Abstract
Background
Frostbite injury occurs when exposure to cold results in frozen tissue. To screen drugs and other field therapies that might improve the outcome for a frostbite victim, it would be helpful to have a reliable and cost-effective preclinical in vivo model.
Objective
We sought to create a novel mouse skin model of induced frostbite injury. This model would allow quantification of the surface area of involved skin, histology of the wound, rate of wound healing, and skin loss in a standardized fashion after the frostbite injury.
Methods
Thirty-six mice were studied. Standardized 2.9-cm diameter circles were tattooed on the mouse dorsum. Magnets frozen in dry ice (−78.5°C) were used to create a frostbite injury on skin within the circle, either as a continuous 5-minute freeze or as 3 repeated freeze (1-minute) and thaw (3-minute) cycles. Appearance, healing rate, skin surface area loss, and histology were recorded until the wounds were healed.
Results
The amount of skin surface area loss was approximately 50% for both freeze methods. Although the time to surface skin healing was similar for both freeze methods, the initial healing rate was significantly (
Conclusions
This novel in vivo mouse model for frostbite allows quantification of affected skin surface area, histology, healing rate, and skin loss and has the potential of being utilized to screen future treatment modalities.
Introduction
Frostbite occurs when exposure to cold external temperature results in freezing of tissue, sometimes leading to extensive tissue loss and disability. High altitude mountaineers, soldiers deployed to frigid environments, and homeless persons exposed to cold temperatures are examples of persons who have frostbite injury. 1 –3 Frostbite is most commonly observed in the fingers/hands, toes/feet, nose, cheeks, and ears because they are exposed, peripheral, and difficult to protect. Exposing tissue to freezing temperature causes vasoconstriction, decreased blood flow, and eventually, microvascular thrombosis. At the cellular level, intracellular ice crystal formation causes protein changes and results in membrane damage. 4 In situations associated with slower tissue cooling, extracellular water is crystallized, which dehydrates cells. Because of this effect, electrolyte concentrations within cells are altered, resulting in modified protein structures. 5 Cellular dehydration and microvascular occlusion cause progressive tissue ischemia, leading to necrosis, severe injury, and disability, including limb loss. 6 –8 Injury is likely compounded by reperfusion effects after the thaw. 9
Other than rapid rewarming, there has not been a dramatic advance in out-of-hospital clinical therapy for frostbite in nearly 50 years. 7 Current primary treatment modalities to minimize frostbite-induced tissue damage are amenable to improvement. One impediment to progress is lack of a preclinical (nonhuman animal) screening method. This situation might be improved by a convenient, reliable, and affordable preclinical method with which to screen new therapeutics. An animal model for screening that efficiently allows quantification of tissue histology, rate of wound healing, and tissue loss after a frostbite event might accelerate research efforts and encourage innovation for prevention and treatment. It could also be used to evaluate current methods to establish or refute efficacy. Finally, a preclinical model might provide a way to expand understanding of the pathophysiology, such as the presence of apoptosis or reactive oxygen species, of frostbite.
For these reasons, we set out to invent a novel mouse skin model of induced frostbite injury. This model would allow quantification of the surface area of involved skin, histology of the wound, rate of wound healing, and skin loss in a standardized fashion after the frostbite injury. Our experiments would also identify the possibilities for refinement and further expansion of data acquisition within our chosen model. Because there is thought to be an increased severity of injury in humans associated with a freeze-thaw-freeze (FTF) event, as compared with a single continuous freeze (CF) event, we included both situations in our experiments. Our goal, then, was to create a novel model of frostbite in the mouse.
Methods
Frostbite Model
Thirty-six 8- to 12-week-old C57Bl/6J male mice (Jackson Laboratories, Bar Harbor, ME) were used in a study approved by the Stanford Institutional Review Board and in accordance with Stanford University Institutional Animal Care and Use Committee Guidelines. The animals were housed 5 per cage before and after surgery in a temperature-controlled facility under a 12-hour light/dark cycle.
Skin Preparation
Twenty-four hours before day 0, mice were individually anesthetized using isoflurane (Baxter, Deerfield, IL). The dorsal skin surface from the base of the neck to the top of the rear haunches was shaved with an electric clipper, after which a depilatory cream (Nair, Church & Dwight, Princeton, NJ) was applied for 2 minutes to remove any remaining hair (Figure 1A). After depilation, the skin was cleansed with a 70% isopropyl alcohol swab. A circle of uniform diameter, 2.9 cm, was gently traced on the skin with a permanent parker using a silicone sheet template (Invitrogen, Grand Island, NY) as a guide (Figure 1B). The marked circle was then permanently darkened by using an animal tattoo machine (Animal Identification and Marking Systems, Hornell, NY [Figure 1C]).
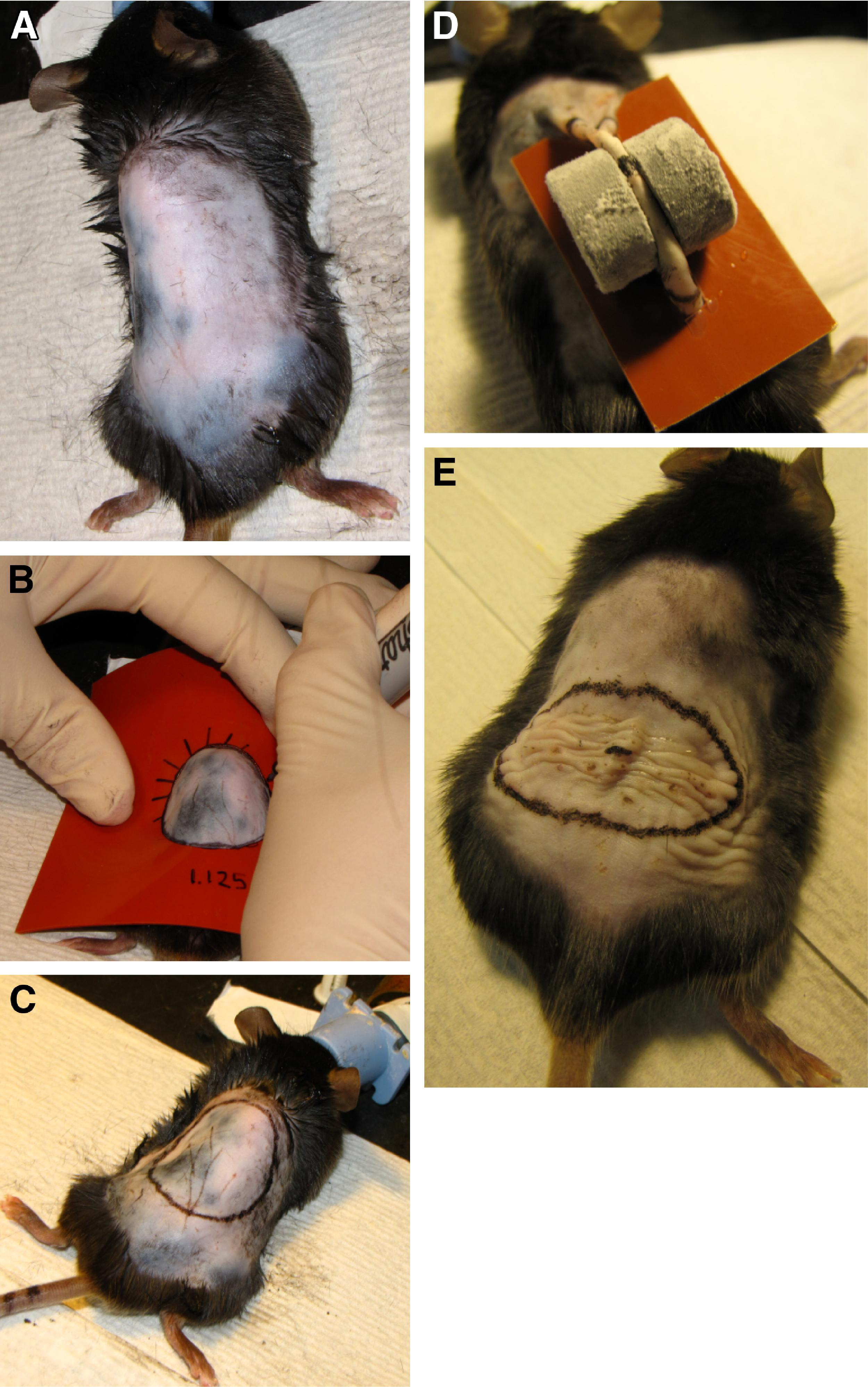
Animal preparation. (A) Hair covering the required dorsal area of the back is completely removed. (B) A circle of diameter 2.9 cm is drawn on the animal's back. (C) The circle is tattooed using indelible black ink. (D) Frozen magnets are placed on opposite sides of the skinfold within the tattooed mark. (E) Skin is shown immediately after thawing.
Continuous Freeze Method
Eighteen mice were studied using the CF method. Twenty-four hours after skin preparation, ceramic (ferrite) magnets (diameter 0.5 inches, thickness 0.219 inches, weight 3.5 g) were placed in crushed dry ice (−78.5°C) and allowed to cool for 15 minutes. The center of the tattooed circle was marked to determine precisely the placement of magnets. Using fingers, the back skin of the mouse was lifted into a skin fold, and then, 2 frozen magnets were placed so that they adhered from opposite sides of the intervening skin fold with the mark at the fold's apex center location. A silicone barrier was then slid underneath the magnets as a thermal shield to limit the mouse's body temperature decline (Figure 1D). Sets of 2 cooled magnets were left in place for 1 minute, then removed to allow new magnets to be immediately placed in the same location against the frozen tissue. The magnet exchange occurred in less than 5 seconds, which did not allow any thaw to occur. The magnet exchange was repeated for a total of 5 applications, resulting in a freeze time slightly longer than 5 minutes.
Injury was intentionally inflicted on a lifted skin fold rather than by applying topical pressure and freezing the tissue in its normal position directly underneath the magnet to simplify the method and avoid between-mouse variability, confine the injury to the skin, create a precise injury, limit systemic hypothermia, and maximize survival. It was not the intention to create a wound below the dermis.
Core temperature was monitored using an infrared thermometer (ThermoWorks, Alpine, UT) applied to the abdomen of the mouse. After 5 magnet placements and removals, the skin was allowed to completely thaw (Figure 1E). Mice were given subcutaneous injections of buprenorphine (0.05 mg/kg) for analgesia after the thaw. No dressings were applied to the wounds.
Freeze-Thaw-Freeze Method
Eighteen mice were studied using the FTF method. Twenty-four hours after skin preparation, as for the CF method, magnets were cooled and placed for 1 minute. However, after 1 minute of application, the magnets were removed, and the skin was allowed to thaw for 3 minutes, achieving a complete thaw with pliable tissue. After the 3-minute thaw, cold magnets were reapplied in the same location. That was done to accomplish 2 additional cycles of freezing (refreezing) for 1 minute and thawing for 3 minutes, for a total of 3 full freeze-thaw cycles. Mice were given buprenorphine (0.05 mg/kg) for analgesia after the final thaw. No dressings were applied to the wounds.
Room Temperature Magnet Placement, Control
To test for possible wound creation based on magnet pressure alone, room temperature magnets were opposed on opposite sides of a dorsal skin fold in a control mouse and removed and replaced each minute for 5 minutes to mimic the CF method conditions, and in a separate experiment in another control mouse for 3 consecutive cycles of 1 minute on, with 3 minutes off, to mimic the FTF method conditions. Skin condition was noted 24 hours and 48 hours after placement.
Wound Surface Analysis and Tissue Loss Quantification
No chemical depilation was performed during the healing period. It was occasionally necessary to gently shave hair with an electric clipper outside the border of the wound to completely visualize the wound and tattoo ring. Digital photographs of the wounds were taken after the thaw on days 1, 4, and 5, and on every other day thereafter until the wounds were completely healed by visual inspection, as previously described. Time to complete healing was defined as the time measured from the initial freeze until the wound bed was completely filled in with new tissue. The silicone sheet with the 2.9-cm diameter hole was centered on the wound before photography to use as a constant area for standardizing wound size. Wound surface area was determined by tracing the wound margin displayed by photograph with a fine-resolution computer mouse. The number of pixels corresponding to a surface area measurement was calculated using ImageJ software (National Institutes of Health, Bethesda, MD). Wound area was reported as percent area of the original maximum frostbite injury ([original wound area minus wound area on day “X”] divided by original wound area). The mice were anesthetized and relaxed during all measurements to mitigate any curvature in their backs and to recreate the same anatomy for each measurement. Tissue loss and contraction was reported as area of the original maximum skin area inside the tattooed circle ([original surface area within the tattooed boundary minus surface area within the tattooed boundary on day “X”] divided by original surface area within the tattooed boundary).
Histopathology Examination
The animals were humanely killed, and the entirety of the frostbite wounds were harvested from 2 mice in each freezing method group on each of days 0, 4, 5, 7, 10, 15, and day 21. The 4 remaining mice in each freezing method group were monitored until day 27 for confirmation that tissue loss had come to an end, which was the case. The tissue samples were fixed overnight in 4% paraformaldehyde and then embedded in paraffin; then, 7-μm sections were cut and stained with hematoxylin-eosin. Analysis was performed by a board-certified dermatopathologist. Tissue necrosis, reepithelialization, healing rate, acute inflammation, chronic inflammation, fibroblast maturation, collagen deposition, neovascularization, and the overall healing score were analyzed based on a modified version of a scoring system suggested by Abramov et al. 10 All measurements were recorded on a scale of 0 to 3, except for the overall healing score. A lower score correlated with less epithelialization, inflammation, and so forth. The overall healing score was based on a scale of 1 to 10: a score of 1 correlated with the most acute wound; a score of 10 correlated with a fully mature wound with scar formation. Examples of hematoxylin-eosin–stained slides of CF method and FTF method skin samples are shown in Figure 2A and B.
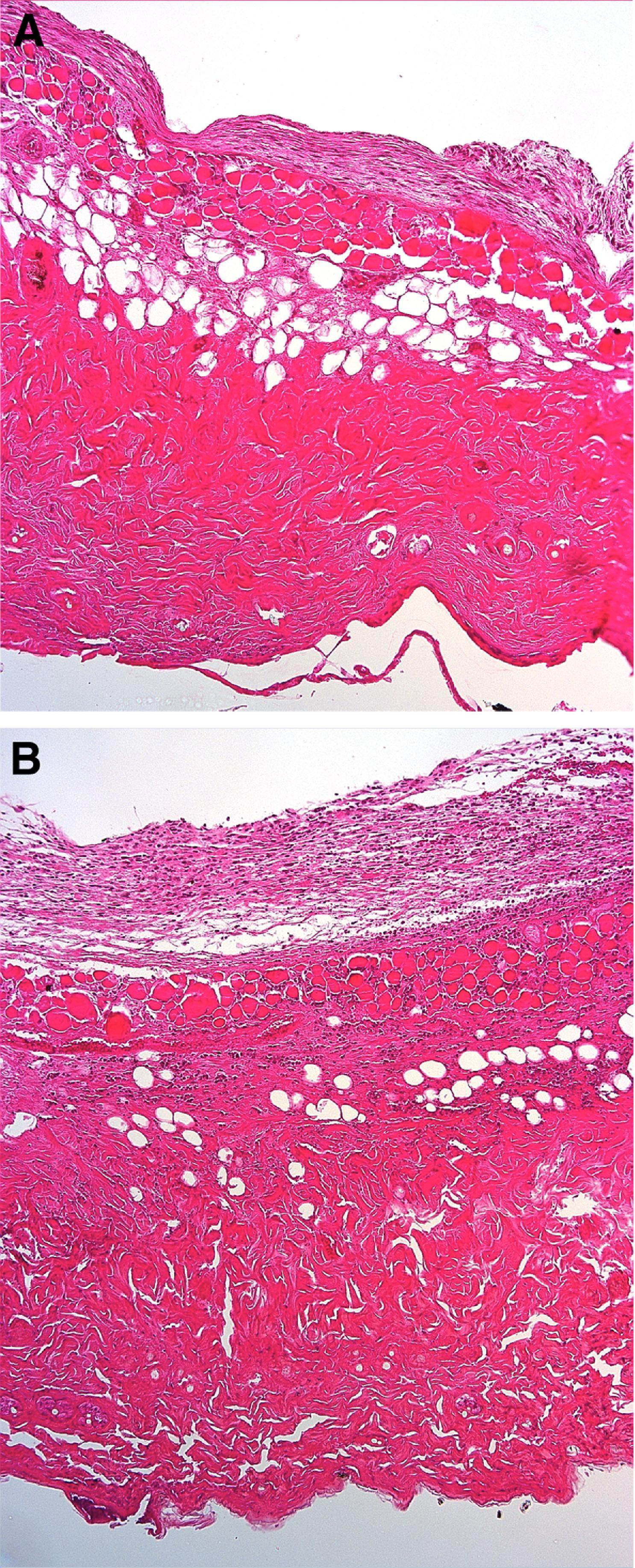
Hematoxylin-eosin–stained skin samples taken from mice 4 days after frostbite, when wounds from both freezing methods were deemed most severe. Magnification ×10. (A) The 5-minute continuous freeze (CF) method: sections show epidermal necrosis with resultant complete loss of the epidermis; there is minimal inflammation throughout the underlying dermis, and there is early fat necrosis with focal lipocyte degeneration. (B) The freeze-thaw-freeze (FTF) method: sections show epidermal necrosis with resultant complete loss of the epidermis; there is mild chronic inflammation in the deep dermis and subcutaneous fat.
Immunofluorescence Examination
Using paraffin-embedded skin samples from days 0, 4, 5, 7, 10, 15, and 21, apoptosis (caspase-3 stain [Abcam, Cambridge, MA]), reactive oxygen species (dihydroethidium [DHE] stain [Invitrogen, Carlsbad, CA]), and DNA fragmentation (terminal deoxynucleotidyl transferase dUTP nick end labeling [TUNEL] stain [ApopTag; Millipore, Billerica, MA]) were assessed to determine feasibility of the method for future investigation of applied therapeutics. All measurements were determined in a nonblinded fashion, with the entirety of the wound examined.
Caspase-3 Stain
Sections were stained with caspase-3. Sections were deparaffinized, then quenched with Red-DEVD-FMK and washed. Stained cells were observed using a fluorescence microscope. Cells that stained bright red, indicating apoptosis, were counted.
Dihydroethidium Stain
Reactive oxygen species were stained using dihydroethidium and mounted with Vectashield mounting medium with DAPI (Vector Labs, Burlingame, CA). Cells containing reactive oxygen species were counted utilizing fluorescence microscopy.
Tunel Stain
Apoptosis and DNA fragmentation were identified using ApopTag peroxidase in situ apoptosis detection kit. Cells were counterstained using 0.5% methyl green. Apoptotic cells were counted using a fluorescent microscope.
Statistical Analysis
Surface area of the wound, tattooed region, and aforementioned silicone stencil were measured in pixels and presented as mean ± SD. The variables between each separate measurement were compared using the unpaired Student's
Results
Room Temperature Magnet Placement, Control
Control mice demonstrated no wounds, skin lesions, irritation, swelling, discoloration, or bruising 24 hours and 48 hours after magnet removal (Figure 3).
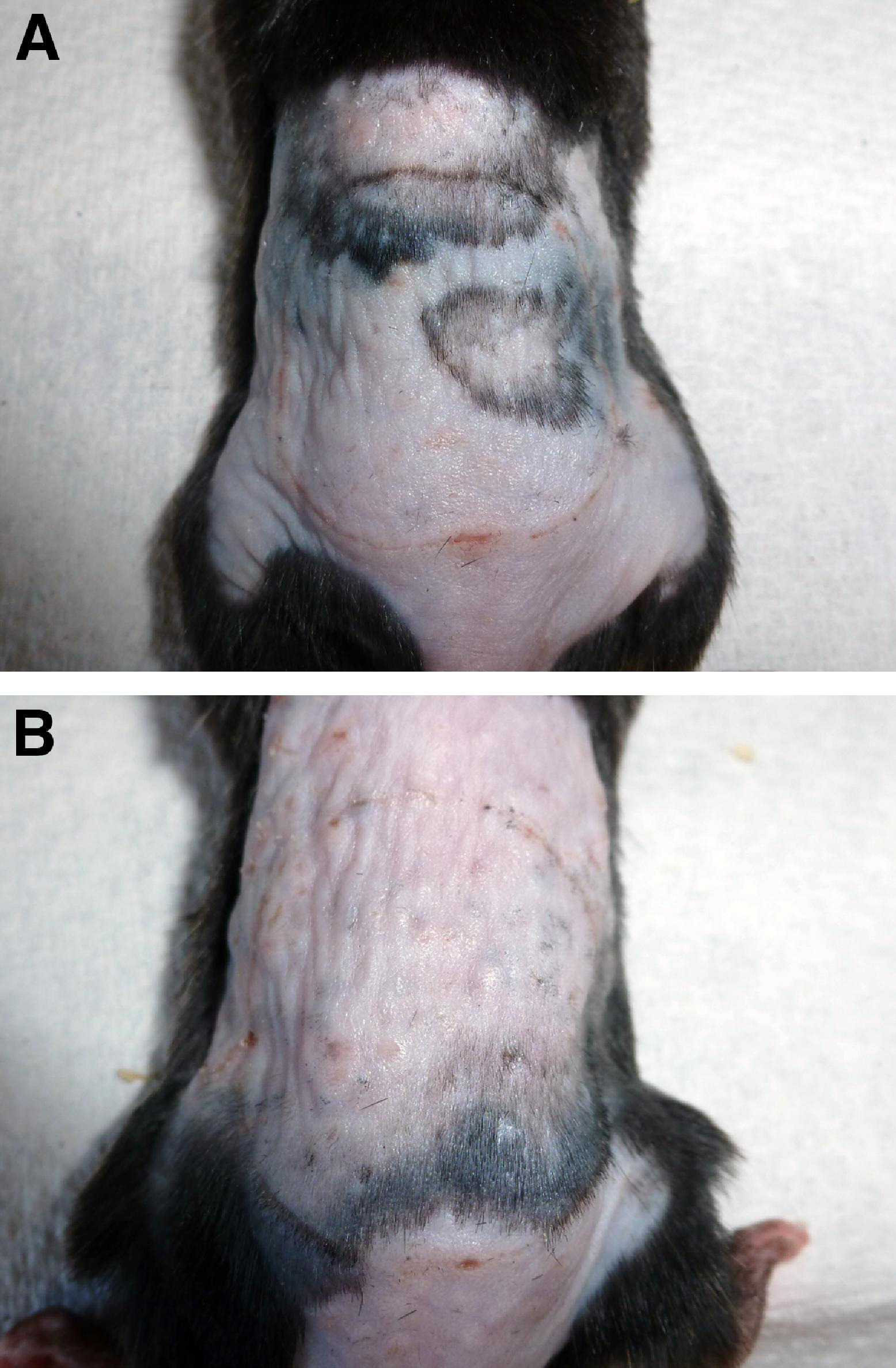
Skin condition 24 hours after control magnet removal. (A) Skin after magnet placement for 5 minutes with intermittent magnet change every minute, mimicking 5-minute continuous freeze conditions. (B) Skin after magnet placement for 1 minute, followed by 3 minutes of rest. Procedure repeated for 3 magnet placements and removals, mimicking freeze-thaw-freeze conditions.
Mouse Body Temperature
Mouse body temperature never fell below 8°C of its initial measured value (Figure 4). During the rewarming period post freeze, the mice shivered for approximately 3 minutes. They fully recovered after this procedure. No mouse died.
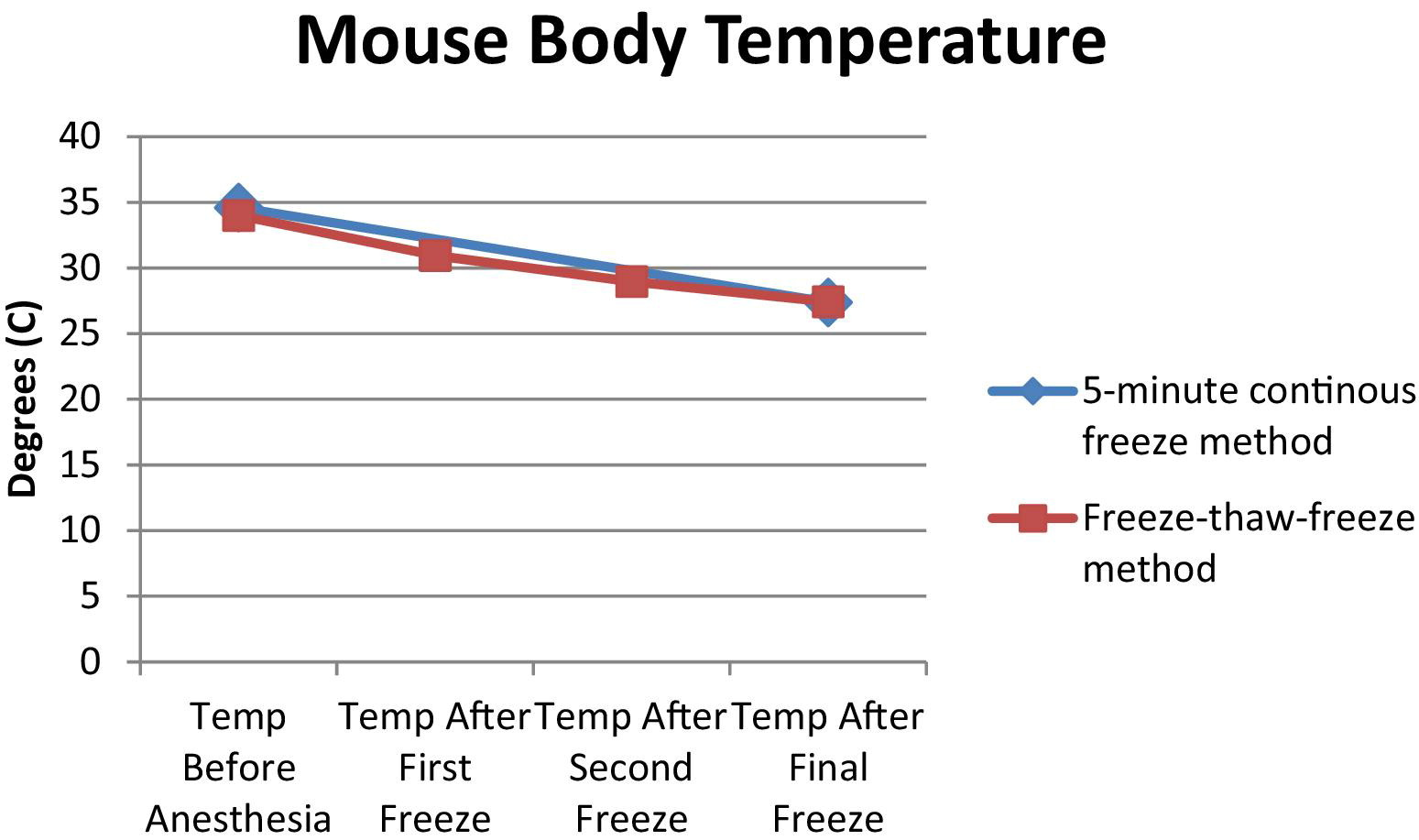
Core body temperature of the mouse at selected time points during the freezing process. Only 2 temperature measurements are taken for the 5-minute continuous freeze (CF) method owing to the single freeze. For the freeze-thaw-freeze (FTF) method, temperature is taken after each magnet removal stage as well as before the initial magnet placement, resulting in more temperature readings. The first and last temperature taken for the FTF method coincides with the temperatures taken for the CF method, which is the reason for their overlap.
Wound Closure and Analysis
Rather than 2 separate freeze wounds, the placement of 2 frozen magnets on opposite sides of the dorsal fold resulted in a single frostbite injury spanning both magnet diameters and the intervening tissue. The skin under and between the magnets, as well as a small perimeter portion beyond the area underlying the magnets, was affected by the cold. This single frostbite injury was subsequently monitored.
Figure 5 shows the progression of wound healing typical for the 2 freezing methods. Wounds for both the CF method and the FTF method experienced no further tissue loss by day 23. Wounds were followed up for another 4 days to visually observe any further healing. That proved to be minimal and was deemed not significant for the purpose of developing this model.
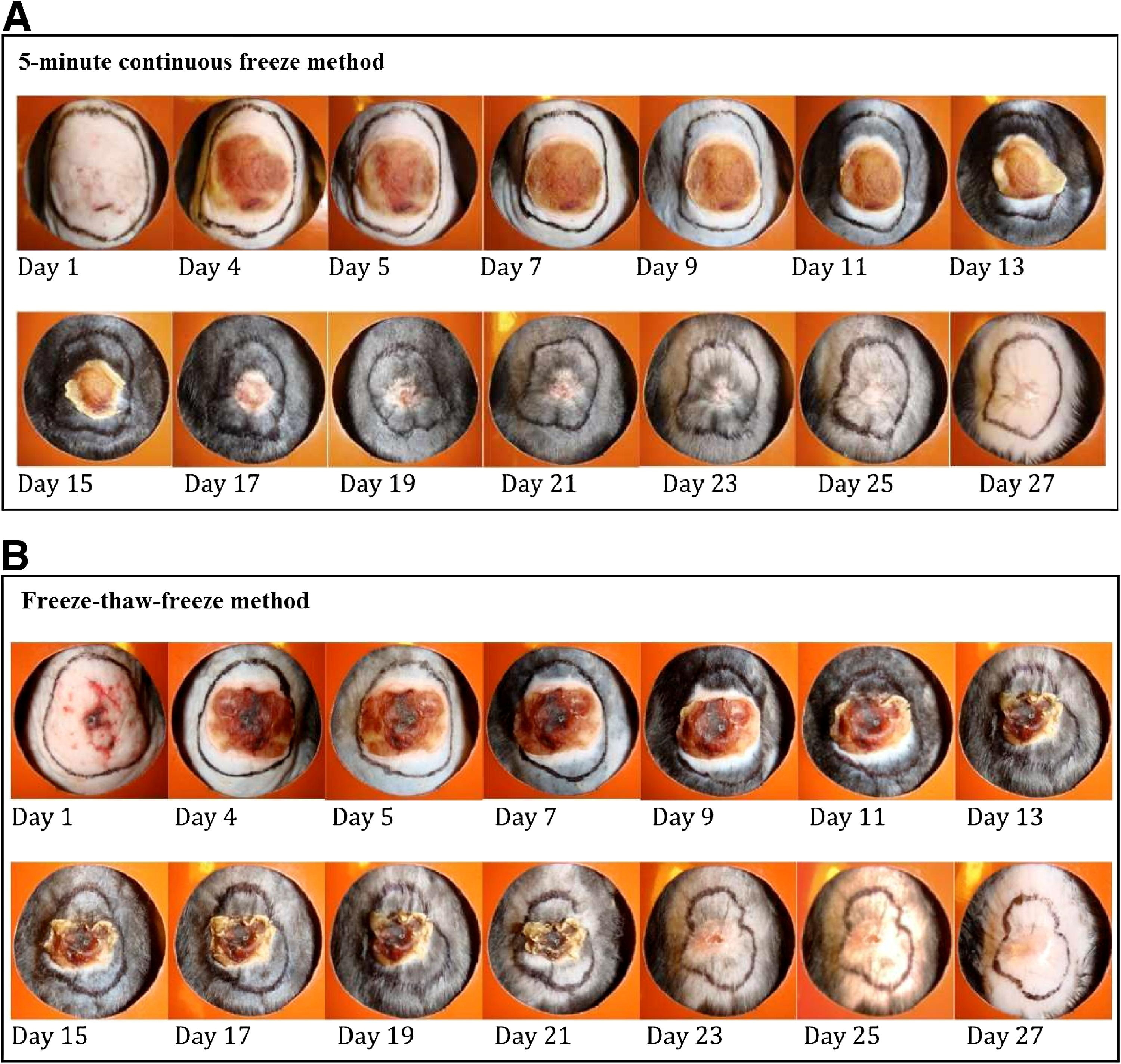
Gross frostbite wound progression after the freeze. (A) Progression of the wound for the 5-minute continuous freeze method in a single mouse. (B). Progression of the wound for the freeze-thaw-freeze method in a single mouse.
Figure 6 shows wound healing rate, as calculated on days that wound area measurements were taken, with trend lines. The numerator was the incremental percent of the wound healed since the previous measurement, and the denominator was the number of incremental days passed. The CF method wounds healed at a more rapid rate than did the FTF method wounds, until day 15, at which point reepithelialization began to slow. Mice with the FTF method wounds healed at a slower rate until day 11, and then began to heal at an increased rate compared with the CF method. The reasons for acceleration of healing rate at day 11 in the FTF method wounds are not definite, but may relate to a more severe initial acute injury. Figure 7 shows the percentage of the wound that was determined to be healed by photo surface analysis. For the mice that underwent the CF method, wounds were approximately 50% healed on day 12, whereas the mice that underwent the FTF method were not approximately 50% healed until day 18. These “50% healed” days are designated on graph analysis, for it is on these days where the lines from each freeze method pass through the “50% healed” threshold on the graph representing wound healing.
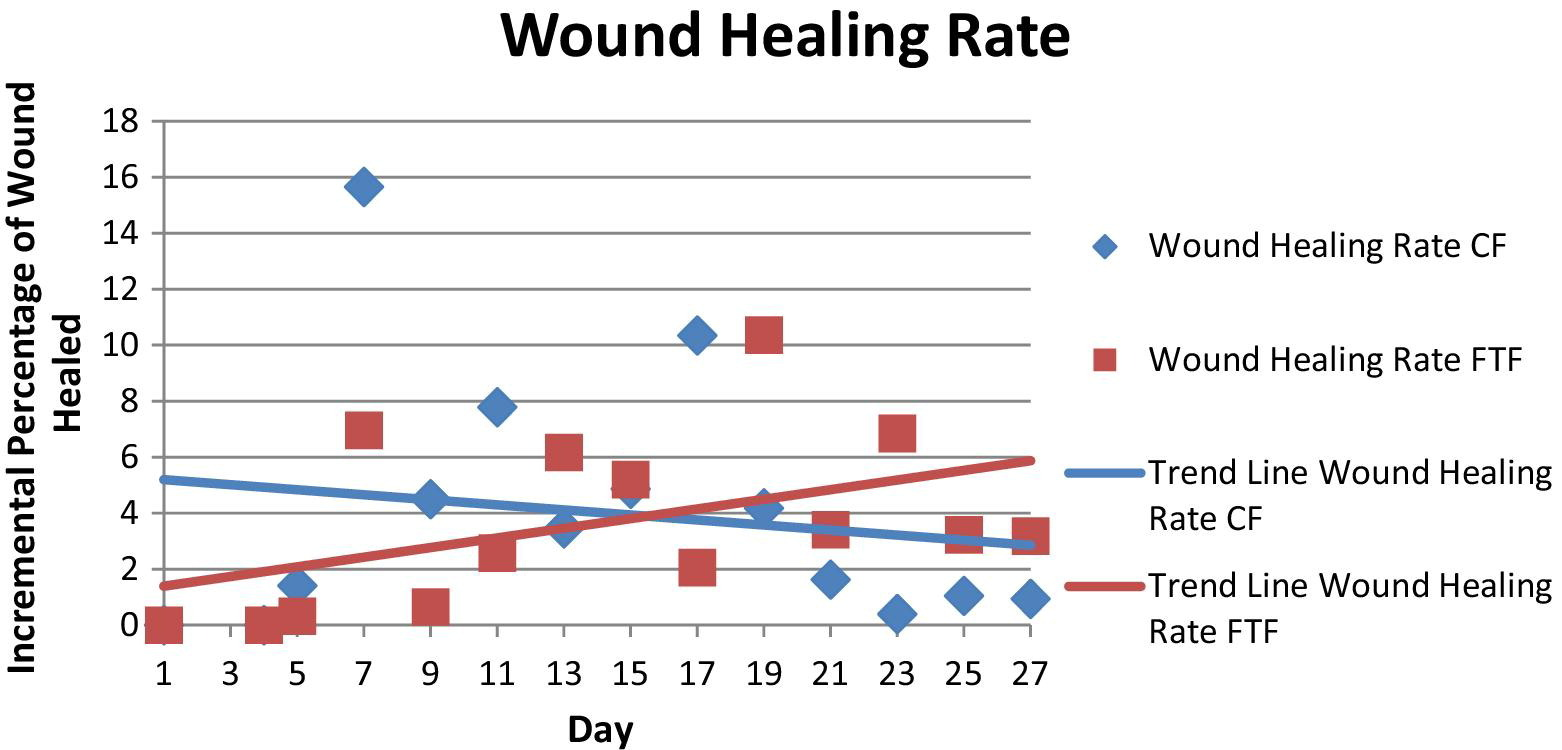
Healing rates of the 5-minute continuous freeze (CF) method and the freeze-thaw-freeze (FTF) method, including trend lines. Initially, healing rate is faster in the CF method wounds, but by day 15, this rate has decreased and the rate of healing in FTF method wounds has increased.
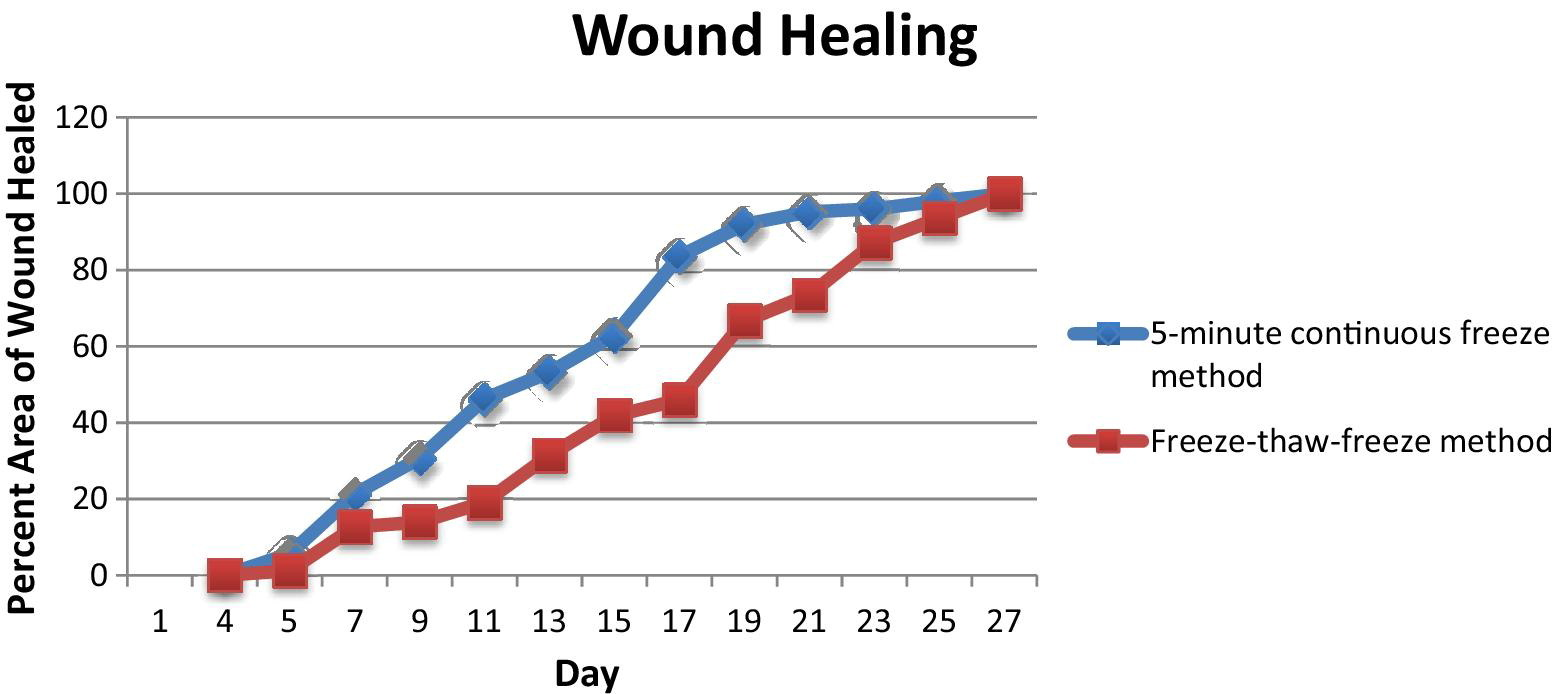
Wound healing chart showing the percentage of the healed wound by day. The continuous freeze method wounds initially heal at a faster rate; wounds of both freeze methods are completely reepithelialized by day 27.
Tissue Loss
The amount of surface tissue loss (approximately 50%, as estimated by surface area measurements) measured after the wounds were completely reepithelialized was nearly the same for the CF and FTF methods (Figure 8). It is likely that wound contraction played a role in this observed phenomenon, but for purposes of this study, the total decrease in surface area was used as a surrogate for tissue loss.
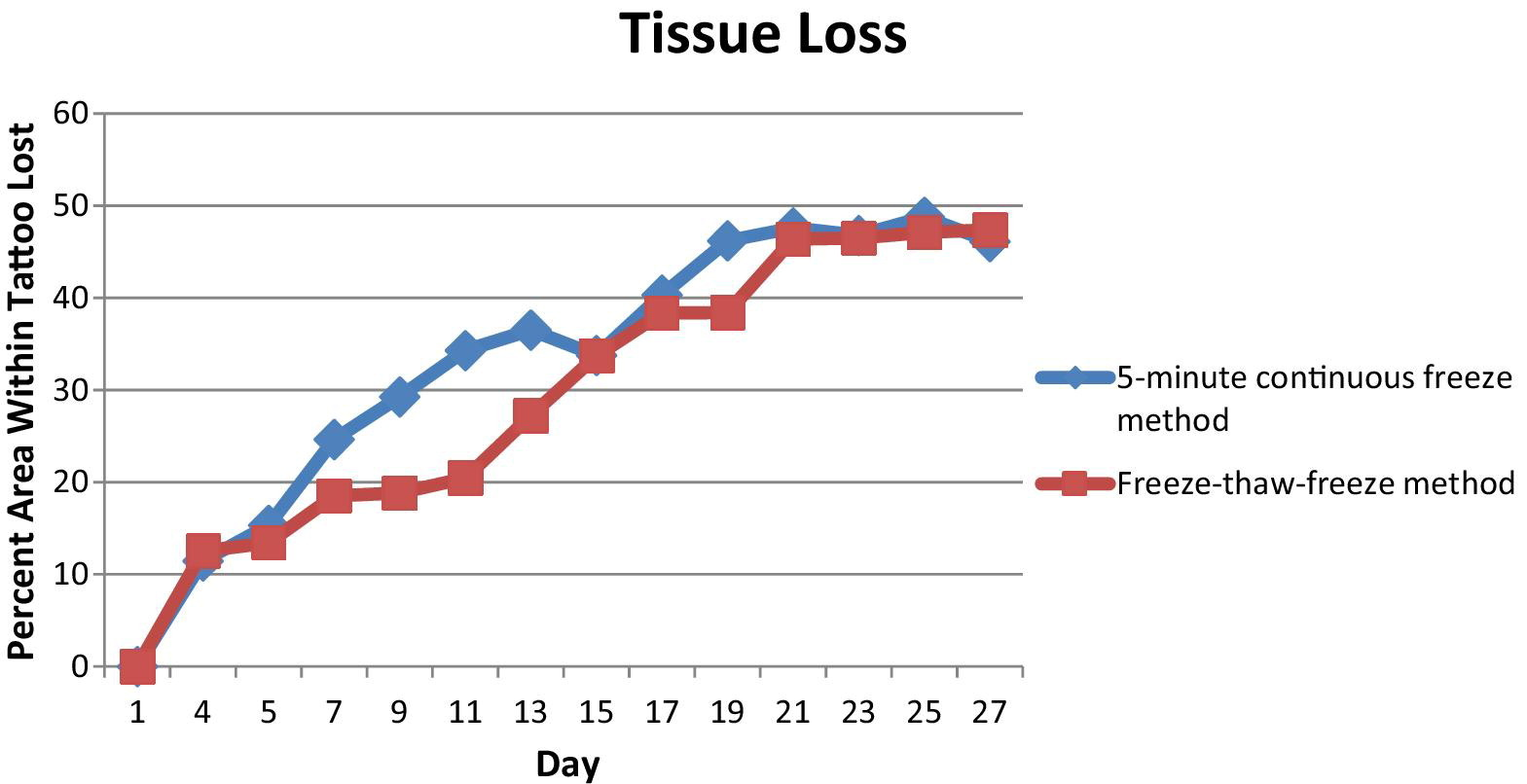
Percentage of lost surface area of tissue within the tattooed ring as compared with the original surface area. Both freeze methods showed the same percentage of surface area lost after they were completely healed.
Frostbite Histology
The main differences between the 2 frostbite methods appear to be early in the healing process (Figure 9). The mice under the FTF method initially have more extensive necrosis, extending through the subcutaneous fat in areas. The FTF method causes complete necrosis of the dermal collagen, whereas the CF method causes incomplete dermal collagen necrosis. There is a higher level of acute and chronic inflammation with the FTF method. Although the intent was to achieve a complete freeze of the skin and not to extend into muscle, necrosis was noted to extend into subcutaneous fat and, in one case, into muscle.
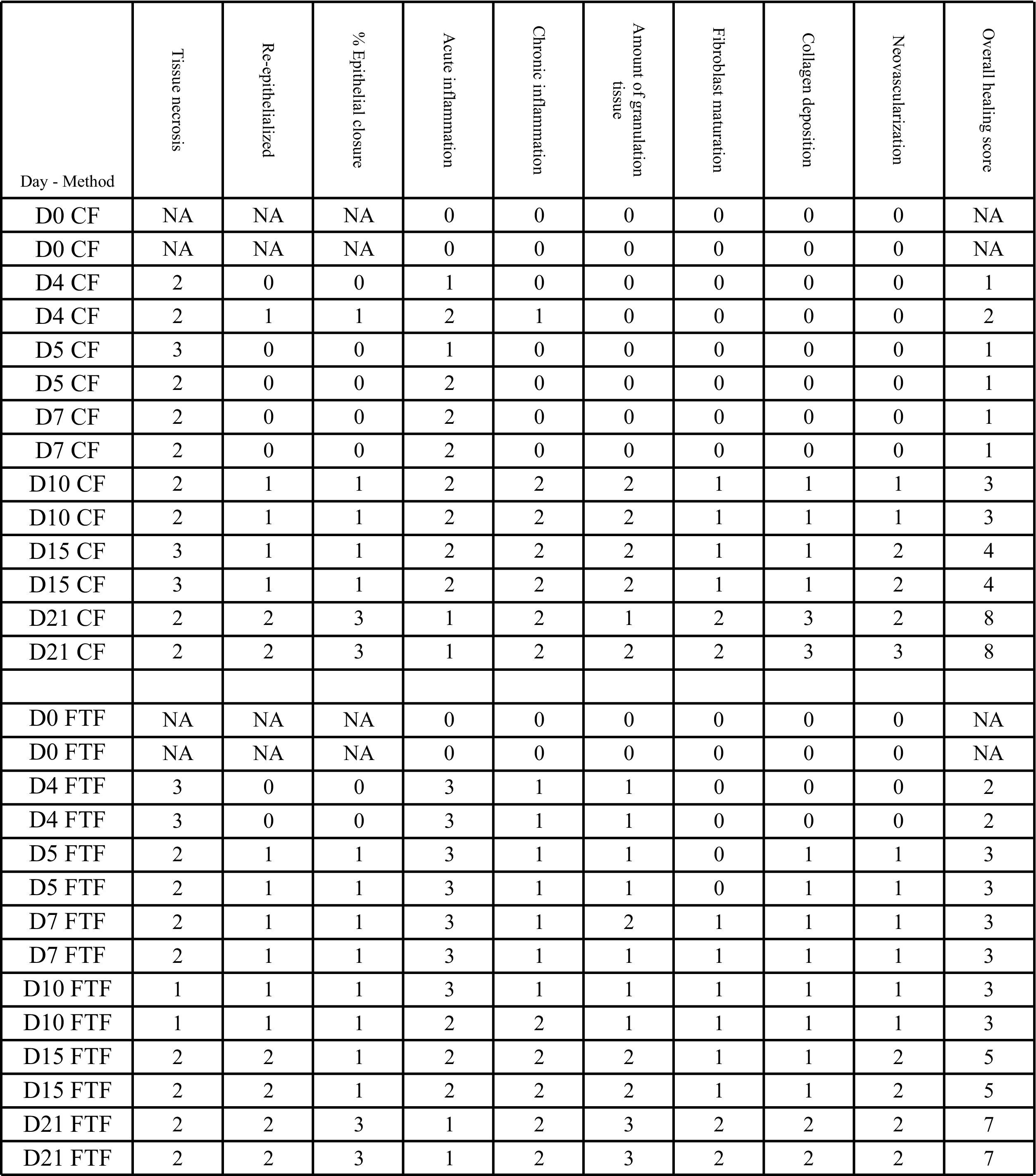
Histology scores (
The mice took 23 days to achieve their maximum tissue loss by visual inspection of the frostbitten sites. The amount of necrosis was substantial and might account for this finding. In a surgically wounded mouse, the wound bed begins to form immediately. In our model, the necrotic dermal tissue needed to be cleared before keratinocytes migrated over the wound bed. Additional epithelialization occurred during the final days of observation.
Immunofluorescence Examination
The DHE, caspace-3, and TUNEL stains did not indicate the presence of reactive oxygen species or apoptosis.
Discussion
One goal of a small-animal frostbite model is to replicate human pathophysiology. Another is to allow the model to be useful for evaluation of new pharmacologic and other approaches to human frostbite treatment and prevention. Ideally, the animal model used for screening would be reliable and affordable in such degrees to allow quantification of the rate of wound healing or amount of corpus tissue lost as a result of freezing tissue. Previous animal models for frostbite include hairless mouse ears,8,11 depilated mouse ears,12,13 rabbit ears, 6 ,9,14–17 dog paws,18,19 rabbit feet, 20 rabbit legs, 21 golden hamster cheek pouch,22,23 rabbit mesenteries, 24 mouse tails, 25 mouse leg muscle, 26 and rat hind limbs. 27 This article describes a novel model of frostbite wounding utilizing ceramic magnets to freeze dorsal dermal tissue, which observes a normal progression of wound healing through granulation and reepithelialization. Our model allows aspects such as core temperature, temperature of skin surface, freeze size and duration, number of freezes, and thaw duration to be controlled, and healing rate and extent to be reasonably quantified.
Previous models are fairly accurate in predicting the amount of tissue loss due to frostbite,28,29 yet are more complicated than ours and not widely used because of overlapping blood vessels and the dangers of utilizing radioactive isotopes. Hamlet et al 20 found that thermographic patterns helped predict the line of demarcation and sloughing, but there is variation among surface heat-loss patterns that confounds interpretation.
Our results show that tissue necrosis attributable to frostbite can be easily induced by application of frozen magnets to a sizeable skin fold on the back of a mouse. Demarcation of macroscopic frostbite injury with thick eschar becomes apparent at days 1 to 4. Figure 9 demonstrates the minimal degree of detail afforded by our method. Visual inspection and histopathology can be readily accomplished and would be expected to objectively show the effect of an applied therapy. Although the results delineated in Figure 9 show comparability between the CF and FTF methods, we showed that multiple cycles of freezing and thawing likely results in an initial greater degree of injury, as represented by the slower initial healing rate. This correlates with clinical reports in humans, in which it has long been noted that refreezing a recently thawed frostbite injury seems to create a markedly worse injury. The cause of this phenomenon is not known, but is likely related to direct physical injury, as well as perhaps to deleterious effects of reperfusion after ischemia. Although it may be simpler to use the CF method, if a proposed therapy possibly diminishes reperfusion injury, it may be prudent to screen it against both the CF method and the FTF method.
The time to complete healing proved to be longer than the previously reported 7 to 9 days in surgically wounded mice. 30 In both methods, the skin began to form a scab as soon as it became necrotic. The time for the scabs to fall off was different between the 2 freezing conditions. Scabs were shed on day 16 for mice exposed to the CF method, whereas they were shed on day 18 for those exposed to the FTF method. That likely contributes to estimation of increased healing rate immediately after those days, as the residual wounds beneath the scabs were significantly smaller than the area of the sloughed scabs.
Study Limitations
We note certain limitations. Our purpose was to keep our model as simple as possible while still obtaining a frostbite injury, not to explore deep tissue or comprehensive limb injury. We selected a well-vascularized area on the back of the mouse that is easy to access and would be predicted to show quantifiable metrics of wound healing. However, mice are not humans, and we did not expect to find absolute comparability in clinical or pathologic presentation. There was not a blistering phase in our model, which has been seen in rabbit ear and leg models.31,32 Absence of blistering is different than what is commonly seen in humans. We do not believe that absence of blistering or proven correlation to human frostbite classifications (eg, “degrees” of depth) negates the potential utility of our model when it is used as a screening method. Because our model was designed to freeze only the skin, we did not estimate tissue effects at depth or total volume of tissue loss represented by subsurface phenomena. Although we scored the degree of neovascularization (angiogenesis), we did not assess the microcirculation in the vascular bed using electron microscopy or other advanced techniques.
For all these reasons, it may be necessary to advance drugs and techniques that are successful at lessening frostbite in our screening model to another animal model, such as rabbit limb, and perform more advanced analysis of subcellular activities, before undertaking human investigation. We observed that the amount of tissue loss was the same for both freezing methods, although the initial degree of injury attributed to the FTF method is worse, and may be due to the anatomy of mouse skin and the process of wound contraction. That might represent a limitation of our model, because it is different than what is typically reported in human frostbite, where tissue loss is thought to be worse when tissue is refrozen. We euthanized 2 animals on each of the days selected for histology evaluation. It is possible that if a larger number of animals were studied, our observations might have been different. The extent to which the wounding, pathophysiology, and healing processes in mice differ from those in humans might impact the interpretation of this model. The major mechanism for wound healing in human skin is by the formation of granulation tissue and reepithelialization, whereas mouse skin lacerations heal in part owing to rapid contraction caused by a subcutaneous muscle layer known as the panniculosus carnosus.33,34 However, the relevance of this observation to our frostbite model is not known, as it has been observed that a freeze wound created by applying a brass disk to a mouse back did not show contraction. 35 This effect of mouse wound contraction, should it become important for future studies, might be mitigated by using tight-skin mice or splinting.34,36
Although DHE, caspace-3, and TUNEL stains did not show reactive oxygen species or apoptosis in our model, that does not absolutely preclude a role for aspects of inflammation or reperfusion injury. Subcellular imaging techniques may be necessary to detect subtle changes.
We believe our new mouse frostbite model meets reasonable criteria for being quantifiable, such that mouse wound contraction and differences in precise mechanisms of granulation and epithelialization do not pose an obstacle for using the model for screening (for promising frostbite therapies) purposes. To be strictly comparable, aspects of mouse experimentation and handling for which there needs to be consistency, and to which we adhered, include the shape of the wound, pain, mouse caging, litter bedding, food and water placement, nutritional status, and other factors influencing well-being. 37
Conclusions
Here, we describe a novel mouse model of frostbite that is quantifiable for rate of skin wound healing and amount of tissue loss. We are able to demonstrate full-thickness skin necrosis, inflammation, and healing with neovascularization. We propose that this technique might become an efficient, reliable, and reproducible model for entry evaluation of new therapeutic modalities and the study of frostbite mechanisms. We are currently using our model to screen novel approaches to frostbite treatment. We hope that other investigators find it useful, and that modifications to it that increase its utility are promptly made known.
Footnotes
Funding
We thank Ms Yujin Park and Dr Evgenios Neophytou for their help with experimental procedures. This work was supported by the National Institute of Diabetes and Digestive and Kidney Diseases (RO1-DK-074095 to GCG), the Howard Hughes Medical Institute Medical Research Fellowship (MGG), and the Division of Emergency Medicine at Stanford.
