Abstract
To assess the capability of 18F-2-tert-butyl-4-chloro-5-{6-[2-(2-fluoroethoxy)-ethoxy]-pyridin-3-ylmethoxy}-2H-pyridazin-3-one (18F-BCPP-EF), a novel positron emission tomography (PET) probe for mitochondrial complex I (MC-I) activity, as a specific marker of ischemia-induced neuronal death without being disturbed by inflammation, translational research was conducted using an animal PET in ischemic brains of Cynomolgus monkeys (
INTRODUCTION
The mitochondrial respiratory chain (complexes I to V) is the major site of ATP production in eukaryotes. This organelle not only generates ATP, but also has an important role in the center of the apoptotic signaling pathway. 1 Among these five complexes, mitochondrial complex I (MC-I; NADH–ubiquinone oxidoreductase, EC 1.6.5.3) is the first enzyme of the respiratory electron transport chain. MC-I takes two electrons from NADH and transfers them to ubiquinone in the inner-mitochondrial membrane. Mitochondrial complex I uses the energy released from this process to move four protons across the membrane, creating charge separation across the membrane.
Mitochondrial dysfunction contributes to the pathophysiology of acute neurologic disorders and neurodegenerative diseases. 2 Mitochondria are considered the main intracellular source of reactive oxygen species (ROS) in cells and also the main target of ROS-mediated damage. Ischemia may cause mitochondrial alterations that would favor ROS production when the oxygen concentration is reestablished by reperfusion. 3 We showed that, immediately after reperfusion from the middle cerebral artery (MCA) occlusion (MCAO) for 3 hours (3-hour MCAO), postischemic hyperperfusion was observed in the neocortical area of monkey brain.4, 5 When ischemic tissue is reoxygenated, electron transport through the respiratory chain is impaired because of depletion of ADP during ischemia, and this leads to a burst of ROS generation during the first minutes of reoxygenation. 3
To image MC-I function in the living brain using positron emission tomography (PET), we recently synthesized several novel PET probes,
6
and confirmed that 18F-2-tert-butyl-4-chloro-5-{6-[2-(2-fluoroethoxy)-ethoxy]-pyridin-3-ylmethoxy}-2H-pyridazin-3-one (18F-BCPP-EF) was most preferable for MC-I imaging in the living brain of rat6, 7 and monkey.
8
Since whole-body PET imaging of rat indicated relatively high uptake and long retention in the brain,6, 7 we hypothesized that PET probes for MC-I could be applicable for the imaging of neuronal damage in living brain. It was suggesting that 18F-BCPP-EF could be useful to detect ischemic neuronal damage at the subacute phase 7 days after ischemic insult (Day-7),
7
at which time unexpectedly higher 18F-fluoro-2-deoxy-
The present study aimed to validate 18F-BCPP-EF, a novel PET probe for MC-I activity, as a specific marker of ischemia-induced neuronal death without being disturbed by inflammation. The binding properties of 18F-BCPP-EF were assessed in an ischemic brain model of living monkey using high-resolution PET, and its cerebral uptake was compared with the regional cerebral blood flow (rCBF), regional cerebral metabolism of oxygen (rCMRO2) measured with 15O-gases, 11C-flumazenil (11C-FMZ) binding to the central-type benzodiazepine receptor (CBR), 11C-PBR28 binding to the translocator protein, and regional cerebral blood flow (rCMRglc) measured with 18F-FDG at Day-7/8 after 3-hour MCAO ischemic insult in the monkey brain.
MATERIALS AND METHODS
Animals and Drugs
Animals were maintained and handled in accordance with the recommendations of the US National Institutes of Health and the guidelines of the Central Research Laboratory, Hamamatsu Photonics. The following experiments were approved by the Ethical Committee of the Central Research Laboratory, Hamamatsu Photonics. Seven male Cynomolgus monkeys (
Isoflurane and pancronium were purchased from Dainippon Pharmaceutical (Osaka, Japan) and Sankyo Co. Ltd. (Tokyo, Japan), respectively. Rabbit anti-Iba1 polyclonal antibody was from Wako Pure Chemical Industry (Osaka, Japan). Mouse anti-NeuN monoclonal antibody and EnVision were obtained from Millipore (Billerica, MA, USA) and DAKO (Carpinteria, CA, USA), respectively. Precursors of 18F-BCPP-EF, 11C-FMZ, and 11C-PBR28, and their corresponding cold compounds, were purchased from NARD Institute (Amagasaki, Japan). Mannose triflate and Kryptofix222 (K[2,2,2]) were obtained from ABX (Radeberg, Germany) and Merck (Darmstadt, Germany), respectively.
Positron Emission Tomography Ligand Syntheses
Positron-emitting carbon-11 (11C), oxygen-15 (15O), and fluorine-18 (18F) were produced by 14N(p, α)11C, 14N(d, n)15O, and 18O(p, n)18F nuclear reactions, respectively, using a cyclotron (HM-18; Sumitomo Heavy Industry, Tokyo, Japan) at Hamamatsu Photonics PET Center. Labeled compounds were synthesized using a modified CUPID system (Sumitomo Heavy Industries, Ltd., Tokyo, Japan). HPLC analyses of labeled compounds were performed on a GL-7400 low-pressure gradient HPLC system (GL Sciences, Inc., Tokyo, Japan) with a radioactivity detector (RLC-700; Hitachi Aloka Medical, Inc., Tokyo, Japan).
11C-PBR28 for translocator protein imaging was labeled by 11C-methylation of its
11C-FMZ for CBR was labeled by 11C-methylation of its
The 18F-BCPP-EF was radiolabeled by nucleophilic 18F-fluorination of its corresponding precursor as reported previously.
6
Radiochemical purity was >99%, and specific radioactivity was 139.6±37.0 GBq/
18F-fluoro-2-deoxy-
Positron Emission Tomography Analysis in Monkey Brain
After overnight fasting, monkeys were tracheostomized, immobilized with 0.05 mg/kg pancronium bromide intramuscularly every 2 hours, and artificially ventilated. Anesthesia was continued with 0.8% isoflurane in a N2O/O2/N2 (1:1:1) gas mixture during the entire experiment. After a transmission scan for 30 minutes using a 68Ge-68Ga rotation rod source, emission scanning with 18F-FDG was conducted for 60 minutes, and scans with 11C-BPR28 and 18F-BCPP-EF were performed for 91 minutes after injection using a high-resolution animal PET scanner (SHR-7700; Hamamatsu Photonics, Hamamatsu, Japan). 15 The PET data obtained were reconstructed by the filtered back projection method with a Hanning filter of 4.5 mm full width at half maximum and attenuation correction using the transmission scan data. Individual PET and MRI images were coregistered. Volumes of interest in brain regions were drawn manually on the MRI referring regional information from BrainMaps.org, 16 and volumes of interest of MRI were superimposed on the coregistered PET images to measure the time-activity curves (TACs) of each PET probe for kinetic analyses. These processes were performed using the PMOD software (PMOD Technologies Ltd., Zurich, Switzerland).
For quantitative analysis of 18F-BCPP-EF, PET scans were conducted with arterial blood sampling as reported previously. 8 Arterial blood samples were obtained every 8 seconds up to 64 seconds, followed by 90, 150 seconds, then 4, 6, 10, 20, 30, 45, 60, and 90 minutes after tracer injection, and these blood samples were centrifuged to separate plasma, weighed, and their radioactivity was measured. For metabolite analysis, methanol was added to some plasma samples (sample/methanol=1/1) obtained at 16, 40, 64 seconds, 6, 10, 30, and 45 minutes after the injection, followed by centrifugation. The obtained supernatants were developed using thin layer chromatography plates (AL SIL G/UV, Whatman, Kent, UK) with a mobile phase of ethyl acetate. The ratio of unmetabolized fraction was determined using a phosphoimaging plate (FLA-7000; Fuji Film, Tokyo, Japan). The input function of unmetabolized 18F-BCPP-EF was calculated using the data obtained by correction of the ratio of the unmetabolized fraction to total radioactivity, which was used as the arterial input function.
As confirmed in our previous study,
8
kinetic analysis of 18F-BCPP-EF was performed by 2-TC analysis,17, 18, 19 which was performed using the PMOD software (PMOD Technologies Ltd.). The unknown rate constants (
The quantitative analyses of 11C-FMZ and 11C-PBR28 were performed by Logan graphical plot analysis 19 using the PMOD software (PMOD Technologies Ltd.). As an input function, the TAC in the cerebellum or pons was applied instead of the TAC of metabolite-corrected plasma.
Assessments of rCBF, rCMRO2, regional oxygen extraction fraction, and regional cerebral blood volume were conducted using the steady-state 15O-gas inhalation method,4, 5 with the successive inhalation of trace amounts of 15O-CO2, 15O-O2, and 15O-CO. The mean values of radioactivity of whole blood and plasma were used for parametric image generation of rCBF, rCMRO2, regional oxygen extraction fraction, and regional cerebral blood volume.20, 21 The CBV correction was performed for rCBF, rCMRO2, and regional oxygen extraction fraction measurements.
During PET measurement with 18F-FDG for 60 minutes, 16 of arterial blood samples were withdrawn for arterial radioactivity measurements. The blood samples obtained at 45 and 60 minutes after injection were also analyzed for blood glucose levels with STAT PROFILE CCX (Nova Biomedical, Waltham, MA, USA). Positron emission tomography images from 40 to 60 minutes after 18F-FDG injection were used to calculate rCMRglc by an autoradiographic method with an operational equation 22 and parameters with lumped constant of 0.56. 23
Brain Ischemic Model of Monkey
Monkeys were tracheostomized, immobilized with 0.05 mg/kg pancronium bromide intramuscularly every 2 hours, and artificially ventilated (Cato, Drager, Germany). Anesthesia was continued with 0.8% isoflurane in a N2O/O2/N2 (1:1:1) gas mixture during the entire experiment. The mean arterial blood pressure, heart rate, rectal temperature, arterial PO2 and PCO2, and pH were continuously or regularly monitored. During the experiments, the animal's body temperature was maintained within normal limits with heating blankets. The right MCA was occluded with two microvascular clips
Regions of interest (ROIs) of inflammatory regions at Day-7 (ROIPBR), defined as areas with >20% increase in 11C-PBR28 binding in the ipsilateral relative to the contralateral hemisphere, were placed on each quantitative image of rCMRO2, 11C-FMZ, 18F-FDG, rCMRglc, and 18F-BCPP-EF. Furthermore, ROIs of infarct regions at Day-7 (ROIInfarct), defined as areas with <40% decrease in rCMRO2 in the ipsilateral relative to the contralateral hemisphere as previously reported, 24 were placed on each quantitative image. The average ROIPBR and ROIInfarct were created from seven animals, and drown on MRI.
Immunohistochemical Assessment
Immunohistochemical assessments were performed with brains sampled at Day-7 after ischemic insults using rabbit anti-Iba1 polyclonal antibody and mouse anti-NeuN monoclonal antibody as reported previously.5, 9 Brains were perfused transcardially with saline, then 4% paraformaldehyde in 0.1 mol/L sodium phosphate (pH 7.4) under anesthesia with overdose sodium pentobarbital. The brains were subsequently removed and postfixed overnight at 4°C in 4% paraformaldehyde, then rapidly frozen in dry ice powder and sliced into 20-
Statistical Analysis
Results are expressed as means±s.d. Comparisons between conditions were performed using the paired, two-tailed Student's
RESULTS
Aided by individual monkey MRI (Figure 1A), several ROIs were set on PET images of the brain of normal animals to obtain the TACs of 18F-BCPP-EF in each region (Figure 1B, upper panel). The TAC of 18F-BCPP-EF in normal animals peaked at ca. 10 minutes after injection, except for the occipital cortex (peak time=ca. 30 minutes), followed by gradual elimination with time (Figure 2A). The uptake levels at 20 minutes after injection were the highest in the temporal cortex and the striatum, intermediate in the frontal cortex, and the lowest in the occipital cortex, the hippocampus, and the cerebellum in the normal brain (Figure 2A).
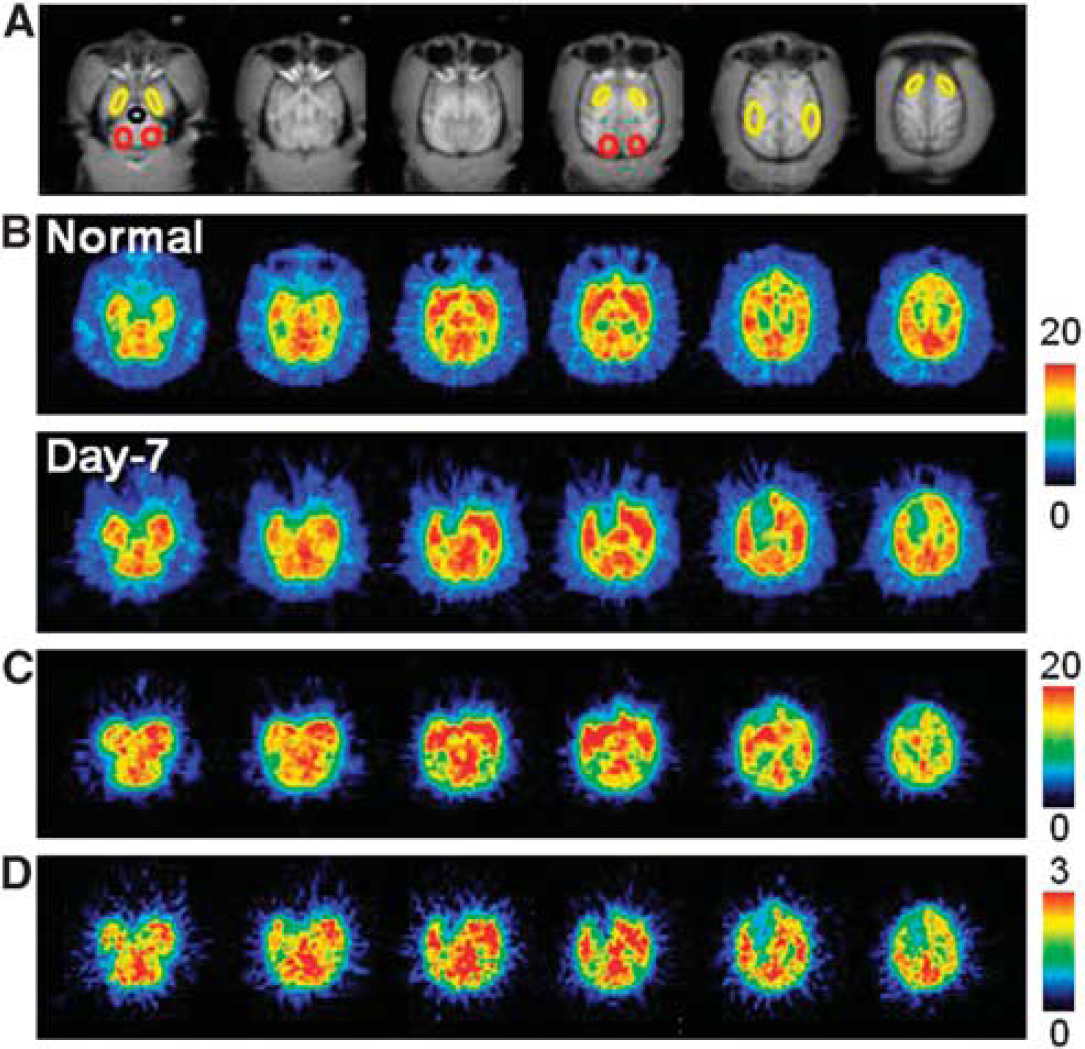
Typical magnetic resonance imaging (MRI) (
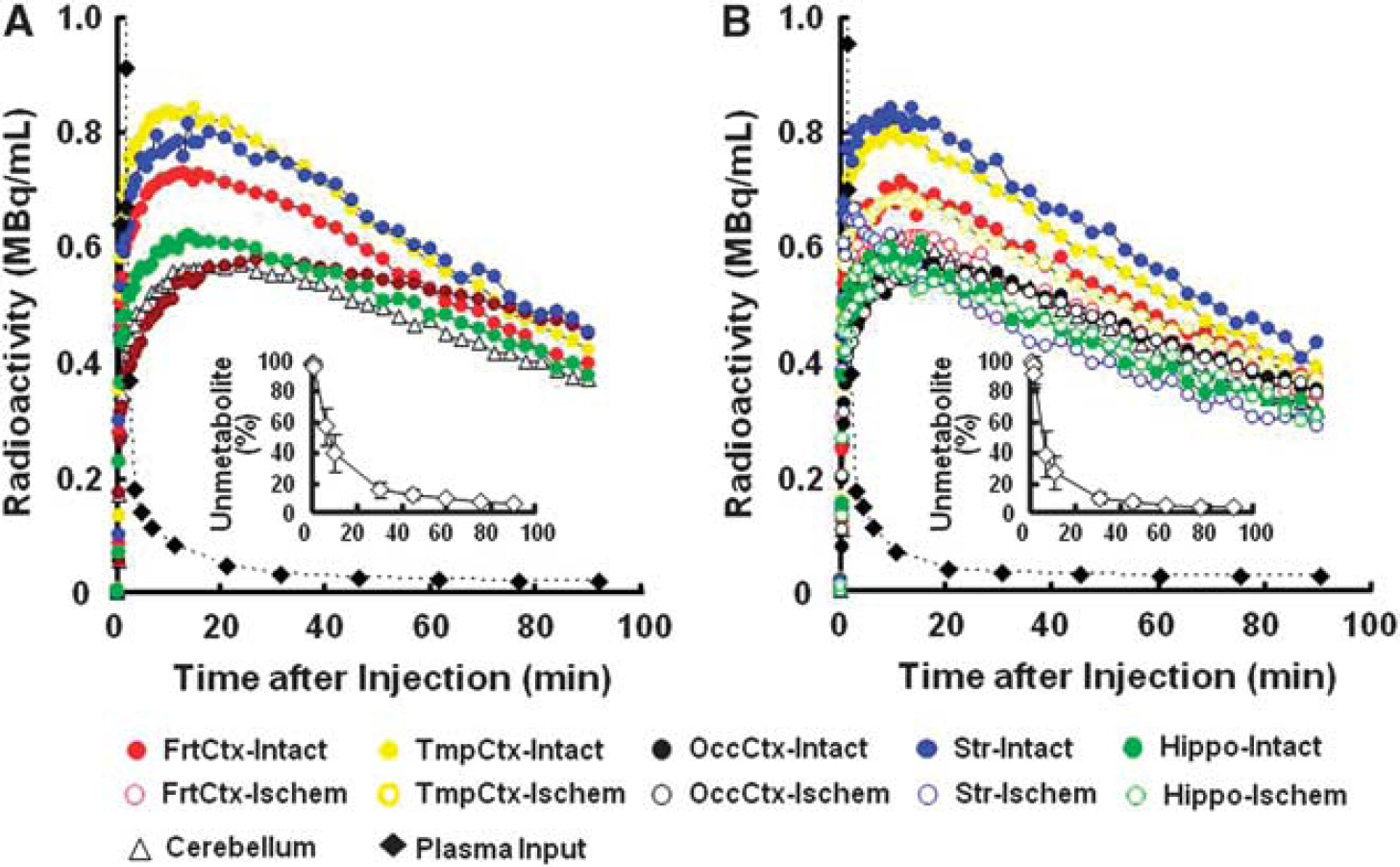
Effects of 3-hour middle cerebral artery (MCA) occlusion (MCAO) ischemic insult on time-activity curves (TACs) of 18F-BCPP-EF in the living brains, metabolite-corrected plasma input, and metabolic profile (insets of
The kinetics and metabolic rate of 18F-BCPP-EF in the plasma were very rapid; only ca. 6% of nonmetabolized parent compound was detected 90 minutes after the injection (insets of Figures 2A and 2B). Although the chemical species of metabolites have not yet been determined in plasma, they were very polar metabolites unable to enter the brain through the blood–brain barrier. With rapid metabolism of 18F-BCPP-EF, no significant radioactivity accumulation was observed in the skull of monkey, suggesting the minimal defluorination metabolism of 18F-BCPP-EF in plasma and brain tissue (Figure 1B).
As reported previously,4, 5 MCAO of the monkey brain for 3 hours acutely decreased rCBF and rCMRO2 mainly in the striatum, which was the focal area for the supply of blood by the MCA, in the present study (data not shown). At Day-7 after 3-hour MCAO ischemic insult, lower rCMRO2 was detected not only in the striatum, but also in the cortex (Figures 1D and 3B), suggesting the induction of ischemic damage including reperfusion injury in the monkey brain. Our previous results showed that, immediately after reperfusion, postischemic hyperperfusion was observed in the neocortical area of monkey brain.4, 5 In these hyperperfused areas, decreased rCMRO2 was observed thereafter. In contrast, rCBF in the ischemia-damaged area indicated almost the same levels as in the intact area (Figures 1C and 3A).
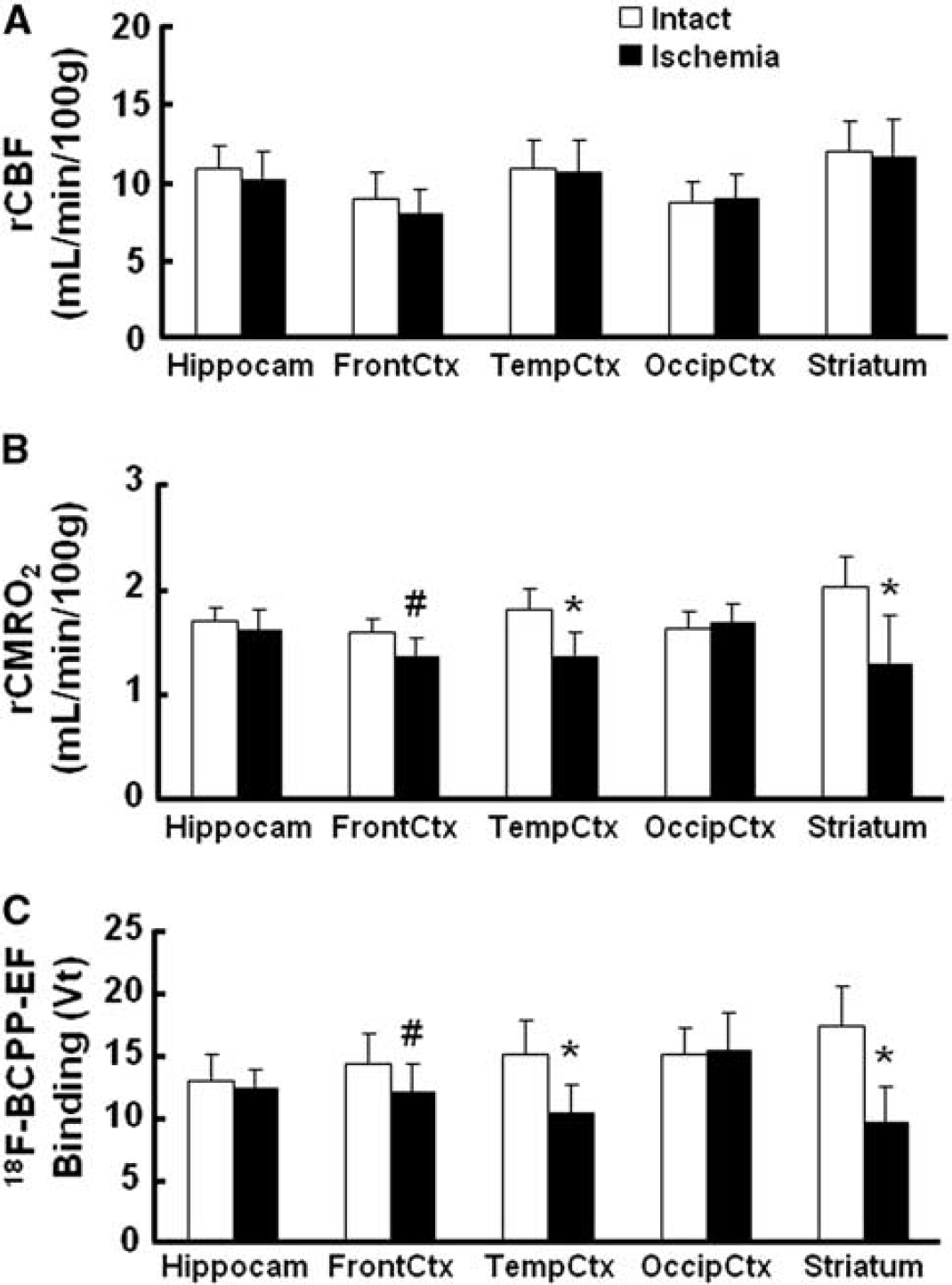
Effects of 3-hour middle cerebral artery occlusion (MCAO) ischemic insult on regional cerebral blood flow (rCBF) (
In the monkey brain after 3-hour MCAO ischemic insult, the TAC of 18F-BCPP-EF also provided a similar TAC pattern as well as uptake levels in the intact side of the brain, showing a peak 10 to 30 minutes after the injection and the uptake levels being the highest in the temporal cortex and the striatum, intermediate in the frontal cortex, and the lowest in the occipital cortex, the hippocampus, and the cerebellum (Figure 2B). In contrast, the TAC in the striatum, and frontal and temporal cortices of the right ischemic hemisphere revealed significantly lower levels than those in the intact hemisphere (Figure 2B). As shown in Figure 1B, the parametric images of
On the PET images of 18F-BCPP-EF (Figure 1B, lower panel), rCBF (Figure 1C), and rCMRO2 (Figure 1D) measured at Day-7 after 3-hour MCAO ischemic insult, the same ROIs, determined with the aid of MRI (Figure 1A), were set on these PET images including both intact and ischemic hemispheres, and then 18F-BCPP-EF data were plotted against rCBF (Figure 4A) or rCMRO2 (Figure 4B). The results showed that the regional
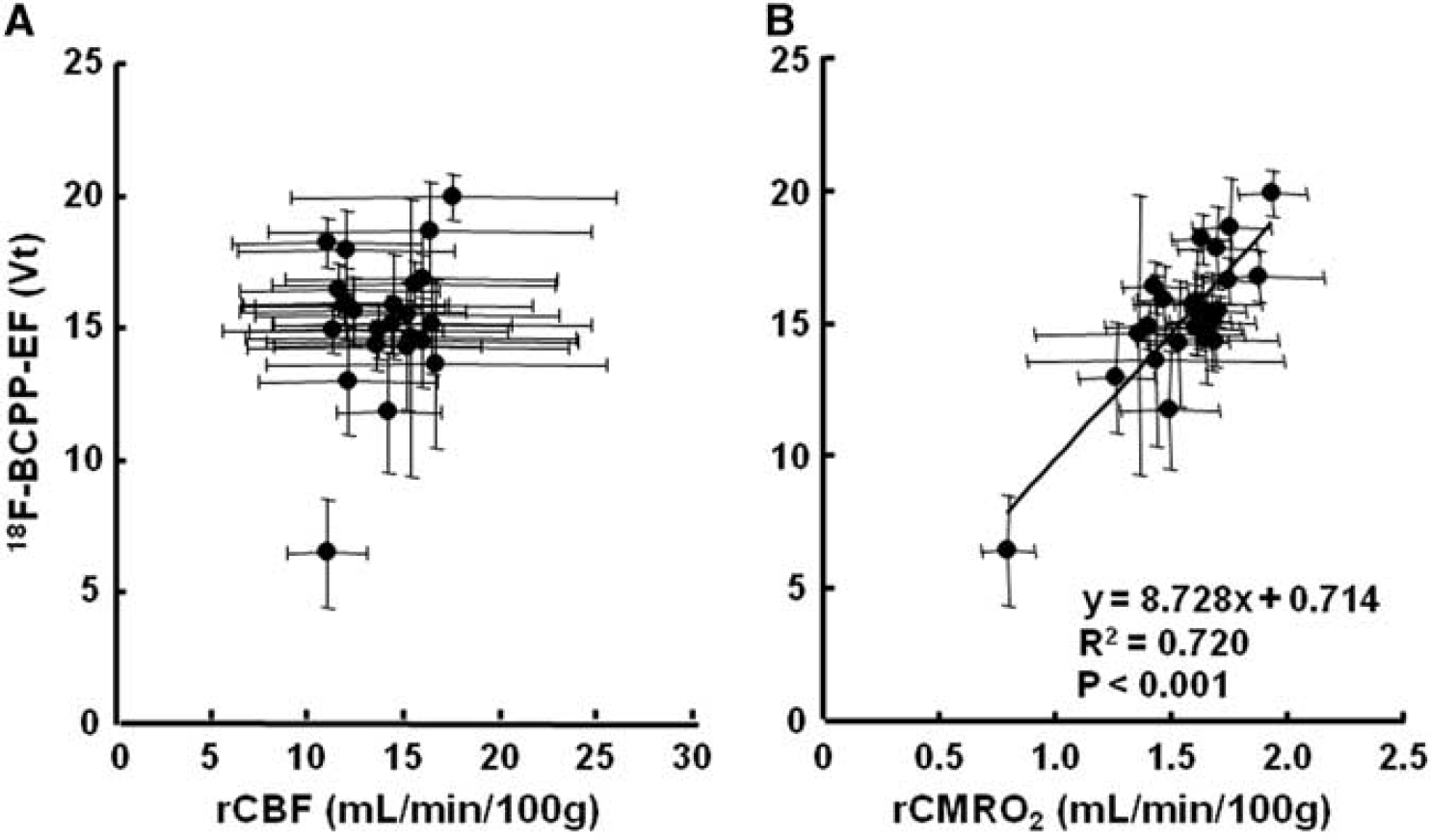
Correlations of total distribution volume (
At Day-7 after 3-hour MCAO ischemic insult, in addition to the ROI-based analyses described above, ROIs of inflammatory regions (ROIPBR), defined as areas with >20% increase in 11C-PBR28 binding in the ipsilateral relative to the contralateral hemisphere (Figure 5A and red and white colored area in Figure 5F), were placed on each quantitative image of rCMRglc, rCMRO2, 11C-FMZ, and 18F-BCPP-EF (Figures 5B to 5E). The rCMRglc was significantly higher in the ROIPBR of the right ischemic side than that on the corresponding intact side (Figures 5B and 5I). In contrast, 18F-BCPP-EF (Figures 5E and 5I) as well as 11C-FMZ (Figures 5C and 5I) and rCMRO2 (Figures 5D and 5I) was significantly lower in the ROIPBR of the right ischemic side than on the corresponding intact side. These areas revealed high immunoreactivity to anti-Iba 1 antibody (Figure 5G) and low immunoreactivity to anti-NeuN antibody (Figure 5H).
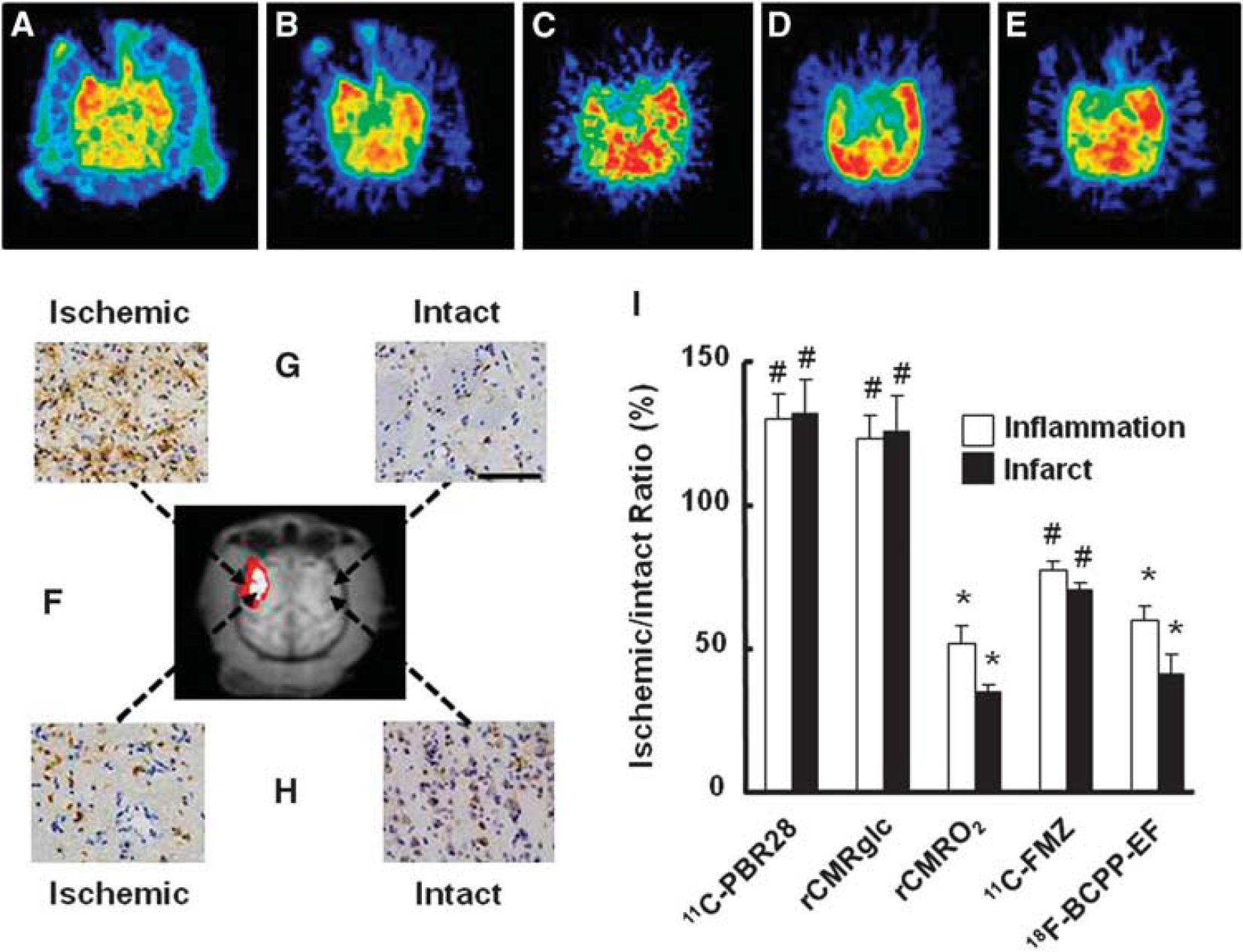
Effects of inflammation with accumulation of activated microglia on 11C-PBR28 binding (
As shown in Figure 5I, the average rCMRO2 value decreased from 51.8% in ROIPBR to 34.6% in ROIInfarct (white colored area in Figure 5F), which was reasonable because the infarct was determined as rCMRO2 of <40% of intact side. The
DISCUSSION
The present results suggest the potential of 18F-BCPP-EF to be used as a PET probe for the assessment of normal and ischemic impaired MC-I in living brain of nonhuman primate (
It has been proposed that an ideal tracer should have high and rapid uptake, rapid washout, followed by reasonably long specific retention in the tissues with the target molecules.
28
From these points of view, the previous6, 7, 8 and present studies suggested that 18F-BCPP-EF could be an ideal PET tracer for MC-I imaging because an appropriate lipophilicity of 18F-BCPP-EF (logD7.4=3.03)
6
could contribute for suitable blood–brain barrier penetration as well as for optimal
It was confirmed that the uptake of 18F-BCPP-EF reflected the specific binding to cellular MC-I,7, 8 which is the first component of four electron transport complexes in the inner mitochondrial membrane. By
18F-fluoro-2-deoxy-
In developed cells like neurons, glucose is converted into pyruvate through a tricarboxcylic acid cycle and ultimately produces energy
The present results showed that the ischemic-induced infarct areas (ROIInfarct), determined as region with rCMRO2 of <40% of intact side at Day-7 as previously reported,
24
showed much lower
It has been reported that ischemic insult induces the diaschisis which is defined as a depression of regional blood flow and metabolism caused by dysfunction in an anatomically separate but functionally related neuronal region, however, may not reflect neuronal death.
36
The present study showed that the
In the present study, 11C-FMZ-PET was also conducted to assess the neural damage in ischemic regions, since CBR was considered to be a superior method for accurate detection of ischemic cerebral damage in primates.29, 37, 38 However, since the CBR is exclusively located in the cortex, not in the white matter and basal ganglia, 11C-FMZ can detect the neuronal damage in the cortex. In contrast, 18F-BCPP-EF can image to detect the neuronal damage in whole brain. In addition, the present study showed less 11C-FMZ binding to CBR at Day-7 after 3-hour MCAO cortical ischemic insult than rCMRO2 and 18F-BCPP-EF binding. It was reported that there were no significant changes in the
One limitation of the present study was that MR imaging could not be conducted after ischemic insult to detect anatomic and pathologic changes because of the Japanese low as described in Materials and methods. If MR imaging data after ischemic insult were coregistered with PET data presented here, then the deeper insight might be obtained for changes in MC-I activity.
In conclusion, the present study succeeded to validate 18F-BCPP-EF, a novel PET probe for MC-I activity, as a specific marker of ischemia-induced neuronal death without being disturbed by inflammation.
Footnotes
The authors declare no conflict of interest.
ACKNOWLEDGMENTS
The authors gratefully acknowledge the technical assistance of Aiko Iwazaki, Kaori Suzuki, and Shigeyuki Yamamoto.
