Abstract
The popularity of pericyte research is increasing, and this was not more evident than at the recent 2019 Brain meeting in Yokohama which featured a large number of presentations focused on brain pericyte research, including the Presidential Symposium. In this article, we will provide a history of brain pericyte research, present the results of our analysis showing a substantial increase in brain pericyte research presented at Brain meetings since 2005, suggest reasons for their increased popularity, and comment on what the future holds for brain pericyte research.
History of brain pericyte research
Eberth and Rouget are often credited as the first authors to describe pericytes. Their 19th century publications included detailed anatomical drawings of cellular and vascular structures and physiological observations in which they identified contractile elements on capillaries displaying pericyte-like morphology.1,2 The general history of pericyte research has been previously reported 3 but the initial observations of brain pericytes have not been extensively discussed.
In 1923, German anatomist K.W. Zimmerman released his seminal work ‘Der feinere Bau der Blutcapillaren’, a culmination of 40 years’ worth of histological observations, including some of the first published literature on brain pericytes. 4 Zimmerman mused on how to better describe Rouget cells, eventually naming them pericytes; referring to the tendency of pericytes to wrap around capillaries.
Zimmerman observed that brain tissue was ‘… more strongly impregnated with pericytes’ compared to other major organs, and noted that brain vasculature contained smooth muscle fibres and precapillary transition forms all the way down to the microvasculature. Interestingly, Zimmerman describes endothelial veiling Rouget cells on the smallest arteries, which appear to have similar morphology to the mural cells with circumferential processes that wrap around the branching vessels off penetrating arterioles. While there remains controversy as to the exact naming of these transitional mural cells, 5 these cells have been described as slightly more elongated than smooth muscle cells, enveloping the entire endothelium, and possessing protruding, ovoid cell bodies similar to those of pericytes. 6 Grubb S et al. 7 propose further characterisation of similar vascular structures as precapillary sphincters, important regulators of cerebral perfusion at the branch point off penetrating arterioles. Further down the vascular tree, Zimmerman describes cells seen in Figure 1(a) to (c) as capillary pericytes, which in recent times have been described as mesh, thin strand or helical pericytes, 6 and are similar to contemporary microscopy images of pericytes within the capillary bed (Figure 1(d) to (f)).
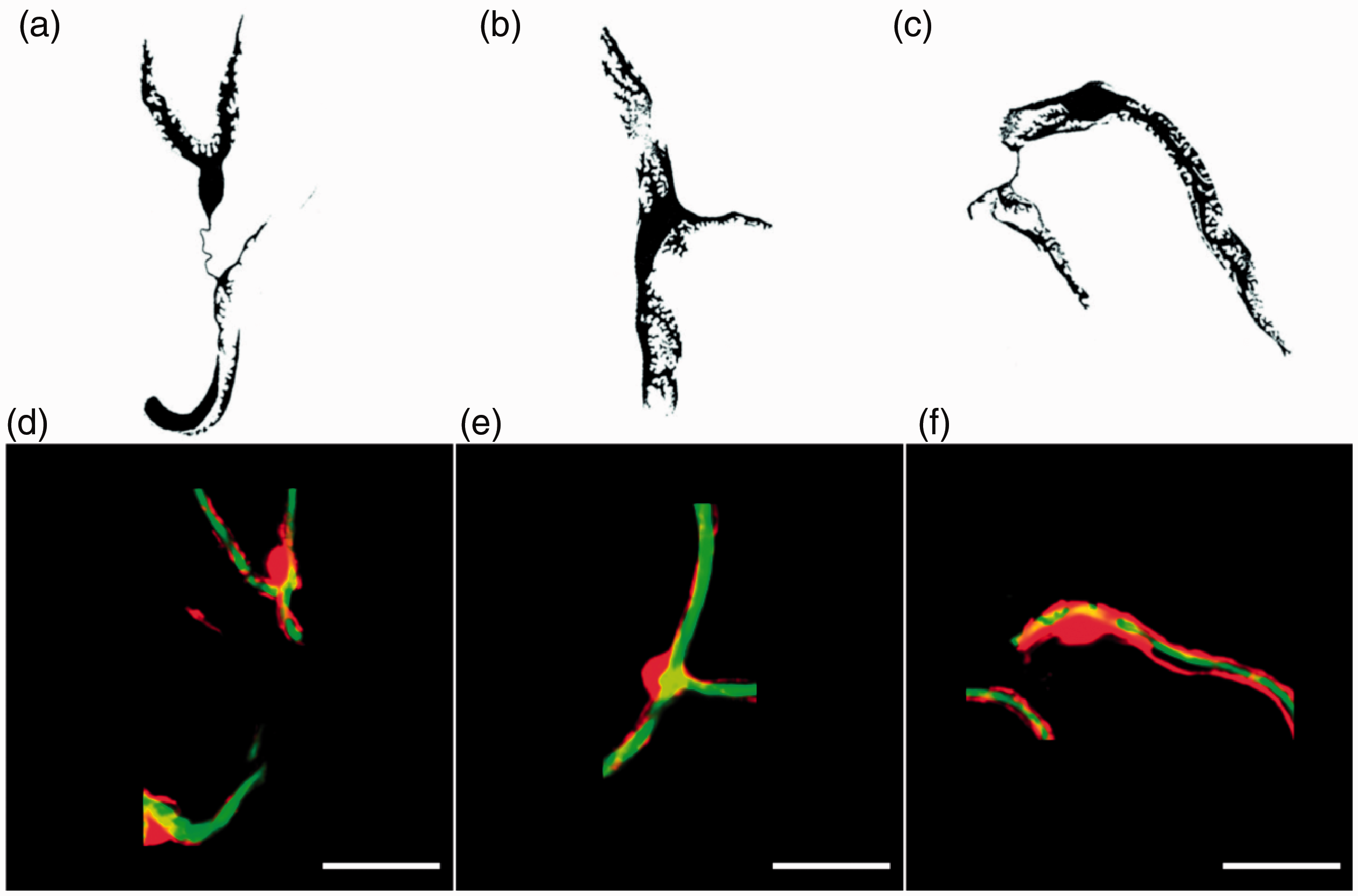
Comparison of Zimmerman’s sketches of brain pericytes with contemporary confocal fluorescent images of brain pericytes. (a–c) Zimmerman’s sketches of cerebellum capillary pericytes with broad primary processes from a young cat. These are some of the first known sketches of brain pericytes, reproduced with permission from Figures 180, 181 and 182 in his 1923 publication.4 (a) and (c) each show an intercapillary process or ‘pericyte bridge’. (d–f) Scanning confocal fluorescent images of cortical pericytes in NG2-DsRed mice (red) associated with FITC-albumin-positive (green) capillary lumens (Brown L.S.). Scale bar = 20 µm.
Zimmerman also observed intercapillary pericyte processes, also called pericyte bridges. Contemporary research has shown that in addition to forming connections with underlying endothelial cells, pericytes can span several endothelial cells, and occasionally connect with endothelial cells in neighbouring capillaries with fine processes that traverse the intercapillary space. 8 Interestingly, this physiology seems to be mainly restricted to retinal, 9 skeletal and cardiac muscle 10 pericytes, with this phenomenon only recently reported in brain, 11 and it is unclear to what extent they occur throughout the brain. The lack of these ‘bridges’ may reflect the collateral redundancy found in the cerebral vasculature. 12 Zimmerman remarked that the endothelial tube is ‘… poorly implanted in the brain and is difficult to generate usable sections for’, 4 which may explain the initial lack of follow up on pericyte characterisation in the brain, alongside the difficulty in obtaining specific staining.
The popularity of brain pericyte research is increasing
Interest in brain pericytes remained low over the next 50 years until advances in electron microscopy allowed researchers to better characterise the cellular makeup of the brain. In early 1970, King and Schwyn observed squirrel monkey brain sections via electron microscopy, 13 concluding that pericytes are truly perivascular and easily identified by the vascular basal lamina which encompasses the cell body and processes. Another early study in the cat brain demonstrated that pericytes could be released from the basement membrane of the capillary and transform into microglia, which required the assistance of astrocytic endfeet, 14 showing the dynamic roles of these cells. Interest in brain pericytes steadily increased during the 1970s and 80 s, with a key word analysis of the NCBI database revealing just under 500 publications that included brain and pericyte in their keywords or title between the years 1970 and 2000. 15 However, only 20% of those publications included brain, pericyte and disease. In the 20 years following, from 2001 to early 2020, pericyte research has exploded with almost 2000 publications including brain and pericyte, while almost 50% of those publications included brain, pericyte and disease. The increase in interest regarding pericytes in the brain during disease highlights our growing understanding of how pericyte and vascular dysfunction can underly and exacerbate many disease processes, including common neurological disorders such as stroke 16 and Alzheimer’s Disease. 17
This rise in interest in brain pericytes culminated in them being the topic of Professor David Atwell’s Presidential Symposium at the recent Brain 2019 meeting in Yokohama, Japan. During his talk, Professor Atwell presented recent results on the role of pericytes in neurovascular coupling and cerebral blood flow (CBF) deficits following cerebral ischemia 18 and Alzheimer’s disease (AD). 19 He concluded by imploring the audience of the importance of these cells that had been relatively neglected for over 100 years and to research them. While attending the Brain 2019 meeting, we noticed the large number of oral and poster presentations as well as a dedicated symposium specifically focusing on brain pericytes.
In order to confirm this increase in pericyte interest, we conducted an analysis of the number of abstracts presenting on brain pericytes in Brain meetings since 2005. All abstracts were freely available through meeting supplements published by the Journal of Cerebral Blood Flow and Metabolism. We included abstracts for all Brain symposia, oral and poster abstracts and unclassified late breaking abstracts. We excluded all Brain PET abstracts and educational course abstracts. We discovered a 10-fold increase in the number of abstracts presenting on brain pericytes between 2005 and 2019, increasing from 0.4% (n = 3) of all abstracts in 2005 to 4% (n = 29) of all abstracts in 2019 (Figure 2(a) and (b)). Interestingly, there was a noticeable jump in the number of abstracts presenting on brain pericytes at the Brain meeting held in Vancouver in 2015, increasing from 1% of all abstracts in 2013 to 3% of all abstracts in 2015 (Figure 2(a) and (b)). The distribution of different topic categories of pericyte abstracts is presented in Figure 2(c). This graph shows that earlier abstracts (2005, 2007) presented predominantly on the anatomy and signalling (e.g. calcium signalling) of pericytes, whereas from 2009 onward abstracts were predominantly presenting on the role of pericytes in ischemia, CBF regulation and BBB regulation. This rise in interest has led to some important paradigm-shifting discoveries about the critical roles of pericytes in brain function in both health and disease.
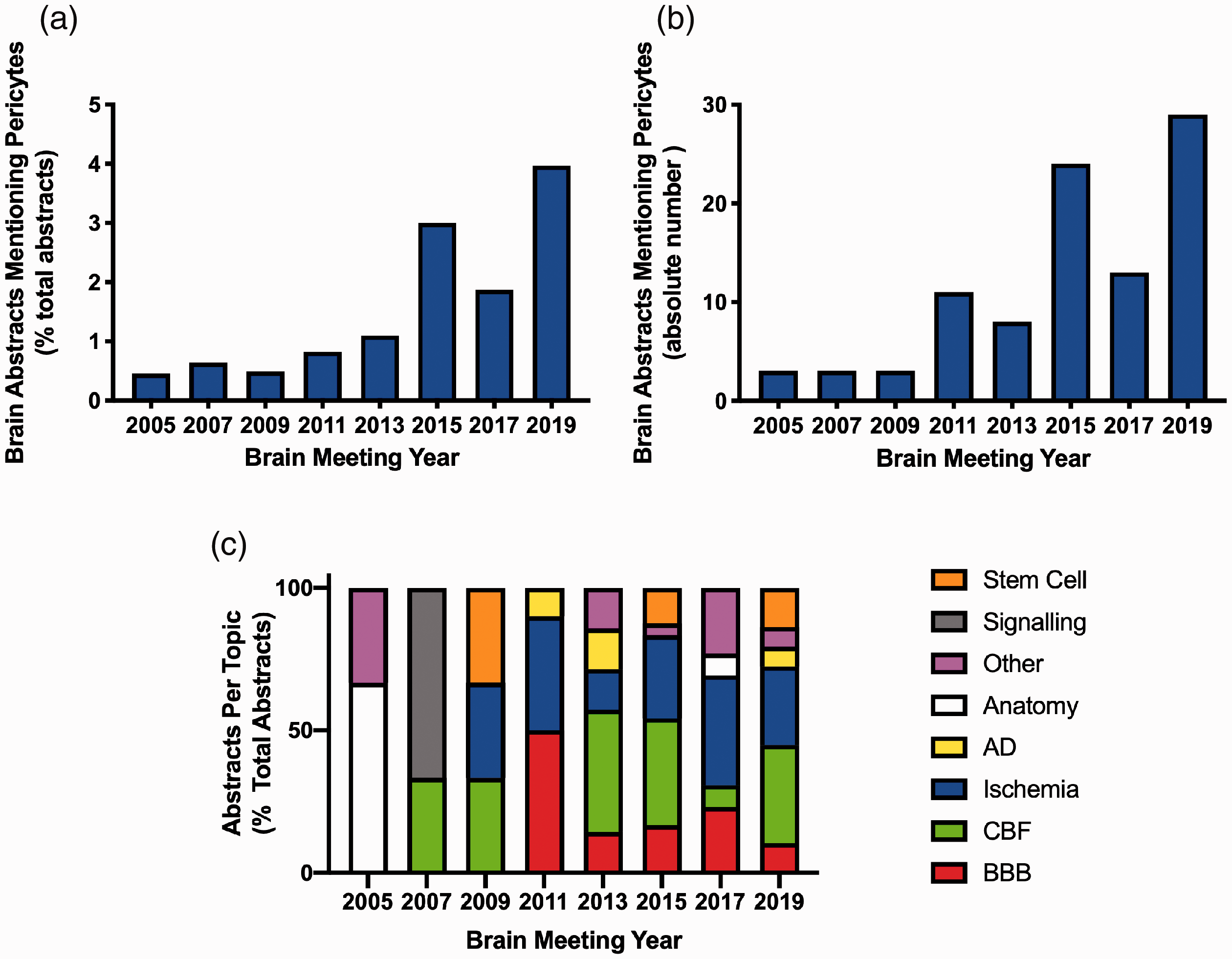
Pericyte abstracts presented at Brain meetings from 2005 to 2019. (a) Number of pericyte abstracts as a percentage of total abstracts. (b) Absolute number of pericyte abstracts. (c) Topic categories of pericytes abstracts as a percentage of total abstracts. AD: Alzheimer’s disease; CBF: cerebral blood flow regulation; BBB: blood–brain barrier; Other: diseases other than ischemia or AD, e.g. diabetes, Huntington’s disease, glioma, traumatic brain injury and multiple sclerosis. Some abstracts presented on a disease and another category, e.g. ischemia and CBF. In these cases, the disease took precedence and that was the category the abstract was assigned.
Functions of pericytes in the healthy and diseased brain
The rise in brain pericyte research could be attributed to the importance of these cells for numerous brain functions that have only become apparent recently, and our improved accuracy and reproducibility to investigate them through imaging and cell specific transgenic mouse technologies. Three of the major roles that pericytes play in the brain, amongst others, 15 are the regulation of CBF, the maintenance of the blood–brain barrier (BBB) and the regulation of immune function. In this section, we will briefly discuss some of the associated key publications that revealed these discoveries.
Regulation of cerebral blood flow in health
Early work by Peppiatt et al. 20 showed that electrical stimulation led to constrictions of capillaries at pericyte locations in the retina, while treatment with glutamate to simulate neuronal activation led to dilation of capillaries at pericyte locations. Hall et al. 18 replicated these findings in vivo by showing somatosensory stimulation evoked capillary dilations at pericyte locations, which preceded the dilation of the parent arteriole. This challenged the long-held understanding that dilation of the penetrating arteriole by VSMCs was the main stimulus for increased blood flow in response to neuronal activation and that capillary dilation occurs passively in response to increases in upstream flow.
Detailed investigation of the relationship between the arteriole dilation induced by VSMCs and capillary dilation by pericytes by Hill et al. 21 revealed that capillary diameter did not change at pericyte locations in response to somatosensory stimuli, and that arteriole dilation by VSMCs was responsible for increased blood flow. Pericytes, in this paper, were defined as alpha-smooth muscle actin (αSMA) negative cells. Any αSMA-positive cells with circumferential processes were defined as VSMCs despite their discontinuous appearance that is inconsistent with the classical notion of arteriolar VSMCs and more consistent with the morphology of a pericyte.5,22
Consensus appears to be emerging that regardless of nomenclature, the primary site of CBF regulation is at small vessels covered by spatially separate, αSMA-positive contractile mural cells. This is reflected in five recent publications. Firstly, Rungta et al. 23 systematically analyzed the response of the entire vascular tree from the pial arteries to capillaries, and identified the primary functional unit that dilated first, in response to odor, was the arteriole and its branching first-order capillary, with VSMCs and pericytes respectively controlling these responses. Kisler et al. 24 showed that pericyte deficient mice have reduced capillary CBF responses to evoked stimuli (neurovascular uncoupling) leading to reduced oxygen supply and metabolic stress. Nikolakopoulou et al. 25 showed that inducible pericyte knockdown in adult mice resulted in basal CBF deficiency and BBB disruption. Grubb et al. 7 report that sphincters at the transition between penetrating arteriole and first order capillary are encircled by contractile mural cells that could not be definitively defined as pericytes or VSMCs, and that these sphincters dilate in response to somatosensory stimuli. Finally, Nelson et al. 26 specifically induced the expression of channelrhodopsin-2 (ChR2) in pericytes in aged mice and showed that excitation of ChR2 with 488 nm light caused contraction of brain capillary pericytes and a subsequent 8% reduction in capillary diameter which led to a 42% reduction in red blood cell velocity. This study further demonstrates that pericytes are contractile cells that can regulate capillary diameter and CBF.
Regulation of cerebral blood flow in disease
Capillary diameters are small enough to require blood cell deformation during passage meaning the effects of dilation or constriction on capillary flow are not accurately estimated by Poiseuille’s law and actually exceed the r 4 effect of vessel radius on flow. 27 Therefore, pericyte constriction of capillaries may also be an important therapeutic target in neurological diseases where CBF is compromised. Yemisci et al. 28 reported pericyte constriction of capillaries at 6 h of reperfusion following 2-h middle cerebral artery occlusion (MCAo), but did not report pericyte cell death in vivo. Hall et al. 18 reported that pericytes die at 24 h after 90 min of experimental MCAo but did not investigate pericyte constriction in vivo. Interestingly, Hill et al. 21 provided some of the most convincing evidence of capillary constrictions at 4 h post MCAo; however, the authors attributed these constrictions to VSMCs despite the convention being to classify these cells as pericytes. 5 In addition to brain ischemia, Nortley et al. 19 reported capillaries being constricted specifically at pericyte locations, but not arterioles and venules in humans developing Alzheimer’s Disease (AD) and in a mouse model of AD. This suggests that the reduction of CBF known to occur in AD30 is produced by capillaries rather than by arterioles.
Regulation of blood–brain barrier in health
The BBB is located at the capillary endothelial cells of the brain, which creates a physical and chemical barrier to maintain tight control of the extracellular environment of the CNS. 30 Although astrocytes and the extracellular matrix have been known to contribute to barrier function for some time, 30 it has only been in the last 10 years that the role of pericytes to barrier function and dysfunction has begun to emerge.
Three papers published in 2010 first revealed the key role that pericytes play in BBB function and development. These studies cleverly used mutations or deletions to disrupt platelet-derived growth factor (PDGF)-B/PDGF receptor β (PDGFRβ) signalling in mice which resulted in pericyte loss and provides a useful tool to assess pericyte function. Armulik et al. 31 showed that Pdgfbret/ret mice, with only 26% pericyte coverage of the vasculature compared to control levels, have increased permeability to water and low molecular weight markers via increased endothelial cell transcytosis. They also showed that pericytes reduce barrier permeability via regulating endothelial cell gene expression and inducing polarization of astrocyte endfeet. Research by Bell et al. 33 extended these findings by demonstrating that a loss of pericytes in Pdgfrβ+/− mice led to an increased accumulation of serum and neurotoxic macromolecules, which preceded neuronal degenerative changes. Similarly, Daneman et al. 33 showed, using both null and hypomorph Pdgfrβ mice, that pericytes are critical for BBB formation, the coverage of capillaries by pericytes can determine vascular permeability, and pericytes can regulate tight junction formation between endothelial cells. More recent work by Berthiaume et al. 34 revealed that a sudden loss of a single pericyte by laser ablation could be compensated for by extension of processes from adjacent pericytes to contact uncovered regions. Interestingly, the uncovered regions of capillaries had normal barrier function but were dilated until adjacent pericyte processes covered the region. Taken together, these studies suggest that pericytes play a critical role in the development and maintenance of the BBB, they may be structurally plastic in vivo and able to compensate for an acute loss of pericyte coverage, while a chronic loss of pericytes leads to pathology.
Regulation of blood–brain barrier in disease
BBB disruption and release of toxic blood molecules into the brain as well as vasogenic edema contribute to neuronal injury and patient deterioration following stroke. 35 While previous work highlighted that pericytes maintain the barrier to protect the normal brain, Underly et al. 36 showed that photothrombotic occlusion of cerebral capillaries resulted in matrix metalloproteinase activation at pericyte somata that was followed by localized plasma leakage. These results suggest that pericytes are responsible for a rapid and localized proteolytic cleavage of the BBB and may be a potential therapeutic target to reduce BBB disruption following stroke.
In addition to stroke, AD is also characterized by an impaired BBB. Work by Sengillo et al. 37 showed that pericyte number in the cortex and hippocampus of AD patients is reduced by 59% and 60% respectively, and this was correlated with BBB disruption. Mechanistic studies carried out by the same group showed that overexpression of APP alone or PDGFR-β deficiency alone in mice was not enough to aggravate AD pathology. 38 However, the combination of APP over-expression and PDGFR-β deficiency had a synergistic effect on accelerating AD pathology. This pericyte loss led to reduced beta-amyloid (Aβ) clearance by pericytes and subsequent Aβ accumulation, which in turn amplifies the loss of pericytes leading to increased BBB damage and neuronal degeneration. 38 This suggests pericytes play an integral role at multiple sites of the AD cascade and may be an important therapeutic target for the treatment of AD.
Regulation of immune function
The location of pericytes at the interface between the brain and its vasculature places them in an ideal position to regulate immune trafficking and function within the brain. Brain pericytes in vitro were shown to be sensitive to tumor necrosis factor alpha (TNF-α), a pro-inflammatory cytokine, by stimulating the release of matrix metalloproteinase-9 (MMP-9) altering BBB permeability and therefore leukocyte trafficking. 39 This was supported by the finding that pericytes regulated the transmigration of neutrophils across the BBB via their expression of interleukin-8. 40 Further investigation showed that brain pericytes had a strong immune activation of chemokines, cytokines and cellular adhesion molecules in response to pro-inflammatory challenges such as TNFα, interleukin-1β, lipopolysaccharide 41 and transforming growth factor beta-1. 42 A more recent study reported in vivo evidence that brain pericytes acted as the initial sensors of systemic insults through secretion of the chemokine CCL2, which increased synaptic transmission of glutamatergic neurons. 43 Another postulated neuroinflammatory role for brain pericytes is phagocytosis. In the context of AD, pericytes are able to phagocytose and clear amyloid beta 40 (Aβ40) through a low-density lipoprotein receptor-related protein 1 (LRP-1)-dependent mechanism. 38 Likewise following ischemic stroke, brain pericytes acquired a microglial phenotype that assisted in the clearance of cellular debris in the lesioned area. 44 Therefore, pericytes have multiple roles in immune regulation in the brain which could be implicated in numerous neurological diseases.
The future of pericyte research
Attempts to modulate pericyte function in neurological diseases have already begun. Administration of adenosine packaged into nanoparticles as well as the blood–brain barrier impermeable S- tert-butyl-α-phenylnitrone can maintain capillary diameters after ischemia and reperfusion without leaving the vascular compartment, which strongly suggests a therapeutic effect on cells of the neurovascular unit (the anatomical and functional structure linking the brain parenchyma to the vasculature composed of the following cell types: neurons, astrocytes, endothelial cells, pericytes and vascular smooth muscle cells) 45 ; however, the role of pericytes in the effect was not specifically investigated.46,47 As well as repurposing existing drugs, the recent development of a commercially available system that allows real-time in vitro assessment of brain pericyte contractility has opened up new avenues for high-throughput screening for novel compounds that can slow down or prevent pericyte constriction to ischemia. 48 Novel compounds that promote pericyte relaxation or prevent pericyte contraction identified by in vitro screening can progress to proof of principle screening for in vivo activity in animal models of stroke. However, due to healthy pericytes in non-disease areas of the brain and body, as well as potential systemic off target effects of pharmaceutical compounds, specific targeting is imperative, for example by packaging of therapeutic compounds into brain pericyte-targeted antibody-coated nanoparticles. Although there are a number of fully characterised and biocompatible nanoparticle carriers (see Landowski et al. 49 for review), there is a lack of unique cell surface markers for pericytes meaning that directing these nanoparticle carriers specifically to brain pericytes is not currently possible.
Brain pericytes have previously been identified in vivo by the expression of platelet derived growth factor receptor beta (PDGFRβ), Desmin, NG2 or αSMA in combination with their perivascular location, discontinuous cellular processes and protruding nucleus.5,22 However, use of these markers in isolation as a means of specifically targeting therapeutics to brain pericytes may not be viable as these markers are also expressed by VSMCs, 50 perivascular fibroblasts 51 and oligodendrocyte precursor cells. 52 Recent advances in single cell RNA sequencing analysis combined with immunofluorescence and morphological analysis have identified abcc9 (Sulfonylurea Receptor 2) and kcnj8 (Potassium Inwardly Rectifying Channel Subfamily J Member 8) as specific brain capillary pericyte markers. 50 However, the expression of these markers in neurological disease states has not yet been investigated.
Pericyte targeting may be complicated by potential changes in the expression profile of pericyte markers in disease. Human brain pericytes, but not VSMCs, shed soluble PDGFRβ in culture media, which is increased by hypoxia. 53 Increased soluble PDGFRβ in the CSF is a biomarker of BBB disruption in humans with early cognitive dysfunction, indicative of pericyte injury.54,55 Identification of healthy and disease specific pericyte markers will provide a better understanding of the phenotype and function of pericytes in vivo as well as pave the way for the development of small chemical molecules, DNA, or antibodies, exploiting an overexpressed marker on the pericyte cellular membrane, that can be used for targeted delivery of therapeutics.
Conclusion
Our understanding of brain pericyte biology has expanded exponentially over the last 15 years. This research highlights that pericytes are multi-functional cells that are embedded in the neurovascular unit that can respond to signals from cells in the brain and the vasculature. Targeted modulation of pericyte function/dysfunction may open up novel therapeutic options for a number of neurological diseases.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: DJB and BAS were funded by the Medical Research Council UK (MR/M022757/1). DJB was funded by the National Health and Medical Research Council Australia (APP1182153). BAS was funded by the Rebecca L. Cooper Foundation and National Health and Medical Research Council Australia (APP1137776). LSB was in receipt of a University of Tasmania Scholarship.
Acknowledgements
We apologise to all of our colleagues whose work we were unable to cite due to space limitations.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
