-NT-1) to test the effects of NT-1 on neovascularization in the adult mouse brain. We first examined if NT-1 overexpression would induce angiogenic changes such as cell proliferation, migration, and tube formation in human cerebral endothelial cells (HCECs) and human aortic smooth muscle cells (HASMCs) in vitro. We then examined if AAV-NT-1 would lead to NT-1 overexpression and promote new vessel formation in the adult mouse brain in vivo.
Materials and methods
Adeno-Associated Virus Vector Construction and Production
Adeno-associated virus vectors were generated according to a previous method but with minor modifications (Su et al, 2000). We first inserted the chicken netrin-1 cDNA (a gift from Dr Tessier-Lavigne, Genentech Inc., South San Francisco, CA, USA) between two ITRs of pAAV-MC plasmid (Strategene, La Jolla, CA, USA) to generate pAAV-netrin-1. Cytomegalovirus promoter was used to control gene expression in this vector (Figure 1A). The recombinant plasmid pAAV-netrin-1 was cotransfected with two helper plasmids into HEK293 cells by the calcium phosphate precipitation method. One helper plasmid, pHelper, contains adenoviral VA, E2A, and E4 regions, which mediate AAV vector replication. The other, pAAV-RC, has rep gene and cap gene of AAV serotype 1. Cell lysates were produced using three freeze-thaw cycles 3 days after gene transfection. Adeno-associated virus vectors were purified by CsCl density gradient centrifugation. Viral titers were determined by dot blot analysis of the DNA content. Adeno-associated virus-lacZ (AAV-lacZ) was prepared as a control. To test the infection efficiency, AAV vectors were added to serum-starved HEK293 cells at the ratio of 500 and 1000 particles per cell. The infected cells were incubated at 37°C in 5% CO2/95% air for 24 h, and gene expression was analyzed 48 h after the AAV transfection. The conditioned medium was collected for the experiments as described below.
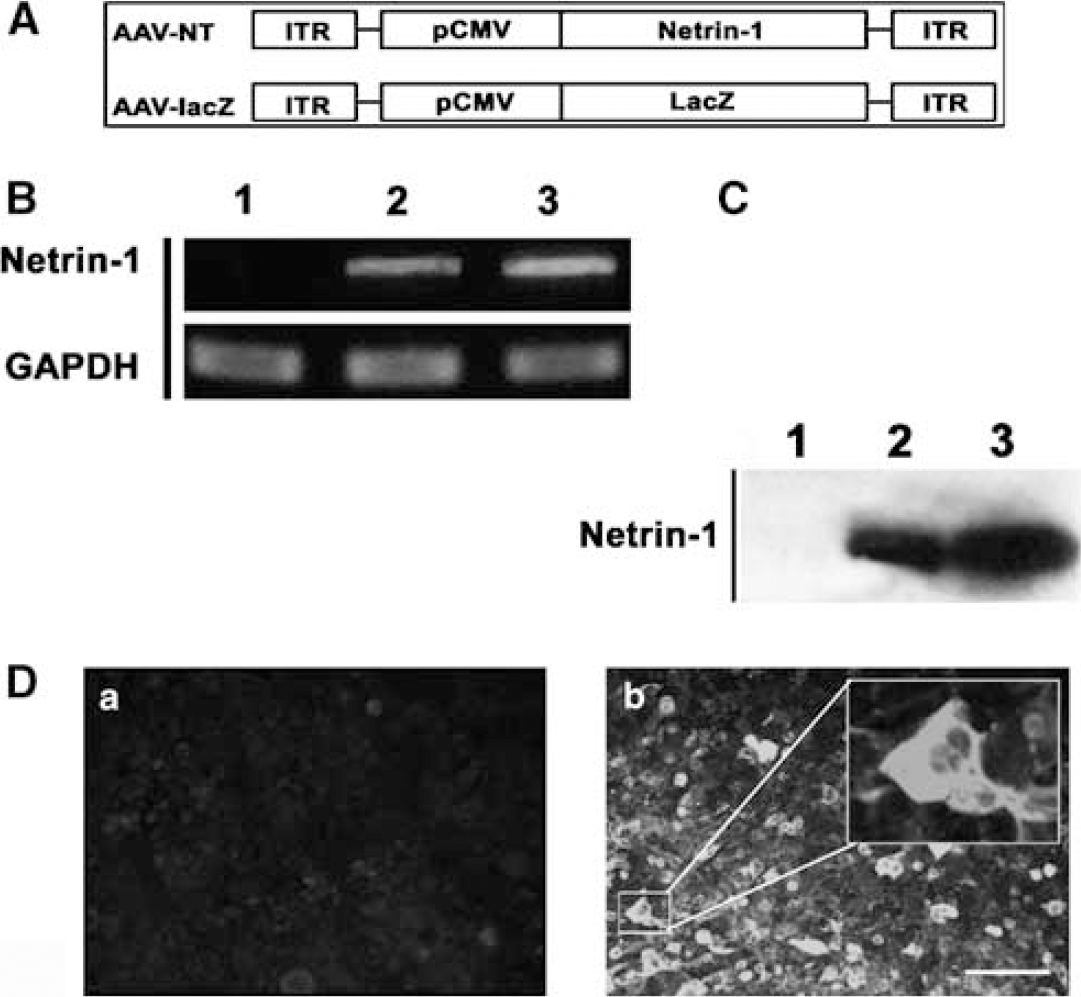
Construction and production of AAV-NT-1. (
In Vitro Assay
HCECs and HASMCs were purchased from Cell Systems (St Katharinen, Germany). The HASMCs were cultured in Dulbecco's modified Eagle's medium supplemented with 10% fetal bovine serum, and the HCECs were cultured in endothelial basal medium 2 (EBM2) supplemented with Single Quote (Cambrex, East Rutherford, NJ, USA) on gelatin (Sigma, St Louis, MO, USA) coated plates at 37°C in 5% CO2 and 95% ambient mixed air, as in our previous study (Yao et al, 2006). The culture medium was changed every 3 days.
For the cell proliferation assay, cells were seeded onto 96-well gelatin coated plate (1 times 104) in EBM2/Single Quote medium. After starvation, bovine serum albumin (BSA) (Sigma), recombinant human VEGF (R&D Systems, Minneapolis, MN, USA), chicken netrin-1 (R&D Systems), or conditioned medium was added to the medium (EBM containing 0.1% BSA for HCECs and Dulbecco's modified Eagle's medium with 0.1% BSA for HASMCs) at the indicated concentration. After treatment for 48 h, cells were labeled with BrdU for 2 h (BrdU Kit; Roche, Mannheim, Germany) and measured for absorbance at 450 nm (reference wavelength 690 nm) after 10 mins using an ELISA reader (E max; Molecular Devices, Sunnyvale, CA, USA).
Cell migration was assayed by using 24-well Tanswell cell culture chambers with 8.0 μm pore polycarbonate filter inserts (Costar, San Diego, CA, USA). Serum-starved HCECs and HASMCs were suspended in the medium with 0.1% BSA at a concentration of 2.5 times 105/mL. A 100-μL cell suspension was applied to precoated insert filters. Test factors were added in serum-free medium and placed in the lower chamber. After incubation at 37°C for 18 h, cells on the upper side of the filter were scraped and the migrated cells on the lower side were fixed and stained with hematoxylin. The membrane was mounted on a slide and examined under a microscope. Migration was quantified by measuring the stained cell in five random areas per membrane.
A matrigel tube formation assay was performed as described by Lee et al (1999). A 96-well culture plate was coated with 50 μL of growth factor-reduced matrigel per well, and then allowed to polymerize for 30 mins at 37°C. Total 2 times 104 cells/well of starved HCECs were seeded on the matrigel in 0.1% BSA/EBM2 medium with testing factors, and incubated for 18 h at 37°C. Pictures were taken at × 4 magnification with a digital output camera (Olympus DP11, Melville, NY, USA). Total tube length was measured by using NIH Image 1.63 software (NIH, Bethesda, MD, USA).
In Vivo Assay
All animal procedures were performed according to a protocol approved by the Institutional Animal Care and Use Committee of the University of California, San Francisco. Fifty-four adult male CD-1 mice weighing 30 to 35 g (Charles River Laboratories, Wilmington, MA, USA) were used for the AAV vector injection. The mice were divided into three groups: AAV-NT-1 (n = 18), AAV-lacZ (n = 18, as a viral control), and saline injected (n = 18, as a control). The animals were killed after 1, 3, and 5 weeks; gene expression and the number and pattern of microvessels in each group were examined using immunohistochemistry.
After anesthesia, the mice were placed on a stereotactic frame with a mouth holder (David Kopf Instruments, Tujunga, CA, USA). A burr hole was drilled in the pericranium 2 mm lateral to the sagittal suture and 1 mm posterior to the coronal suture. A 10-μL Hamilton syringe was stereotactically inserted into the lateral ventricular and caudate putamen. An 8-μL viral suspension (AAV-NT-1 or AAV-lacZ) containing 8 times 109 particles was injected into the right hemisphere at a rate of 0.2μL/min (Yang et al, 1997). Control animals received the same amount of saline injection. The needle was withdrawn 15 mins after injection, the bone hole sealed, and the wound closed. The animals were allowed to recover and return to their home cages.
The number of microvessels was assessed by CD-31 staining (Lee et al, 2004; Yang et al, 2003). Two coronal brain sections, 1 mm anterior and 1 mm posterior from the needle track, were chosen. Images in three areas immediately adjacent (left, right, and bottom) to the needle track were taken using a × 10 objective microscopy. The total number of microvessels and the enlarged microvessels (defined as vessels with diameter greater than 10 μm) were calculated as the mean of the microvessel counts obtained from the three images.
Immunohistochemistry
Frozen brain sections were blocked with 5% goat serum after primary antibody incubation at 4°C overnight. The antibodies included goat anti-chicken netrin-1 (1:200 dilution; R&D Systems), rat anti-mouse CD-31 (1:200 dilution; BD Bioscience, San Jose, CA, USA), mouse anti-mouse NeuN (1:500 dilution; Chemicon, Temecula, CA, USA), anti-SM-α actin (1:200 dilution; Chemicon), and Cy3-conjugated glial fibrillary acidic protein (GFAP; 1:1000 dilution; Sigma). The sections were incubated with avidin—biotin enzyme reagent (ABC kit; Vector Labs, Burlingame, CA, USA) for 1 h, and were washed and reacted with stable DAB after hematoxylin counterstaining. For fluorescent staining, the sections were incubated for 1 h with fluorescence-conjugated secondary antibodies at room temperature. Sections were observed through a fluorescent microscope (Nikon Microphot-SA). Appropriate positive and negative controls were performed for each batch of slides.
Western Blot Analysis
Protein from HEK293 cells was extracted with the RIPA buffer (Invitrogen, Carlsbad, CA, USA). The protein concentration was determined using the BCA protein assay kits (Pierce, Rockford, IL, USA). An equal volume of proteins (40 μg) was loaded on 10% acrylamide gel for electrophoresis. Subsequently, proteins were electro-blotted onto a polyvinylidene fluoride membrane (Bio-Rad, Richmond, CA, USA). The membrane was blocked in 5% nonfat milk Tris-buffered saline with 0.1% Tween-20 for 1 h, then immunoprobed with anti-netrin-1 antibody (1:400 dilution; R&D Systems) overnight at 4°C. After washing, the membrane was incubated with HRP-conjugated anti-rabbit antibody (1:10,000 dilution; Pierce Inc.), and then reacted with an enhanced ECL (Pierce). Finally, the membrane was exposed to Kodak film.
Statistical Analysis
All data are presented as mean ± s.d. Parametric data among different groups were analyzed using a one-way analysis of variance followed by the Fisher's PLSD test. A probability value of less than 5% was considered to be statistically significant.
Results
Adeno-Associated Virus Vector-Mediated Netrin-1 Expression In Vitro and In Vivo
To determine whether AAV-NT-1 was capable of mediating netrin-1 expression, we examined netrin-1 expression both in the HEK293 cells infected with AAV-NT-1 in vitro and in the mouse brain in vivo. After infection of HEK293 cells with AAV-NT-1, netrin-1 was detected in both RNA (590 bp; Figure 1B) and protein levels (Figures 1C and 1D). When AAV-NT-1 or AAV-lacZ was injected stereotactically into the adult mouse brain, numerous netrin-1 positive cells were detected around the needle track after 1, 3, and 5 weeks of gene transfer. No netrin-1-positive cells were detected in the AAV-lacZ-transduced brain after the same time points. Netrin-1 expression was maintained for at least 5 weeks after AAV-NT-1 transduction (data not shown). Double-label immunostaining showed that netrin-1-positive cells were mainly expressed in the neurons and astrocytes rather than in the endothelial cells (Figures 2A–2C).
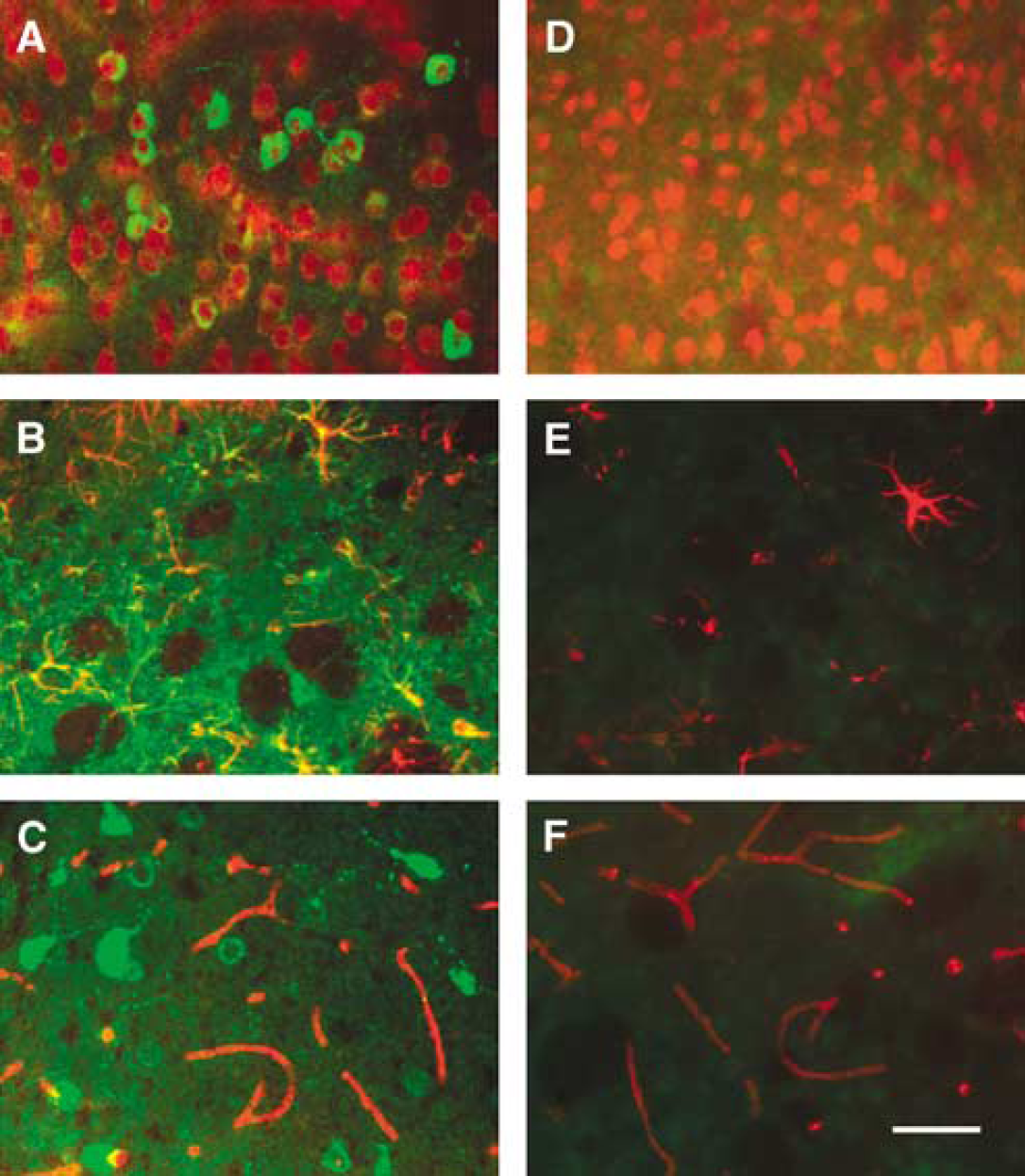
Netrin-1 was expressed in astrocytes and neurons. Photomicrographs show NT-1 expression in the mouse brain 3 weeks after AAV-NT-1 (
Netrin-1-Induced Angiogenic Changes In Vitro
To determine whether netrin-1 stimulated HCEC and HASMC proliferation, we first performed a BrdU incorporation assay. Proliferation of HCECs and HASMCs was increased in cultures treated with AAV-NT-1 conditioned medium compared with the cells treated with AAV-lacZ-conditioned medium (Figures 3A and 3C; P < 0.05). To determine if netrin-1 was directly responsible for this effect, we further examined the effect of netrin-1 protein on HCEC and HASMC proliferation. The results showed that netrin-1 protein could stimulate HCEC and HASMC proliferation in a dose-dependent manner with an optimal effect at 50 ng/mL (Figures 3B and 3D). The effect of netrin-1 on stimulation of HCEC and HASMC proliferation was similar to that of VEGF in vitro.
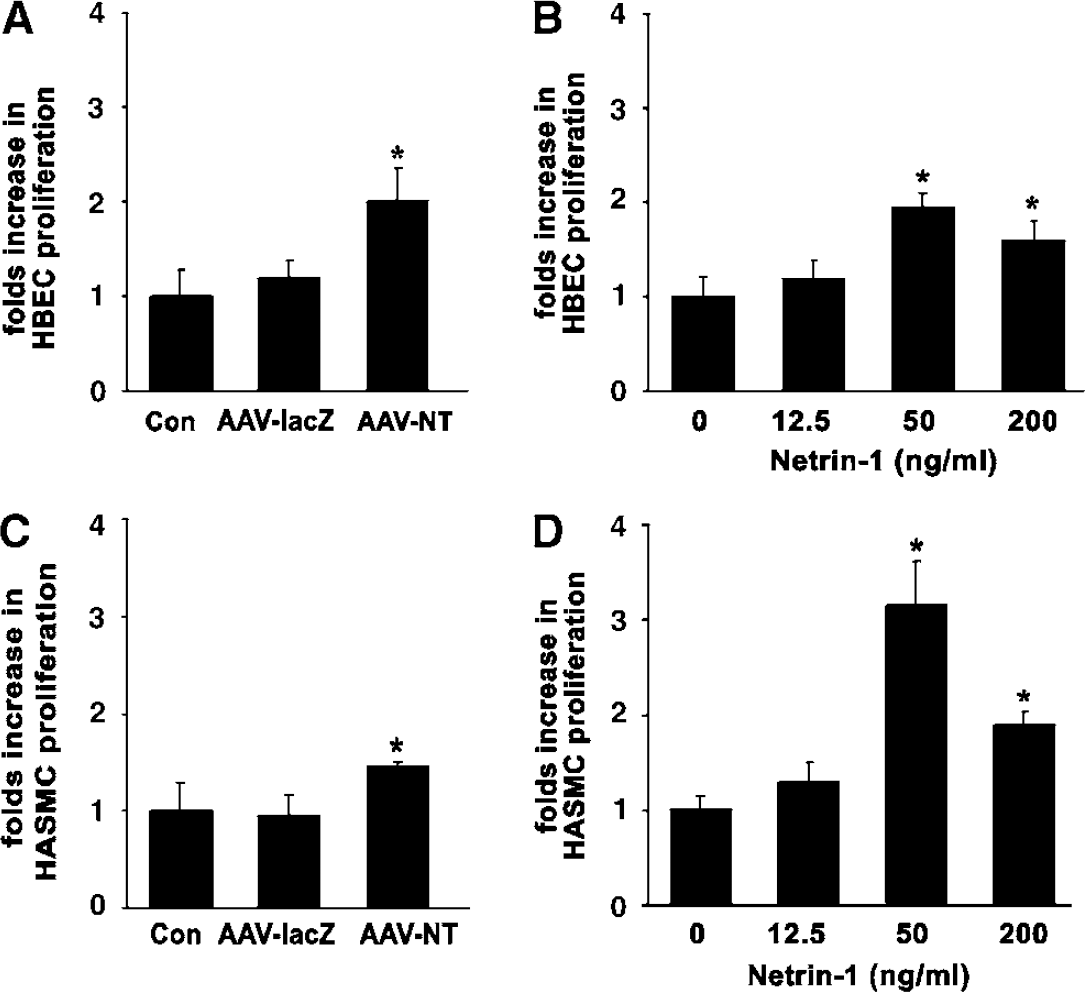
Netrin-1 stimulated HCEC and HASMC proliferation. Bar graphs show BrdU incorporation of HCEC and HASMC after application of NT-1. VEGF (20 ng/mL) acted as a positive control and AAV-lacZ-conditioned medium or PBS acted as negative controls. Data are presented as fold increase in BrdU incorporation relative to the negative control cultures. After 48 h of treatment, AAV-NT-1-conditioned medium significantly increased HCEC (
To examine if netrin-1 enhanced HCEC and HASMC migration, we counted cells moving through an insert filter in a chamber filled with testing factors. The AAV-NT-1-conditioned medium significantly increased HCEC and HASMC migration compared with the AAV-lacZ medium (Figures 4A and 4C; P < 0.01). Again, to determine if netrin-1 was directly responsible for this effect, we applied netrin-1 protein; the result showed that netrin-1 protein could induce HCEC and HASMC migration. Both cell types responded in a dose-dependent manner with optimal effects observed at 50 ng/mL for HASMCs and ≥ 200 ng/mL for HCECs (Figures 4B and 4D). Although not statistically significant, the effect of netrin-1 on promotion of HCEC and HASMC migration appeared stronger than the effect of VEGF in vitro.
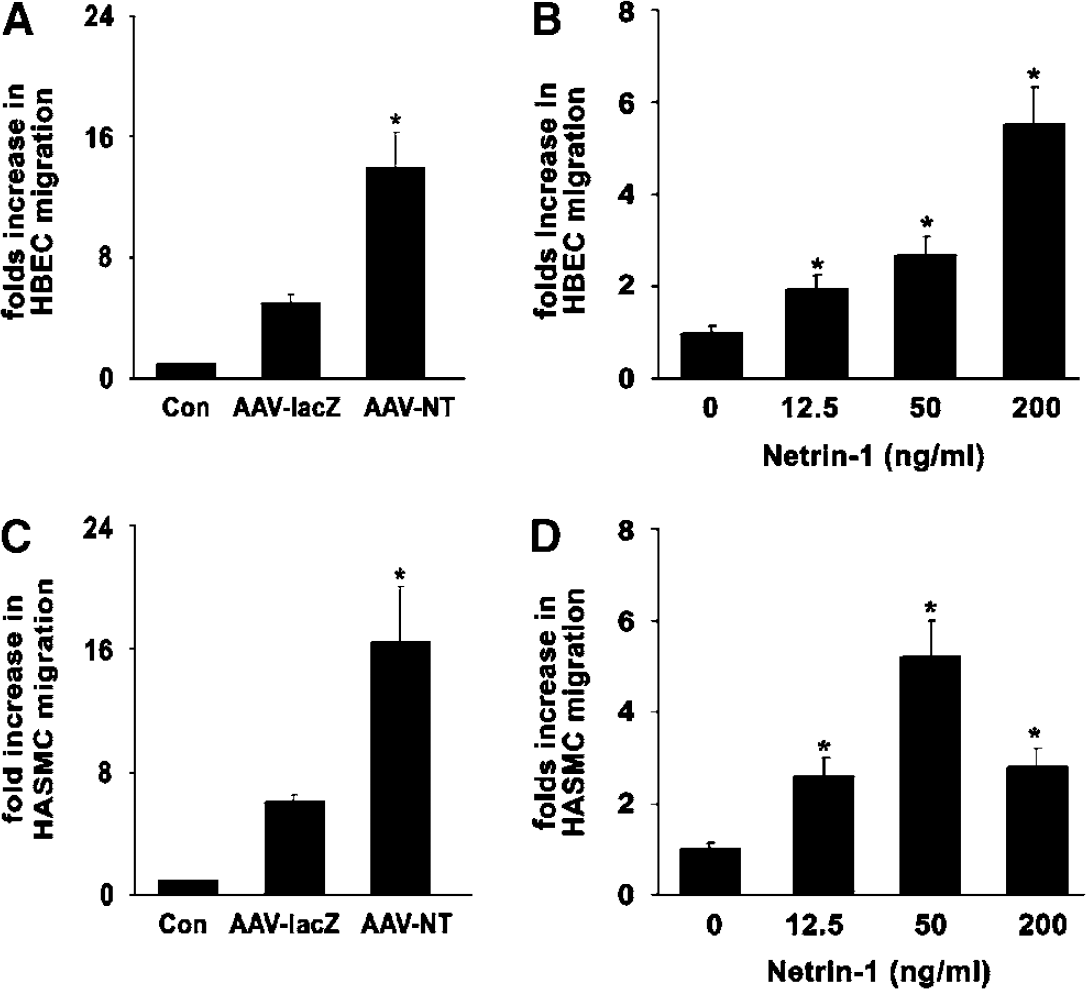
Netrin-1 induced migration of HCECs and HASMCs. A Boyden chamber assay was used to assess the ability of NT-1 to stimulate HCECs (
Next, we examined if netrin-1 stimulated and stabilized tube formation using HCECs cultured on matrigel. Exposure of HCECs to either AAV-NT-1-conditioned medium or netrin-1 protein led to the formation of a capillary-like structure on the matrigel. The microvascular-like structure formed at 6 h, and persisted for up to 18 h after netrin-1 treatment (Figure 5A). Interestingly, the effect of netrin-1 on stimulating HCEC tube formation was similar to the effect of VEGF (Figure 5B). Netrin-1-induced tube formation in a dose-dependent manner with optimal effects observed at 50 ng/mL (Figure 5C).
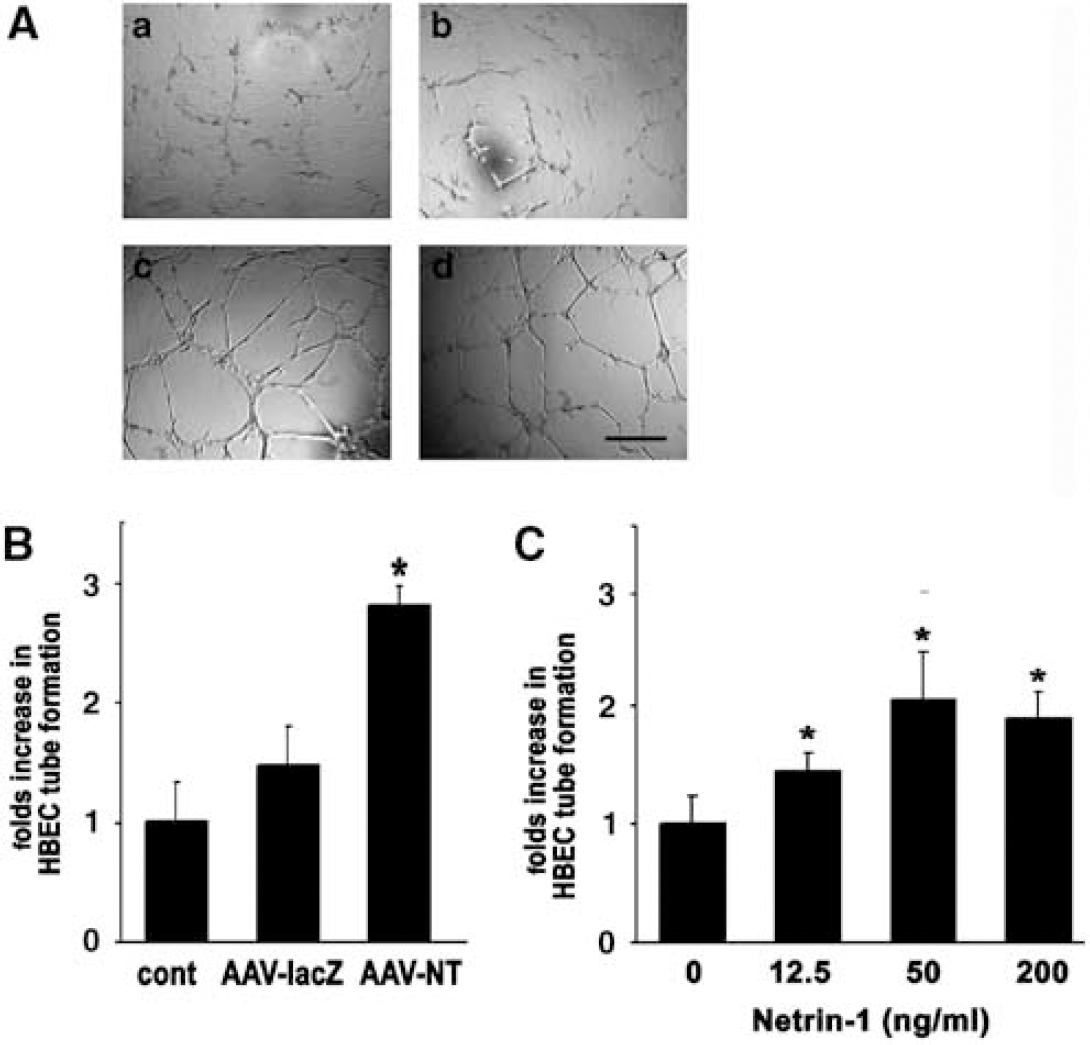
Netrin-1 promoted matrigel HCEC tube formation. A matrigel assay was used to assess the ability of NT-1 to induce HCECs to form microvessel-like tubes. VEGF (20 ng/mL) acted as a positive control and AAV-lacZ-conditioned medium or PBS acted as negative controls. (
Netrin-1-Induced Neovascularization Different from VEGF-Induced Angiogenesis in the Adult Mouse Brain
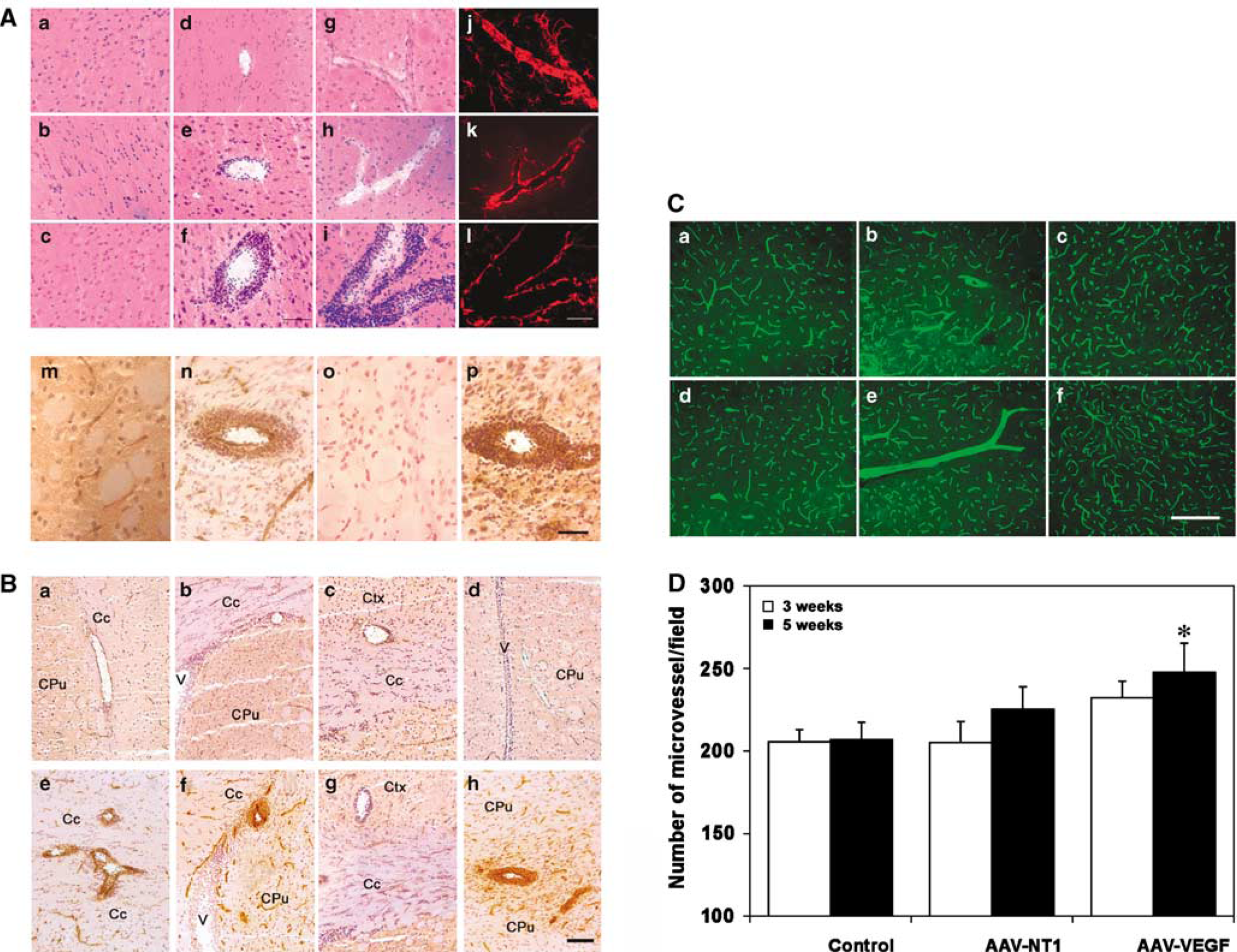
To determine if netrin-1 stimulated angiogenesis in the adult mouse brain in vivo, we transduced AAV-NT-1 gene into the mouse brain parenchyma. The number and the morphology of microvessels were examined after 1, 3, and 5 weeks of AAV-NT-1 gene transfer. Unexpectedly, there was no significant difference in the total number of microvessels between the AAV-NT-1- and the AAV-lacZ-transduced or saline-treated mice (Figures 7C and 7D). However, numerous enlarged microvessels, which were defined as vessels with a diameter greater than 10 μm in this study, were greatly increased in the AAV-NT-1-transduced mice compared with the AAV-lacZ-transduced or saline-treated mice (Figures 6A and 6C). These enlarged microvessels are BrdU positive (Figure 6B) and could be observed as early as 1 week and sustained until at least 5 weeks after netrin-1 gene transfer (Figure 6D).
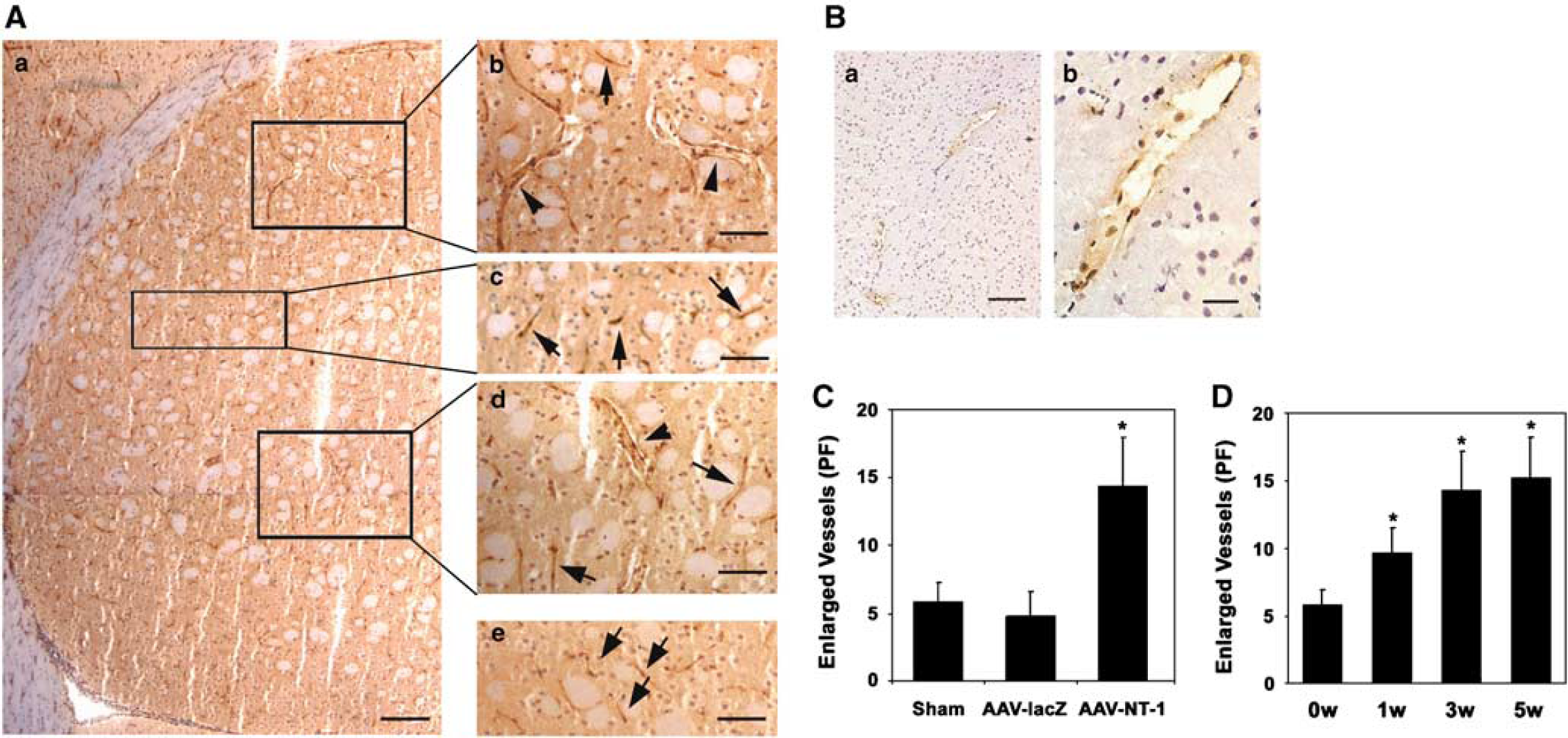
Netrin-1 induced enlarged vessels after AAV-NT-1 gene transfer. (
The patterns of the enlarged vessels after AAV-NT-1 gene transfer. (
We observed the development of the new vessels in the AAV-NT-1-transduced mouse brain (Figure 7A). The vessel wall was composed of a monolayer of cells 1 week after AAV-NT-1 gene transfer (Figures 7Ad and Ag), but grew thicker and contained two to three layers of cells at 3 weeks after AAV-NT-1 gene transfer (Figures 7Ae and 7Ah). At 5 weeks, it included an inner endothelial cell inner layer, an astrocyte-connected outlayer, and multiple layers of cells between them (Figures 7f and 7i). To explore the role of astrocytes during netrin-1-induced neovascularization, we performed GFAP fluorescent staining (Figures 7AJ–7Al). There was strong GFAP staining around the enlarged microvessels 1 week after AAV-NT-1 gene transfer (Figures 7Aj). This staining was gradually reduced with microvessel development (Figure 7Ak), until only a single layer of astrocytes appeared to wrap these vessels after 5 weeks (Figure 7Al), suggesting that astrocytes have a supporting role in the early stage of neovascularization. Moreover, immunostaining showed that all the enlarged vessels were α-actin-positive stained, suggesting that smooth muscle cells formed in the wall of these vessels, endowing them with artery-like characteristics (Figures 7Ao and 7Ap). In addition, we found that the enlarged vessels were distributed throughout the AAV-NT-1-transduced hemisphere, including the cerebral cortex, the corpus callosum, and the caudate putaman (Figure 7B). These changes were not observed in the AAV-lacZ or saline-injected control groups. Neutrin-1-induced new vessels absolutely differed from VEGF-induced angiogenesis; the latter mainly increased microvessel number but did not change vessel morphology (Figures 7C and 7D; Shen et al, 2006b).
Discussion
The present study provides evidence that netrin-1, either in conditioned medium or as purified protein, can stimulate HCEC and HASMC proliferation, migration, and tube formation in vitro. Furthermore, we have successfully constructed AAV-NT-1 and transduced this gene into the mature mammalian brain via AAV-mediated gene transfer; overex-pressed netrin-1 promoted new vessel formation, in a pattern distinct from the one we previously observed for VEGF (Shen et al, 2006b). These results indicate that netrin-1 not only plays a key role in neuronal development, but also participates in neovascularization and vessel remodeling in the adult brain.
Because the recombinant AAV vector is a reliable tool for the gene transfer in vivo, we chose AAV vector for the netrin-1 gene delivery. The advantages of using recombinant AAV vector include, but are not limited to, eliciting little or no inflammatory responses, infecting both dividing and nondividing cells, and mediating long-term gene expression for months to years in vivo (McCown, 2005; Xiao et al, 1996). After infecting HEK293 cells with AAV-NT-1, netrin-1 expression increased significantly both at the mRNA and protein levels. Similar to netrin-1 protein, the conditioned medium from HEK293 cells infected with AAV-NT-1-stimulated HCEC and HASMC proliferation, migration, and tube formation, which indicated that AAV-NT-1-transfected cells could express and secrete the functional NT-1 with bioactivity the same as the recombinant protein; the secreted NT-1 bound its receptors on HCEC or HASMC and then induced the proangiogenic changes. We further demonstrated by immunofluorescent staining that both neurons and astrocytes expressed netrin-1 after AAV-NT-1 transfer, which is consistent with the finding that neurons and astrocytes are transducible by the recombinant AAV1 vectors (Tenenbaum et al, 2004; Wang et al, 2003). Similar to in vitro studies, the infected cells (neurons and astrocytes) expressed and secreted functional netrin-1 and acted on cells in brain tissue, including endothelial cells.
Endogenous netrin-1, predominantly expressed by the floor plate cells in the central nervous system, induces axonal outgrowth, axonal orientation, and neuronal migration during neuronal development (Barallobre et al, 2005; Xie et al, 2006). Besides the aforementioned roles it plays in the central nervous system, NT-1 has also been implicated in regulating salivary gland migration (Kolesnikov and Beckendorf, 2005), regulating invasion and migration of mouse mammary epithelial cells (Strizzi et al, 2005), promoting lung branching morphogenesis (Liu et al, 2004), and mediating pancreatic epithelial cell adhesion (Hebrok and Reichardt, 2004). Further, netrin-1 can stimulate the proliferation and migration of human microvascular endothelial cells, and induce angiogenesis on chorioallantoic membrane and murine cornea in vivo (Park et al, 2004). In the current study, we confirmed that netrin-1 stimulated normal HCEC proliferation, migration, and tube formation in vitro. Furthermore, we showed that overexpression of netrin-1 in the adult mouse brain induced numerous enlarged vessels (Figure 7). These vessels clearly show the smooth muscle cells present in the wall of enlarged vessels, suggesting that the newly formed vessels include small arteries or veins. These newly developed vessels were distributed within the AAV-NT-1-transduced hemisphere, including the cortex, subventricular zone, corpus callosum, and caudate putamen. These changes are evidently different from VEGF-induced angiogenesis in that VEGF potently promotes microvessel growth and branching, but it does not change the vessel morphology in the mouse brain (Shen et al, 2006a). Our findings suggest that netrin-1, a strong angiogenic factor, can remodel the preexisting brain vasculature and promote new artery-like vessel formation in the adult rodent brain, instead of branching microvessels into much smaller vessels. Although netrin-1-induced angiogenesis is physiologically significant, its pathological impact warrants further investigation.
Astrocytes in the gray matter of the brain or spinal cord are usually GFAP negative, apart from those that specifically contact blood vessels strongly expressing GFAP (Bignami and Dahl, 1974). Interestingly, we found that many GFAP-positive astrocytes enclosed the enlarged microvessels that we observed 1 week after AAV-NT-1 transduction. As the wall grew thicker, the GFAP-positive astrocytes were reduced gradually to monolayer thickness. This result suggests the importance of perivascular astrocytes in the development and stabilization of these enlarged vessels. It has been reported that epoxyeicosatrienoic acids from astrocytes are mitogenic and angiogenic (Zhang and Harder, 2002), and that astrocytes express integrin αVβ8 through which they interact with endothelial cells to promote brain vessel differentiation and stabilization (Cambier et al, 2005). In addition, reciprocal feedback between blood vessels and astrocytes stabilizes the retinal vascular network (West et al, 2005).
We found that netrin-1 primarily promoted larger vessel formation, whereas VEGF generally increased microvessel growth (Shen et al, 2006a). This difference suggests that netrin-1 and VEGF promote angiogenesis through independent pathways. It has been reported that the signaling through the known netrin receptors such as DCC, Neogenin, or UNC5H family members is not detected in endothelial cell types (Park et al, 2004). Although controversial, it has also been reported that netrin-1 induces neovascularization through a DCC-dependent ERK1/2-eNOS pathway, because DCC antibodies, DCC small-interfering RNAs, and specific inhibitors can inhibit angiogenesis and the production of nitric oxide in aortic endothelial cells (Nguyen and Cai, 2006). Nevertheless, the molecular mechanism of netrin-1-induced angiogenesis and neovascularization remains uncertain. More comprehensive studies, especially on identifying the specific netrin-1 receptors in the endothelial or smooth muscle cells both in vitro and in vivo, are required to address the related mechanism.
