Abstract
As effective stroke treatment by thrombolysis is bound to a narrow time window excluding most patients, numerous experimental treatment strategies have been developed to gain new options for stroke treatment. However, all approaches using neuroprotective agents that have been successfully evaluated in rodents have subsequently failed in clinical trials. Existing large animal models are of significant scientific value, but sometimes limited by ethical drawbacks and mostly do not allow for long-term observation. In this study, we are introducing a simple, but reliable stroke model using permanent middle cerebral artery occlusion in sheep. This model allows for control of ischemic lesion size and subsequent neurofunctional impact, and it is monitored by behavioral phenotyping, magnetic resonance imaging, and positron emission tomography. Neuropathologic and (immuno)-histologic investigations showed typical ischemic lesion patterns whereas commercially available antibodies against vascular, neuronal, astroglial, and microglial antigens were feasible for ovine brain specimens. Based on absent mortality in this study and uncomplicated species-appropriate housing, long-term studies can be realized with comparatively low expenditures. This model could be used as an alternative to existing large animal models, especially for longitudinal analyses of the safety and therapeutic impact of novel therapies in the field of translational stroke research.
Introduction
Acute cerebral vessel occlusion causing ischemic stroke and leading to subsequent loss of cerebral tissue and function is one of the leading causes of death and disability in industrial nations (Kolominsky-Rabas et al, 2006). Systemic thrombolysis using tissue plasminogen activator or other agents can provide significant clinical benefits for stroke patients. However, only a minority of stroke victims can be treated with this approach, mainly because of the narrow time window for successful and safe intervention (Lapchak and Araujo, 2007). Consequently, the development of novel therapeutic strategies is essential to improve the situation in acute stroke treatment.
Current studies on experimental stroke therapies, evaluating the efficiency of neuroprotective agents, cell-based approaches, or other strategies, primarily use rodent models of permanent or transient focal cerebral ischemia. These animal models offer numerous advantages such as reproducible lesion size, detailed, but not capacious knowledge about pathophysiologic mechanisms of stroke, broad availability, and high flexibility for different experimental requirements. However, lesion size and reproducibility of sensorimotor deficits might differ between common rat strains (Traystman, 2003).
In the past, a noticeable number of experimental protocols based on neuroprotective drugs, which proved to be effective in rodents, have failed in clinical trials (Del Zoppo, 1995; Heiss, 2002). This indicates a clear demand for an additional experimental step between common rodent models and human patients in evaluating novel stroke therapies before entering clinical applications. Consequently, the STAIR criteria strongly recommend the use of appropriate, close-to-practice large animal models of focal cerebral ischemia (Schabitz and Fisher, 2006). In this regard, modern brain imaging techniques such as magnetic resonance imaging (MRI) and positron emission tomography (PET) are crucial for online monitoring of novel treatment protocols.
Human-relevant primate models, which are essential for investigating acute stroke, are restricted to specialized breeding facilities and, the application of marmoset models notwithstanding, do not often allow for long-term observation of experimental subjects because of high mortality (1999).
Therefore, the aim of this study was to develop a robust model of focal cerebral ischemia in sheep, which can easily be housed in a species-appropriate environment. Because of the
Materials and methods
Experimental Animals
All experimental procedures were approved by the Experimental Animal Committee of the Regional Council of Leipzig. A total of 30 healthy outbreed adult hornless merino rams weighing 42 to 65 kg were obtained from the experimental farm of the Faculty of Veterinary Medicine of the University of Leipzig without any kind of selection. Subjects were kept in small flocks with
Behavioral assessment

Sheep were examined regularly for neurological dysfunctions according to the items given. The score point system is based on a common veterinary neurological investigation technique (Oliver et al, 1997). In the case of stupor and coma, animals received 25 and 27 score points, respectively. No further investigation could be performed. It should be noted, that stupor was detected once on day 1, while a comatose state was not caused by MCAO in any animal observed in this study.
Induction of Stroke by Middle Cerebral Artery Occlusion
Rams were randomly assigned to one of five groups: transcranial 1-branch- (
Anesthesia was performed according to Supplementary Table 1. Animals were intubated after induction of anesthesia. An intraesophageal tube was used to avoid ruminal tympania. Throughout the surgical procedure, tracheal respiration with 1.5% to 2.0% isoflurane in pure oxygen was performed. An electrocardiogram was recorded, and blood pressure was continuously measured using an intra-arterial probe placed in the dorsal pedis artery of the left hind limb.
The relevant anatomic structures and the surgical procedures are shown in Figure 1. Briefly, the skin between the ear and eye was incised on the left side of the head to expose the branches of the superficial temporal artery and the accompanying vein. After occlusion of these vessels by high frequency bipolar forceps (Liga Sure, Valleylab, Boulder, CO, USA), the origin of the temporalis muscle was incised at the temporalis line and the muscle was carefully elevated from the parietal bone. After exposure of the skull bone surface, a hole was drilled into the parietal plate using a set of neurosurgical burrs and was then extended using Kerrison rongeurs. On local incision of the dura mater, MCA or its branches (according to the experimental group) were permanently occluded. Then, the temporalis muscle was fixed on the temporalis line and the skin was sutured using 2-0 absorbable filaments (Ethicon Ltd, Norderstedt, Germany). After termination of surgery, animals were disconnected from artificial ventilation and immediately taken to a specialized awakening box. Intratracheal and intraesophageal tubes were removed when the swallowing reflex was restored. A postsurgical antibiotic treatment (Supplementary Table 1) was performed for 7 days.
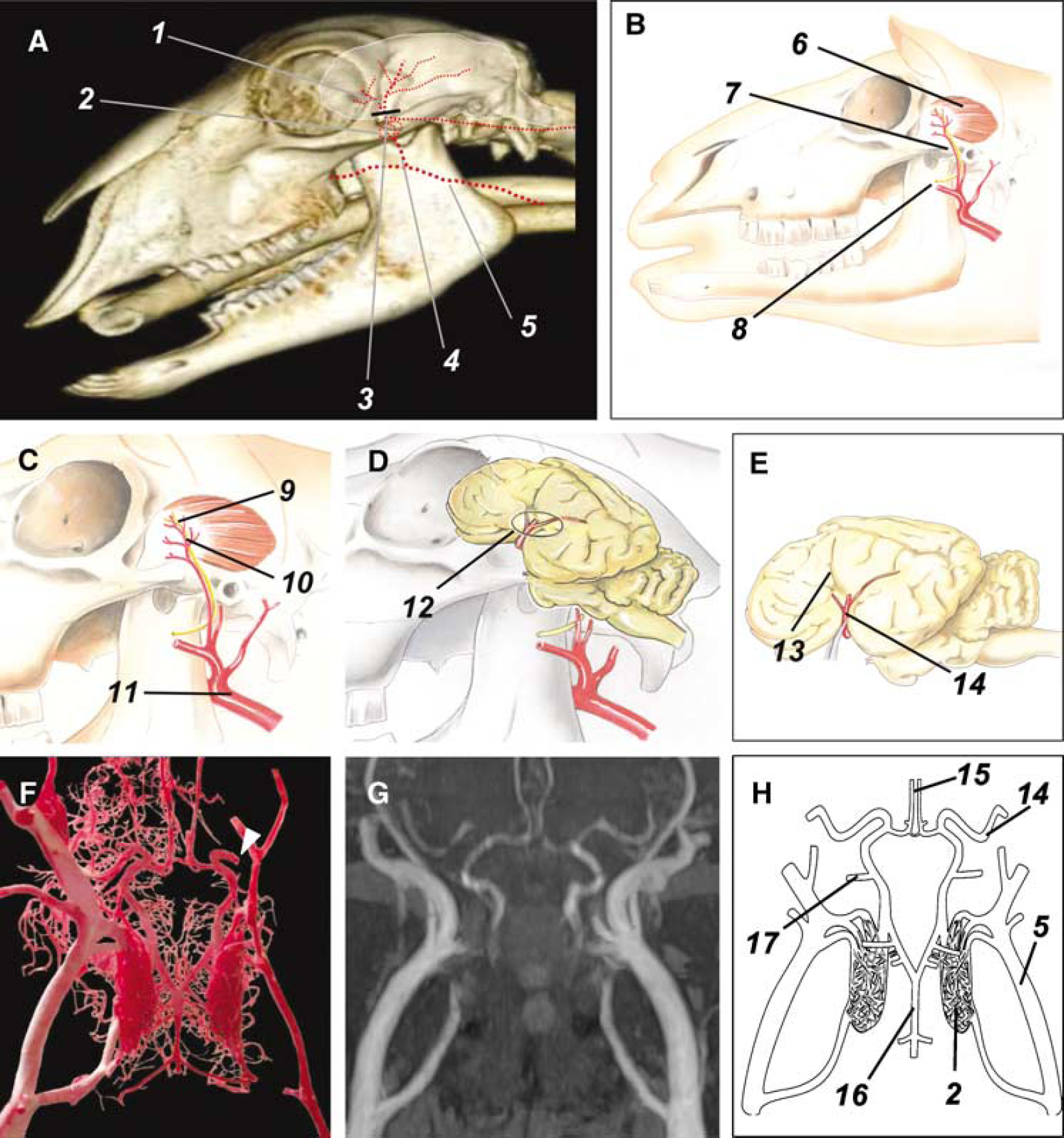
Intraoperational situs, topographical anatomy and cerebral blood supply. Principle of the surgical procedure and the most important anatomic structures in the operational field. (
Imaging Procedures
Magnetic resonance and PET examinations were performed on days 1, 14, and 42 after onset of stroke. Magnetic resonance imaging was performed using a 1.5 T clinical whole body scanner (Gyroscan Intera; Philips, Koninklijke, The Netherlands) with a flexible double loop RF-coil (Sense Flex M; Philips). Animals were anesthetized as described in Supplementary Table 1. Spontaneous ventilation of sheep was monitored by a pneumatic respiration probe connected to the scanner. Magnetic resonance imaging was performed according to protocols shown in Supplementary Table 2. Altogether, T1 turbo spin echo, T2 turbo spin echo, T2 diffusion weighted (DWI), T2 FLAIR, T2* gradient echo, and time of flight MR angiography (MRA) sequences were recorded. To diminish the susceptibility artifacts in the DWI and to shorten the examination duration, a parallel imaging protocol was used (SENSitivity Encoding; Philips) with a SENSE factor of 1.5 or 1.3 (MRA only). Three diffusion directions and two
For MRI volumetry, T2 weighted sequences were analyzed by ImageJ 1.34 secs software (Wayne Rasband; National Institutes of Health, Bethesda, MD, USA), whereas the YAWI 2D 1.3.2. software (open source) served as the selection tool. The sizes of the ischemic lesion (area of cytotoxic edema as verified by decreased apparent diffusion coefficient values of the DWI), of the ipsilateral and contralateral side ventricles, and of both hemispheres were all calculated. Infarct volume was assessed by calculating the size of the lesion observed in T2W MR images on day 1. On days 14 and 42, the difference between the volumes of both side ventricles was added to the size of the lesion (if observed) to compensate for occasionally occurring difficulties in discrimination between the lesion area and side ventricle in T2 weighted images.
Tissue loss because of ischemic damage was represented by the hemispherical atrophy. For this process, the quotient between the size of the ipsilateral and contralateral hemispheres minus the associated ventricle volumes was calculated on days 14 and 42.
Positron Emission Tomography Imaging
In addition to MRI, serial PET imaging was performed in a subgroup of five sheep with total proximal MCAO. This was performed using a high-resolution clinical scanner (ECAT EXACT HR+; Siemens/CTI, Knoxville, USA). The scanner characteristics include: axial field of view 15.5 cm, 63 parallel transverse slices, slice thickness 2.4 mm, final reconstructed image resolution 7.1 mm (transverse), and 6.7 mm (axial). Cerebral blood flow (CBF) was visualized and quantified in the sheep using 15O-H2O. Further, on day 42 cerebral metabolic rate of glucose (CMRGlu), a surrogate measure of brain viability was obtained by means of 18F-FDG PET. In addition to these five sheep with total MCAO, a series of one subject each with 1-branch and 2-branch MCAO and sham surgery underwent brain CBF PET with 15O-H2O 1 day after surgery. The anesthetized sheep were i.v. administered with ∼1000MBq 15O-H2O and ∼370MBq 18F-FDG, respectively. In the case of 15O-H2O, dynamic PET data were acquired in 3D-mode over 5 mins, and in the case of 18F-FDG in 2D-mode over 60 mins. Further details on the PET acquisition parameters are given in Supplementary Table 3. After standard image data reconstruction and correction for measured tissue attenuation, the resulting PET image datasets were transferred to a Hermes workstation (Hermes Medical Solutions, Stockholm, Sweden). By using the multimodality software, irregular regions of interest (ROI) were manually defined by an experienced investigator in all MCAO-affected transverse slices of the five sheep brains after total MCAO for the areas with uptake deficits. Slice-wise, these ROI were horizontally mirrored to the contralateral (nonaffected) brain hemisphere to define reference ROI. From these ROI, the volume as well as the relative (as related to the mirrored contralateral VOI) severity of the MCAO-induced activity deficits were calculated.
Behavioral Phenotyping
All animals were examined for neurofunctional disabilities before surgery (sham and MCAO) as well as on days 1, 4, 7, 10, 16, 25, 32, and 42 thereafter. For this process, a neurologic score point system was developed. The system is based on the common test system for neurologic dysfunctions in large animals (Oliver et al, 1997), however, focuses on the specific functional deficits observed in most animals after MCAO. A detailed description of the parameters investigated is given in Table 1. Interestingly, sheep rapidly habituated to weak or medium nociceptive stimulation. Therefore, sensor function could not be tested reliably.
Neuropathology, Immunohistochemistry and Immunofluorescence
Animals were killed 43 days upon MCAO by a single i.v. injection of 15 mL pentobarbital-Na (Eutha 77; Essex Pharma Ltd, Munich, Germany). After immediate decapitation heads were perfused through internal carotid arteries with 1 L of phosphate buffered saline solution (pH 7.4) followed by 14 L of 4% paraformaldehyde (Sigma Aldrich Ltd, Taufkirchen, Germany)/0.1 mol/L phosphate buffered saline. Occipital and parietal skull plates were removed followed by immersion fixation in 4% paraformaldehyde/0.1 mol/L phosphate buffered saline overnight before the brain was removed.
After gross examination, the whole brain was coronally cut into equally spaced slices (approximately 4 mm) for macroscopic and microscopic examination. Two specimens were taken from three defined sites of the left hemisphere (Figure 5A): the macroscopically assessed frontal and occipital borders of the infarct and the level of the left MCA. From the intact right hemisphere, two samples corresponding to locations 1a and 1b were taken. The coronal sections were fixed in buffered 4% paraformaldehyde for another 2 days, processed routinely, embedded in paraffin, and cut in 4 μm thick sections. For conventional light microscopy sections were stained with hemalaun and eosin or pikrosirius red by standard methods and facing cut surfaces the consecutive specimens were histologically examined.
For immunohistochemistry, the peroxidase antiperoxidase method was used. Sections were dewaxed, rehydrated, and treated with hydrogen peroxide 0.5% in methanol for 30 mins at room temperature (RT). After washing in Tris-buffered saline (TBS), blocking with 50% pig serum in TBS at RT was performed for 10 mins. The polyclonal antibody rabbit anti-bovine glial fibrillary acid protein (GFAP; 1:500) was applied to the sections overnight at 4°C and the slides were incubated for 30 mins at RT with pig anti-rabbit immunoglobulin G secondary antibody (DAKO Cytomania GmbH, Hamburg, Germany; 1:100) thereafter. This was followed by incubation for 30 mins at RT with the rabbit-peroxidase antiperoxidase-complex (DAKO; 1:100). After signal detection with a freshly prepared solution of 3,3‘-diaminobenzidintetrahydro-chloride (Fluka Feinchemikalien, Neu Ulm, Germany) for 10 mins at RT, the sections were counterstained with Papanicolaou's stain. For control purposes the primary antibody was replaced by normal rabbit serum (DAKO). Histologic and immunohistochemical investigations were performed using a standard microscope (Olympus).
For fluorescence microscopy perfusion fixated coronal sections taken from the reactive zone next to the lesion and a corresponding area in the intact hemisphere were cryopreserved in 30% sucrose (Sigma Aldrich)/0.1 mol/L phosphate buffered saline. Free floating sections from cryopreserved specimens were extensively rinsed with 0.1 mol/L TBS, pH 7.4. Nonspecific binding sites for subsequently applied antibodies and lectins were then blocked with 5% normal donkey serum in TBS containing 0.3% Triton X-100. Next, the sections were incubated with primary antibodies and biotinylated lectins (see Supplementary Table 4) overnight at RT. For triple fluorescence labeling, markers were simultaneously applied in two main series: (1) mouse-anti-CD11b + rabbit-anti-GFAP + biotinylated RCA or (2) biotinylated tomato (or potato) lectin + rabbit-anti-GFAP + mouse-anti-NeuN. After rinsing with TBS, the sections were processed by concomitant use of appropriate carbocyanine (Cy)2-, Cy3-, and Cy5-conjugated secondary immunoreagents (all Dianova, Hamburg, Germany; supplier for Jackson Immuno Research, West Grove, PA, USA) and used at 20 mg/mL for 1 h. Finally, sections were extensively washed with TBS, briefly rinsed with distilled water, mounted onto fluorescence-free-slides, air-dried and coverslipped with Entellan (Merck, Darmstadt, Germany).
In control experiments, the omission of primary antibodies or lectins resulted in the expected absence of any cellular labeling, switch of fluorophores caused no alteration of staining patterns.
All fluorescence slides were inspected with a confocal laser-scanning microscope LSM 510 Meta (Zeiss, Jena, Germany) equipped with an argon laser exciting Cy2 at 488 nm and two helium-neon lasers (543 nm for Cy3 and 633 nm for Cy5). Cy5-labeled structures were color-coded in blue.
Statistics
Statistical analysis was performed using the SigmaStat 3.0 software (Systat Inc., San Jose, CA, USA). Differences in neurofunctional outcome between different MCAO groups and controls were evaluated using the one-way ANOVA test. Differences in MRI and PET findings size were analyzed by the Student's
A
Results
General Observations and Animal Welfare
No animal died because of reasons related to the surgical procedure or stroke. There was a reversible reduction of vigilance and food intake for a time period of up to 4 days after MCAO. Although movement disturbances including hemiparesis were observed, especially in the total MCAO group, hemiplegia was not caused in any animal. Hence, the rams were able to stand, move about, and join other sheep. Outdoor housing was possible from day 5 onwards.
Diffusion Disturbances and Hemispherical Atrophy in Relation to Numbers of Middle Cerebral Artery Branches Occluded
Middle cerebral artery occlusion but not sham surgery caused diffusion disturbances and atrophy in the sheep brain.
The development of diffusion disturbances and hemispherical atrophy is illustrated in Figures 2 and 3. In the control or sham-treated animals, MRI revealed no signs of ischemic damage. However, in three of the five sham animals diffusion disturbance (Figure 2A, white arrow heads) were observed on day 1, but not thereafter. There was no indication for an ischemic origin of these disturbances in the corresponding apparent diffusion coefficient maps. Furthermore, these areas appeared as small cortical contusion damages in histologic examinations.
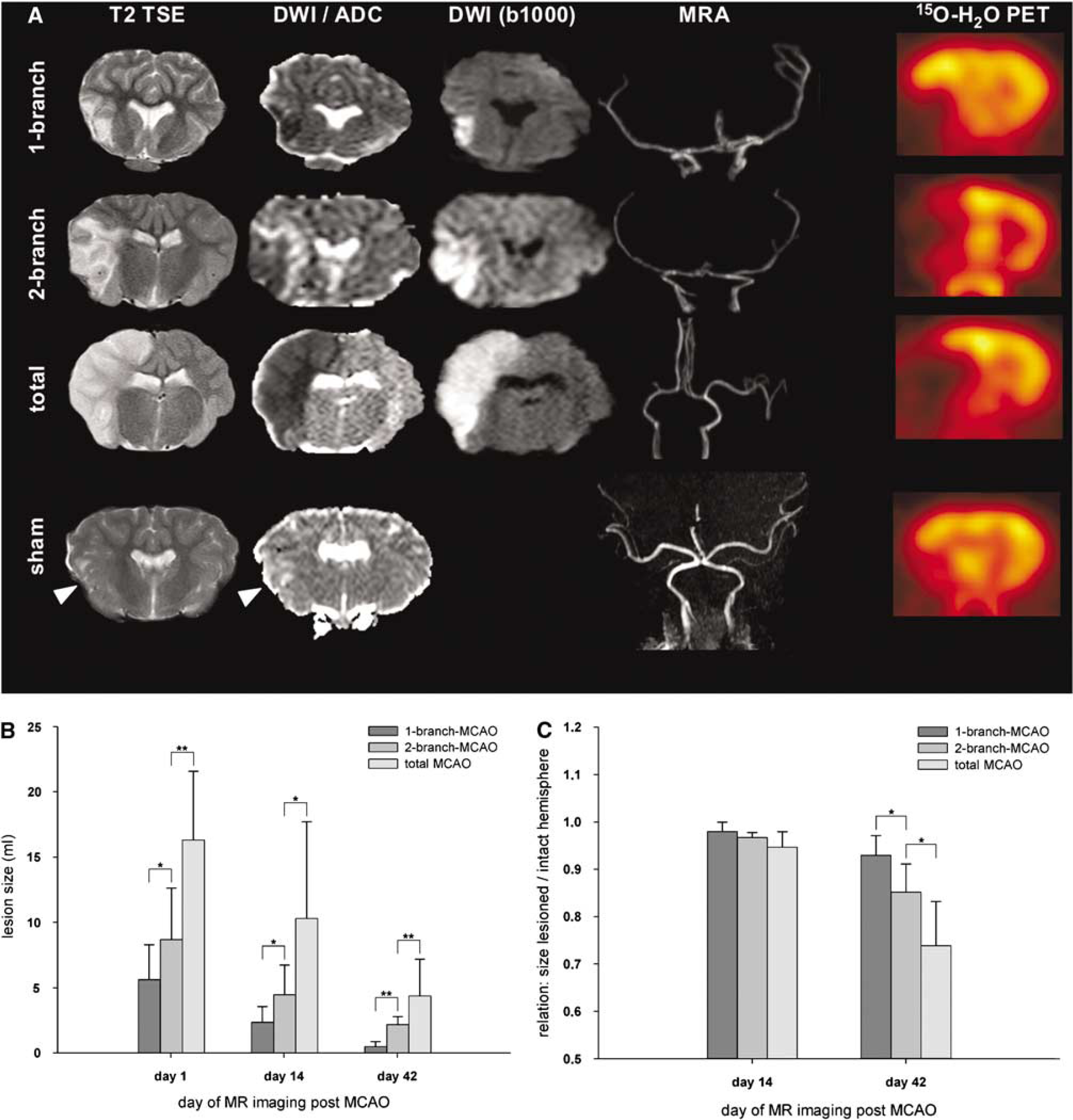
Lesion size and hemispherical atrophy in relation to MCAO modality. (
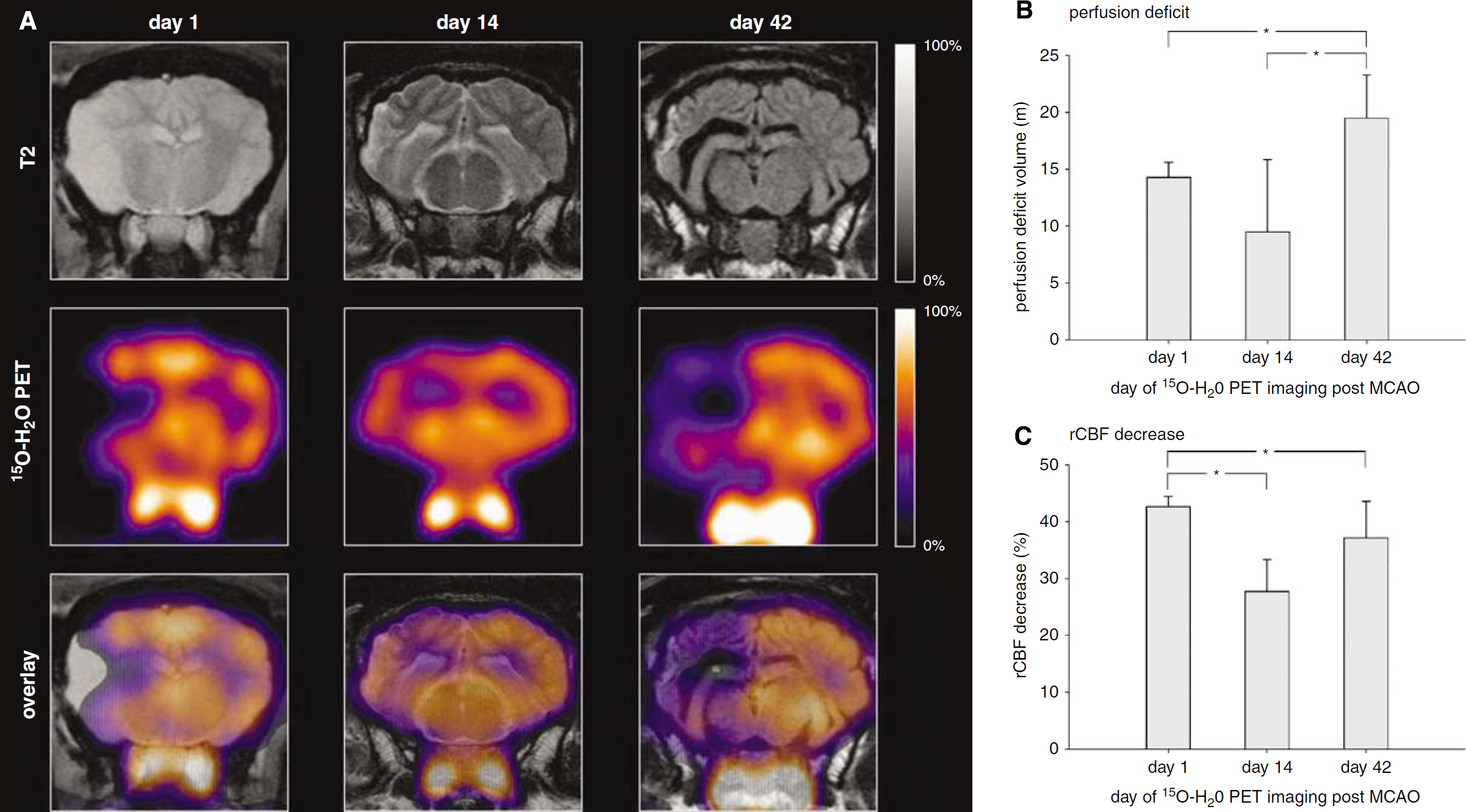
Follow-up 15O-H2O PET imaging. (
Magnetic resonance imaging analyses revealed that the volume of diffusion disturbances reflected the number of MCA branches occluded. Thereby, the initial (day 1) lesion volume in diffusion-weighted MRI after total MCAO was significantly larger compared with the volume after 2-branch-MCAO (16.3±5.2 versus 8.7±3.9 mL,
Concerning the ratios between the volumes of both hemispheres (lesion versus intact; Figure 2C), there was no statistically significant difference between the experimental subgroups on day 14 (
15O-H2O and 18F-FDG Positron Emission Tomography Show Characteristic Middle Cerebral Artery Occlusion-Related Deficits
Figure 3 shows the influence of the extent of MCA surgery on the stroke-related CBF deficits as visualized by 15O-H2O PET. In accordance with the MRI data, the deficit extent increased in the order sham surgery < 1-branch MCAO < 2-brach MCAO < total MCAO (Figure 2A). For the five sheep with total MCAO which underwent serial brain perfusion PET imaging, the extent of the CBF deficits defined by the described ROI technique increased over time from 14.3±1.3 mL on day 1 (over 9.5±6.3 mL on day 14; not significant) to 19.5±3.7 mL on day 42 (
Behavioral Phenotyping Shows Functional Disabilities Correlated to Lesion Size
Assessment of motor functions and vigilance resulted in reproducible defect patterns corresponding to the size of the ischemic lesion induced by MCAO (Figure 4). Total MCAO was accompanied by more severe dysfunctions compared with other groups throughout the observation period (
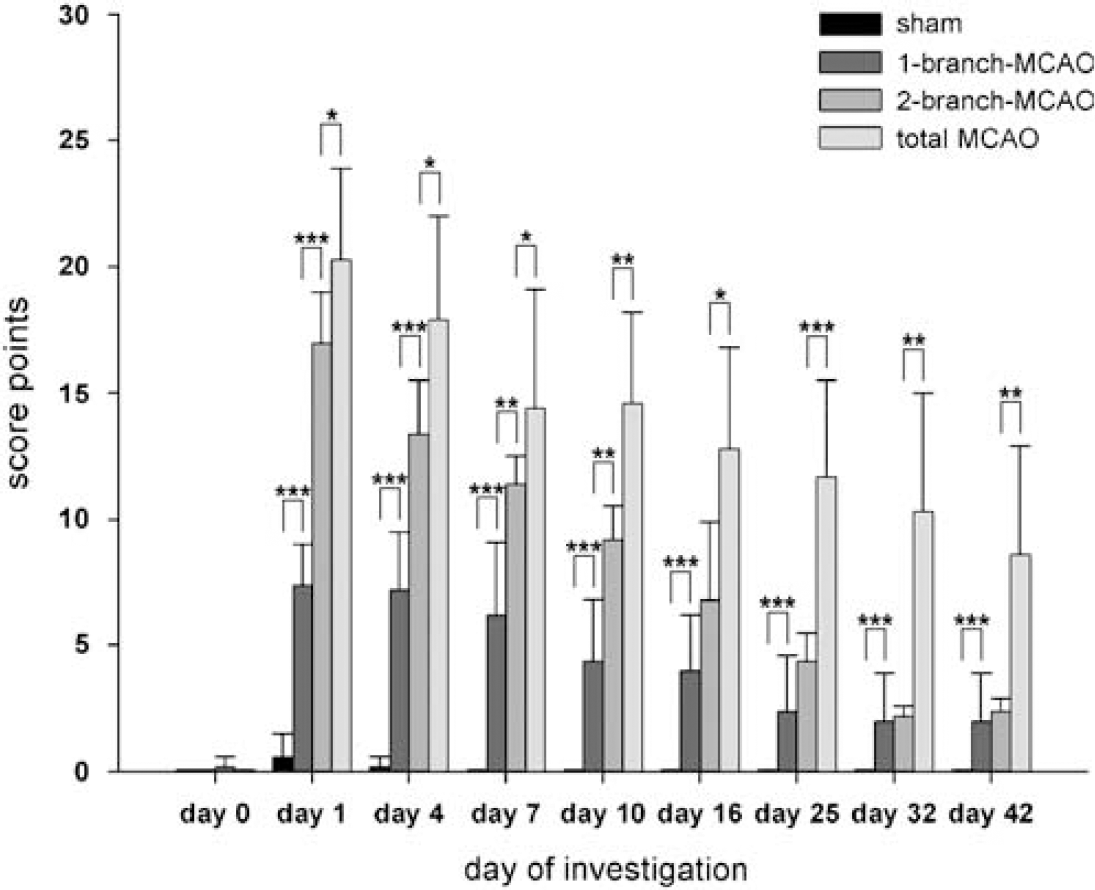
Clinical assessment using neurologic score point system. Behavioral phenotyping after MCAO showed statistically significant differences between the experimental groups in relation to the number of MCA branches occluded. There was a certain recovery in all occlusion groups, without reaching baseline levels. Sham-operated animals showed only mild and transient dysfunctions on days 1 and 4, whereas 1- and 2-branch-MCAO yielded significantly different clinical scores until day 10 (
In sham-operated animals, minimal transient dysfunctions were observed only on days 1 and 4. Those dysfunctions were rather unspecific, mainly including a positive item 2 (Table 1) and a reduced state of activity. Therefore, they could not be discriminated from common postsurgical effects, not related to MCAO. The transient dysfunctions in sham-operated animals were significantly less (
Gross pathology, Immunohistochemistry, and Fluorescence Microscopy
The infarct area in the left hemisphere was clearly discernible as a concave light brown area (Figure 5B) of softer consistency with a slightly thickened and opaque leptomeninx. The cerebral gyri and sulci were hardly visible or already missing. In the coronal sections tissue shrinkage accompanied by a varying enlargement of the associated lateral ventricle (Figure 5C) was obvious. The sharply demarcated infarct area, consistently involving cortex and medullary layer, showed variable cavitations. Apart from the extension, histopathologic findings did not differ between the MCAO groups and can be described as follows.
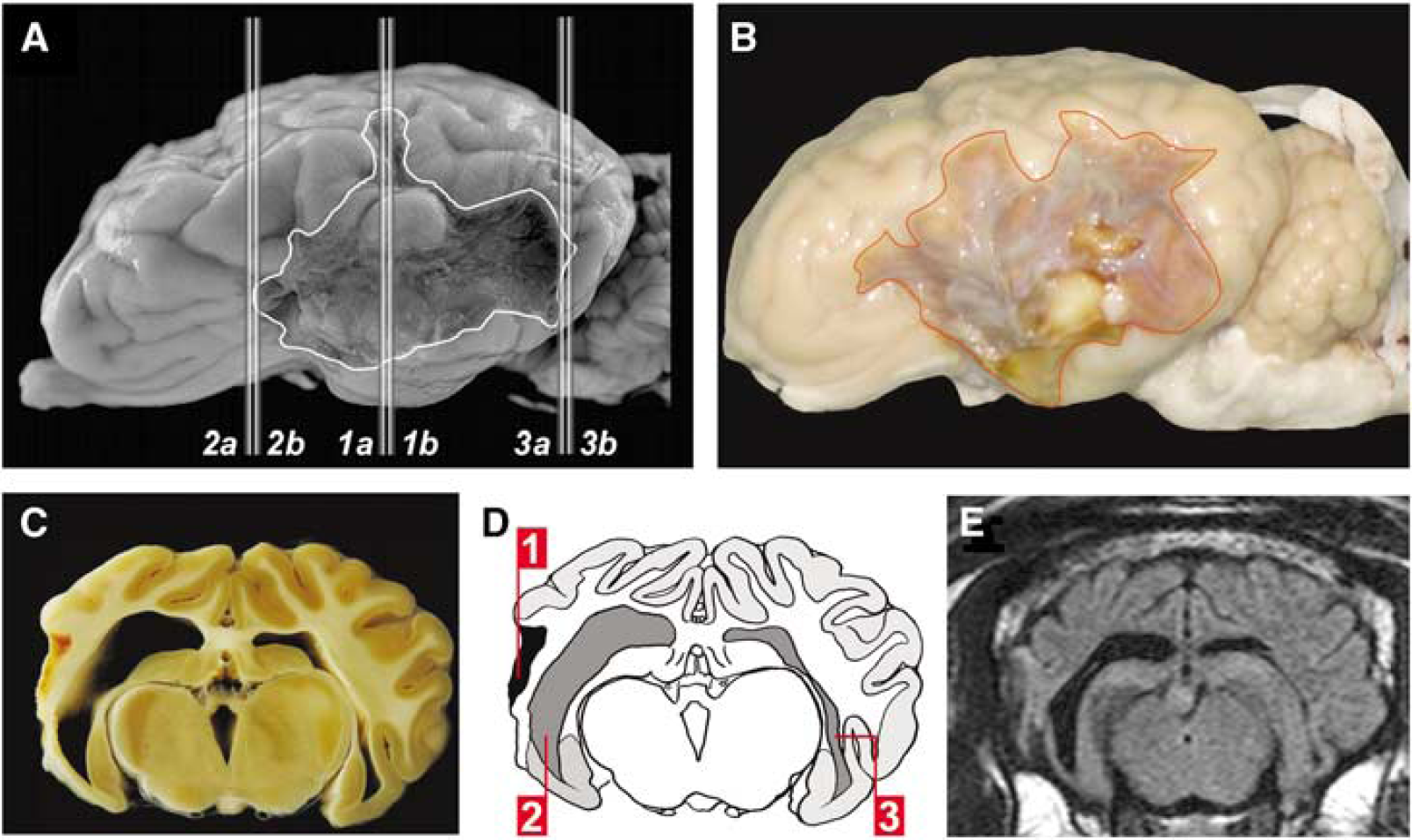
Histopathologic sampling procedure and gross pathology. All brain specimens investigated were obtained after killing the animals on day 43 after MCAO. (
In the area of MCAO, the leptomeninx displayed a mild to focally moderate active fibrosis with scattered mononuclear infiltrations. In the infarct border, fibrotic changes were moderate and proliferation of capillaries was visible. The classification of the infarct area into a central zone (necrosis), a reactive zone (directly bordering the necrosis), a marginal zone (transitional zone with both intact and affected brain tissue), and a remote zone was adapted from the classification of Garcia and Kamijyo (1974).
The central zone (Figures 6A and 6B) consisted of abundant foamy fat granule cells, scattered lymphocytes and plasma cells, few siderocytes, minimal to moderate angiogenesis, and connective tissue proliferation, increasing next to the leptomeninx. Cavitation was noted in the depth of infarct (Figure 6A). The formation of connective tissue and angiogenesis increased to the margins of the central zone.
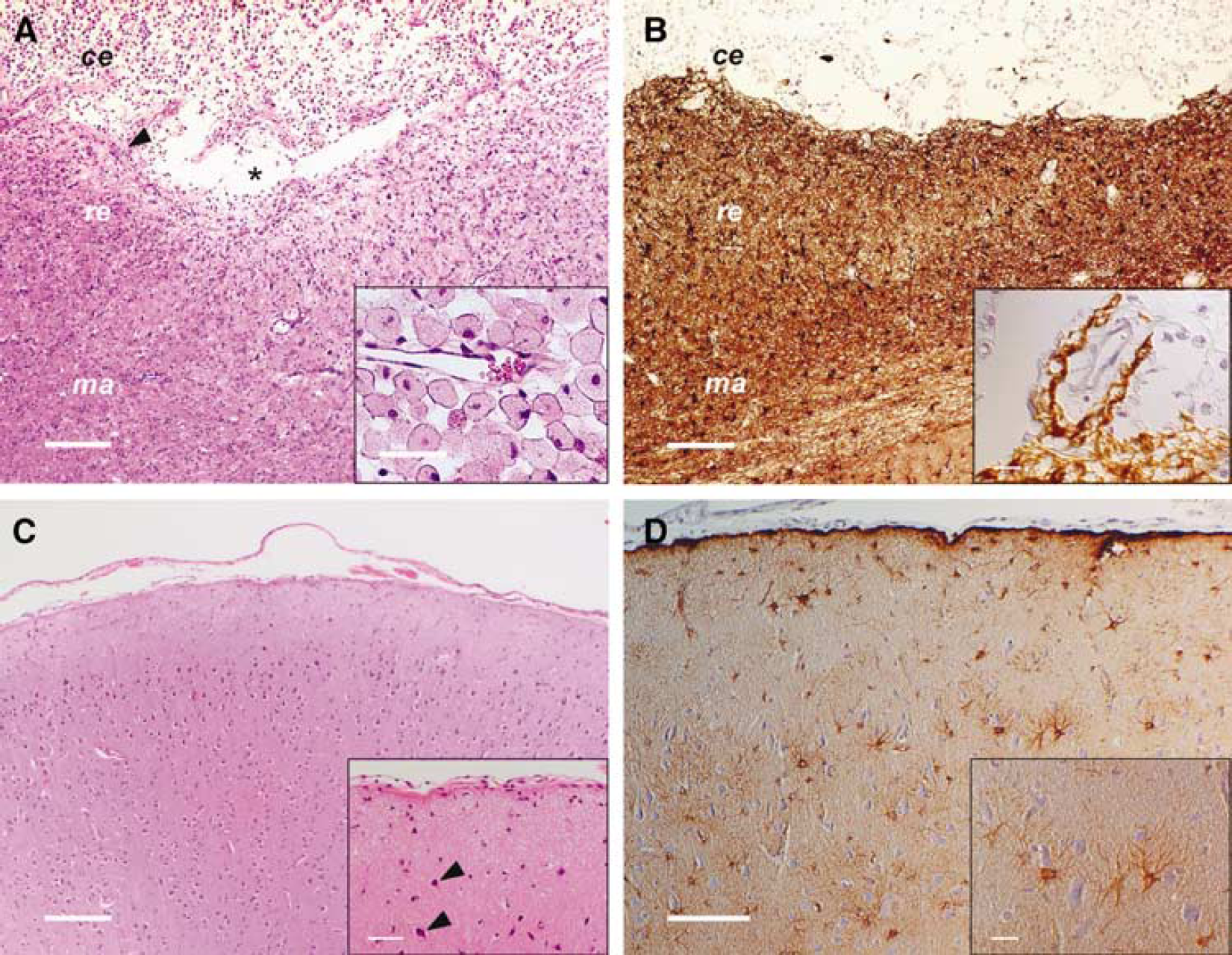
Histopathologic findings. Histopathologic evaluation was performed from brain specimens obtained on day 43 on induction of ischemia. (
In the reactive zone (Figures 6A and 6B; see also Figure 7) moderate augmentation of astrocytes and microglia as well as moderately increased capillarization was observed. In close proximity to the central zone neurons were completely missing. More distant, neurons showed mild satellitosis and signs of hypoxic cell changes. In the medullary layer, spheroids occurred in mild to moderate, focally high numbers.
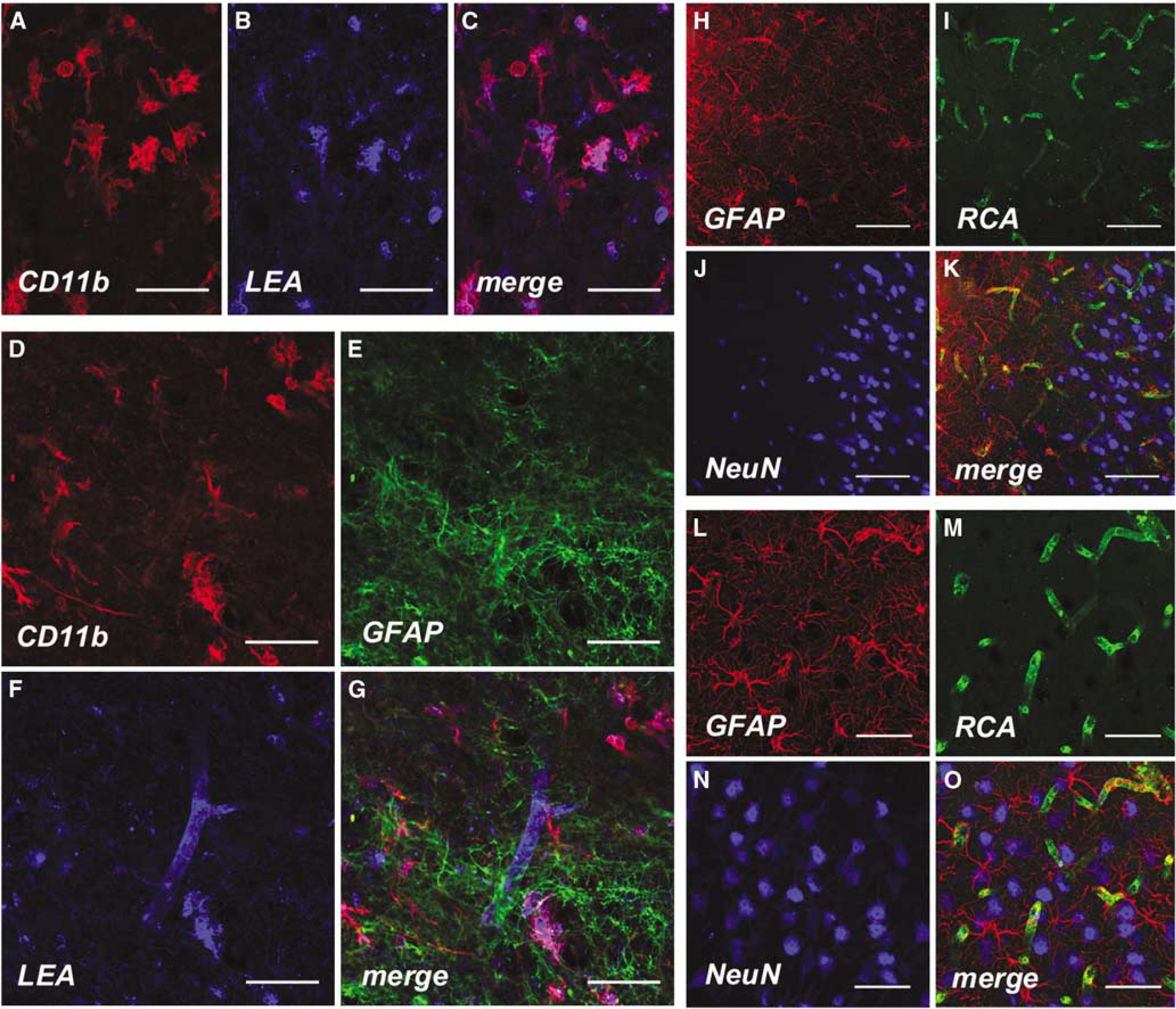
Confocal fluorescence microscopy. Fluorescence immunohistochemistry was performed using brain tissue that was cryopreserved immediately after animal euthanasia on day 43. Augmentation of cD11b+ microglial cells (lb>
The cortex of the marginal zone (Figures 6A and 6B) showed, hinted to minimal satellitosis, mild hypoxic neuronal changes, astrocytosis, as well as moderate microgliosis. In the medullary layer, showing mild astrogliosis and microgliosis, spheroids were rare.
In the remote zone (Figures 6A and 6B), the cortex displayed mild satellitosis, hypoxic neuronal cell changes and slightly increased numbers of astrocytes and microglia.
The intact right hemisphere showed mild leptomeningeal fibrosis with few mononuclear infiltrations. The cortex and medullary layer displayed alterations already described for the remote zone of the affected left hemisphere.
All antibodies and lectins used in the described protocols labeled distinctive cellular structures in sheep brain specimens. Investigation of the reactive zone using fluorescence microscopy showed a moderate increase of CDl1b+ cells, colocalized with vessel structures (LEA; Figures 7A–C). Triple staining experiments including antibodies directed against GFAP indicated enhanced astrocytosis in areas where augmentation of CDl1b+ cells was detected (Figures 7D–G).
In border zone areas next to the core of the lesion, reactive astrocytosis was indicated by strong GFAP-staining (Figure 7H) representing glial scar tissue. Although the density capillaries was slightly increased as revealed by RCA (Figure 7I) or using
In corresponding areas of the intact hemisphere and regions more distant form the central lesion (Figures 7M–P), there were no local differences between the density of NeuN+ cells (Figures 7O and 7P) whereas slight astrocytosis indicated by GFAP + (Figure 7M) cells could be observed. However, astrocytosis was much less prominent in corresponding areas of the intact hemisphere than next to the lesion.
Discussion
The treatment protocols for ischemic stroke, which are currently available for routine clinical application and are based on thrombolysis, are efficient but limited by a narrow time window. It is well known that several alternative approaches, including neuroprotective and cell based strategies, have been shown to improve stroke outcome and possibly to widen the therapeutic time-window in rodents (Newcomb et al, 2006; Daadi et al, 2008); however, these approaches have still not been successfully translated into routine clinical protocols and additional research, especially elucidating underlying mechanisms, will be urgently needed to assess the value of these experimental approaches for human stroke patients. It is further known from previous trials involving neuroprotective substances that appropriate animal models are crucial for verifying novel, promising approaches. An optimal preclinical stroke model should reflect the size and complexity of the human brain and in parallel allow for the monitoring of clinical, pathophysiologic, morphologic, and histologic parameters of stroke development (Green, 2002).
In principle, most primate models fulfill these requirements and are of outstanding importance in acute stroke research but are often accompanied by a limited observation time because of high mortality rates. However, studies lasting for 10 weeks using total MCAO in marmosets (Marshall et al, 2003), one of the smallest primate species, were not able to predict clinical ineffectiveness of the neuroprotective agent NXY-059 (Shuaib et al, 2007). This might possibly be related to the small size of the marmoset brain. Thus, neuroprotective agents (as in rodents) could potentially reach large relative proportions of hypoxic or ischemic tissue by diffusion and/or active molecular transport processes within the therapeutic time frame after permanent MCAO, resulting in an average volume of protected tissue of 90 mm3, also accompanied by reduced functional deficits (Marshall et al, 2003). This absolute amount of rescued brain tissue would probably not be of that particular functional relevance in larger species or humans also experiencing extended stroke because of total MCA blockage.
Limited observation times after MCAO which are seen in many currently available large animal models could be resolved by using ruminants such as sheep, which also exhibit important similarities to primates in terms hematologic parameters and blood grouping (see Supplementary Table 5). The absent of stroke-related mortality in our study was most probably because of the partial removal of parietal skull bone thus limiting the increase of intracranial pressure in the phase of the subacute cytotoxic brain edema. Also, the induction of cortical (distal) but not total (proximal) MCAO as performed in this study, effectively limits the increase of intracranial pressure.
However, it has to be stated that more methodological tools for ovine studies, especially those aiming on metabolic and molecular processes after cerebral ischemia, have to be developed. This is crucial to augment the value of the model for stroke research and to bring the level of knowledge on these processes in sheep to a level that is common in other, well-established large animal models, in particular primates. Although sheep models of global cerebral ischemia are broadly used for pathophysiologic (Riddle et al, 2006) and methodical (Martinez-Coll et al, 2003) investigations, to our knowledge, this is the first description of an ovine model of focal cerebral ischemia.
Different investigation modalities were used to evaluate the consequences of MCAO in sheep, including behavioral testing, MR and PET imaging, and histopathologic methods.
In behavioral tests, long-lasting, severe motor functional consequences were only observed after total MCAO. Thus, total MCAO might be required to detect beneficial therapeutic effects on clinical outcome in sheep at later time points. However, even after total vessel occlusion, none of the 10 animals died whereas all subjects were able to join their flock on a meadow within 5 days upon MCAO. However, clear signs of cerebral infarction in brain imaging as well as histologic and cellular consequences could also be induced without permanent major clinical consequences by 1- or 2-branch occlusions. These occlusion modalities might be used to study the pathophysiology of stroke without the necessity of focusing on clinical outcome parameters. A possible limitation of the sheep model is the fact that the animals rapidly habituate to nonendangering nociceptive stimuli. This aggravates assessment of MCAO-induced sensory deficits. However, these deficits can be at least partially evaluated indirectly by observing ataxic movements and disturbed startle reflexes.
In clinical routine, MRI is currently the imaging method of choice in diagnosis (Chalela et al, 2007) and follow-up of ischemic stroke in humans and animals (Back et al, 2004). Size and weight of sheep provide an easy access to clinical MR scanners. Using not only morphologic, but also functional MRI tools, it is possible to verify successful induction of stroke by DWI (van Everdingen et al, 1998) at an early time point. Brain MR imaging of sheep indicated diffusion disturbances, which in their extent, localization, and time-dependency during a 42-day-timecourse are comparable to those observed in human patients (Forschler et al, 2007, who for comparison referred to Ritzl et al, 2004). Magnetic resonance imaging is, furthermore, able to depict infarct size and associated vessel occlusion type
As with the extent of the brain diffusion disturbances (as seen by MRI), it was possible to control the extent and severity of the CBF deficits by varying the number of occluded MCA branches. This was clarified by means of 15O-H2O PET imaging on day 1 after MCAO (Figure 2A). Follow-up brain perfusion PET on day 14 and 42 in a subset of five sheep after complete MCAO revealed a final (after 42 days) increase of the CBF deficit volume, whereas the relative severity of the CBF deficit in the stroke-related lesion returned to the baseline level on day 1. In the subacute stage after stroke (14 d), the CBF deficits showed a marked improvement of both defect extent and severity. The additional PET imaging of brain glucose consumption with 18F-FDG, a surrogate marker of brain viability, after 42 d revealed CMRGlu deficits which were 55% of the size and 84% of the severity of the corresponding CBF deficits. Our PET results are a good approximation of the known CBF time course and CMRGlu outcome after ischemic stroke in human patients (Huber et al, 1992). In particular, our finding of a temporal CBF improvement in the MCAO-related brain tissue might fit to the established concept of reversible postischemic hyperperfusion (Marchal et al, 1999).
Overall, a possible limitation of the sheep model at this point is that extensive anesthesia, performed for 8 h or longer is accompanied with reduced recovery rates in sheep thus possibly limiting the investigation of the development of the ischemic lesion in sheep beyond this time window after MCAO in case recovery is needed for subsequent investigations.
Recording of electroencephalograms or somato-sensory-evoked potentials was not performed in this study but was previously shown to be feasible in nonanesthetized sheep (Voss et al, 2006, Marcus et al, 1997), thus allowing electrophysiologic studies using the species to be performed as well.
Macroscopic
As sheep are not frequently used for medical research at the present time, there is a lack of species-specific antibodies. However, all antibodies used in this study (Supplementary Table 4) showed sufficient cross-reactivity and could, therefore, be used to evaluate cellular alteration in the adult sheep brain after ischemic stroke induced by MCAO. Although the usefulness of other antibodies, for example directed against oligodendroglia, still has to be elucidated, the immunolabeling shown is feasible for investigating at least basic processes after ischemic stroke in sheep with or without experimental therapeutic interventions. Double and triple fluorescence staining revealed typical cellular reaction patterns expected in the mammalian brain on ischemic stroke such as neuronal loss close to the lesion, astrogliosis, and microglial augmentation.
A disadvantage of the model is the longer generation time of sheep compared with laboratory rodents and the dependency on the reproductive season. The merino rams used in this study needed to mature for approximately 12 to 18 months before they were considered to be adult animals. This limits the flexibility of the sheep model. However, the animal species used in existing feline and canine models show similar growth times, whereas adulthood in most primates is reached even later. Another disadvantage is the necessity of a reliable, but rather complex transcranial surgical approach for MCAO because of the
It has been shown that the ovine stroke model provides major advantages, making it an attractive tool especially for long-term evaluation of novel therapeutic concepts. As such, this new large animal model could be appropriate for verifying new stroke therapies in translational medicine and could, for example, be of particular value for evaluation of the safety and efficacy of autologous cell therapies, which can hardly be evaluated in small species because of the limited number of obtainable cells per animal.
Overall, the sheep model of stroke, by overcoming some major limitations of other large animal models, has the potential to improve our understanding of stroke pathophysiology and to support the development of novel stroke therapies.
Disclosure/conflict of interest
No conflict of interest is evident with respect to this study.
Footnotes
Acknowledgements
The authors thank the MRI, PET and cyclotron crews at the Departments of Neuroradiology (Professor Kahn, Dr Lobsien) and Nuclear Medicine (Professor Sabri, Dr Grofimann) at the University of Leipzig. Furthermore, the authors especially thank the Departments of Veterinary Anatomy and Large Animal Surgery for support; Dr Grosche for confocal fluorescence micrographs and Mrs Ute Bauer for technical assistance. The authors are grateful to Mr Scott Krausen, a zoologist and artist who drew the illustrations in ![]() .
.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
