Abstract
Vascular growth and redistribution of flow can compensate for arterial occlusion and possibly reduce the effects of hypoperfusion. As yet there is limited information on the age-dependent nature of vasculature remodelling. In this study, we have monitored the vascular and morphologic changes using magnetic resonance imaging and histology in a chronic bilateral common carotid artery occlusion (BCCAO) model in both newborn and adult rats. Acutely, cerebral blood flow (CBF) decreased immediately after BCCAO, producing a state of oligaemic hypoperfusion. At 6 months after BCCAO in both adult and neonatal rats, the CBF had normalised at control values. To investigate the underlying mechanism for the return of CBF to control values, intra- and extracerebral magnetic resonance angiograms (MRAs) were acquired. As expected, signal from the common carotid arteries was present in the sham-operated rats, but was absent in the BCCAO animals. India ink angiograms demonstrated more tortuous basilar arteries in the adult rats post-BCCAO and MRAs demonstrated more extracerebral midline collaterals in the neonatal rats post-BCCAO, indicating different modes of vascular adaptation dependent on the age at onset of the insult. Both groups had collateral vessels arising from the vertebral arteries, and BCCAO was also associated with increased diameter of basilar, posterior cerebral, posterior communicating, internal carotid, middle cerebral and anterior cerebral arteries. Our study suggests that the developing and mature animals exhibit different patterns of vascular remodelling and that the BCCAO hypoperfusion model will be useful for investigating age-dependent vascular events in response to vasoocclusive disease.
Introduction
Occlusive cerebral arterial disease and cerebral hypoperfusion are seen most commonly in adults, but may also be found in children, for example in moyamoya disease (Calamante et al, 2001). As yet, however, few experimental studies have addressed the vascular and haemodynamic responses to arterial occlusion in the developing brain, either in humans or in animals models of brain disease. Arterial occlusion causes haemodynamic perturbations and a redistribution of blood flow, which the circulatory system can compensate for by various processes, for example, utilising collateral circulation and/or autoregulatory mechanisms (Liebeskind, 2003). However, the reserve capacity of the circulatory system is limited, and when these processes are insufficient, then inevitably tissue will enter a state of hypoperfusion. Complete occlusion of a large artery causes redistribution of blood flow due to the change in pressure gradients across the collateral circulation. This redistribution increases flow and intravascular shear forces through intact vessels, ultimately leading to adaptive growth in vessels. This vascular adaptation is known as arteriogenesis (Busch et al, 2003; Buschmann et al, 2003a). If the adaptive mechanisms are insufficient, then the tissue may, depending on the severity of hypoperfusion, become hypoxic, which is the main driving force for another form of vessel growth, namely angiogenesis. Angiogenesis is a complex phenomenon resulting in sprouting of capillaries and is regulated by hypoxia-inducible factor (HIF). Upregulation of HIF increases development of new vessels via angiogenic factors such as vascular endothelial growth factor (VEGF) (Buschmann et al, 2003b). Previous studies have shown that arterial occlusion in adult rats, for instance in the permanent bilateral common carotid artery occlusion (BCCAO) model, is associated with an initial decrease in blood flow, which subsequently returns towards normal over a period of months (Tsuchiya et al, 1992; Ohta et al, 1997). It has been suggested that the mechanisms underlying normalisation of blood flow involve intracerebral vascular adaptation, that is, angiogenesis and arteriogenesis (Coyle and Panzenbeck, 1990; Busch et al, 2003). However, the extracerebral contribution to this flow normalisation is not known.
Existing data suggest that the sequelae of arterial occlusion are age-dependent. For example, De La Torre et al (1992) found that 9 weeks after permanent arterial occlusion, cerebral perfusion returned to normal in adolescent rats but remained low in aged rats. Also, He et al(1997) reported an age-related CBF redistribution after arterial occlusion when comparing suckling, adult and aged rats. Furthermore VEGF, which is a proangiogenic factor, is far more potent in the neonate than in the adult (Robertson et al, 1985). Age-dependent acute responses to occlusive pathology have been demonstrated by Canese et al (2004) in a rat model of transient global ischaemia using MRI. Measuring apparent diffusion coefficient of water (ADC) and spin-spin relaxation time (T2), they reported a higher incidence of MR-detected lesions in aged rather than in young rats (Canese et al, 2004). Also, T2-weighted changes in the hippocampus of aged but not young rats have been seen in a model of cerebrovascular insufficiency 9 weeks after surgery (De La Torre et al, 1992).
The multiparametric capabilities of MRI can provide a number of indices of pathology routinely used in clinical diagnosis; although the precise pathologic substrates of these measures are not completely understood, their variation commonly indicates physiologically relevant changes in the tissue (Thomas et al, 2000). The relaxation times T1 and T2, and diffusion MRI each reflect aspects of the microscopic tissue environment and are established indicators of both acute and long-term consequences of brain injury (Van Bruggen et al, 1994; Hoehn et al, 2001; Lythgoe et al, 2003; Dijkhuizen and Nicolay, 2003). Susceptibility (T*2)-weighted imaging during transient hypercapnia can provide an indication of the cerebrovascular reactivity of the blood vessels (Harris et al, 2001). Cerebral haemodynamics and angioarchitecture may be assessed by perfusion imaging and MR angiography, respectively (Calamante et al, 1999b; Besselmann et al, 2001). Against this background, the aim of this study was to use MR angiography, perfusion, diffusion, T1, T2 and T*2 imaging to characterise the age-dependent vascular, haemodynamic and tissue response to bilateral common carotid occlusion in the rat.
Materials and methods
Experimental Design
All animal care and procedures were carried out in accordance with the UK Animals (Scientific Procedures) 1986 Act. Two experimental protocols were used in this study: (1) to investigate the immediate effects of BCCAO in adult rats; and (2) to investigate long-term age-dependent vascular and haemodynamic changes after permanent BCCAO in neonatal (p3) and adult rats.
Protocol 1 involved 2-month-old animals (n = 8). Each animal was anaesthetised, positioned inside the bore of the magnet and imaged in the pre-occluded state. Next, both common carotid arteries were occluded from outside the MRI scanner (i.e. remotely) using snares (see surgical methods) and the animal was imaged again. The remote occlusion method ensured good registration of pre- and post-occlusion images (Allen et al, 1993).
Protocol 2 involved four groups of animals. Neonatal and adult rats underwent either BCCAO or sham operation and were subsequently imaged 6 months after the surgery before being killed for histopathologic examination. The groups were: (A) neonatal sham operated 3-day-old (6 to 8g) rats (n = 8); (B) neonatal BCCAO 3-day-old (6 to 8g) rats (n = 8); (C) adult sham operated 3-month-old (320 to 370g) rats (n = 6) and (D) adult BCCAO 3-month-old (320 to 370 g) rats (n = 7).
Surgical Methods
Protocol 1: Eight male Sprague-Dawley rats (∼200g; Harlan, UK) were anaesthetised with 2.5% halothane in 1 L/min O2 and maintained on a 2% halothane and 40/60 O2/N2O mixture that was supplied via a nose cone. The animals, which were allowed to breathe spontaneously throughout the experiment, were prepared for remote controlled BCCAO in the manner described by Allen et al (1993). Briefly, using an operating microscope, the carotid arteries were isolated and dissected from the vagus nerves. A 12 cm length of nylon (Ethilon 2/0, Ethicon), bent by heat into a ‘V’ shape to act as a snare, was passed under the carotid artery. The free ends of the snare were passed through two small holes in the sealed end of a curved plastic tube (snare holder), which was rigidly fixed to the animal holder. The ends of the snare were attached to a perspex rod, which could be manipulated remotely (from outside the magnet) by means of a drive screw. Operation of the drive screw resulted in the nylon snare being drawn against the snare holder, thus occluding the artery. This arrangement was replicated for the other carotid artery.
Protocol 2: Male Sprague–Dawley rats (Harlan, UK) (neonatal and adult) were anaesthetised with 2% halothane in 1 L/min oxygen. Animals were allowed to breathe spontaneously throughout the surgery. Body temperature was monitored using a rectal probe and maintained between 36.5°C and 37.5°C. Animals were randomly selected for BCCAO or sham surgery. A ventral midline incision was made to expose the common carotid arteries, which were carefully separated from the vagus nerves. Both common carotid arteries were tied off with a single 5 to 0 suture (Mersilk, Ethicon) (adult group) or 8 to 0 suture (Mersilk, Ethicon) (neonatal group) under a surgical microscope. The wound was closed with absorbable suture (Vicryl rapide, Ethicon) and the rats were allowed to recover from anaesthesia before returning back to their cage (adult) or dam (neonatal). Sham surgery was conducted in control animals, which is the common carotid arteries were exposed under a surgical microscope but not occluded and the wound was sutured.
Magnetic Resonance Imaging Methods
Magnetic resonance imaging data were acquired before and after remote occlusion (protocol 1) or 6 months after surgical preparation (protocol 2). The combination of anaesthesia, surgery and MRI scanning in the neonates is likely to have a detrimental impact on the neonatal animals, thus precluding an acute imaging time point. The animals were anaesthetised with 2.5% halothane in 1 L/min oxygen and maintained on 2% halothane in a 40/ 60 O2/N2O mixture supplied via a nose cone. The rats were allowed to breathe spontaneously throughout the study and body temperature was recorded with a rectal thermometer and maintained between 36.5°C and 37.5°C by blowing warm air into the magnet bore. The respiratory rate and ECG were also monitored throughout the experiment.
Magnetic resonance imaging was performed in a 2.35T horizontal bore magnet (Oxford Instruments, Oxford, UK) interfaced to an SMIS console (Farnham, UK). Coronal images were acquired approximately 3.3 mm from bregma using a volume transmitter radiofrequency coil with a length of 6 cm, and a separate 3-cm-diameter surface coil for signal reception. Unless otherwise stated, the images were acquired with: field of view (FOV) = 40 × 20 mm, slice thickness = 2 mm, image matrix = 128 × 64 pixels. Total imaging time = 78 mins.
Cerebral blood flow: For noninvasive CBF measurement, we used the continuous arterial spin labelling (CASL) method (Alsop and Detre, 1996), based on spin-echo echo-planar imaging (EPI) with interleaved adiabatic fastpassage inversion and control measurements. The centre of the inversion plane was situated 2 mm behind the back of the cerebellum in all animals (approximately 10kHz frequency offset); for the control image the same offset value was used, but with the sign reversed. A postlabelling delay time of 500 ms was used to minimise transit time effects and intravascular artefacts. The echo time (TE) was 36ms, the repetition time (TR) 1000secs, and 44 averages were acquired. A nonslice selective inversion recovery (IR) EPI sequence was used to obtain the tissue T1 and the spin density (M0) parameters necessary for subsequent CBF quantification. Images were acquired at eight inversion times ranging between 200 and 4000 ms, TE was 36 ms, TR 2,000ms, and 22 averages were acquired. The IR data were fitted to obtain T1, α0 and M0 assuming a mono-exponential recovery function. Subsequently, these values were used to calculate CBF from the difference between the labelled and control image (Alsop and Detre, 1996). Assumptions were T1 of arterial blood = 1.5 secs, blood:brain partition coefficient for water (λ) = 0.9, efficiency of the spin labelling pulse = 0.71 (previous results; data not shown), tissue transit time = 250ms, arterial transit time=200ms. Duration of acquisitions: T1 = 11 mins and CASL = 8 mins.
Cerebrovascular reactivity: The sensitivity of T*2-weighted imaging to the paramagnetic properties of deoxyhaemoglobin is well-documented (Ogawa et al, 1993) and has been exploited extensively in functional imaging studies (Howseman and Bowtell, 1999). As CO2 is a potent vasodilator in mammals, increasing levels in the inspired gas leads to changes in blood flow and volume, and a decrease in deoxyhaemoglobin. The consequent modulation in T*2-weighted image intensity can provide an indication of the cerebrovascular reactivity of the blood vessels (Harris et al, 2001). Standard gradient-echo EPI (TE = 45ms, TR = 1500 ms, 40 averages) images were acquired before and after administration of 15% CO2. In total, 21 images were acquired sequentially for a total of 21 mins, the initial six images being used to determine the baseline after which 15% CO2 was administered via the nose cone for 5 mins. The percentage signal change from baseline after administration of CO2 provided an indication of the cerebrovascular reserve. Lack of change would imply maximal cerebral vasodilatation at rest. Duration of acquisition = 21 mins.
Magnetic resonance angiography: 3D TOF-MRA (time-of-flight magnetic resonance angiography (MRA)) was performed using a three-dimensional flow-compensated gradient echo sequence (matrix size = 128 × 128 × 128, FOV = 25 × 25 × 30 mm, TE = 12 ms) (Besselmann et al, 2001). Angiograms were acquired centred on the head, to detect cerebrovascular changes, and on the neck, to investigate local vascular changes at the site of BCCAO. The following empirically optimised values for flip angle and TR were used: for cerebrovascular MRA, TR = 100 ms and pulse angle α = 55°; and for the neck, TR = 100 ms and pulse angle α = 70°. Duration of acquisition = 25 mins.
T2 and ADC maps: T2 maps were obtained using the multiple acquisition of spin and gradient echoes with an interleaved echo planar imaging (MASAGE-IEPI) sequence (Thomas et al, 2002). The parameters used were: TR = 1,500 ms, 16 averages, with TE1 = 24 ms, TE2 = 65 ms, TE3 = 106 ms (for definitions of TE1/2/3, see Thomas et al, 2002). Quantitative ADC maps were calculated from traceweighted single shot spin-echo EPI images. The parameters were as follows: TR = 1500 ms, TE = 56 ms, 48 averages, b = 38 and 872 s/mm2. Duration of acquisitions: T2 = 4 mins and ADC = 9 mins.
Image Processing and Analysis
T1, T2 and ADC maps were calculated using in-house software written in IDL (Research System Inc., Boulder, CO, USA). Three regions of interest (ROI) were selected for image analysis corresponding to the cortex, hippocampus and the thalamus. A whole-brain ROI was drawn for the gradient-echo assessment of cerebrovascular activity. The raw data from the MRA acquisitions were zero-filled to a 256 × 256 × 256 pixel matrix size before Fourier-transformation. Angiograms were created using the maximum intensity projection function in ImageJ (open source software from NIH). The MRAs were assessed by one observer who was unaware of the status of the animals. The observer was required to assess (i) whether or not the common carotid arteries were present or absent; (ii) changes in individual artery signal intensity when compared with other surrounding vessels; (iii) changes in morphology in the intracranial or cervical arteries; and (iv) the presence of any intracranial or cervical collateral vessels.
Post-mortem Cerebral Angiograms
Four animals from each age-group (neonatal and adult) were anaesthetised with 2% halothane in 1 L/min oxygen. A lethal dose of papaverine hydrochloride (40 to 50 mg/kg, Sigma) was intravenously injected to induce maximal dilatation and minimise cerebrovascular resistance (Coyle and Jokelainen, 1982). India ink was injected in the ascending aorta, and the external jugular vein was cut to facilitate venous outflow. The brains were carefully removed and suspended in 4% paraformaldehyde. An observer who was unaware of the status of the animals measured the diameters of the basilar, superior cerebellar, posterior communicating, posterior cerebral, internal carotid, middle cerebral and anterior cerebral arteries under a microscope with a calibrated eye piece micrometer to assess gross morphologic vascular changes and measure the vessel diameters.
Data Analysis
All data are given as sample means ± s.e.m. A paired t-test was used to analyse the CBF data from Protocol 1. For Protocol 2, exploratory analyses using a repeated measures (degrees of freedom-adjusted mixed-model) three-way ANOVA was conducted on the CBF, T1, T2 and ADC data. The main effects for these analyses were region (cortex, thalamus, hippocampus), age (neonatal, adult) and surgery (sham, BCCAO). For the vessel diameter data (India ink angiograms), the main effects were the vessel (basilar, superior cerebellar, posterior cerebral, posterior communicating, internal carotid, middle cerebral and anterior cerebral arteries), age and surgery. A two-way ANOVA was used to investigate the cerebrovascular reactivity data; the two factors were age and surgery. The Fisher exact test was used as a test of association in relation to the observed changes in MRA data. All analyses were performed in SPSS for Windows.
Results
We performed MRI measurements of cerebral haemodynamics, intra- and extracranial vasculature, tissue relaxation times and diffusion, together with post mortem measures of vessel diameter, to assess the effects of BCCAO in adult and neonatal rats. Cerebral blood flow (mL/min/100 g) was calculated using a noninvasive arterial spin labelling MRI technique. Vascular reactivity was determined via T2*-weighted MRI, using a CO2 challenge test. Vascular size and distribution were visualised using MRA and post-mortem india ink cerebral angiograms. MRI relaxation times (T1 and T2) and diffusion (ADC) measurements were used to examine tissue pathophysiology
Cerebral Blood Flow
Protocol 1: Cerebral blood flow before and immediately after bilateral common carotid artery occlusion: Cerebral blood flow was measured in the cortex, hippocampus and the thalamus before and immediately after remote BCCAO. A decrease in mean CBF was observed across all regions after remote occlusion: cortex (202 ± 11 to 68 ± 7 7mL/ 100g/min), hippocampus (156 ± 12 to 91 ± 11mL/ 100g/min) and the thalamus (213 ± 16 to 148 ± 15 mL/100g/min) (Figure 1).
Protocol 2: Cerebral blood flow at 6 months after bilateral common carotid artery occlusion: Cerebral blood flow was measured in three regions (cortex, hippocampus and the thalamus, Figure 2) and the following sample mean CBF estimates were obtained for the adult, neonatal, sham and BCCAO groups. Adult sham mean CBF was: cortex 190 ± 11; hippocampus 131 ± 12; thalamus 182 ± 16 mL/ 100 g/min and in the adult BCCAO group regional mean CBF was: cortex 176 ± 10; hippocampus 135 ± 6; thalamus 194 ± 10 mL/100 g/min. Neonatal sham regional mean CBF was: cortex 182 ± 5; hippocampus 136 ± 3; thalamus 184 ± 6 mL/100 g/min and in the neonatal BCCAO group regional mean CBF was: cortex 176 ± 12; hippocampus 121 ± 6; thalamus 179 ± 13 mL/100 g/min. No significant difference in CBF was demonstrated between the sham and the BCCAO groups (F = 0.315, P = 0.58), or between the neonatal and the adult groups (F = 0.186, P = 0.67) at 6 months after surgery (degrees of freedom-adjusted ANOVA main effects tests).
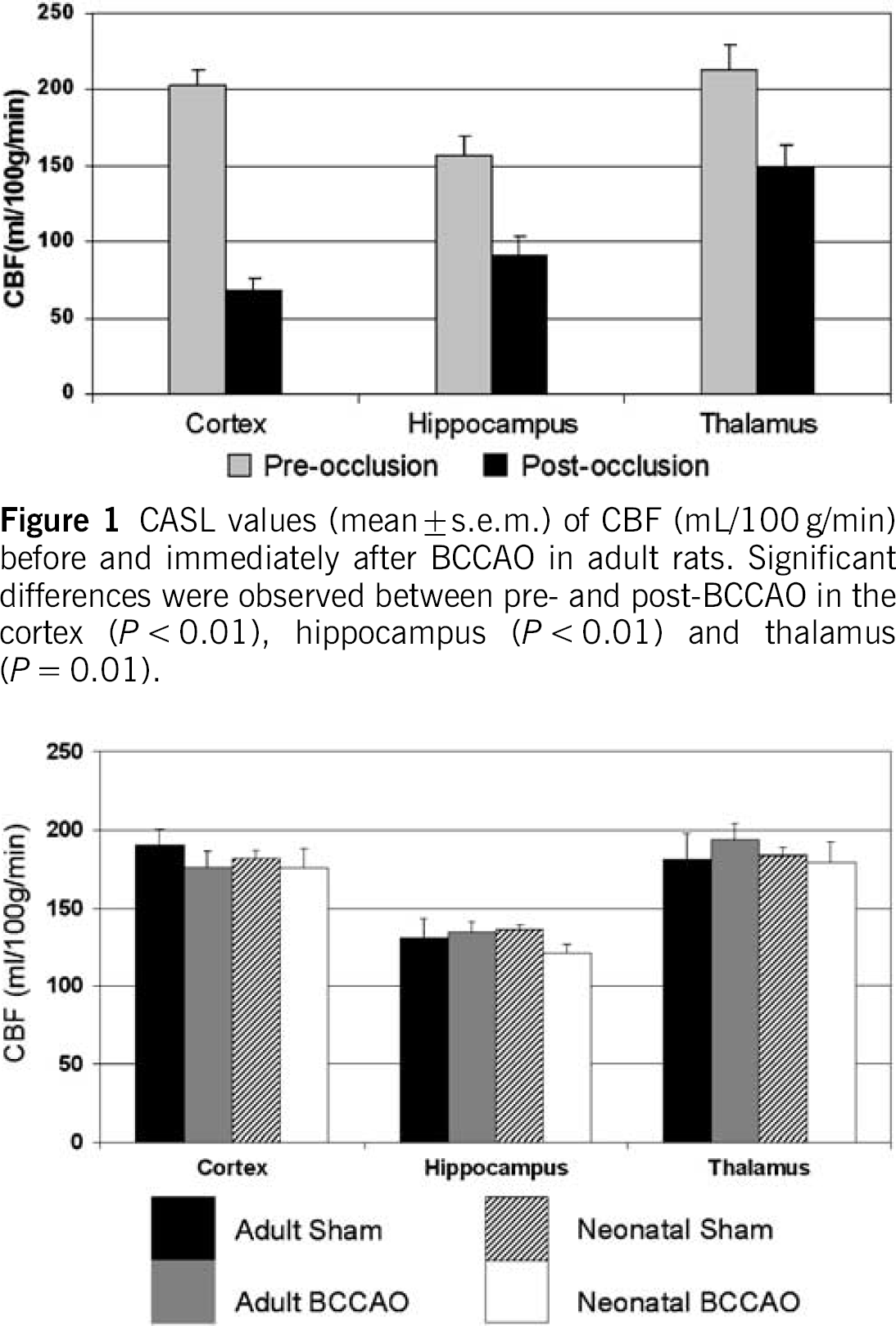
CASL values (mean ± s.e.m.) of CBF (mL/100 g/min) before and immediately after BCCAO in adult rats. Significant differences were observed between pre- and post-BCCAO in the cortex (P < 0.01), hippocampus (P < 0.01) and thalamus (P = 0.01).
CASL CBF values (mean ± s.e.m.) (mL/100 g/min) in adult and neonatal rats 6 months after BCCAO. No differences were observed between the sham and operated groups (P = 0.67).
Cerebrovascular Reactivity
Maximal vasodilatation was induced by a CO2 challenge and assessed using a gradient-echo sequence.
The percentage signal change from baseline was used to give an indication of cerebrovascular reactivity. No significant difference in the percentage signal change between sham-operated (3% ± 0.3%) and BCCAO (3% ± 0.3%) animals was demonstrated (F = 0.006, P = 0.94).
Magnetic Resonance Angiography
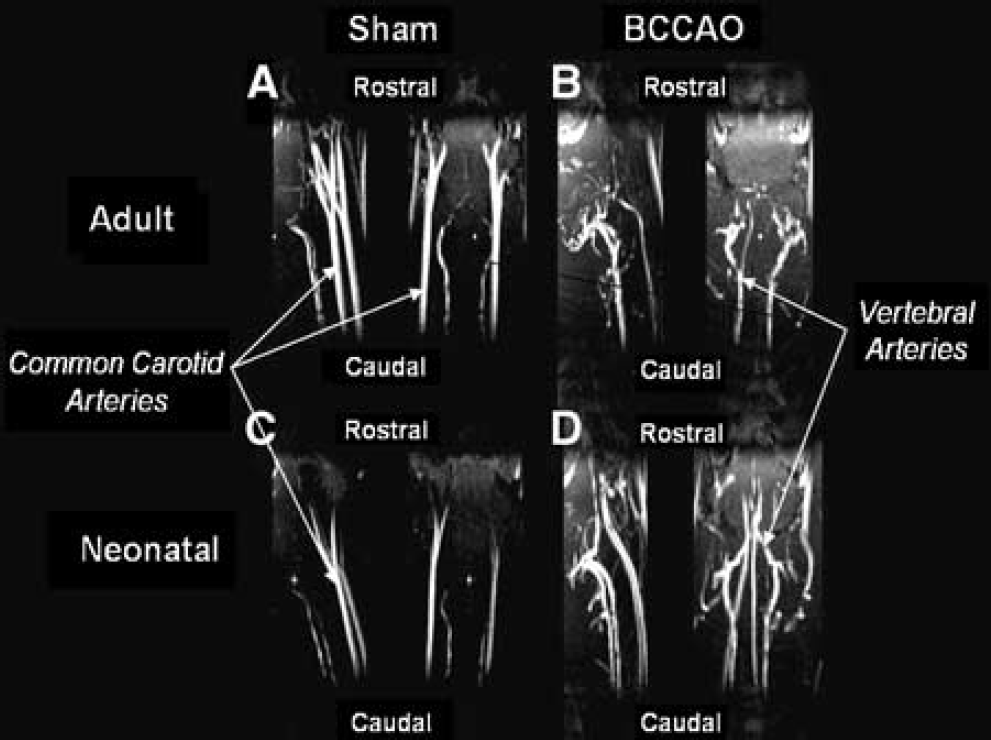
Comparison of bilateral common carotid artery occlusion and sham groups: One observer who was unaware of the status of the animals assessed the MRAs (Table 1). The absence of MR signal in the common carotid artery was used to confirm effective occlusion (Figure 3). The presence of signal from the common carotid arteries was observed in both the sham groups, adult (7/7) and neonatal (8/8), but was absent in the BCCAO animals (adult 0/6 and neonatal 0/8). Other differences between the BCCAO and sham groups were: (i) more animals in the BCCAO groups (adult and neonatal) exhibited an increase in signal intensity from the posterior circulation than sham groups (adult BCCAO 5/6, adult sham 1/7 P = 0.026; neonatal BCCAO 7/8, neonatal sham 1/8, P = 0.01) and (ii) all animals from the BCCAO groups (adult and neonatal) exhibited cervical collateral vessels, whereas they was not visible in the sham group (adult BCCAO 6/6, adult sham 0/7, P < 0.001; neonatal sham 8/8 neonatal BCCAO 0/8, P < 0.001).
Summary of reported features on MR angiography
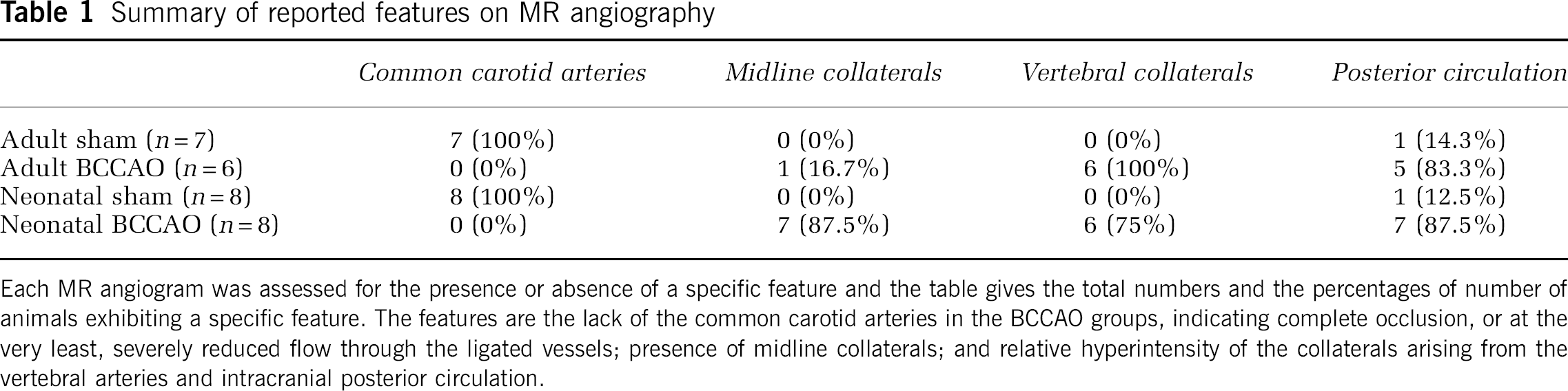
Each MR angiogram was assessed for the presence or absence of a specific feature and the table gives the total numbers and the percentages of number of animals exhibiting a specific feature. The features are the lack of the common carotid arteries in the BCCAO groups, indicating complete occlusion, or at the very least, severely reduced flow through the ligated vessels; presence of midline collaterals; and relative hyperintensity of the collaterals arising from the vertebral arteries and intracranial posterior circulation.
Typical 3D TOF-MRA centred on the neck 6 months after surgery. Note the presence of the characteristic bifurcation of the common carotid arteries in the sham-operated rats, which is absent in the BCCAO animals.
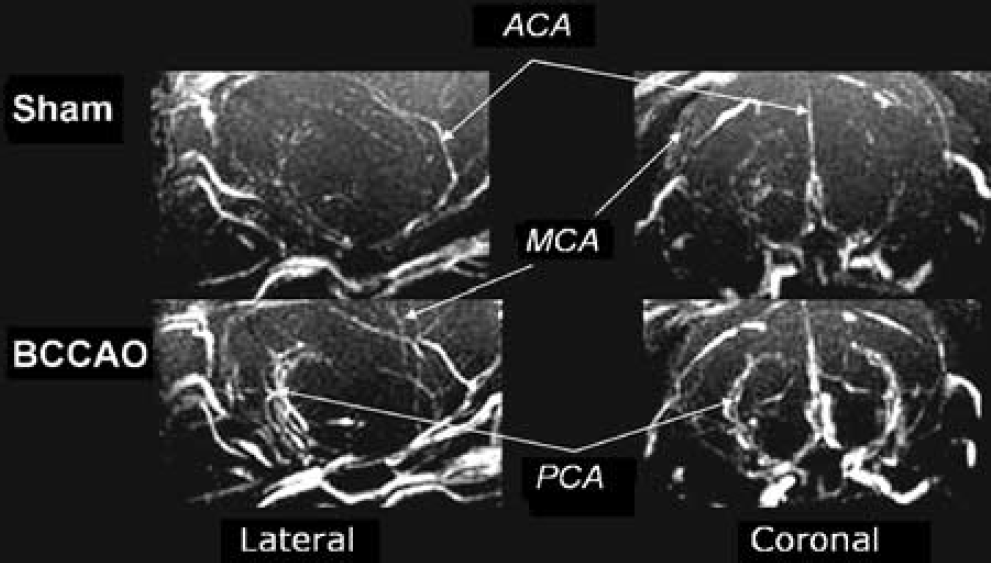
Comparison of adult and neonatal groups: Although both adult and neonatal BCCAO groups were found to have collateral vessels arising from the vertebral arteries (adult 6/6, neonatal 6/8, P = 0.5), the neonatal BCCAO group was associated with a greater number of midline collateral vessels than the adult (adult 7/8, neonatal 1/6, P = 0.03) (Figure 4 and Table 1).
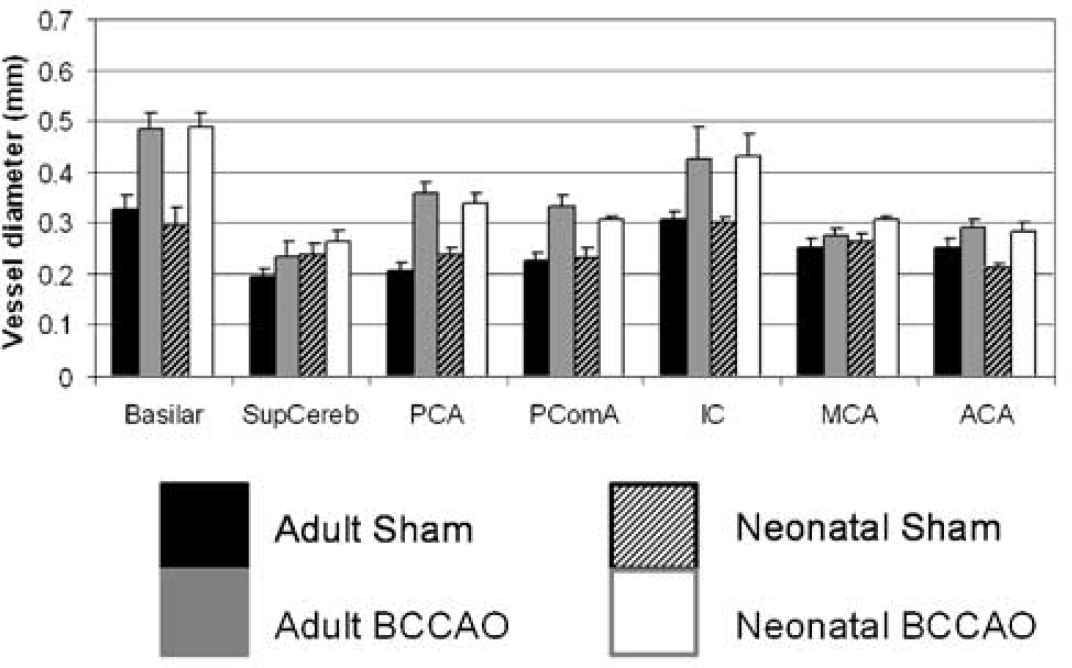
Typical 3D TOF-MRA. The cerebral vasculature of a sham and a BCCAO rat. The major cerebral vessels are indicated: ACA = anterior cerebral artery, MCA = middle cerebral artery, PCA = posterior cerebral artery.
Post-Mortem Cerebral Angiograms
Comparison of bilateral common carotid artery occlusion and sham groups: Significant differences were observed in all mean artery diameters except the superior cerebellar artery (F = 2.028, P = 0.18) after BCCAO (Figure 5).
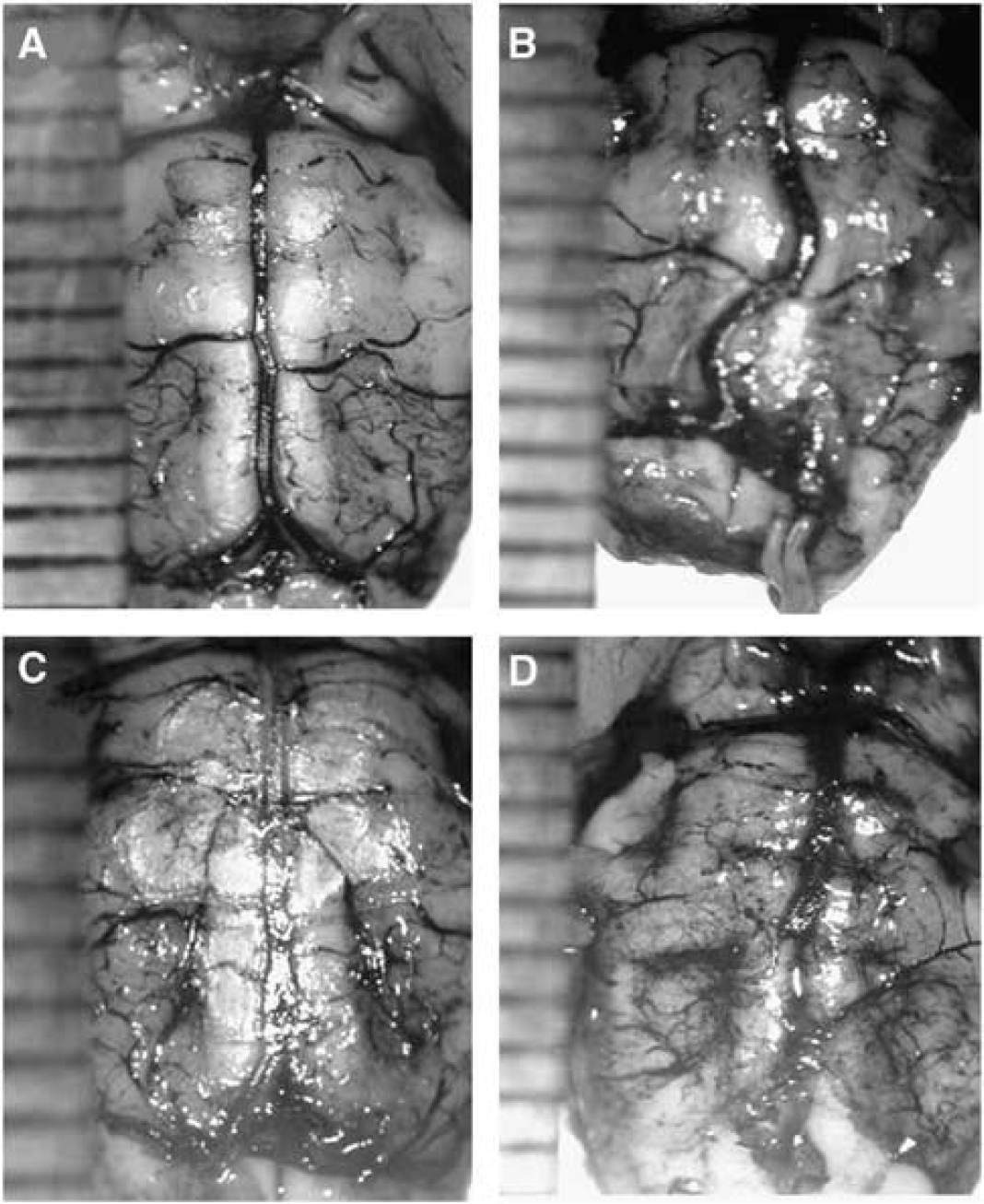
Post-mortem diameter of the vessels (mean ± s.e.m.) in the circle of Willis, visualised by infusion of India ink. SupCereb=superior cerebellar artery, PCA=posterior cerebral artery, PComA = posterior communicating artery, IC = internal carotid artery, MCA = middle cerebral artery, ACA = anterior cerebral artery.
Comparison of adult and neonatal groups: The basilar artery in the adult BCCAO group (Figure 6) appeared more tortuous than in either the adult shams or any of the neonatal animals. No significant differences in mean diameter were observed between the adult and the neonatal groups for any of the vessel diameters.
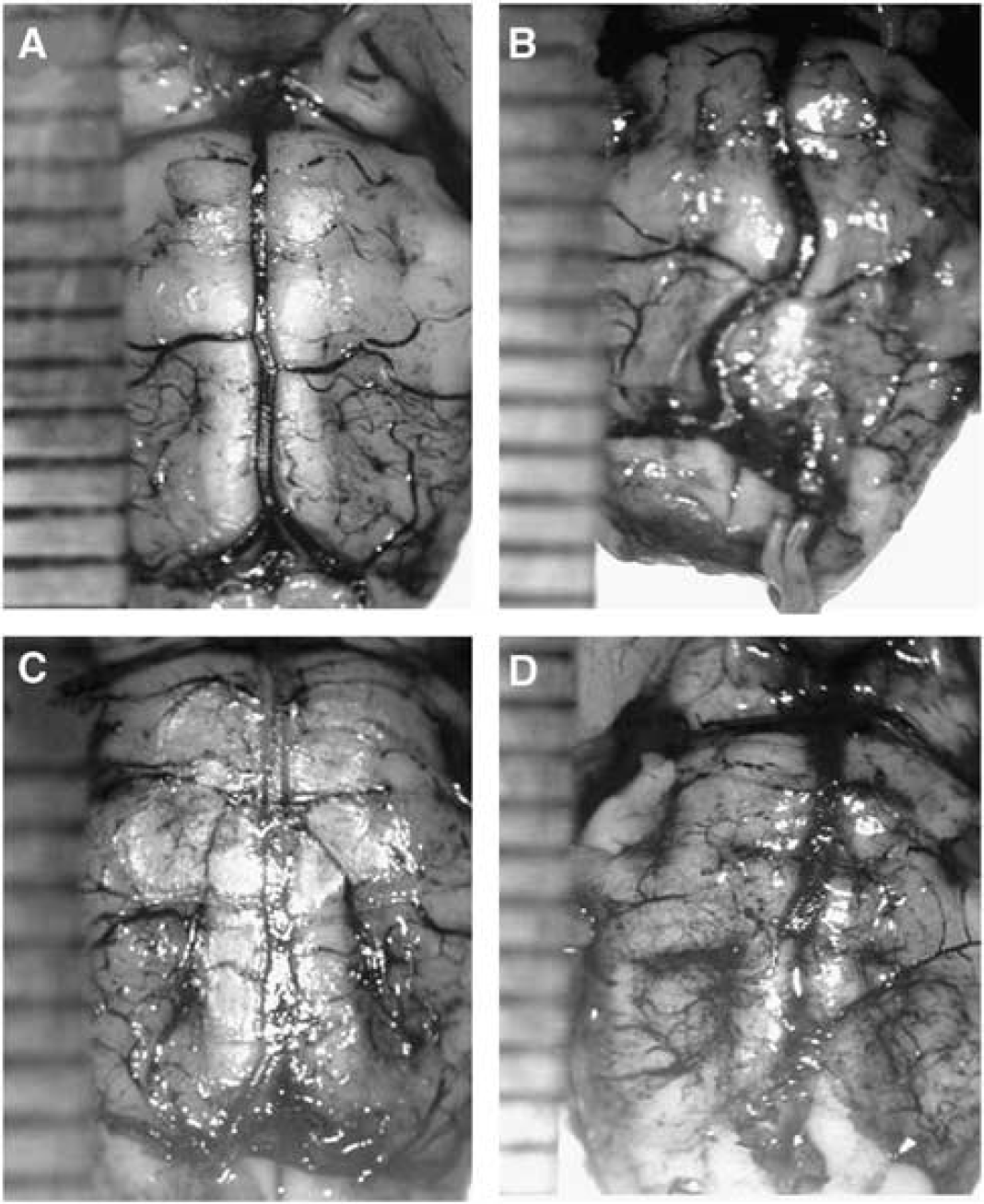
The basilar arteries (post mortem) perfused with India ink 6 months after BCCAO: (
T1, T2 and ADC 6 months after bilateral common carotid artery occlusion
A comparison of the neonatal and adult groups (main effect, test on age, F = 1.752, P = 0.2) failed to show a statistically significant difference on mean T1. However, a comparison of the BCCAO and sham groups (main effect, test on treatment) provides evidence for a statistically significant difference in mean T1 (F = 5.819, P = 0.024, Table 2). The T2 data did not demonstrate any differences between the neonatal sham and BCCAO animals, although the adult group demonstrated a small difference in the T2 of the thalamus of the BCCAO animals when compared with the sham group (difference between BCCAO and sham mean T2 values in the thalamus 1.9 ± 0.7 ms, F = 5.654, P = 0.034 Table 3). Our ADC measurements did not indicate any significant differences between the sham and BCCAO animals (F = 0.74, P = 0.40).
T1 measurements of adult and neonatal animals 6 months after sham or BCCAO surgery in the cortex, hippocampus and thalamus (mean ± s.e.m.)
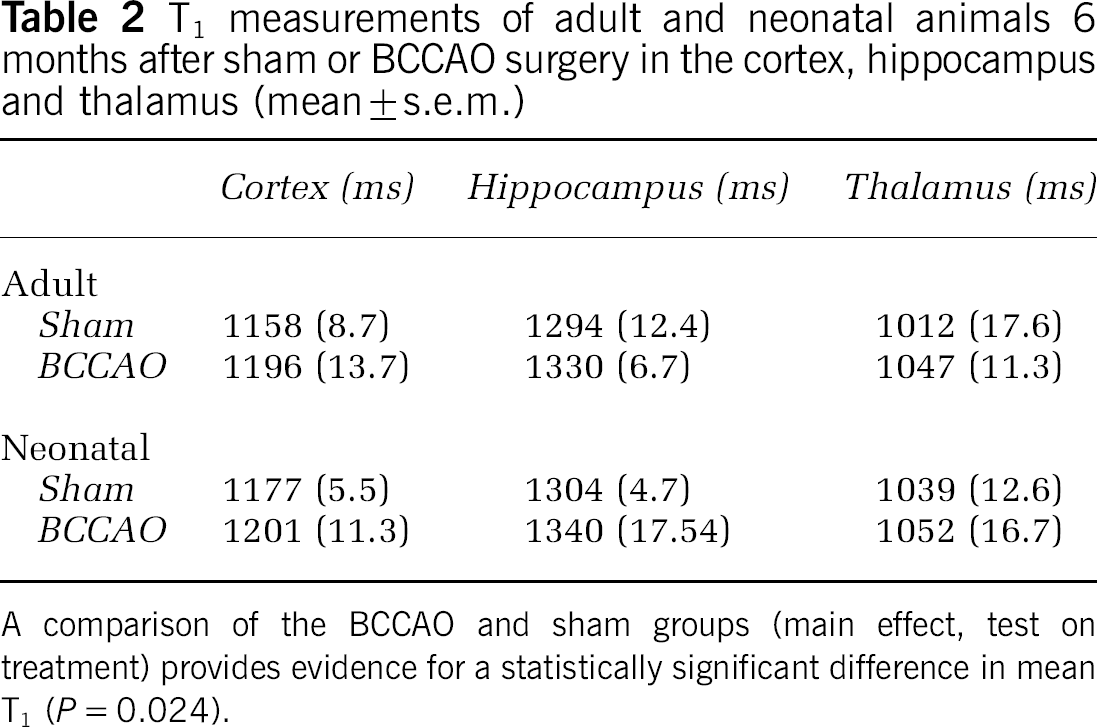
A comparison of the BCCAO and sham groups (main effect, test on treatment) provides evidence for a statistically significant difference in mean T1 (P = 0.024).
T2measurements of adult and neonatal animals 6 months after sham or BCCAO surgery in the cortex, hippocampus and thalamus (mean ± s.e.m.)
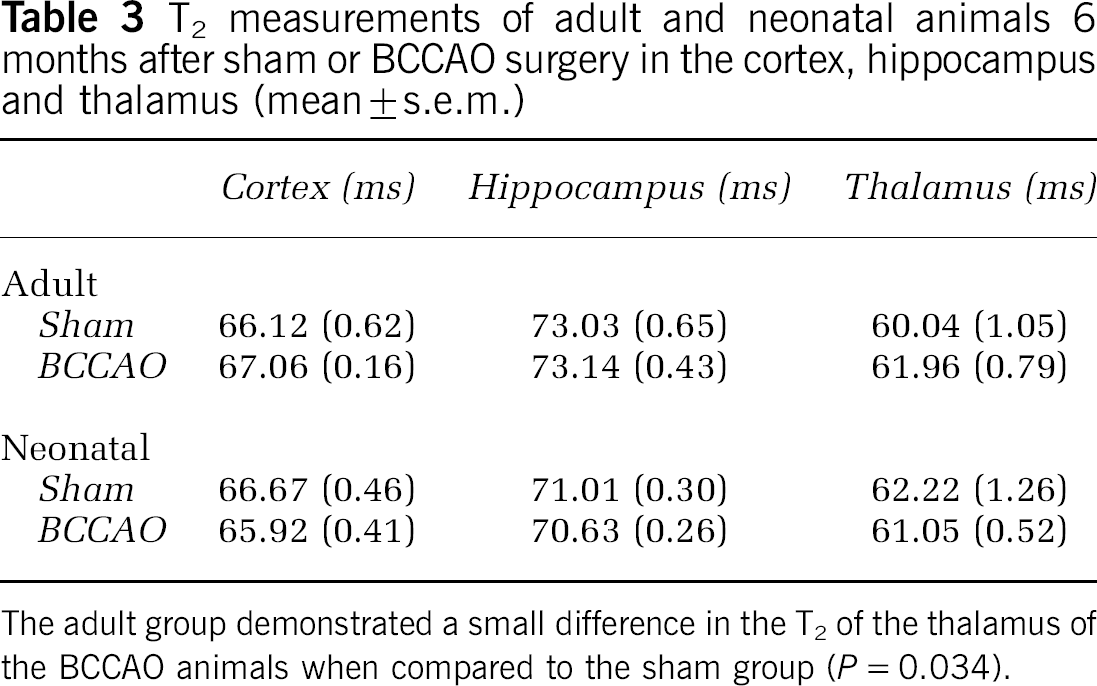
The adult group demonstrated a small difference in the T2 of the thalamus of the BCCAO animals when compared to the sham group (P = 0.034).
Discussion
The arterial spin labeling MRI technique, CASL, allows a noninvasive, quantitative and regional assessment of CBF. Using this technique we have demonstrated a decrease in CBF in the cortex, hippocampus and thalamus immediately after occlusion of both common carotid arteries in the rat. Overall, these measurements compare well with previous CBF studies, which have used alternative techniques such as hydrogen clearance (Eklof and Siesjo, 1972) and autoradiography (Tsuchiya et al, 1992) where decreases of around 25% to 50% in CBF have been reported immediately after BCCAO.
In our chronic study we investigated the effects of permanent BCCAO on the neonatal and the adult cerebral vasculature using MR and India ink angiograms. To our knowledge, there have been no other studies that investigate the haemodynamic changes 6 months after neonatal BCCAO. In this study we have demonstrated that despite ligation of both carotid arteries, CBF had returned to control values at the 6-month time-point in both neonatal and adult groups. Using the same model, Tsuchiya et al (1992) observed that CBF was returning towards control values after 1 week and Otori et al (2003) reported that CBF had returned to normal levels by 8 weeks in the adult. Taken together, these studies indicate that CBF recovers over time, which prompts the question as to the mechanism for this recovery in blood flow.
Dilatation of the cerebral vasculature is a well-defined autoregulatory mechanism for maintaining CBF after reduced perfusion pressure. If postocclusion CBF was being maintained at normal values by some degree of vasodilatation, effectively compensating for a reduced perfusion pressure due to the occlusion, we hypothesised that the BCCAO group would have a reduced capacity to vasodilate on a cerebrovascular reactivity test compared with controls. Following a CO2 challenge, the sham controls and BCCAO animals demonstrated a similar T*2 change, suggesting a similar capacity to vasodilate. This would suggest that vasodilatation is not the primary mechanism for CBF recovery in our model.
Following arterial occlusion, there is a redistribution of flow leading to an increase in intravascular shear forces, which have been implicated as a mechanism for the adaptive growth of pre-existing arteries, a process know as arteriogenesis (Busch et al, 2003). In our model, the occlusion of the common carotid arteries may cause increased flow through the vertebro-basilar network, resulting in a change in vessel diameter possibly accounting for the normalisation of CBF. We have therefore used MR angiography to investigate the changes in the intra-and extracerebral angioarchitecture. Common carotid arteries were not visible 6 months after BCCAO on the cervical MR angiograms. Nonetheless, normal regional CBF was observed, suggesting some redistribution of blood flow to the brain. The MR angiograms indicate that there are changes in the morphology of both the intracranial and extracranial vasculature by 6 months.
The intracranial MR angiograms demonstrate a haemodynamic change in the posterior cerebral arteries, which was present in both the adult and neonatal BCCAO animals. This is perhaps not surprising as the primary pathway for blood flow to the brain would be via the vertebral arteries after ligation of the common carotid arteries. Post-mortem comparison with India ink cerebral angiograms confirmed increases in vessel diameter (in both adult and neonatal BCCAO groups) of the majority of main intracerebral arteries, suggesting that there had been some vascular remodelling, although no differences between adults and neonates were noted.
Extracranial collateral vessels were observed in all of the adult and neonatal BCCAO animals and were absent in both the sham-operated groups. Furthermore, although both adult and neonatal BCCAO rats were observed to have collateral vessels arising from the vertebral arteries, the neonatal BCCAO animals had larger calibre midline collaterals than the adults, suggesting that these morphologic changes may be influenced by age.
Oldendorf (1989) has also reported intracranial vascular changes and the appearance of multiple cervical collateral vessels in the adult rat 15 weeks after BCCAO. Similar vessel diameter changes were also observed 5 weeks after BCCAO in a rabbit model (Masuda et al, 2000). Busch et al (2003), using a 3-vessel occlusion model, suggested that even 3 weeks after occlusion vascular remodelling had occurred in the mature rat brain. However, Oldendorf noted enlargement of only the proximal segment of the posterior communicating, vertebral and posterior communicating arteries whereas we observed addition changes (an increase in vessel diameter) in the anterior cerebral artery. This discrepancy could be because of differences in time scale: 15 weeks in Oldendorfs study compared with our 6 months. Our results demonstrated further changes in the anterior segment of the circle of Willis, which may indicate that vascular remodelling continues after 15 weeks of occlusion. Overall, this study suggests that the angioarchitecture has altered in response to BCCAO, and furthermore that the adaptive mechanisms have an intra- and extracranial component with some regional age and time dependence.
The age at insult may be a determining factor in vascular plasticity. Vascular endothelial growth factor has an essential role for vessel growth in early postnatal life (Gerber et al, 1999), and there is a close correlation between cerebral angiogenesis and VEGF expression in the postnatal developing brain (Ogunshola et al, 2000). The ability of VEGF to induce maximal capillary proliferation peaks between 5 and 9 postnatal days, when it is 40 times more potent than in the adult (Robertson et al, 1985). Interestingly, VEGF mRNA has been shown to be upregulated in a model of chronic cerebral hypoperfusion in adult rats (Hai et al, 2003). Thus, it may be hypothesised that BCCAO induces the upregulation of VEGF, which, in turn, could lead to the development of collateral vessels, and its greater potency in the developing brain may account for some of the differences found between the age groups, such as the midline collaterals. However, this may not explain why the basilar artery is more tortuous in the adult BCCAO than in the neonatal BCCAO animals. The increased tortuosity of the basilar artery has been observed previously (Oldendorf, 1989) and it has been postulated that this is due to vertebral artery elongation, which leads to a rostral movement of the vertebrobasilar junction and subsequently basilar tortuosity. The question remains as to why the basilar arteries become more tortuous in adults than in the neonatal group. One possibility is that whole body growth in the neonatal group negates the effects of relative vascular elongation. It is also possible that the increased tortuosity is due to ageing of the vessel wall leading to loss of longitudinal stiffness, which combined with changes in shear forces after BCCAO leads to greater tortuosity (Tao et al, 2004).
We also investigated ADC, T1 and T2 at 6 months after surgery as an indication of the morphologic sequelae of BCCAO. We demonstrated a subtle increase in T1 in the BCCAO animals when compared with the sham animals, but did not observe any significant differences between the adult and the neonatal group. We also observed a small increase in T2 in the thalamus of the adult BCCAO group when compared with the sham-operated adult animals; no differences were observed in any of the regions in the neonatal group. No significant ADC differences were noted. Previous studies have reported T2-weighted changes in a model of cerebrovascular insufficiency confined to the hippocampus of aged rats but not young rats 9 weeks after surgery (De La Torre et al, 1992). This MRI hyperintensity, corresponding to increased T2, showed excellent correlation with histopathologic data and was presumed to reflect local tissue oedema (De La Torre et al, 1992). Although the origins of both acute and delayed T1 and T2 changes after MCAO have been discussed previously (Van Bruggen et al, 1994; Fujioka et al, 1999; Calamante et al, 1999a), we are unsure of the exact pathologic significance of our small T1 and T2 changes. These may reflect, for example, an increase in total tissue-water content due to oedema. Another possibility is that due to the differences between blood and tissue T2 values (Kalisch et al, 2001), our T2 changes may reflect an increase in cerebral blood volume (CBV) via an increase in vascular density. Previous work has shown that CBV changes can be associated with changes in vascular density after transient focal cerebral ischaemia (Lin et al, 2002) or changes in vessel size, leading to changes in T2 (Kalisch et al, 2001).
In summary, we have demonstrated that occlusion of the common carotid arteries in our animal model causes an initial decrease in CBF, which is subsequently restored by 6 months. This CBF change is associated with vascular remodelling, which may account for the normalisation of CBF. Both MR and India ink angiograms suggest that the specific adaptive response is age-related, and has both an intra- and extracranial component. Our study indicates that MRI is an effective tool to investigate age-related vascular responses in models of vasoocclusive disease.
Footnotes
Acknowledgements
The authors gratefully acknowledge the Wellcome Trust and the Biotechnology and Biological Sciences Research Council for their support and the Child Health Research Appeal Trust for pump-priming support.
