Abstract
Chronic stroke is a highly important but under-investigated scientific problem in neurologic research. We have reported earlier that stem cell factor (SCF) in combination with granulocyte-colony stimulating factor (G-CSF) treatment during chronic stroke improves functional outcomes. Here we have determined the contribution of bone marrow-derived cells in angiogenesis and neurogenesis, which are enhanced by SCF + G-CSF treatment during chronic stroke. Using bone marrow tracking, flow cytometry, 2-photon live brain imaging, and immunohistochemistry, we observed that the levels of circulating bone marrow stem cells (BMSCs) (CD34+ /c-kit +) were significantly increased by SCF +G-CSF treatment. In addition, live brain imaging revealed that numerous bone marrow-derived cells migrate into the brain parenchyma in the treated mice. We also found that bone marrow-derived cells, bone marrow-derived endothelial cells, vascular density, and bone marrow-derived neurons were significantly augmented by SCF + G-CSF. It is interesting that, in addition to the increase in bone marrow-derived endothelial cells, the number of bone marrow-derived pericytes was reduced after SCF + G-CSF treatment during chronic stroke. These data suggest that SCF + G-CSF treatment can enhance repair of brain damage during chronic stroke by mobilizing BMSCs, and promoting the contribution of bone marrow-derived cells to angiogenesis and neurogenesis.
Introduction
Stroke, a leading cause of long-term disability in adults, is an enormous public health problem and a heavy public financial burden worldwide. Stroke progression is generally classified into three phases based on the timing after stroke onset: the acute phase (within 48 h), the subacute phase (up to 6 weeks), and the chronic phase (beyond 3 to 6 months) (Parsons et al, 2000; Kang et al, 2004). In contrast to most research focusing on rescuing neurons from a very limited treatable window in the acute phase, little attention has been paid to developing new therapeutic strategies to repair the brain during chronic stroke.
Stem cell factor (SCF) and granulocyte-colony stimulating factor (G-CSF) belong to the hematopoietic growth factors and play major roles in the regulation of blood cell production (Galli et al, 1994; Welte et al, 1996). However, convincing evidence suggests that SCF and G-CSF also play roles in the central nervous system (Hirata et al, 1993; Jin et al, 2002; Kirsch et al, 2008). Recently, accumulating evidence has shown that both SCF and G-CSF have therapeutic effects in brain ischemia. The administration of G-CSF alone (Schabitz et al, 2003; Komine-Kobayashi et al, 2006; Zhao et al, 2007b) or in combination with SCF (Zhao et al, 2007b) during the acute phase of brain ischemia, or the subcutaneous injection of SCF + G-CSF during the subacute phase of brain ischemia (Kawada et al, 2006) results in infarction size reduction and functional improvement. We have recently shown that systemic administration of SCF + G-CSF during the chronic phase of brain ischemia, initiated 3.5 months after ischemia for 7 days, leads to improved functional recovery with reduced size of infarct cavities (Zhao et al, 2007a). However, the mechanisms by which SCF + G-CSF help to repair the damaged brain during chronic stroke remain entirely unknown.
It has been characterized that CD34+ cells are hematopoietic stem cells/hematopoietic progenitor cells (Krause et al, 1996; Vodyanik et al, 2005). Considerable evidence shows that SCF in combination with G-CSF has a synergistic effect on the mobilization of CD34+ cells from the bone marrow (BM) to the blood (de Revel et al, 1994; Hess et al, 2002). In addition, CD34+ cells contribute to both neurogenesis (Shyu et al., 2006) and angiogenesis in the brain (Taguchi et al, 2004a). Moreover, SCF + G-CSF augment BM-derived cells that express neuronal marker, NeuN, in intact brains (Corti et al, 2002) as well as in acute and subacute ischemic brains (Kawada et al, 2006). The aim of this study is to determine whether SCF + G-CSF can mobilize BM stem cells (BMSCs) and promote the differentiation of BM-derived cells that contribute to angiogenesis and neurogenesis during chronic stroke.
Materials and methods
All procedures have been approved by the Institutional Animal Care and Use Committee of Louisiana State University Health Sciences Center and are in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Experimental Design
Mice were subjected to cerebral cortical ischemia. Two ischemic groups, SCF + G-CSF (n = 5) and a phosphate-buffered saline (n = 4) (PBS) control, and one intact control group (n = 5) were utilized for this study. Twelve weeks after ischemia, both intact and stroke mice were irradiated, and the recipient's BM was reconstituted with the BM of the mice carrying green fluorescent protein (GFP). Four weeks after BM transplantation (16 weeks after ischemia), the stroke mice were randomly divided into two groups, and SCF + G-CSF or PBS was injected subcutaneously for 7 days. One day after the final injection, the number of CD34/CD117-positive hematopoietic stem cells/hematopoietic progenitor cells in peripheral blood (PB) was determined using flow cytometry. Thereafter, a cranial window was installed and BM-derived GFP+ cells were observed using a 2-photon microscope in live animals 1 and 5 weeks after the final injection. Eight weeks after treatment (25 weeks after ischemia), the mice were killed, and the fate of BM-derived cells in the brain was examined by immunohistochemistry (Figure 1).

Experimental design of this study.
Focal Cerebral Ischemia
C57BL/6J mice (female, 3 months old) were anesthetized with Avertin (240 mg/kg, i.p.). Brain ischemia was induced by ligation of the right common carotid artery with a 6–0 silk surgical suture and coagulation of the right middle cerebral artery with a cauterizer (World Precision Instruments, Sarasota, USA), under a microscope. Body temperature was maintained at 37°C with a warming pad until the mice woke up from anesthesia.
Bone Marrow Transplantation and Hematopoietic Growth Factor Administration
The mice were irradiated with a lethal dose of 900 rad X-ray to destroy the BM. The ends of each femur of C57BL-GFP mice (8 to 10 weeks old) (The Jackson Laboratory, Bar Harbor, USA) were clipped to expose the marrow. The BM was flushed out by 10 ml ice-cold Hanks Balanced Salt Solution in a syringe with a 21-gauge needle, followed by filtration through a 70-µm nylon mesh to obtain a single-cell solution. The BM cells were centrifuged, resuspended with Hanks Balanced Salt Solution, and transplanted to the recipients through the tail vein. Bone marrow transplantation (107 cells per mouse) was performed within 24 h after irradiation. Mouse recombinant-SCF (200 µg/kg) (Pepro-Tech, Rocky, USA) with human recombinant-G-CSF (50 µg/kg) (Amgen, Thousand Oaks, USA) or an equal volume of PBS was administered subcutaneously for 7 days, beginning 16 weeks after brain ischemia.
Flow Cytometry
Blood was collected from the tails of mice and labeled with phycoerythrin-conjugated anti-mouse CD34 antibody (1:50), APC-conjugate anti-mouse CD117 antibody (anti-ckit, 1:100), or an equal amount of isotype-matched phycoerythrin and APC antibodies (eBioscience, San Diego, USA) on ice for 30 min. Red blood cells were then lysed with a FACS lysis buffer (BD Pharmingen, Franklin, USA) and washed with PBS containing 0.5% fetal bovine serum. Then 5 × 106 cells were analyzed by flow cytometry (FACSCalibur, BD, Franklin, USA).
Live Brain Imaging
Brain imaging was performed through a cranial window that was installed by making a hole (6mm in diameter) in the skull with a dental drill, covered with a sterilized-coverslip (8mm in diameter), and sealed with a mixture of cement and Krazy Glue. The animals were anesthetized with Avertin, and immobilized with a custom stereotaxic stage. Body temperature was maintained at 37°C during imaging. Images of GFP+ cells were obtained using a Zeiss 2-photon microscope (Zeiss, LSM510 NLO) (Carl Zeiss, Thornwood, USA). The 2-photon fluorescence imaging was generated with an excitation wavelength of 860nm and a range of 505 to 555nm for collecting fluorescence emission. Images were acquired using a × 40 water-immersion objective (0.8 numerical aperture). High-resolution image z-stacks, originating at the cortical surface, were taken 1 µm apart and at 512 × 512 pixels with 12-bits per image frame. Each z-stack contained 8 to 16 serial optical sections, and three sites per brain in the periinfarct cortex were scanned. Image stacks were analyzed with Zeiss LSM510 4.0SP2 software and ImageJ 1.38W.
Charge-Coupled Device Recording
BM-derived cells (GFP+) in the blood vessel wall and in the blood stream were visualized with an upright Nikon fluorescence microscope (Nikon Inc. Melville, USA) through the cranial window using a ×20 water immersion lens 6 weeks after treatment. The images were acquired with a 3-charge-coupled device color video camera (Sony Corporation, Tokyo, Japan) in live animals.
Immunohistochemistry
The mice were perfused with 4% buffered formaldehyde. The brains were cryoprotected by overnight treatment with 30% sucrose. Coronal brain sections, 35-µm thick, were cut with a microtome. The free-floating method was used for immunohistochemistry. Briefly, nonspecific binding was blocked by 10% normal goat serum diluted in 1% bovine serum albumin (IgG-free) (Jackson ImmunoResearch Labs, West Grove, USA) with 0.25% Triton X-100. The sections were then incubated with primary antibodies, rabbit or mouse anti-GFP (1:200) (Santa Cruz Biotechnology, Santa Cruz, USA), mouse anti-CD31 (1:200) (BD Pharmingen, Franklin, USA), mouse anti-NeuN (1:500) (Chemicon, Temecula, USA), mouse anti-GFAP (1:500) (Sigma, St Louis, USA), or rabbit anti-Iba 1 (1:500) (Wako, Richmond, USA) antibodies at 4°C overnight. Thereafter, the sections were incubated with Cy2- or Cy3-conjugated goat anti-mouse or rabbit antisera (Jackson ImmunoResearch, West Grove, USA) for 2 h at room temperature in the dark. Counterstaining was performed with 4',6-diamidino-2-phenylindole (DAPI) (1:5000) (Sigma, St Louis, USA). Sections to which the primary antibodies were not added served as a negative control. Four to five sections per brain were processed for the immunohistochemistry.
Cell Count
Images were captured from brain sections with a Zeiss 2-photon confocal microscope (Zeiss, LSM510 NLO) (Z-stacks, 11 to 13 serial optical sections with 2-µm intervals) in periinfarct areas to detect the co-expression of GFP, tissue-specific cell markers, and nuclear dye (DAPI). The images taken from the four areas surrounding the infarct cavities (indicated in Figure 6A) were used for determining the number of GFP+ cells, GFP+/NeuN+ cells, GFP+/GFAP+ cells, and GFP+ /Iba-1+ cells. The images taken from the ipsilateral frontal cortex (indicated in Figure 4D) were employed for identifying the number of GFP+/CD31+ endothelial cells and for determining blood vessel density. Cell counting was based on the 3D images with high magnification. The number of counted cells is presented as the average per section. The process of cell counting was performed in a blinded manner.
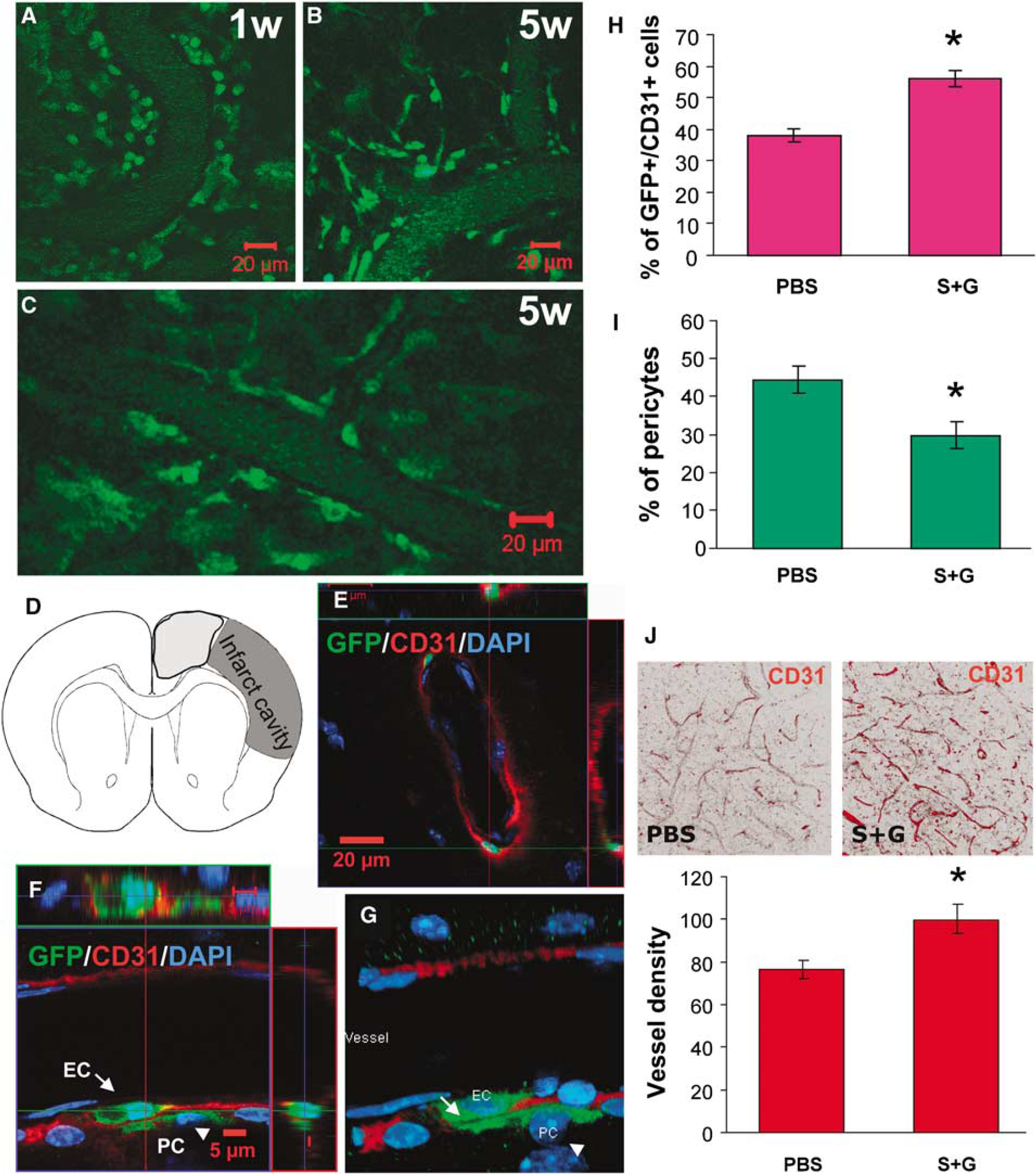
Angiogenesis promoted by SCF+G-CSF treatment during chronic stroke. (
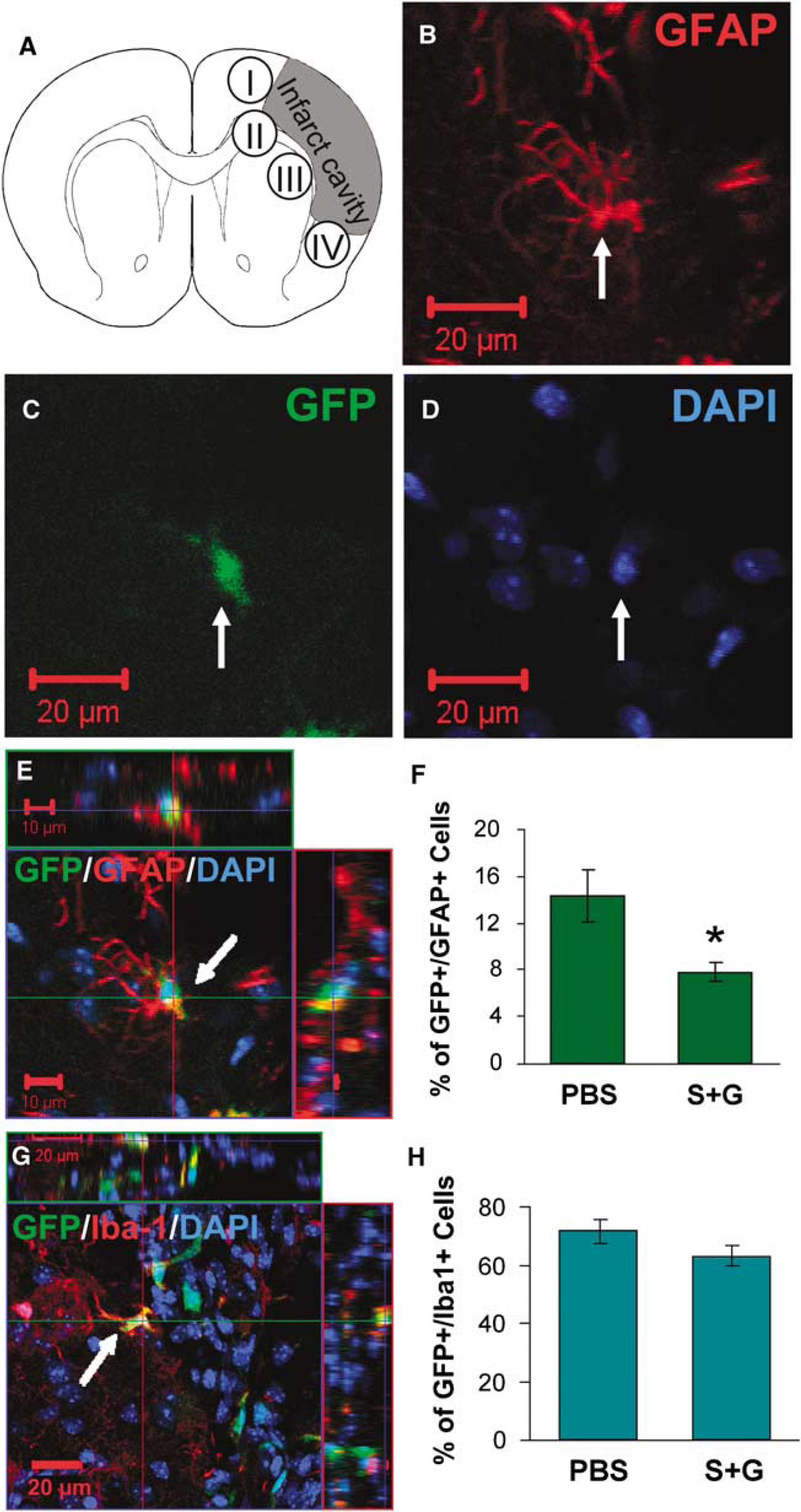
Bone marrow-derived astrocytes and microglia in periinfarct areas in chronic stroke. Confocal images are acquired from the areas bordering the infarct cavities (I, II, III, and IV indicated in
Statistical Analysis
One-way analysis of variance was used for data analysis, and multiple comparisons (for three group data) were corrected with Bonferroni post hoc test. P > 0.05 was considered as significant difference. Data are expressed as mean±s.e.
Results
Stem Cell Factor plus Granulocyte-Colony Stimulating Factor Mobilized Bone Marrow Stem Cells into the Blood during Chronic Stroke
Peripheral blood was collected 1 day after the final injection of SCF + G-CSF, and the levels of CD34+/c-kit + cells were examined using flow cytometry. We found that the percentage of CD34+/c-kit + cells was increased 2.15-fold in SCF + G-CSF-treated mice (P > 0.01) (Figure 2), indicating that the treatment paradigm mobilized CD34+ /c-kit + BMSCs into the blood during the chronic phase of stroke.
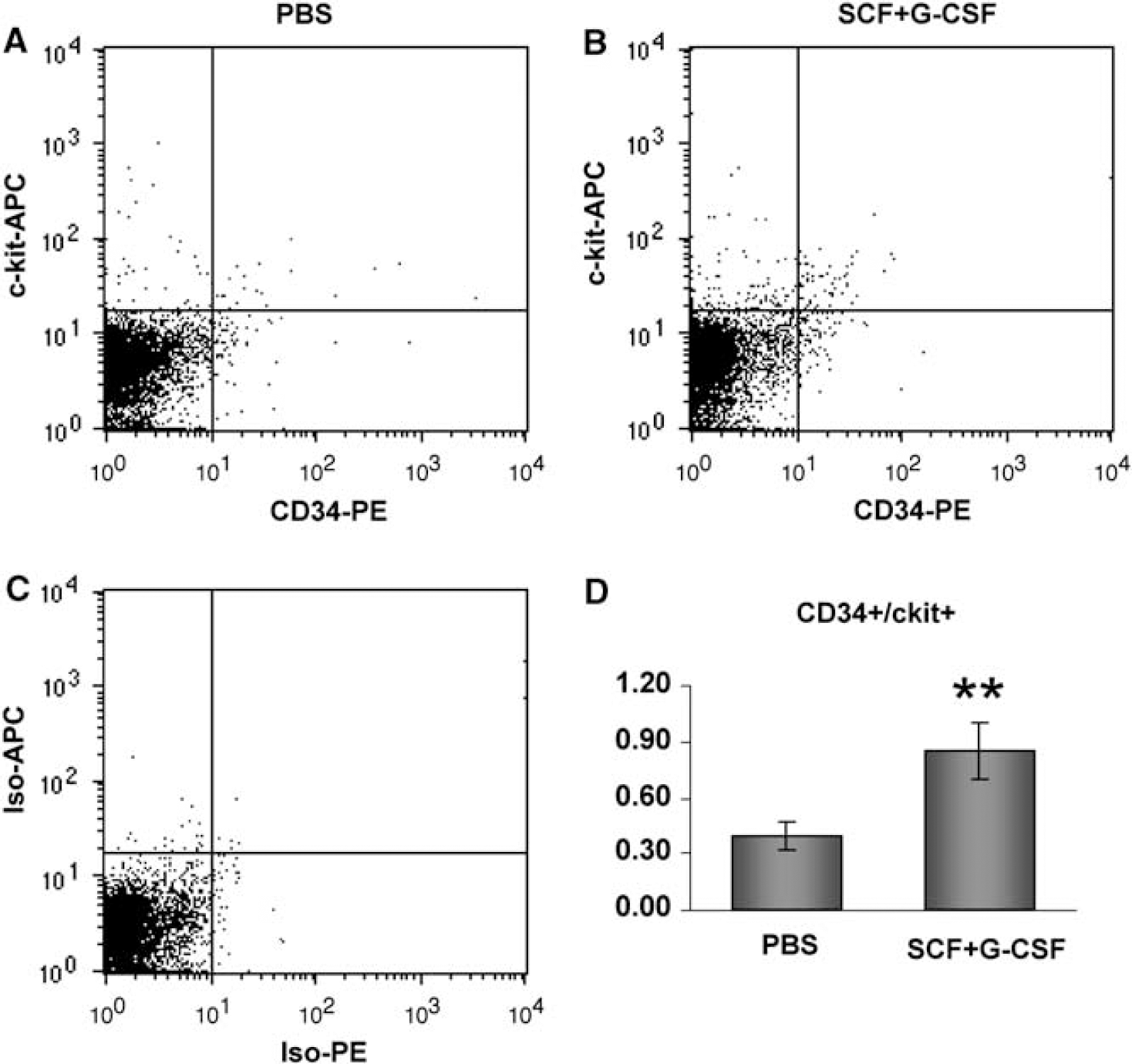
The levels of BMSCs in the peripheral blood after SCF+G-CSF treatment during chronic stroke. Circulating BMSCs (CD34+/ckit+) of PBS-(
Stem Cell Factor plus Granulocyte-Colony Stimulating Factor Facilitated the Entry of BM-derived Cells into the Chronic Stroke Brain
Next, we sought to determine whether homing BM-derived cells were enhanced by SCF +G-CSF treatment during chronic stroke. One week after the final injection, a cranial window was prepared. Two-photon brain imaging was performed in live animals 48 h after surgery. We observed many BM-derived cells (GFP expressing cells) in the brains of both PBS control mice and SCF + G-CSF-treated mice. However, the morphology of the GFP+ cells was quite different between the two groups: GFP+ cells with long processes were observed in SCF + G-CSF-treated mice, whereas, round cells with no processes were found in PBS controls (Figure 3). This morphologic feature suggests that the GFP+ cells were stimulated by SCF + G-CSF. On the basis of an earlier study in which neural stem cell (NSC) movement was recorded by time-lapse movies using a 2-photon microscope, cells with long processes are migrating cells (Zhao and Nam, 2007). Does the presence of GFP+ cells with long processes also suggest that SCF + G-CSF treatment promotes BM-derived cell migration into the brain? If so, the number of GFP+ cells could be increased by SCF + G-CSF treatment.
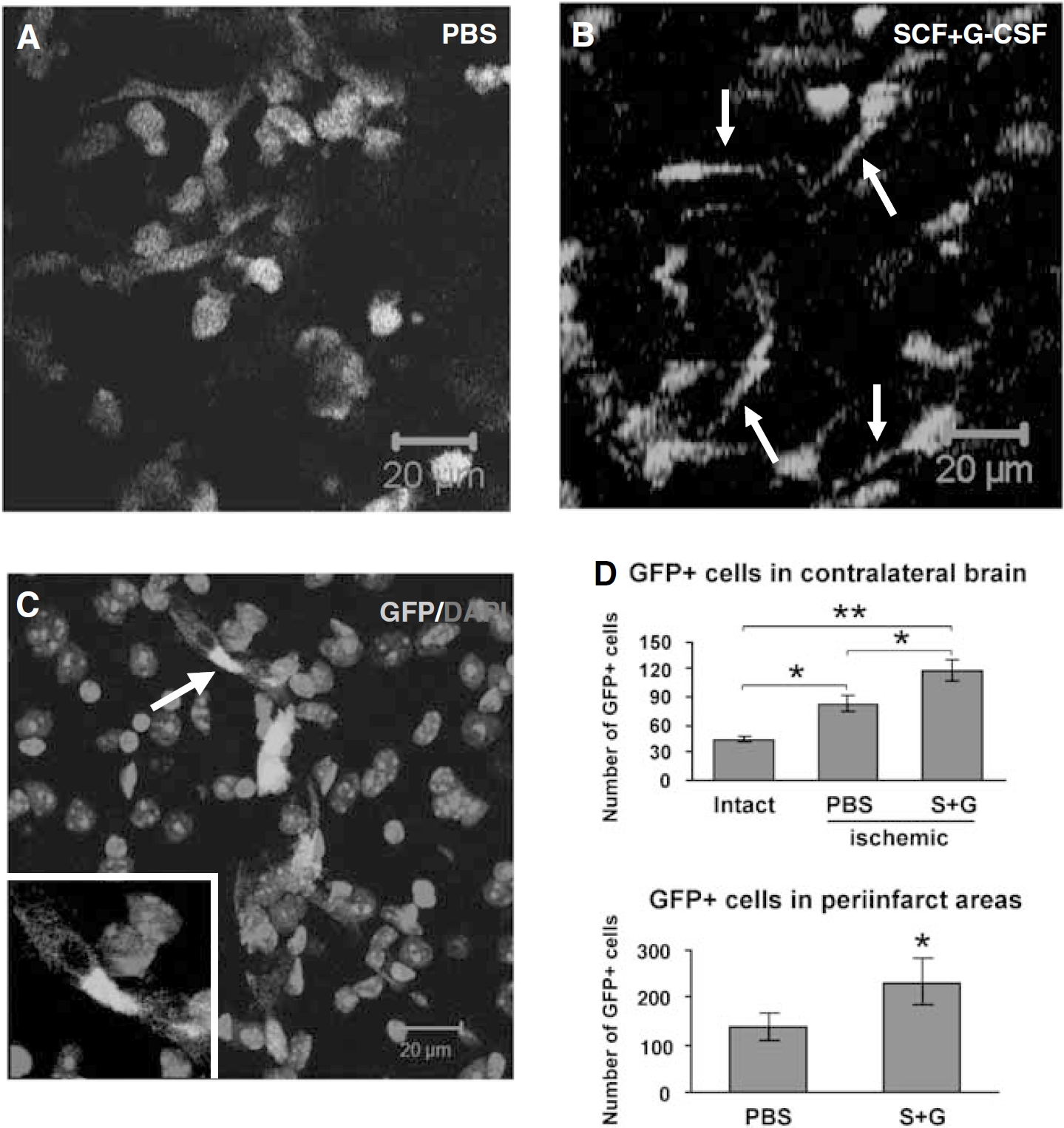
Bone marrow-derived cells (GFP+ cells) in the brain. (
To determine whether SCF + G-CSF treatment during chronic stroke leads to homing of BM-derived cells into the brain, 2 months after treatment, brain sections were processed for immunohistochemistry. We found that BM-derived cells penetrated into both intact and ischemic brains, and that the entry of BM-derived cells (GFP+) significantly increased in the ischemic mice as compared with those in the intact mice (intact versus PBS, P > 0.05; intact versus SCF + G-CSF, P > 0.01) (Figure 3D), indicating that BM-derived cells may be involved in both brain physiologic function and brain repair during chronic stroke. In addition, we observed that the number of GFP+ cells in both the contralateral hemisphere and the ipsilateral hemisphere adjacent to the infarct cavity was significantly higher in the mice treated with SCF + G-CSF than in the PBS controls (P > 0.05) (Figure 3D), indicating that penetration of BM-derived cells into the brain was enhanced by SCF + G-CSF treatment during chronic stroke.
Stem Cell Factor plus Granulocyte-Colony Stimulating Factor Augmented Bone Marrow-derived Endothelial Cells and Promoted Angiogenesis in the Chronic Stroke Brain
In 2-photon live brain imaging, we observed that many GFP expressing cells had accumulated in surrounding blood vessels in SCF + G-CSF-treated mice 1 week after treatment (Figure 4A). One month later, these cells were incorporated into or firmly attached to the blood vessel wall (Figures 4B and 4C). This observation was further confirmed by brain live images recorded with a charge-coupled device video camera (Movie 1 in the Supplementary Materials).
To further determine the cell types of the GFP+ cells on the blood vessel wall, immunofluorescent double staining was performed 2 months after treatment. GFP+ cells coexpressing CD31, the marker for endothelial cells, were considered as BM-derived endothelial cells (Figures 4E and 4F, Movie 2 in the Supplementary Materials). As there is no specific marker available for identifying all pericytes (Lamagna and Bergers, 2006), the GFP+/CD31−cells that immediately contact the CD31+ endothelial cells (Figures 4F and 4G) were considered as pericytes, as originally described (Bergers and Song, 2005). The identification of BM-derived endothelial cells and pericytes was performed with 3-dimensional (3D) 2-photon confocal images (Zeiss, LSM510) in the frontal cortex ipsilateral to the infarct cavity as indicated in Figure 4D. We observed that GFP+/CD31+ cells rarely appeared in intact brains, whereas there were many GFP+/CD31+ cells in the brains of ischemic mice. Further, SCF + G-CSF-treated mice showed a significant increase in the number of GFP+ /CD31+ cells when compared with the PBS controls (Figure 4H) (P = 0.01). In addition, the density of blood vessels was also significantly increased in SCF+ G-CSF-treated mice (Figure 4J) (P > 0.05). It is interesting that SCF + G-CSF-treated mice showed a significant reduction in the number of pericytes (Figure 4I) (P > 0.05). These data suggest that BM provides the source of both endothelial cells and pericytes contributing to brain repair after ischemic injury, BM-derived endothelial cells and pericytes may share the same stem/progenitor cells, SCF + G-CSF may promote differentiation of the stem/progenitor cells into endothelial cells and inhibit the differentiation into pericytes, and SCF +G-CSF enhance angiogenesis when administered during chronic stroke.
Stem Cell Factor and Granulocyte-Colony Stimulating Factor Augmented Bone Marrow-derived Neurons in the Chronic Stroke Brain
Next, we asked whether BM-derived neurons increased by SCF + G-CSF treatment during chronic stroke. GFP+/NeuN+ cells in the periinfarct areas were determined with 3D-confocal imaging. We observed that the total number of GFP+/NeuN+ cells was significantly higher in SCF + G-CSF-treated mice (16.8±3.5) than in PBS controls (7.9±1.5) (P > 0.05). Interestingly, approximately 50% of GFP+/NeuN+ cells in the SCF +G-CSF-treated group were located near the corpus callosum bordering the infarct cavities. We noted that GFP+/NeuN+ cells in this particular area were also significantly increased by SCF + G-CSF treatment during chronic stroke (P > 0.01; Figure 5). These data indicate that the corpus callosum may provide a favorable environment interacting with SCF + G-CSF to direct BMSC differentiation into neurons.
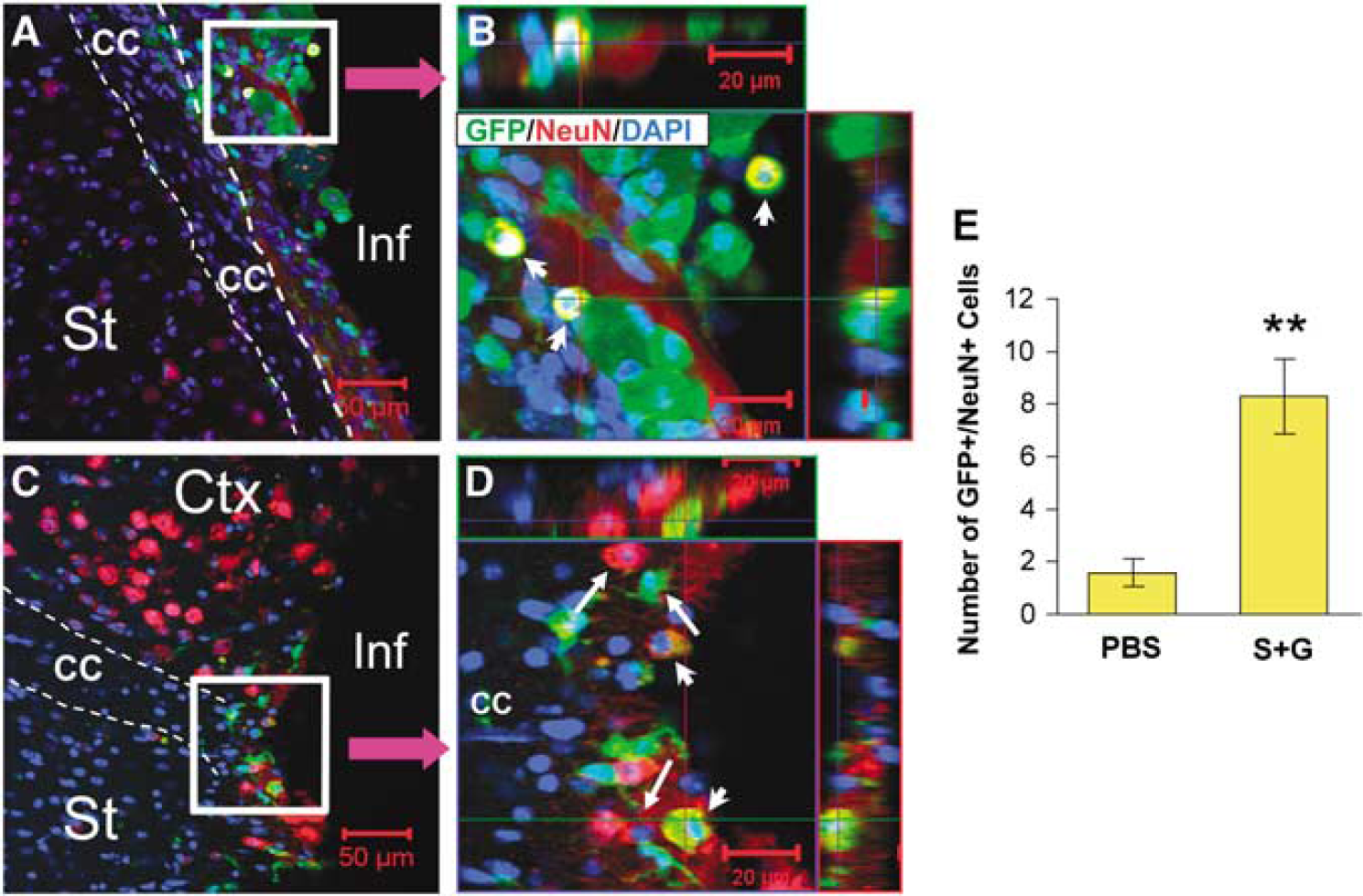
Bone marrow-derived neurons adjacent to the corpus callosum (CC) bordering the infarct cavities in chronic stroke. (
To determine whether BM-derived glial cells were also changed by SCF+G-CSF treatment, immunofluorescent double staining for identifying GFP+/GFAP+ cells (BM-derived astrocytes) and GFP+ /Iba-1+ cells (BM-derived microglial cells) was performed 2 months after the treatment. We found that SCF+G-CSF treatment during chronic stroke led to a significant reduction in BM-derived astrocytes (Figure 6F) (P< 0.05), whereas there was no difference between the treated and the nontreated animals in the number of BM-derived microglial cells (Figure 6H). These data, together with the data for BM-derived neuron-like cells, suggest that SCF+ G-CSF may play a role in the differentiation of BMSCs to neural cells.
Discussion
The main findings of this study are that systemic administration of SCF +G-CSF during chronic stroke leads to the mobilization of CD34+ BMSCs into PB, the enhancement of entry of BM-derived cells into the brain, the augmentation of BM-derived endothelial cells and BM-derived neuron-like cells, and the increase in angiogenesis.
BM contains both CD34+ and CD34− stem cells (Krause et al, 1996; Huss, 2000). The population of CD34+ hematopoietic stem cells is 1.5% in the BM and > 0.5% in the PB (Krause et al, 1996). However, many studies have shown that SCF in combination with G-CSF significantly increases the levels of CD34+ cells in the PB when compared with SCF and G-CSF alone, suggesting a synergistic effect of SCF + G-CSF in the mobilization of CD34+ cells from BM to blood (de Revel et al, 1994; Hess et al, 2002). In agreement with these earlier studies, we observed that systemic administration of SCF + G-CSF during chronic stroke elevated CD34+ cells in the blood, indicating the treatment paradigm is sufficient to mobilize BMSCs.
In addition to mobilizing CD34+ stem cells, SCF + G-CSF treatment enhances the entry of BM-derived cells into the brain during chronic stroke. Ischemic brain tissue-released chemoattractant, stromal-derived factor-1, has been shown to cause BMSC migration (Kucia et al, 2006). However, SCF + G-CSF-induced penetration of BM-derived cells into the brain is not related to brain ischemia-induced chemokines. We and other investigators found similar results using the same technique to track BM-derived cells: replacing the recipient's BM with the BM of transgenic mice expressing GFP. Using intact animals, Corti et al (2002) reported that GFP + cells in the brain were increased in the mice receiving SCF +G-CSF treatment. Kawada et al (2006) found that SCF + G-CSF injection either during 1 to 10 days (acute phase) or during 11 to 20 days (subacute phase) after brain ischemia caused an increase in the number of GFP+ cells in the brain. Here we observed that GFP+ cells also increased in the brain after SCF + G-CSF administration during the chronic phase of brain ischemia. Thus, all these findings suggest that SCF + G-CSF-induced entry of BM-derived cells into the brain is dependent on the biological function of SCF + G-CSF but is not brain lesion related. It is interesting that using 2-photon live brain imaging, we also observed that many GFP+ cells in the brains of SCF + G-CSF-treated mice appeared to have the features of migrating cells. This observation indicates that SCF + G-CSF treatment may change the microenvironment in the brain, resulting in enhancement of the penetration of GFP+ cells. Exactly how SCF +G-CSF treatment modifies the brain microenvironment requires further studies to clarify.
Bone marrow stem cells have been shown to contribute to neurogenesis in the brain. Although the ability of BMSCs to transdifferentiate into neurons remains controversial, many investigators in different laboratories have observed BMSC-derived neuron-like cells in the brain (Brazelton et al, 2000; Mezey et al, 2000; Weimann et al, 2003). Evidence shows that it may be a natural event that BMSCs form neurons through both cell fusion (Alvarez-Dolado et al, 2003) and noncell fusion (Cogle et al, 2004). However, when we counted GFP+ /NeuN+ cells in the periinfarct areas, we only selected the single nuclear cells coexpressing both GFP+/NeuN+ and considered them to be BM-derived neurons. We observed that SCF + G-CSF treatment augmented the BM-derived neurons in periinfarct regions, and it was apparent that neuronal regeneration was more prolific in the areas adjacent to the corpus callosum. Similar to our findings, Kawada et al (2006) reported that BM-derived neurons in periinfarct areas were increased by SCF + G-CSF treatment during the acute or the subacute phase of brain ischemia. Carmichael (2006) also observed that brain ischemia-induced neurogenesis occurred in the periinfarct areas and that the microenvironment in the periinfarct areas favored neuronal regeneration within 1 month after ischemia. If, in the chronic phase of brain ischemia, the favorable changes in the microenvironment have passed, why do the BM-derived neurons still occur in the periinfarct zone? We postulate that SCF + G-CSF treatment may reestablish the ‘favorable microenvironment’ in the periinfarct areas, especially surrounding the corpus callosum during chronic stroke to permit BMSCs to generate neurons. We noted that a recent study did not find an increase in BM-derived neurons by SCF + G-CSF when the treatment was performed during 1 to 5 days after brain ischemia (Toth et al, 2008). Notably, the brain microenvironment is quite different between the acute and chronic stages of stroke. Inflammatory mediators, extensive glutamate release, and dying cell-released neurotoxins during the early phase of stroke may affect newborn neuron survival. Although Kawada et al (2006) found a slight increase in BM-derived neurons by SCF +G-CSF injected during 1 to 10 days after ischemia, the increase was only observed in a limited area.
It is worth noting that SCF + G-CSF-induced neuronal regeneration from BM is not limited to the condition of brain ischemia. Corti et al (2002) observed that systemic administration of SCF + G-CSF also augmented GFP+/NeuN+ cells in intact brain. This observation, together with our data shown here and the findings reported by Kawada et al (2006), suggests that SCF + G-CSF intervention itself has the capability to stimulate BMSCs to form neurons.
Interestingly, in addition to the increase in neurogenesis, we also found that the number of GFP+/GFAP+ cells was reduced by SCF + G-CSF injected during chronic stroke. It has been shown that microenvironmental alterations regulate the determination and differentiation of NSCs into neurons or glial cells. In an in vitro study, Qian et al (1997) discovered that a low level of basic fibroblast growth factor directs NSC differentiation into neurons with a reduction of glial cell fate differentiation. Using NSC cultures, we also observed that SCF +G-CSF treatment promotes neuronal fate differentiation and inhibits differentiation of NSCs into astrocytes (Piao et al, unpublished data). However, whether SCF + G-CSF treatment can regulate differentiation of BM-derived progenitor cells into neural cells in the same manner as NSCs, and how this process is regulated, remains to be determined.
During embryogenesis, hematopoietic cells and endothelial cells are derived from the common progenitor cells (Lamalice et al, 2007). In adults, CD34+ cells have been considered to be both hematopoietic stem cells and endothelial progenitor cells (Brugger et al, 1995; Asahara et al, 1997). Increasing evidence shows that circulating CD34+ cells contribute to angiogenesis after brain ischemia. Taguchi et al, 2004a, b reported that intravenous injection of CD34+ cells 48 h after brain ischemia in mice reduced infarction size, enhanced angiogenesis and neurogenesis, and improved functional outcome. It is interesting that the angiogenesis provided a favorable environment for neuronal regeneration. When blocking the CD34+ cell-induced angiogenesis, the neurogenesis was reduced. In addition, intracerebral injection of CD34+ cells 7 days after brain ischemia induced an increase in angiogenesis and neurogenesis (Shyu et al, 2006). In stroke patients, high levels of circulating CD34+ cells showed angiographic features in the brain (Yoshihara et al, 2008), with an increase in regional cerebral blood flow (Taguchi et al, 2004b). An fMRI study showed that an increase of regional cerebral blood flow in chronic stroke patients was linked to a significant recovery in speech and limb motor function (Mountz, 2007). We have shown earlier that administration of SCF + G-CSF during chronic stroke improves the functional outcome (Zhao et al, 2007a). Here we have observed that the use of the same treatment paradigm led to an increase in the levels of circulating CD34+ cells, in the numbers of BM-derived endothelial cells, and in the vascular density. In line with our data, Toth et al (2008) also observed that SCF +G-CSF enhanced the contribution of BM-derived cells to angiogenesis when administered during the acute phase after brain ischemia. We postulate that SCF + G-CSF-induced mobilization of CD34+ cells may contribute to angiogenesis, which provides a supportive microenvironment for neuronal regeneration, and that both the angiogenesis and the neurogenesis by SCF + G-CSF treatment play roles in functional restoration during chronic stroke.
Pericytes are the cells directly contacting endothelial cells. Unlike endothelial cells, pericytes have a complex ontogeny during development. Although several markers, such as alpha-smooth muscle actin, desmin, neuron-glial 2, and platelet-derived growth factor receptor-β, are considered as the markers for pericytes, none of these are specific for pericytes and none of them recognize all pericytes (Lamagna and Bergers, 2006; Bergers and Song, 2005). Therefore, in this study, we have identified BM-derived pericytes by their location using high-resolution 3D-confocal imaging. We observed a reduction of BM-derived pericytes by SCF + G-CSF treatment during chronic stroke. Although the source of pericytes is heterogeneous, substantial evidence has shown that one of the origins for pericytes is BM (Song et al, 2005) and that endothelial cells and pericytes share a common progenitor cell during development (Lamagna and Bergers, 2006). Here we have found that both pericytes and endothelial cells are derived from adult BM. The most interesting finding is that SCF + G-CSF treatment during chronic stroke leads to the elevation of BM-derived endothelial cells and a decrease in BM-derived pericytes, suggesting that endothelial cells and pericytes share the same stem cells in the adult BM, and that SCF + G-CSF treatment guides BMSC differentiation into endothelial cells by inhibiting pericyte fate differentiation during chronic stroke. This modulation by SCF + G-CSF may contribute to angiogenesis in the chronic phase of stroke, because it has been shown that pericytes inhibit endothelial cell proliferation and angiogenesis (Hirschi et al, 1999).
In summary, in this study we have shown that systemic administration of SCF +G-CSF during chronic stroke can mobilize BMSCs into PB, enhance homing BM-derived cells into the brain, promote differentiation of BM-derived stem cells into endothelial cells and neuron-like cells, and increase angiogenesis. Our data provide new evidence to support the possibilities that (1) a brain permanently damaged by stroke can be repaired by BM-derived cells that are mobilized and homed into the brain by SCF + G-CSF; (2) BM is the source for both endothelial cells and pericytes in adults; (3) SCF +G-CSF govern the fate of BM-derived cells in the brain during chronic stroke; and (4) SCF + G-CSF enhance the contribution of BM-derived cells to angiogenesis and neurogenesis during chronic stroke. These observations provide insight into understanding the role of SCF and G-CSF in stimulating BMSCs/BM-derived cells to repair brain tissue during chronic stroke. The outcome of this study also provides evidence to help in developing a new SCF + G-CSF-based therapy for treatment of chronic stroke, a clinical condition for which there is currently no effective treatment available.
Footnotes
Acknowledgements
This work was supported by an American Heart Association grant (to LRZ) (0665522B), Malcolm Feist Endowment for Cardiovascular Research, CADASIL foundation of America, Louisiana Gene Therapy Research Consortium, and The John Franks Imaging laboratory. The authors thank Kathleen LIorens, Lijia Yin, and Janice Russell for their technical assistance, and Dr David S Knight for editing the manuscript.
The authors state no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
