Abstract
Objective
Outbreaks of dermatitis linearis have been documented worldwide. We present a case series of dermatitis linearis from Latin America to highlight the importance of this clinical entity. Clinical, historical, epidemiological, and pathological aspects of the condition are discussed, and a concise current approach to the management and treatment of this morbidity is presented.
Methods
We present a series of 4 selected cases reflecting the clinical spectrum exhibited in dermatitis linearis by Paederus along with a review of the literature.
Results
In this review we demonstrate the need for awareness of dermatitis linearis as a clinical entity that must be considered in the broad list of differential diagnosis embracing vesicating linear lesions.
Conclusions
Capture of the insect, epidemiologic features, and a high clinical suspicion can aid in making the correct diagnosis. Primary prevention through public awareness, decreased use of artificial lighting, and mosquito nets can limit the extent and severity of outbreaks.
Introduction
Dermatitis linearis, also known as Paederus dermatitis, is a contact dermatitis characterized by erythematous and vesicular lesions of sudden onset on exposed areas of the body. The disease is provoked by vesicating toxins found in the endolymph of Paederus beetles belonging to the class Insecta, order Coleoptera (beetles), family Staphylinidae (rove beetles), subfamily Paederinae, tribe Paederini, subtribe Paederina.1,2 The subtribe Paederina (Paederus and close allies) contains more than 622 species, but only about 30 have yet been shown to cause dermatitis linearis or to contain toxic pederin, or both. 1 Paederus species are distributed throughout all continents except Antarctica.2,3
Dermatitis linearis is caused by the release of toxins after injuring or crushing a Paederus beetle. The characteristic linear lesions are most commonly caused by the victim inadvertently crushing the beetle and reflexively brushing away the insect. The insects do not sting, bite, or attack their victims, as some authors had incorrectly suspected in the past. 2 Serious dermatologic, ophthalmologic, and systemic symptoms highlight the clinical importance of dermatitis linearis.2,4 This case series discusses the most prominent historical, epidemiological, and clinical therapeutic aspects of dermatitis linearis and reviews the current knowledge of dermatitis linearis as a clinical entity and its management based on the Venezuelan experience.
Summary of Cases
Case 1
A 46-year-old man from the town of Nirgua in the agricultural state of Yaracuy, Venezuela, was referred to our clinic because of a rash of acute onset. The patient was a farmer who developed the lesions while collecting oranges and exposing himself to numerous beetles on the trees. Several other farmers were affected at that time with identical presenting symptoms. The lesions started as erythematous and edematous patches that later evolved into multiple blisters and subsequent erosions (as a result of intense scratching). The surrounding area was extremely erythematous, fading gradually toward the unaffected skin. The patient also complained of weakness and generalized joint pain. Identification was possible because of the recovery of several specimens of Paederus columbinus, recovered in orange (Citrus aurantium/sinensis) and mandarin (Citrus reticulata) trees from the patient's workplace, which the other farmers also identified as the causative agent of their illness, referring to them as “cantáridas.” The patient agreed to have a biopsy, which revealed findings consistent with an arthropodlike pattern of reaction. The patient was treated with clobetasol 0.05% ointment twice daily to the affected areas and acetaminophen 500 mg 3 times a day and desloratadine 5 mg daily. The skin lesions improved after 72 hours; however, joint pain persisted for a couple of weeks, requiring aspirin 325 mg twice daily for a week.
Case 2
A 38-year-old man attended our clinic as an emergency because of an insect bite. The patient reported that a few hours before, while waiting at night in a bus station in the town of Ospino, Portuguesa state (in the central western Venezuelan plains), he noticed what looked like an ant crawling on his hand. In an attempt to get rid of it, he accidentally crushed it against the skin. Erythema immediately developed without significant discomfort; however, a few hours later the lesions evolved into an erythematovesicular mildly pruritic rash. Initially, clinical examination revealed multiple pustules on a background of severe erythema localized on his left hand. Even though the distribution did not follow a dermatomal pattern, the arrangement highly resembled those of herpes zoster grouped in crops or herpes simplex. After 24 hours the lesions started to erode with scant visible vesicles and intense erythematous borders (Figure 1a). Tzanck smears for herpetic cells were negative. Lesions were biopsied, and both microscopic examinations and cultures for bacteria and fungi also proved to be negative. A sample of tissue was sent for histological examination (Figure 2). The patient immediately applied calamine lotion with modest improvement but shortly after developed severe headache and fever, which resolved with the administration of dipyrone 500 mg every 8 hours for 24 hours. Pruritus persisted for a few days and resolved with loratadine 10 mg by mouth daily and topical mometasone furoate 0.1% ointment.
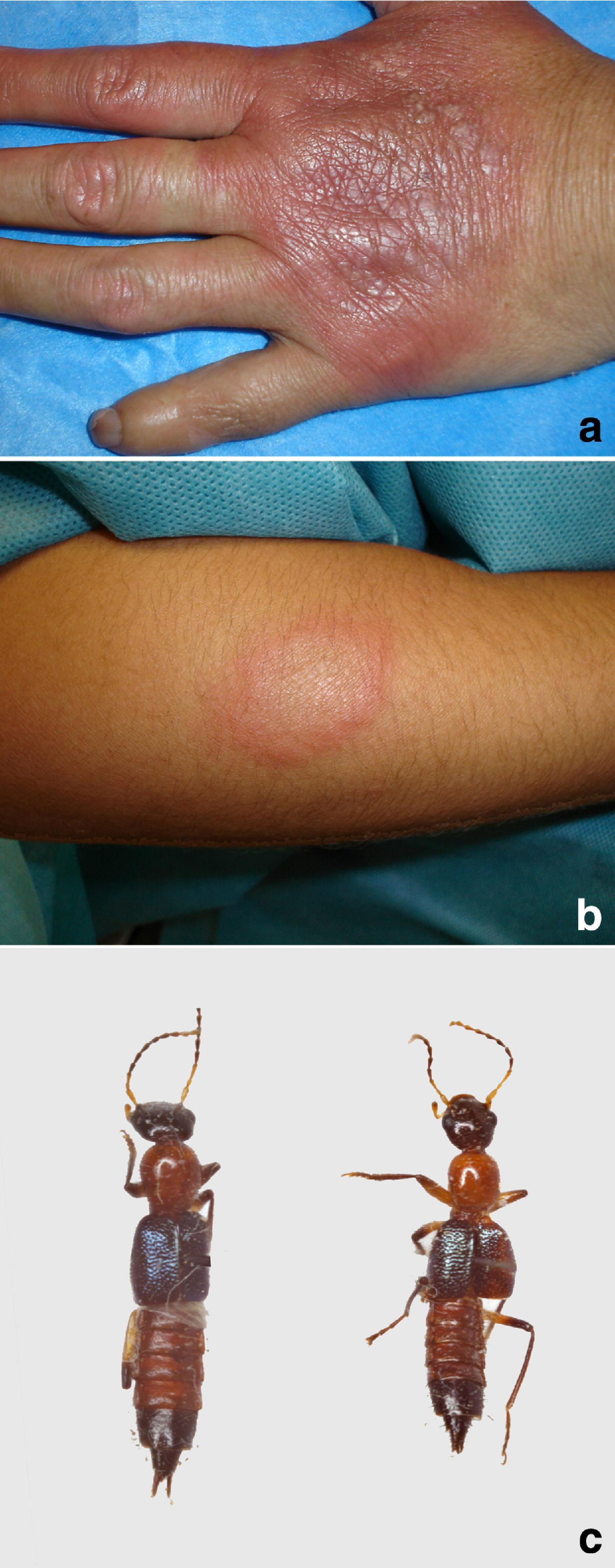
Dermatological features of dermatitis caused by P columbinus. (a) Multiple pustules on a background of severe erythema localized on left hand of the patient—case 2. (b) Multiple vesicular-pustular lesions—case 4. (c) Adult Paederus spp.
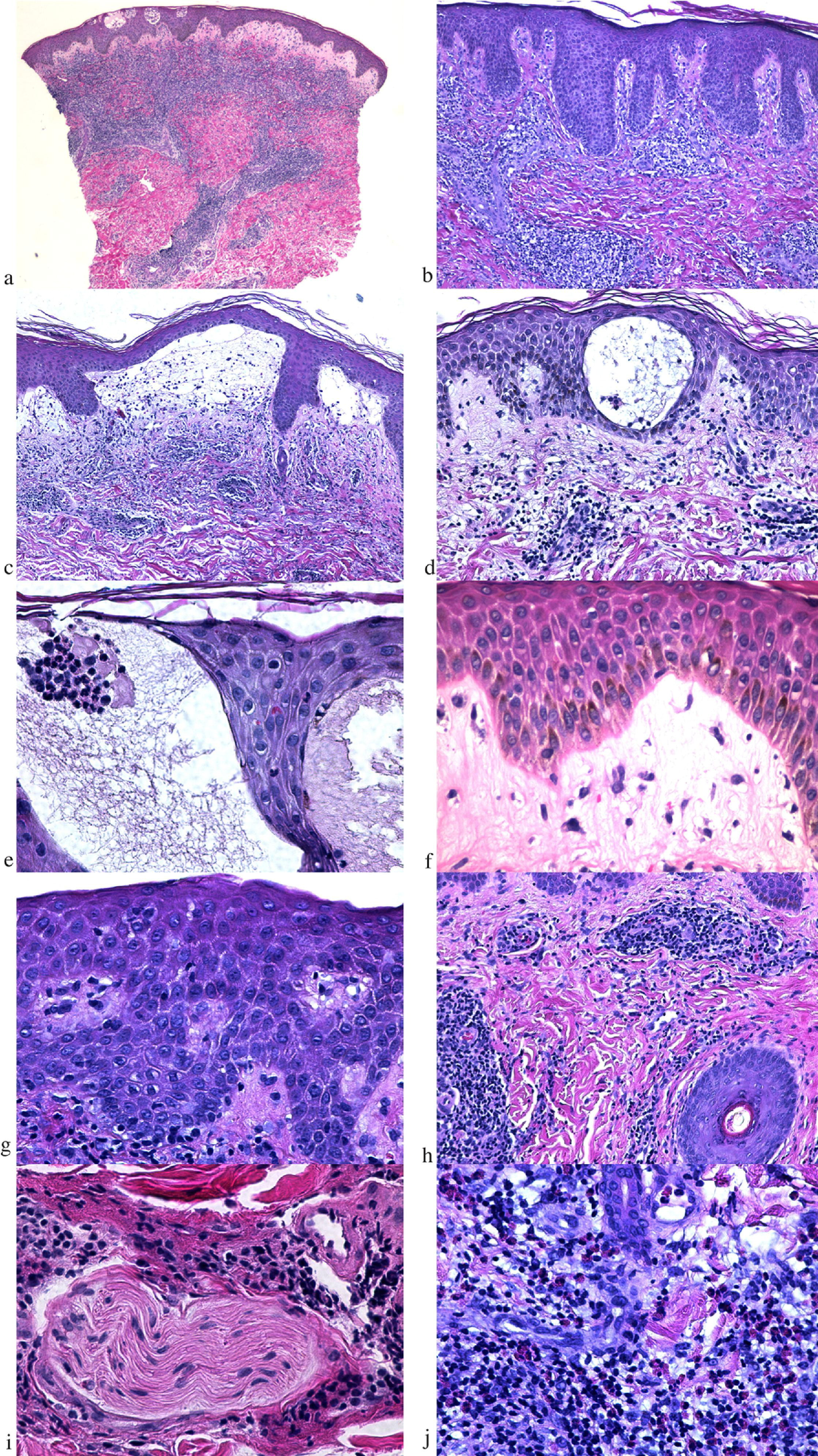
Histopathological features of dermatitis linearis. (a) Dense wedge-shaped perivascular lymphocytic infiltrate admixed with eosinophils (hematoxylin and eosin [H&E], ×4). (b) Irregular acanthosis and underlying dense lymphocytic infiltrate in a late-stage lesion (H&E, ×10). (c) Subepidermal blister formation (H&E, ×10). (d) Intraepidermal blister with marked spongiosis (H&E, ×20). (e) Neutrophilic and acantholytic cell aggregate within an intraepidermal blister (H&E, ×40). (f) Papillary dermal edema (H&E, ×40). (g) Spongiosis and lymphocyte exocytosis (H&E, ×40). (h) Dense periadnexal infiltrate (H&E, ×20). (i) Perineural lymphocytic infiltrate (H&E, ×40). (j) Dense eosinophilic aggregate (H&E, ×40).
Case 3
A 22-year-old woman from the state of Cojedes (central eastern Venezuela) was brought to the dermatology clinic because of a dermatitis localized to the right arm. She reported that the day before she had been placing some garments on a clothesline pole outdoors when she suddenly felt an insect on her arm, which she accidentally squashed while trying to brush it off her forearm. The dermatitis suddenly appeared on the same evening when she noticed localized erythema and edema. The lesion was somehow annular in shape, and of variegated coloration, which ranged from yellowish-pink to bright red; scarce small erosions developed later as a consequence of severe itching. The patient also complained of a burning sensation and flulike symptoms. All laboratory tests done were within the normal range. The diagnosis of linear dermatitis was confirmed because the patient had captured and brought the beetle with her, which was later identified as P columbinus. Mild fever persisted, which responded to a single dose of 500 mg of acetaminophen. Applying cool compresses, emollients, and zinc oxide ointment controlled the burning sensation. However, a stinging sensation remained for weeks, but disappeared after a short 3-week course of 25 mg of pregabalin twice daily.
Case 4
A 36-year-old man, who was traveling through the central western region of Venezuela, felt a sudden crawling sensation on the back of his neck caused by an insect while he was taking a rest and refueling at a gas station in the vicinity of Tinaquillo (Cojedes state). The patient reported a myriad of insects flying around the lights of the station. A few minutes after the contact with the arthropod, he felt a stinging sensation that soon became pruritic; around 6 hours later, a wide edematous and erythematous plaque appeared on the skin. The patient was evaluated the next morning when he complained of headaches, dizziness, and intense pruritus on the affected skin area. Multiple vesicular-pustular lesions were present (Figure 1b). A biopsy was performed to rule out herpes zoster. Improvement of symptoms was achieved by intravenous administration of 10 mg of chlorpheniramine maleate, metoclopramide 15 mg 3 times a day, and 500 mg of acetaminophen. The arthropod was recovered by the patient in a container and later identified as P columbinus. Mild to moderate pruritus persisted at the affected site and spontaneously disappeared after a week.
Discussion
Paederus
Adults of most Paederus species are active in broad daylight and climb on vegetation, especially in moist habitats.2,5 Some Paederus species adults are winged and can be seen flying at night when they are attracted to incandescent and fluorescent light sources, and others are brachypterous or apterous, and flightless. Adults of a few Paederus species are at times very abundant, especially in tropical countries, while others are known only from a few specimens and appear to be rare. 2
Paederus species have life cycles similar to those of other Staphylinidae, with egg, larval, and pupal stages. The adult Paederus beetle has a distinctive color pattern consisting of a black head, orange prothorax, iridescent blue elytra, and orange abdomen with black apex (Figure 1c). Adult Paederus beetles are usually 7 to 10 mm long and 0.5 to 1 mm wide, and are often mistaken for ants. Definite species-level identification of Paederus is insufficient on phenotypical grounds alone, usually requiring dissection at least of the genitalia. The key to the diagnostic features of each species was described in detail by Scheerpeltz. 6
Like other members of the subfamily Paederinae, the larvae have only 2 developmental stages; in contrast, most other Staphylinidae have 3. Paederus beetles in temperate regions may have only 1 generation per year, whereas those in tropical regions may have several. The timing and number of generations varies with climate. 2 Of the 622 species described to date worldwide, 9 species of the Paederus genus have been reported in Venezuela: Paederus conspicuus (Erichson 1840), Paederus ferus (Erichson 1840), Paederus laetus (Erichson 1840), Paederus salvini (Sharp 1876), Paederus signaticornis (Sharp 1886), Paederus tempestivus (Erichson 1840), Paederus yucateca (Sharp 1886), Paederus brasiliensis (Erichson 1840), and P columbinus (Laporte 1835). 6
Epidemiology
Worldwide, dermatitis linearis has been found in Turkey, Iran, Italy, Nigeria, Egypt, Tanzania, Australia, Sri Lanka, and Malaysia. 3 ,7–9 Outbreaks in the Americas have been documented in Peru, Guatemala, Panama, Ecuador, Venezuela, Brazil, and Argentina.2,10 In Venezuela, Paederus activity has been documented from May to January, 2 with the peak occurring in the spring and summer (between July and September). 5 On the northern coast of Brazil, dermatitis linearis is observed particularly during the rainy months of April and May. 11 Winter hibernation does not appear to occur in these species, with limitations in activity arising from low humidity during the dry season. Rice fields are ideal breeding grounds, and their activity coincides with that of the rainy season. 3 In Venezuela, farmers involved in the collection of oranges (C aurantium) and lemons (Citrus limon) are most commonly exposed. 12
Most frequently, cases occur at night when the beetles are attracted to artificial lights. In the tropics, outbreaks are more common among western expatriates than the indigenous population, likely owing to their higher use of artificial lights at night. 13 Paederus beetles are more sensitive to ultraviolet and white light, and are relatively insensitive to orange and yellow light. 14 An outbreak in Tanzania was halted after mercury tube lights, which emit ultraviolet light, were replaced with incandescent light bulbs that emit yellow light. 15
Fewer than 4% of the known species of Paederus have been associated with incidents of dermatitis linearis. These species include, but are not limited to, Paederus fuscipes in Asia and Europe, Paederus sabaeaus in Africa, and P columbinus and P brasiliensis in South America. 2 The pattern from documented epidemics is one of explosive population increases of one or more Paederus species, virtually simultaneously and over large areas, most likely stimulated by heavy rainfall. 2 Peru, in 1998, experienced an outbreak of dermatitis linearis as a result of El Niño rainfall, which promoted the growth of dense vegetation in areas that were historically dry. 10 Large outbreaks can become a major public health issue as evidenced by one incident that forced the evacuation of an entire Australian aboriginal community. 16
In Venezuela, dermatitis linearis was first described in the 1940s by Talamo 17 and Hómez and Franko 18 in the western states of Trujillo and Zulia, respectively, followed by the description of almost 400 cases by Dao19,20 and Kerdel-Vegas and Goihman-Yahr 21 between 1963 and 1966. Interestingly, the study by Dao was the first to describe ophthalmologic involvement concomitantly with skin lesions. Subsequent reports have followed with the description of cases extending into a broader geographical area embracing the central plains, as well as the coastal highlands of northeastern Venezuela. Recently patients from the state of Cojedes, Venezuela, were found to have dermatitis linearis caused by P columbinus. 12 In most areas of the country, dermatitis linearis is known with the common names of brasas, quemas, or culebrilla, and people often refer to the Paederus beetle as cantáridas, piqui-hulle, puri-puri, or tar-tari. 12 ,17–21
Clinical Diagnosis
Dermatitis linearis results from crushing and smearing a Paederus beetle on the skin, which gives the lesions its characteristic linear appearance. Past misconceptions attributing these lesions to the beetles biting or stinging their human victims have been proven to be unjustified. Skin contact is inadvertent on the part of the insect. 2 Symptoms typically begin between 24 and 48 hours, with the most common being itching and burning, and eventually progress to erythematous and edematous lesions. 5 ,22,23 Vesicles generally appear toward the center of the plaque and frequently become pustular. This is in contrast to cantharidin dermatitis, which is characterized by noninflammatory vesicles and bullae. 22
Clinical symptoms are graded from mild to severe, and progress from an erythematous phase to a vesicular phase and finally a squamous phase. 2 Mild cases exhibit only the erythematous phase with slight erythema beginning at 24 hours and resolving after 48 hours. Moderate cases have marked erythema and pruritus beginning at 24 hours, followed by a vesicular stage with blisters at 48 hours and gradually enlarging to maximal development at 96 hours. The squamous stage occurs during the next 7 to 10 days, characterized by drying out and umbilication of the vesicles, which then exfoliate and leave hyperpigmented scars that persist for a month or more. Severe cases resemble moderate cases but have more extensive skin involvement, and have additional symptoms such as fever, neuralgia, arthralgia, and vomiting. Erythema in some instances persists for months. Secondary infections, rhinitis, tympanitis, and ocular injury have also been documented.2,4
Exposed areas of the body such as the face, neck, and arms are most affected. 2 ,3,23 Typically the palms of the hands and soles of the feet are spared. 2 The lesions are usually linear; however, a “kissing” lesion may appear at areas where damaged skin apposes previously healthy skin, such as the flexure of the elbows and adjacent surfaces of the inner thigh. 23 Transfer of the toxin by fingers has been attributed to ocular and genital symptoms. 2
The human eye is particularly sensitive to the Paederus toxins. Contact may occur as a result of toxin transfer from the fingers or directly by collision with the insect. Pain and lacrimation are nearly immediate, followed by progressively worsening erythema and edema. Periocular and periorbital lesions are typical in these cases, which may progress to conjunctivitis, keratitis, and temporary vision loss. 4
Differential Diagnosis
The clinical differential diagnosis of dermatitis linearis includes acute allergic or irritant contact dermatitis, thermal burns, chemical burns, herpes zoster, herpes simplex, and bullous impetigo. 3 ,13,22 An important differential diagnosis to consider is phytophotodermatitis as there are many similarities between the 2 conditions including linear asymmetric erythema, blister formation, and dyspigmentation. 24 Other entities that must be considered are cutaneous larva migrans, 25 dermatitis herpetiformis, 21 pemphigus foliaceus, 26 and caterpillar27,28 or moth-related urticaria. 29 Typically, the diagnosis can be made clinically without the assistance of histopathologic confirmation. Capture of the offending insect, the characteristic linear appearance of the lesions, their predilection for exposed areas, the presence of kissing lesions, and epidemiologic features should all assist the clinician in making the correct diagnosis.
Histopathological Diagnosis and Pathogenesis
Because it had long been known that the Meoidae and Oedemeridae families of beetles released cantharidin as a defensive response, early authors mistakenly assumed that the toxin responsible for dermatitis linearis was also cantharidin. Studies on the characteristics of the Paederus toxin eventually led to the conclusion that the substance is in fact a different chemical compound.2,24 The offending agent is one or a combination of the vesicant toxins pederin, pseudopederin, and pederone. 13 These toxins are produced by an endosymbiotic bacteria of the genus Pseudomonas and can be found primarily in the endolymph of females. 30 Larvae and subsequent generations acquire the symbiotic bacteria through the ingestion of egg shells produced by “infected females.” 31
Dermatitis linearis may be considered as an example of irritant contact dermatitis rather than a true allergic contact dermatitis. 32 Pederin (C25H45O9N) is an amide with 2 tetrahydropyran rings 33 –35 that is contained in the hemolymph of the beetle and is released when crushing the insect onto the skin as a result of the reflex attempt to brush it away2,3; it is thought to act as a vesicant that blocks mitosis at levels as low as 1 ng/mL, apparently by inhibiting protein and DNA synthesis but not affecting RNA synthesis.36,37
The dermal host response often reveals a wedge-shaped perivascular lymphoid infiltrate with eosinophils (Figures 2a, 2b). The acute lesion is characterized by an irregular, multilocular dermal (Figure 2c) and intraepidermal blister (Figure 2d), with marked necrosis of the epidermis, intercellular and intracelluar edema, reticulate necrosis of the epidermis, and grouped acantholytic cells in the bulla fluid (Figures 2e, 2f).7,32 Acantholysis is most likely caused by the release of epidermal proteases 7 and consequent disruption of tonofilaments in the desmosomes. The inflammatory cell pattern is characterized by superficial and deep wedge-shaped perivascular (Figure 2a) and periadnexal (Figure 2h) lymphocytic infiltrate and by interstitial mixed cell infiltrate, with a predominance of eosinophils both in the dermis and the bulla. Epidermal lymphocytic exocytosis is common (Figure 2g). Eosinophils are scattered throughout the remnants of the dermis (Figure 2j), and epidermis and nuclear dust is a prominent finding.
Subacute changes show a superficial confluent necrosis of the epidermis with a predominance of eosinophils and neutrophils, overlaying a newly formed, irregularly acanthotic epidermis. At this stage, papillary edema (Figure 2f) fades away, but a mixed, prominent inflammatory infiltrate is present around the superficial and deep vessels. In the late stage, a small irregular acanthosis (Figure 2b) and reappearance of the stratum granulosum are the main features. There is persistent superficial and deep perivascular and periadnexal dermatitis, with the presence of lymphocytes (Figures 2i, 2j). A desquamating scale-crust is usually evident. 32 Mitotic figures and apoptotic changes such as chromatin condensation and DNA fragmentation have been identified in the basal and suprabasal layers both histologically and ultrastructurally. 23
Curiously, in the biopsy sections examined from all our patients, a perineural lymphocytic infiltrate (Figure 2i) was present, being most prominent in case 3, thus possibly reflecting the underlying pathological substrate for the patients' dysesthesias. To the best of our knowledge, perineural lymphocytic infiltration has never been histologically reported before in dermatitis linearis and supports the clinical expression of neuropathic signs (pain, tingling, and stinging) observed in most cases. Although several exogenous endopeptidases and proteases along with pederin may induce pain, itching, and inflammation, the potential role of neuropeptides in this particular clinical scenario has yet to be elucidated and may constitute a promising target in the therapeutic approach of this condition, as well as other arthropod-related assaults exhibiting a dysesthetic and pruritogenic nature.
Treatment
Although at least 25 pharmaceutical preparations have been used to treat dermatitis linearis, there are limited data on their effectiveness. 2 Experts agree that affected patients should be managed as irritant contact dermatitis—with removal of the pederin toxin by washing with soap and water. This can be followed by application of cold wet compresses, oral antihistamines, and topical steroids. 3 ,22,38 Antibiotics can be considered for the prophylactic treatment of secondary bacterial infection. One study in Sierra Leone found that patients treated with a topical steroid, an antihistamine, and oral ciprofloxacin had shorter healing times than those treated with steroid and antihistamine alone. The authors concluded that this may be related to Pseudomonas the Paederus beetles harbor. 38 Tincture of iodine has been shown to destroy pederin, and has been used in the treatment of dermatitis linearis; however, its application may be too late once the reaction has developed. 2
In our experience patients usually present with variable signs and symptoms, and therapy must be individualized. Cooling agents such as calamine, alcohol, and menthol can provide temporary relief of itching and burning sensations; these options are usually the first-line agents available to the patient over the counter, before they consult a specialist. Emollients can also provide significant relief of itching.
Antihistamines including promethazine and diphenhydramine are useful for relieving the pruritus, with loratadine (a second-generation H1 histamine antagonist) and desloratadine (also a selective and peripheral H1-antagonist) being the most effective in our experience. Anesthetics agents are rarely used, but are very effective in decreasing pain and tingling sensations. Topical preparations of camphor, lidocaine, and benzocaine have been used by our group and other colleagues with an extraordinarily antipruritic and anesthetic effect.
Among other topical agents that are pivotal in the therapeutic intervention of dermatitis linearis are the highly effective topical corticosteroids. Corticosteroids exert a variety of mechanism of actions locally to the skin such as suppression of histamine release and mast cell inhibition. Effectiveness can be improved by placing the corticosteroid on a hydrocolloid or plastic film occlusion dressing to enhance penetration. Mid- to high-potency corticosteroids are preferred for a 7- to 10-day cycle.
Systemic therapy is reserved only for severe cases or those recalcitrant to topical intervention. Even though systemic corticosteroids are excellent anti-inflammatory drugs, they do not provide immediate itch relief, thus requiring its association with an antihistamine. Also, their use must be for a limited time. In our experience, only 1 case of dermatitis linearis with involvement of the genitalia prompted the use of a short-course treatment with prednisone at a taper dose of 60 mg/3 days, 40 mg/3 days, and 20 mg/3 days. Typical doses are calculated at a range of 0.5 to 1.0 mg · kg–1 · day–1.
Arthralgias, cephalea, and fevers are usually controlled with nonsteroidal anti-inflammatory drugs, which also have an indirect effect on ameliorating inflammatory-associated pruritus. 39 Nausea and vomiting, although infrequent, usually respond to metoclopramide and dimenhydrinate; however, if severe, ondansetron might be an option also because of its known anti-itching effects. 40
Lastly, in our experience dysesthesias is a common acute and chronic complaint. Even though it can be mitigated with the initial therapeutic intervention, chronic dysesthesias (such as seen in case 3 of this series) might require the use of low-dose anticonvulsants such as gabapentin and pregabalin. We have used a 3-week course of 25 mg of pregabalin with complete resolution of symptoms.
Primary prevention by increasing public awareness during outbreaks, decreasing the use of artificial lights at night, and using mosquito nets is advocated by several authors. 3 ,5,13
With ever increasing trends toward urbanization and globalization, the profile of affected individuals by dermatitis linearis has gradually changed. Additionally, there has been increased occurrence in rare and nonendemic regions such as the United States, where affected individuals are predominantly travelers or military personnel deployed to endemic regions. 13 Although outbreaks of dermatitis linearis typically occur throughout rainy seasons, recently there have been descriptions of aberrant increases in populations and out-of-phase biological cycles in some regions as a consequence of climatic changes. Such is the case of the Piura outbreak (which affected Peru in 1998), in which the growth of abundant vegetation in previously dry areas, as a consequence of the climatic phenomenon of “El Niño,” provoked an increase in the natural growth of the Paederus population and its relocation to urban areas. Owing to the abovementioned factors, it is important for physicians to be aware of dermatitis linearis as an entity that must be considered in the broad list of differential diagnoses embracing vesicating linear lesions.
