Abstract
Cryptococcus neoformans meningoencephalitis (CNME) is a leading fungal cause of death among acquired immunodeficiency syndrome patients. Innovative preclinical systems that permit high throughput in vivo evaluation of novel agents are desperately needed. Magnetic resonance imaging (MRI) was evaluated as a tool to develop a rat model of CNME and to quantify noninvasively blood—brain barrier (BBB) disruption secondary to this disease. The aim of this study was to identify MRI changes compared with histopathology and fungal burden measurements as potential biomarkers. A well-characterized strain of C neoformans (CN) var grubii was used to infect rats using intravenous inoculation. An inoculum-finding study was performed by infecting rats with 103, 105, and 107 colony-forming units (CFUs). Animals underwent dynamic MRI on days 4 and 7 after inoculation. An inoculum-confirming study was performed by infecting rats with 105, 106, and 107 CFU and fungal burden was determined in the brain, lung, and spleen. Animals infected with 107 CFU of CN developed lesions that appeared hyperintense on T2-weighted images on day 4. The histopathology results correlated well with MRI data. Diffusion weighted and permeability estimates were 1.4 and 6.1-fold higher, respectively, in lesions compared with healthy tissue. Magnetic resonance imaging is a promising preclinical tool to evaluate effects of antifungal and adjunctive agents.
Introduction
Cryptococcus neoformans (CN) is an encapsulated yeast that has been classified into three capsular serotypes (A, D, and AD; Bicanic and Harrison, 2004). The serotype A isolates are referred to as CNvar grubii and account for 95% of cryptococcal infections worldwide (Bicanic and Harrison, 2004; Lakshmi et al, 2007; Lui et al, 2006). The microorganism is ubiquitous and can be acquired by inhalation to cause a pulmonary infection (Kuroki et al, 2004). Impaired cell-mediated immunity as seen in patients with acquired immunodeficiency syndrome (AIDS) permits CN to spread from the lung and infect the brain. The greatest incidence of cryptococcosis is in sub-Saharan Africa, where an estimated 25 million patients are infected with HIV (McCarthy et al, 2006). Cryptococcosis has been characterized as the AIDS defining illness in 40% to 68% of HIV patients in southern Africa (McCarthy et al, 2006; Wong et al, 2007). A mortality rate of 20% to 100% is associated with this disease and is more likely to occur in patients that develop brain lesions (Bicanic and Harrison, 2004; Dromer et al, 2007; Kisenge et al, 2007; Mwaba et al, 2001).
The most common clinical presentation of cryptococcosis is meningitis with typical symptoms of severe headache, fever, and a depressed level of consciousness (Bicanic and Harrison, 2004). However, the infection affects both the subarachnoid space and brain parenchyma and thus is more appropriately termed C neoformans meningoencephalitis (CNME). Several studies have evaluated the radiologic features of patients with proven CNME using magnetic resonance imaging (MRI; Berkefeld et al, 1999; Gultasli et al, 2007; Miszkiel et al, 1996; Narai et al, 2001). The largest series to date included a retrospective evaluation of 25 AIDS patients with microbiologically proven CNME (Miszkiel et al, 1996). All patients showed abnormal dilatation of Virchow—Robin spaces (hyperintense on T2-weighted (T2w) images), cryptococcomas that were greater than 3 mm in size and were hyperintense on T2w images. Abnormal dural or leptomeningeal enhancement and other mass lesions were also identified. Unfortunately, noninvasive methods such as MRI have not been exploited to discriminate disease severity or as a tool to monitor outcomes associated with pharmacological interventions. Magnetic resonance imaging offers several advantages for its application to animal research, it is a noninvasive imaging technique that provides good soft tissue contrast, allows longitudinal study of disease process in animal models, and testing of novel pharmaceutical agents as part of newer treatment strategies. In this study, T2, diffusion weighted (DW), and blood—brain barrier (BBB) permeability estimation methods have been used to study brain lesions of infected animals. The choice of this particular imaging strategy was based on the hypothesis that lesions produced by cryptococcus infection are visible as regions with high signal on T2w images (suggesting they are fluid filled) and thereby could be uniquely identified and studied using DW MRI. In addition, it was hypothesized that the invasive nature of the CN pathogen can be attributed to its ability to cause BBB damage that allows it move from the vascular into the tissue compartment. Moreover, the BBB permeability estimation would allow us to use permeability coefficient as a potential quantitative neuroimaging biomarker for studying the efficacy of novel pharmaceutical molecules for treating CNME. The MRI-based multiple-time graphical analysis (MTGA) method for estimating BBB permeability was used in this study. The technique was recently proposed by Ewing et al (2003) and has been used to investigate the BBB blocking effect of novel pharmaceutical molecules (Sood et al, 2008).
The current pharmacological approach to CNME includes a combination of amphotericin B and 5-fluorocytosine for 2 weeks followed by prolonged fluconazole maintenance therapy (Saag et al, 2000). Although effective, this pharmacological approach has been too complex to benefit patients in sub-Saharan Africa (Lortholary, 2007). Development of animal models to study newer antifungal agents could lead to more practical and effective treatment strategies for AIDS patients with CNME. Several mammalian models including the mouse, rat, guinea pig, and rabbit have been used to study CNME (Carroll et al, 2007). Almost all models used the less virulent serotype D strain of CN and not the clinically dominant serotype A strain to infect animals. Current animal models of CNME also use an intracisternal route of CN inoculation to induce disease (Carroll et al, 2007). This method of inoculation has a 15% procedural mortality rate and does not mimic pathologic assessment noted in patients with AIDS (Goldman et al, 1996). As a result, we sought to characterize the ability of a CN serotype A strain to cause CNME in a rat model using an intravenous route of inoculation.
Thus, the aims of this study were (1) to define using MRI, the optimal size of inocula to induce CNME; (2) to correlate MRI results with histopathology findings and brain fungal burden measurements; and finally, (3) to identify qualitative and quantitative (apparent diffusion coefficient (ADC) and permeability coefficient) MRI-based parameters that would be useful to follow the longitudinal course of the disease process and BBB integrity in an animal model.
Materials and methods
Animal Preparation
The study was approved by the University of New Mexico Institutional Animal Use committee and conformed to the National Institutes of Health guidelines for the ethical use of animals. Male 250 to 350 g Fischer F344 (Charles River Laboratories, Wilmington, MA, USA) rats were used. Animals were acclimated in a 12 h dark—light cycle vivarium with free access to food and water for at least 1 week before and during experiments.
Organism Preparation
We used a single CN serotype A strain (H99, ATCC 208821) given extensive genetic and phenotypic characterization of this strain. A 48-h old culture of CN on Sabouraud dextrose agar was used to prepare the inocula with an optical density of 1.0 at 530 nm in phosphate-buffered saline. We had previously showed that this process generated viable inocula of 1 × 108 colony-forming units (CFUs) per mL. The inocula was serially diluted 1:10 using phosphate-buffered saline to achieve 1 × 106, 1 × 105, and 1 × 104 CFU/mL.
Infection Model
Animals were restrained using a Tailveiner device (Braintree Scientific, Braintree, MA, USA) and a 24-gauge 5/8-inch catheter was used to achieve lateral tail vein access. A 0.1 mL volume of inocula of CN was injected followed by 0.5 mL bolus of normal saline though this access. Animals were divided into three per group based on an injected inocula of 1 × 103, 1 × 105, and 1 × 107 CFU, respectively (experiment 1). On the basis of initial results, experiments were repeated in a similar design with inocula of 1 × 105, 1 × 106, and 1 × 107 CFU (experiment 2). The final animal model development study included use of nine animals inoculated at 1 × 107 CFU (experiment 3), which was repeated (experiment 4). Assessment of changes in BBB integrity was determined in five animals inoculated at 1 × 107 CFU (experiment 5). The experimental duration was up to 15 days for experiment 1, 28 days for experiments 2 to 4, and 5 days for experiment 5. Three animals were killed on days 7, 14, and 28 to quantify brain, lung, and spleen fungal burden during experiments 3 and 4 over time. The overall study design was to first complete an inoculum-finding study (experiment 1), an inoculum-confirming study (experiment 2), an organ fungal burden time-course study (experiments 3 and 4), and an in vivo evaluation of BBB integrity study (experiment 5).
Animal Monitoring
Animals were weighed daily and evaluated for signs and symptoms of distress. Animals who showed a >20% weight reduction, marked diminished activity, hunched back posture, shivering, chromodacryorrhea, or deemed to be in distress were evaluated for early killing. Killing was performed using intraperitoneal injection of 0.25 mL of SleepAway (sodium pentobarbital, 7.8% isopropyl alcohol).
Animal Imaging Timeline
Rats in experiment 1 underwent the structural MRI study on day 4 after inoculation. The MR images acquired on day 4 showed lesions in the brain because of the fungal infection and as a result the rat infected with 107 CFU underwent MRI on day 7 to assess disease progression. In addition, rats infected with 105 CFU were reimaged on day 7 to evaluate a potential time delay in the formation of brain lesions. Animals that underwent the MRI study were subsequently killed and their brains were harvested for further histologic analysis (day 7). Rats in experiment 5 underwent a structural MRI study at baseline and completed the DW and MTGA-based BBB permeability estimation study on day 4 after inoculation. These rats were killed on day 5 and their brains were harvested for histologic analysis.
MRI
The MRI study was performed in two steps. In step 1, MRI was performed to determine the optimal inocula that would enable visualization of CNME lesions on MR images. In step 2, MRI was performed in rats injected with the optimal inocula determined in step 1 of the study. In step 2 of the MRI study, the MR protocol consisted of T2, DW, and BBB permeability measurement techniques.
Magnetic resonance imaging was performed on a 4.7 T Biospec dedicated research MR scanner (Bruker Biospin, Billerica, MA, USA), equipped with 500 mT/m (rise time 80 to 120 μs) gradient set (for performing small animal imaging) and a small bore linear RF coil (ID 72 mm). Animals were anesthetized using isoflurane gas (induction dosage 2% to 3%; maintenance dosage 1.5% to 2%) in 70% N2O and 30% O2 and real-time monitoring of physiological parameters (heart rate and respiratory rate) was performed during the entire duration of the study. The level of anesthesia was adjusted to maintain a respiratory rate of 40 to 50 breaths per minute. Initial localizer images were acquired using the following parameters: two-dimensional FLASH, TR/TE 10/3 ms, matrix 256 × 128, field of view (FOV) 6.4 cm, 1 slice per orientation. After the localizer images were acquired, T2w and diffusion-weighted imaging was performed with the following parameters; T2w two-dimensional rapid acquisition with relaxation enhancement, TR/TE 4,000/65 ms, FOV 3.2 × 3.2 cm, slice thickness 2 mm, slice gap 1 mm, number of slices 11, matrix 256 × 128, number of averages 15, receiver bandwidth 100 kHz; diffusion-weighted imaging (two-dimensional DW rapid acquisition with relaxation enhancement), TR/TE 2,000/31.2 ms, FOV 3.2 × 3.2 cm, slice thickness 2 mm, slice gap 1 mm, number of slices 11, matrix 64 × 64, number of averages 22, receiver bandwidth 100 kHz, d=5 ms, D=20 ms, b-value of 0 and 900 secs mm−2, diffusion gradient along the slice select direction. The slices for the T2w and DW protocols were prescribed such that they were exactly matched in location, slice thickness, and FOV.
After determining the optimal inocula in step 1 of the study, rats infected with the optimal dose were brought to the MRI suite at day 4 after inoculation, for step 2 of the imaging study. A 24-gauge 5/8-inch catheter was used to achieve lateral tail vein access and locked with 100 IU/mL of heparin sodium to maintain patency for the MRI study duration. A 24-inch tubing set was connected to the catheter to permit distal injection of Gd-DTPA without removal of the animal from the magnet. Initial localizer images were acquired using the following parameters: two-dimensional FLASH, TR/TE 10/3 ms, matrix 256 × 128, FOV 6.4 cm, 1 slice per orientation.
After acquiring the localizer images, T2w and diffusion-weighted imaging was performed with the parameters as described in the previous paragraph. The slices for the T2w and DW protocols were prescribed such that they were matched in location and FOV. The MR protocol for acquiring data for the MTGA method was then implemented. In this acquisition, a reference baseline acquisition using the fast T1 mapping protocol was obtained. Gd-DTPA (0.2 mmol/kg; Magnevist-Bayer HealthCare Pharmaceuticals, Montville, NJ, USA; molecular weight 938 Da) was injected as a bolus into the tail vein through the in-dwelling catheter, followed by imaging with rapid T1 mapping protocol for a total time of 45 mins, resulting in 14 time points (15 including the preinjection baseline scan). The following optimized MRI parameters were used for this protocol: axial plane, two-dimensional IR-SE-EPI, TR/TE 7.9 secs/19.4 ms, FOV 4.0 × 4.0 cm, slice thickness 2 mm, slice gap 1 mm, number of slices 2, matrix 64 × 64, number of averages 2, receiver bandwidth 250 kHz. Nonslice-selective magnetization inversion was performed using a hyperbolic secant (sech) RF pulse with pulse width 4 ms. The T1 mapping-specific parameters for this protocol were time for inversion (TI), TI=(100+600 × n) ms where n=0, 1, 2, … 11, number of TI points (n) is 12. The total scan time was 3 mins and 12 secs for each time point.
The acquired data were transferred to a dedicated computer workstation for postprocessing. Postprocessing of the raw data involved generating ADC maps from diffusion-weighted images. Data processing was performed using MRVision software (version 1.67; Winchester, MA, USA) implemented on a 64-bit processor (AMD64) workstation running Red Hat Enterprise Linux version 3 (64 bit). Image analysis was performed using ImageJ (NIH, Bethesda, MD, USA) and MRVision (version 1.67) software. The average ADC value for the healthy brain tissue was 0.68±0.08 × 10−3 mm2 per second. The processing of the MTGA data for estimating permeability coefficient requires an input for the plasma concentration of the injected tracer. In this study, the time profile for plasma concentration of the tracer was obtained from the individual rat sagittal sinus. In the permeability coefficient maps reconstructed using MRI data, pixel signal intensity values are proportional to permeability coefficient values. Please find the details of the underlying theory and data processing used in this study for the MTGA method, in Sood et al (2007).
Brain Fungal Histology and Burden
Killed animals were decapitated and the brains were harvested and immediately fixed in 10% buffered formalin acetate solution for 48 to 72 h at room temperature. A vibratome was used to generate 5 μm coronal slices of brain tissue. Tissue specimens were stained with hematoxylin and eosin, Mayer's mucicarmine, and periodic acid—Schiff stains, respectively. Histology was performed on each animal that underwent MRI in experiments 1 and 5. For fungal burden assessment, organs (brain, lung, and spleen) were harvested within 15 mins of killing, weighed, and homogenized in cold phosphate-buffered saline using the Bead Beater System (Biospec, Bartlesville, OK, USA). A 100 μL aliquot was serially diluted and a 20 μL sample was plated in triplicate on Sabouraud dextrose agar. The agar plates were incubated for 24 to 48 h at 35°C and CFUs were enumerated.
Statistical Analyses
The organ fungal burden between inocula was compared using one-way analysis of variance with post hoc comparisons using Tukey's test. Change in fungal burden over time was compared using repeated-measures analysis of variance with time as the repeated factor. A P-value <0.05 was considered statistically significant.
Results
Inoculum-Finding Study (Experiment 1)
Magnetic resonance images acquired on day 4 after infection showed multiple, diffuse, well-circumscribed intracerebral lesions that appeared hyperintense on T2w images. An increase in ADC (mean±s.d., 1.15±0.06 × 10−3 mm2 per second) was observed in the cryptococcal brain lesions on the ADC maps. Intracerebral lesions were only visible on T2w images in animals that were inoculated with the highest CFU, that is, 107 CFU of CN. There was good correlation between the brain lesions seen on T2w images and histopathology staining method (Figure 1). Qualitative assessment of MR images acquired on day 7 after inoculation, showed an increase in number and size of visible intracerebral lesions that had similar characteristics to lesions on day 4. Animals inoculated with 103 and 105 did not have visible lesions on histopathology and also matched MR results. The brain fungal burden was below the limit of detection (102 CFU/g) in rats infected with 103 CFU, and was detectable (102.2 CFU/g) in 1 rat infected with 105 CFU. In contrast, both rats infected with 107 CFU had fungal burdens that exceeded 104 CFU/g. Furthermore, necropsy data revealed that the lung, liver, and spleen were grossly infected (visible miliary lesion) in rats infected with 107 CFU. Histopathology (Figure 2) revealed noninflammatory cystic lesions that were densely filled with CN (Mayer's mucicarmine, periodic acid—Schiff) and had a classic ‘soap bubble’ type appearance (hematoxylin and eosin). These features mimicked histopathology noted in previous studies of AIDS patients with CNME (Kalra and Bansal, 2004; Miszkiel et al, 1996).
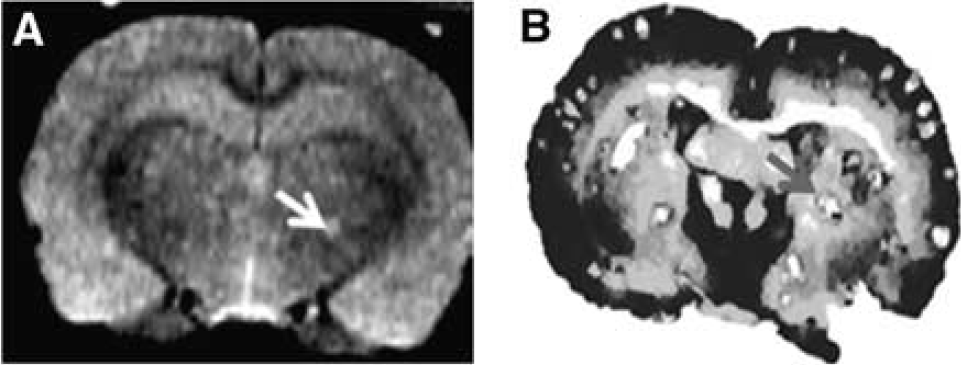
T2w image (
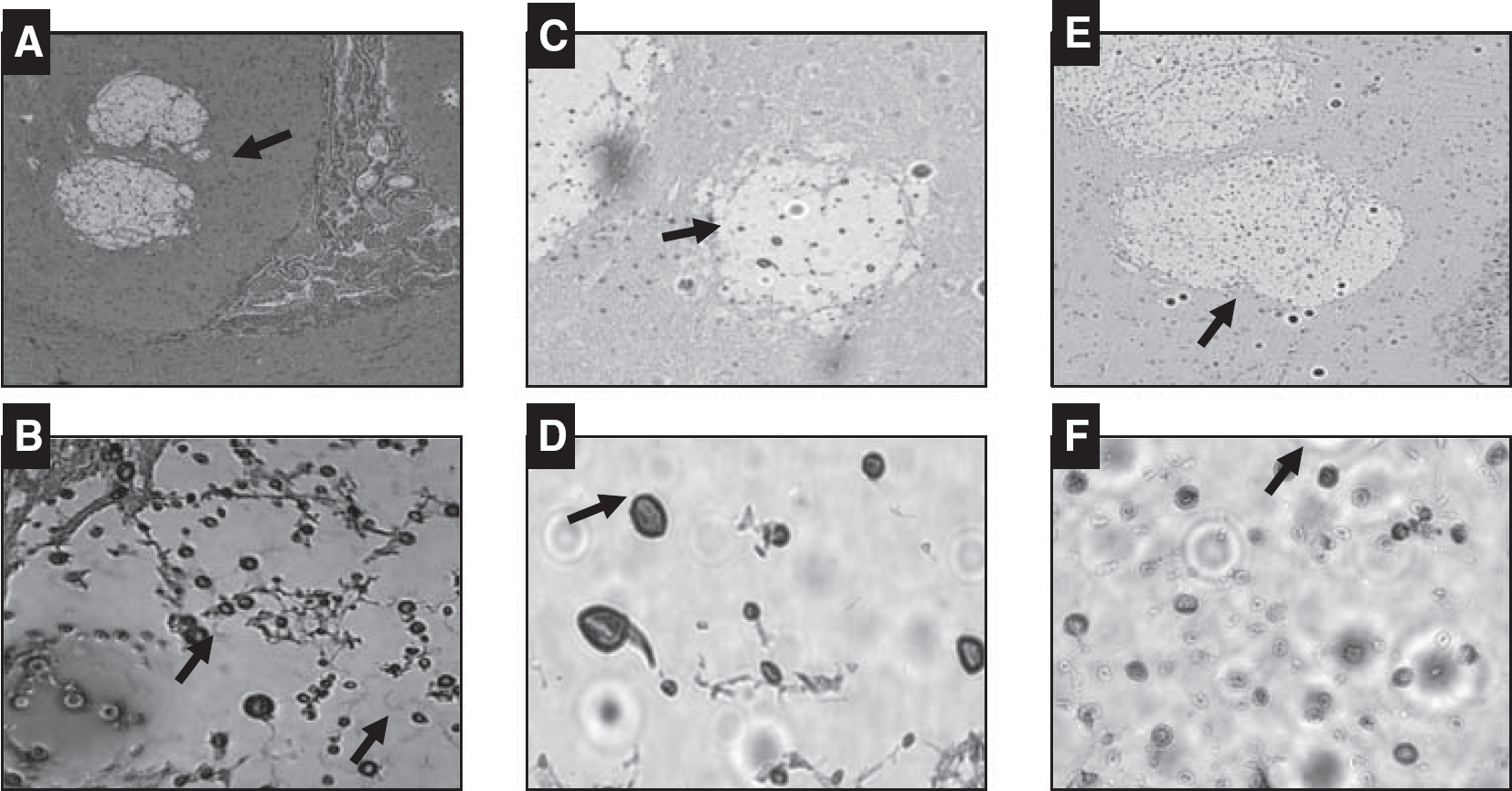
Brain histology showing ‘soap bubble’ noninflammatory cystic lesions with visible C neoformans cells and capsular structure based on Mayer's mucicarmine stain (
Inoculum-Confirming Study (Experiment 2)
Given that the previous study identified a difference in the brain fungal burden between 107 and 105 CFU inocula. The aims of this experiment were to confirm the difference in effect relative to inocula, evaluate the effect of inoculation at 106 CFU, and evaluate animals over a 28-day period to evaluate survival. Two rats (n=6) were killed on day 14, one each in the 107 (n=3) and 106 (n=3) CFU inoculation groups. Both rats elicited a 7% to 9% weight loss over this time period, had a hunched back posture, were shivering, had a slow gait, and a general depressed level of activity. The log10 CFU per gram mean±s.d. brain fungal burden was 6.52±0.71, 4.17±1.93, and undetectable in the animals infected with 107, 106, and 105 CFU, respectively. Similarly, the log10 CFU/g mean±s.d. lung fungal burden was 6.15±0.21, 5.36±2.93, and 4.9 (1 rat, below limit of detection in 2 rats) in the animals infected with 107, 106, and 105 CFU, respectively. Fungal burden was undetectable in the spleen of all rats with the exception of the two rats that were killed on day 14, which had a burden (log10 CFU/g) of 2.8 (107 inocula) and 2.1 (106 inocula). The brain and lung fungal burdens for rats infected with 106 and 107 were not significantly different (P>0.05) compared with one another but were significantly higher (P<0.05) than the 105 inocula group. However, animals infected with 107 CFU had a 100% probability of developing brain and lung cryptococcosis with documentation of lesion formation by MRI. As a result, the 107 CFU inocula were selected as the infection-generating dose.
Fungal Burden Time Course (Experiments 3 and 4)
The mean±s.e.m. change in brain, lung, and spleen fungal burden is illustrated in Figure 3. The fungal burden in brain relative to lung was not statistically significant for day 7 (P=0.16), day 14 (P=0.10), or day 28 (P=0.87). As previously noted, the fungal burden in the brain and lung was significantly higher on days 7 and 14 (P<0.05) but not at day 28 (P=0.08) compared with fungal burden in the spleen. The fungal burden in brain and lung was not significantly different (P=0.65) between days 7 and 14 but was significantly lower on day 28 (P<0.05) relative to day 7. No time-dependent change in spleen fungal burden was identified. This natural decline in organ fungal burden over time matched original published data from a rat model of CNME (Goldman et al, 1996).
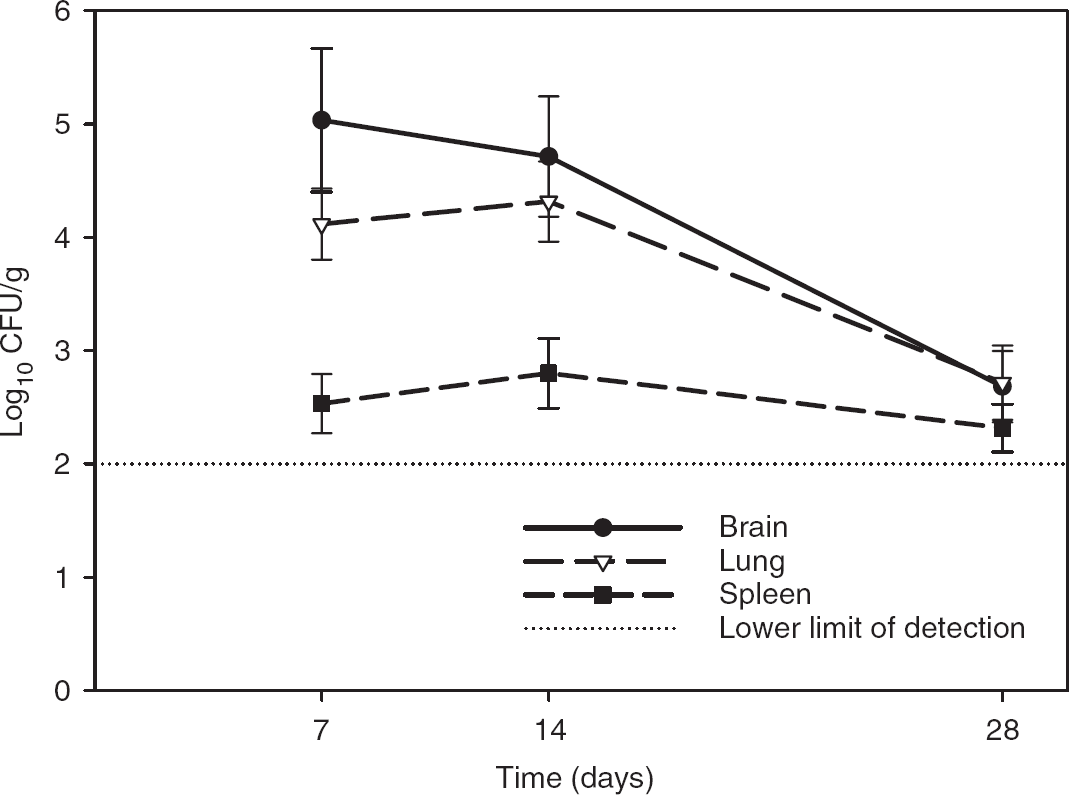
Change in mean±s.e.m. organ fungal burden in colony-forming units (CFUs) per gram over time in rats infected with 107 CFU by tail vein injection of Cryptococcus neoformans var grubii (H99).
MRI Study (Experiment 5)
The cryptococcal lesions appeared as regions with high ADC values on the ADC map with the average ADC values in the lesions being approximately 39% higher than the healthy brain tissue. The average ADC values estimated in the intracerebral lesions in all 5 rats was 1.14±0.03 × 10−3 mm2 per second (mean±s.d.). There was a significant difference (P<0.05) in the ADC values between the intracerebral lesions and healthy brain tissue. T2-weighted image and color-coded permeability coefficient maps for one representative rat model of cryptococcal meningitis are illustrated in Figure 4. The average permeability coefficient estimate in the intracerebral lesions and normal brain tissue in all 5 rats was 2.06±0.42 × 10−3 and 0.34±0.1 × 10−3 mL/g-mins, respectively. For reference, the permeability coefficient values for healthy brain tissue typically are in the range 0 to 1 × 10−3 mL/g-mins. The lesions appeared as regions with high permeability on the color-coded permeability maps. A significant difference (P<0.05) in permeability coefficient values was obtained between the lesions and healthy brain tissue. The interquartile range for permeability values in the intracerebral lesions and healthy brain tissue was in the range 0.15 to 0.3 mL/g-mins and 0.01 to 0.07 mL/g-mins, respectively (Figure 5). The upper (75th percentile) and lower (25th percentile) hinges of the box in control group have the limits between 0.1 and 0.98 mL/g-mins whereas in the lesions it is 1.0 and 4.3 mL/g-mins, suggesting that the majority of the cryptococcal lesions had some amount of increase in permeability coefficient, that is, the BBB was open and they were ‘leaky’.
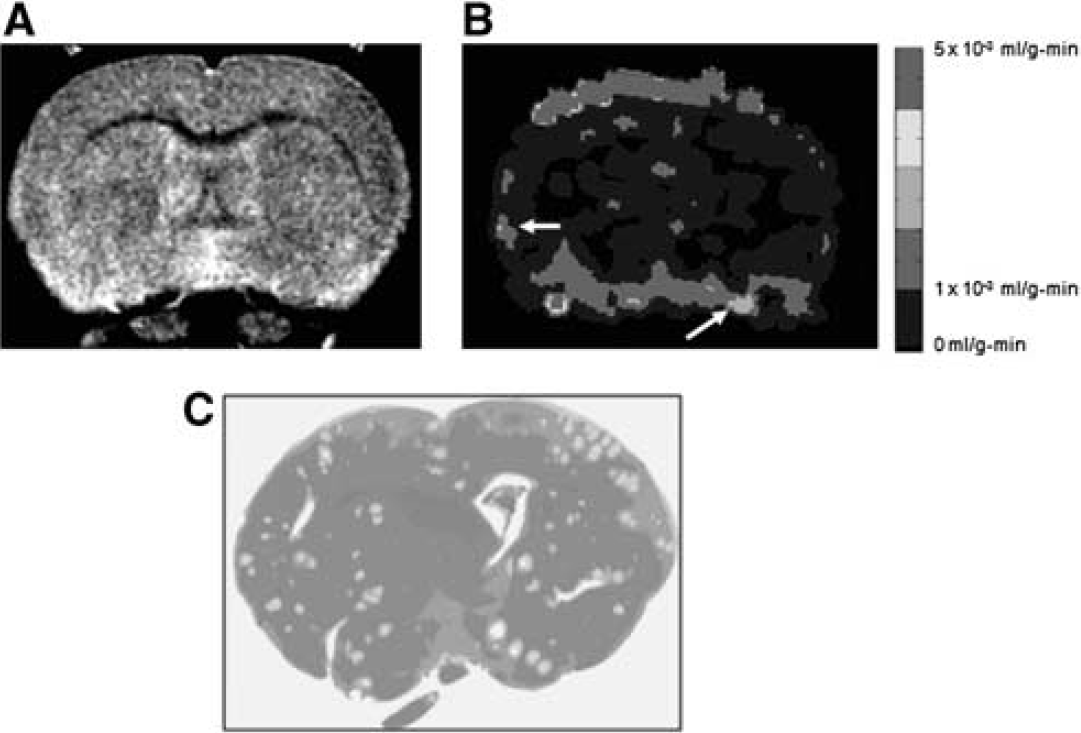
T2 (
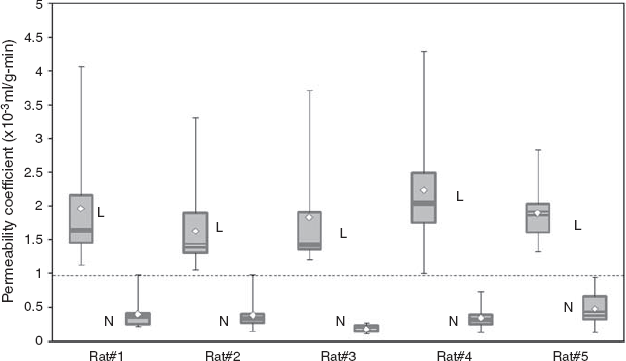
Box and whiskers plot for permeability coefficient estimates in five rats. L, lesions; N, normal healthy tissue. The plot shows that the CNME lesions clearly show high BBB permeability compared with the normal healthy tissue. The permeability estimates were obtained by drawing an ROI on the lesions and healthy tissue. Please see text for details.
Discussion
Cryptococcosis is a major cause of morbidity and mortality in patients with AIDS (Mitchell and Perfect, 1995; Stevens et al, 1999). The incidence of cryptococcosis has declined in the United States but remains high in sub-Saharan Africa and Southeast Asia (Iyer and Banker, 2002; McCarthy et al, 2006). In Atlanta, Georgia for example the incidence of cryptococcosis declined from 66 to 7 per 1,000 patients with AIDS between 1992 and 2000 (Friedman et al, 2005). In contrast, population-based surveillance from Gauteng province in South Africa revealed the incidence of cryptococcosis to be 14 per 1,000 AIDS patients (McCarthy et al, 2006). The median age was 34 years (1 month to 74 years) and a mortality rate of 27% was documented. Although, population-based surveillance data suggest incidence rates of this disease to be twofold higher in South Africa compared with the United States, the true rates may be higher. Wong et al (2007) reviewed 8,421 autopsy records of deceased South African miners between the years 1996 and 2000. The median age of the deceased miners was 39 years (range, 23 to 88 years). A total of 589 cases (7%) of cryptococcal pneumonia were identified, which translated to 31 per 1,000 in 1996 and 91 per 1,000 in the year 2000. CNME was present in 51.9% of cases of cryptococcal pneumonia, and 53.1% of patients were not diagnosed antemortem with cryptococcosis despite having had the disease. As a result, the prevalence of cryptococcosis may be 10-fold higher than that predicted by population surveillance (Wong et al, 2007).
The limited access to antiretroviral in developing countries implies that CNME will continue to be major cause of mortality among AIDS patients (Rosen et al, 2005). As an example, a 100% rate of mortality was documented among 230 Zambian patients with AIDS-related CNME because of limited access to antifungal and antiretroviral therapy (Mwaba et al, 2001). Despite increased access to current antifungal therapy, mortality secondary to CNME has not been substantially altered (Collett and Parrish, 2007). Additional variables such as male sex and abnormal neurology or brain imaging have been associated with increased mortality (Dromer et al, 2007). Dromer et al showed that patients with abnormal brain imaging results at baseline have a survival probability (95% CI) of 0.74 (0.53 to 0.83) relative to patients with normal results. The high morbidity and mortality associated with CNME demand exploration of novel methods to improve current outcomes.
The pharmacological management of CNME has not changed in over a decade and was based on a pivotal trial among AIDS patients with CNME in the United States (van der Horst et al, 1997). This management approach has not been feasible in developing countries facing the true burden of this illness (Lortholary, 2007). Investigations of novel and existing antifungal agents against CNME in animal models of this disease can lead to practical treatment regimens to remedy this problem. Multiple animal models of CNME have been developed but require intracisternal administration of CN (Carroll et al, 2007). Most investigators have used animal models to establish pathogenesis of CNME by using the less virulent serotype D strain of CN and not the more clinically relevant serotype A strain (Carroll et al, 2007). The goals of the this study were to begin to address these deficiencies.
We established the inoculum size necessary to induce CNME in rats to be 107 CFU by tail vein injection. Magnetic resonance imaging was an integral component during the exploratory analysis that provided a noninvasive approach to identify infected animals and allowed longitudinal follow-up of these animals. Limited data evaluating the role of MRI as a tool to develop animal models of CNME currently exist in the literature (Fries et al, 2005). Therefore, one of the goals of this study, among others, was to use a powerful and robust noninvasive neuroimaging modality such as MRI, to establish an animal model of cryptococcus CNS infection that would be available to the research community for testing newer treatment strategies. Moreover, the wide use of MRI as a diagnostic imaging modality in a clinical setup allows translation of research methods and techniques from basic science to the clinics.
In this study, the brain lesions in cryptococcus-infected animals were visible as well-circumscribed, discrete hyperintense regions with a global distribution on T2-w MR images, an observation in that is in good agreement with the previously published data in humans. In addition, a high ADC finding in these regions confirms that the intracerebral lesions are fluid-filled, that is, they are cystic lesions. On the structural T2w images, a qualitative analysis of the images showed that the intracerebral lesions were of variable size, varying from subpixel to approximately 10 pixels (pixel size 0.5 × 0.25 mm). On further visual examination of T2 and DW images, the intracerebral lesion distribution was global with a relatively higher distribution in the region of the caudate and hippocampus. However, more rigorous analysis will be required to confirm these findings. The correlation between the intracerebral lesions visible on MRI and histology findings, yet again confirms that the intracerebral lesions are cystic and widely disseminated in the brain giving a miliary appearance. The average permeability coefficient estimates were sixfold higher in intracerebral lesions compared with normal brain tissue. This finding suggests that the intravascular CN pathogen crosses into the tissue compartment by breaching the BBB and thereby confirms the previous findings based on histopathologic studies (Charlier et al, 2005). It has been postulated that the CN pathogen crosses the BBB through weaknesses in the Virchow—Robin spaces. However, our finding suggests that there may be a physical damage to the BBB induced by some unknown mechanism by the CN pathogen. The increase in BBB permeability observed in this study also suggests that large molecular weight (1 to 2 kDa) antifungal agents may be able to penetrate into these lesions. In addition, ADC and permeability coefficient can be used as potential neuroimaging biomarkers to investigate the efficacy of novel pharmaceutical molecules for treatment of CNME infection.
The inocula strength of 107 CFU was identified as ideal for the development of an animal model of cryptococcus meningitis infection. The demonstration of the intracerebral lesions on T2w and DW images and quantification of ADC changes in the lesions suggest that both T2 (qualitative), ADC (quantitative) and BBB permeability coefficient will be useful markers of cryptococcus CNS infection. However, it is important to note the lesions on the histology slides do not perfectly match with the permeability maps. This can be explained by the fact that there was a 24 h delay between the histology study (done on day 5 after inoculation) and the BBB permeability studies (done on day 4 after inoculation). Another reason could be because of the difference in the slice thickness between the histology slides and permeability maps. The thicker slices and low spatial resolution images used to generate permeability maps result in partial volume effects that could be potentially an additive factor.
The excellent correlation of histopathology between our rat model compared with data from AIDS patients with CNME increases the probability for clinical translation of future pharmacological investigations. The present animal model also showed high fungal brain burden (105 CFU/g) as reported in AIDS patients (Brouwer et al, 2005). The rat provides an optimal model to investigate antifungal agents. The current rabbit model of CNME is expensive to maintain and require immunosuppression to develop disease (Perfect et al, 1980). The mouse model is limited in that animals do not survive for greater than 7 days after inoculation (Charlier et al, 2005). Renal clearance of antifungal agents (fluconazole, flucytosine) is also very rapid in mice compared with rats, which limits simulation of human concentration—time profiles in mice. Further work with this model could use noninvasive techniques such as MRI to quantify lesion load and assess development and longitudinal change in lesions with pharmacological interventions. Refining the present animal model using MRI has the potential of reducing the number of animals used to identify improved pharmacological interventions against CNME.
Conclusion
Magnetic resonance imaging was applied as a useful tool to replicate a rat model of CNME using a novel but simple route of intravenous inoculation. This model of CNME results in the formation of brain lesions that mimic the pathologic assessment of severe infection in AIDS patients with this disease. CNME also disrupts rat BBB and is detectable by Gd-DTPA-enhanced MRI. This suggests that novel antifungals agents with high molecular weights should not be discounted as possible treatments of CNME. Further evaluation of MRI to track changes in brain lesions over time could also lead to a useful noninvasive tool to quantify drug effects. Exploration of single and combination antifungal agents in this model may improve treatment options for patients with this high mortality disease.
Contributions
MPP, RS, and CRL conceived the study and study design. US performed ROI drawings and data analysis. RS performed all MRI-related experimentation and interpretation. MPP and SLP performed fungal burden and histology-related work. MPP and RS wrote the paper. All authors read and approved the final paper.
