Abstract
Objectives:
Lucilia sericata (Diptera: Calliphoridae) is used in larval therapy for wound healing. Netrin-A is an enzyme secreted from the salivary glands of these larvae, and has a central role in neural regeneration and angiogenesis. This study aimed to produce the recombinant Netrin-A protein from Lucilia sericata larvae by the baculovirus expression vector system in the Sf9 insect cell line.
Methods:
The coding sequence of Netrin-A was cloned, amplified in the pTG19 vector, and then cloned in the pFastBac HTA vector. It was then transformed into DH10Bac, and the recombinant Bacmid was subsequently transfected into Sf9 cells. The recombinant Netrin-A was purified by Ni-NTA agarose. The evaluation was done using SDS-PAGE and western blot, respectively. Finally, its concentration was calculated with the Bradford assay.
Results:
The molecular weight of this protein was 52 kDa with 404 amino acids. The signal peptide was located between amino acids 24 and 25. The concentration of Netrin-A was calculated to be 48.8 μg/ml. It reaffirmed the characterized gene codes of Lucilia sericata Netrin-A in a previous study.
Conclusions:
The generation of recombinant Netrin-A could be used in larval therapy, and as a biomarker in certain diseases. The netrin-A of Lucilia sericata was unprecedentedly cloned and expressed in a eukaryotic cell line. Given that this larva is FDA-approved, and non-pathogenic, it conduces to research on the development of maggot therapy in future.
Introduction
Most people are exposed to multiple wounds throughout their lives. The most common traumas are superficial lesions limited to the outer layers of skin. 1 At the same time, some wounds are more profound, damaging subdermal tissues and organs. Depending on their cause, location, and depth, wounds can range from simple to severe injury in people. Injuries, burns, accidents, and diseases such as diabetic foot and venous ulcers can lead to acute and chronic wounds in individuals. 2 It was estimated that 8.2 million people had infected and uninfected wounds globally, and treatment costs summed up to $96.8 billion.3–7
However, antibiotics, surgical interventions, and various other medical methods are implemented to treat and cure injuries, but the critical issue is resistance to multiple antibiotics. Attempts to use alternative and complementary procedures in wound healing are thus contemplated. 8 In the last several years, maggot therapy has been one of the most biocompatible routes of managing chronic wounds. 9 It has had a practical outcome so far; the FDA first approved this protocol in 2004. 10 In this way, the greenbottle fly, Lucilia sericata (Diptera: Calliphoridae), larvae crawl and wriggle through the ulcer and secrete protease enzymes,11,12 antimicrobial peptides (AMPs),13,14 and other influential factors in wound tissue regeneration leading to their cleansing and disinfection.15–17 The triple (debridement, disinfection, and growth leading to wound healing) therapeutic effects of medicinal maggots have been reviewed elsewhere. 18
While the patient’s health is in danger and his body cannot tolerate antibiotics and other wound healing methods, sterile larval therapy as a productive alternative way can lead to wound resolution in these people. 19 There are also efforts to use bagged maggots in patients’ wounds to avoid the problem of their direct exposure to larvae. This controlled intervention requires sufficient training and skills. 20 Larval excretory/secretory (ES) factors, degrading extracellular matrix, include a variety of proteolytic enzymes (collagenase, leucine, carboxypeptidase, aminopeptidases, and netrins),12,21 growth factors, 22 AMPs, 23 anti-inflammatory, 24 chemokines, 25 and other unknown compounds in wound healing. For example, some of them, such as collagenase, cause wound debridement, and some, such as growth factors, accelerate wound healing.11,26
Netrin-A is a secreted protein from L. sericata. It is in the family of Laminins with the most salient role in axon guidance. It was first described in the nematode Caenorhabditis elegans in 1990 and named UNC-6, Netrins were also identified in Drosophila melanogaster, frog, mice, and human. 27 Researchers discovered two Netrin-A and Netrin-B (NetA, NetB) types in the fruitfly(D. melanogaster). 28 Netrins are enzymes with significant roles in wound repair, neural regeneration, cell migration, and angiogenic activity. 29 They have many diagnostic, therapeutic, and research applications in medical sciences.30–34
Netrins are the main components of the peripheral nervous system extracellular matrix and play a role in adhesion, differentiation, migration, resistance to apoptosis, and determination of the stable phenotype. 29 Therefore, the extraction of effective proteins in wound healing and recovery is of particular importance, and is one of the principal priorities of biotechnological science in medicine. 35 Netrins can repair neural tissues and cellular processes.36,37 This enzyme has a role in improving wounds and various injuries caused by diabetes, burns, and osteomyelitis. 38 The generation of recombinant proteins is one of the significant achievements of biotechnology in the twentieth century. Using the natural power of prokaryotic and eukaryotic host cells to express these proteins has led to research on medicinal constituents. Recombinant proteins have become very crucial for clinical applications. 39 Protein production in the insect cell line has increased due to its cost-effectiveness and successful expression of many products. It has been reported in culture media, whether intracellular or secretory. 40 For example, most cell lines are derived from the ovaries of the fall armyworm, Spodoptera frugiperda (Sf9, Sf21) and Trichoplusia ni, (Tn5 or High Five TM). These cells are used to produce recombinant proteins. 41
Baculovirus was identified as a tool for overproducing recombinant proteins in eukaryotic cells. 42 The baculovirus expression vector system (BEVS) is a powerful recombinant protein generation method. Numerous factors which contribute to their popularity have helped. They take advantage of the eukaryotes, protein changes, processing, and transport systems in excellent eukaryotic cells . However, significant recombinant compounds have been produced from the Calliphoridae family of flies. 11 Nevertheless, the number of recombinant proteins produced from the secretions of L. sericata larvae is an understudied area of research. 43 Netrin-A is one of the most mysterious proteins of L. sericata flies. Due to its vital role in medicine and pharmaceutics, the aim of the present study was the cloning, expression and molecular analysis of recombinant Netrin-A protein of L. sericata Meigen (Diptera: Calliphoridae) larvae.
Materials and methods
This experimental study was conducted with the ethics code IR.SUMS.REC.1399.1155 at Shiraz University of Medical Sciences. The entire mRNA of Netrin-A was previously identified by the authors and the gel-extracted cDNA of L. sericata Netrin-A was stored at −70°C. 44 The following steps were undertaken to amplify and produce the Netrin-A protein. In the author’s previous study, netrin-A mRNA was identified using the rapid amplification of cDNA ends and rapid amplification of genomic ends method, 44 and it has been registered in the gene bank with accession numbers MG009433.1, MZ358388.1 and MG891747.1
Primer design and ligation
Initially, TA cloning was performed to amplify the Netrin-A gene. The primers were designed based on BamH1 and EcoR1 enzymes on the pTG19-T cloning vector (Table 1).
The sequence of primers designed to clone Netrin-A gene in pTG19-T cloning vector.

At this stage, the fragment of the Netrin-A gene was inserted into the pTG19-T cloning vector by mixing the following solutions in a ligation reaction: 1 µl of T4 DNA ligase buffer (10×), 0.2 µl T4 DNA ligase enzyme, 1 µl of the pTG19-T cloning vector. Distilled water was added to reach a total volume of 10 µl. The volume of the DNA template was variable (based on OD computing and proportion 3:1 insert and vector). The prepared solution was then incubated for 1 h at room temperature. The inserted fragment in the pTG19-T cloning vector was transformed into DH5α competent cells according to the relevant protocol. They were cultured on plate LB agar containing ampicillin (50 μg/ml), IPTG (50 μg/ml), X-gal (100 μg/ml) and incubated at 37°C overnight. The so-formed colonies were blue and white. Polymerase chain reaction (PCR) was performed on several white colonies with specific and pUC/M13 primers to verify the incorporation of the desired fragment into the vector (Table 1). Positive colonies were then grown on a 5 ml LB broth culture medium overnight. Plasmid extraction was performed using a GeneALL plasmid extraction kit (Bioneer, South Korea). Plasmids containing Netrin-A were digested using BamH1 and EcoR1 restriction enzymes.
Digested pFastBac HTA and ligation of coding sequence (CDS) of L. sericata Netrin-A gene
The double digestion method dissolved the pFastBac HTA plasmid using BamH1 and EcoR1 restriction enzymes. Subsequently, 3 µl of CDS of L. sericata Netrin-A were ligated into 1 µl of linearized pFastBac HTA vector (proportion 1:3 insert and vector). The ligation product was then transformed into DH5α competent cells. To confirm the correct placement and orientation of the inserted fragment into pFastBac HTA vector, PCR was performed on the grown colonies (white colonies) using pFastBac HTA vector primers and specific primers of the inserted component (Table 2).
General primers designed for pFastBac HTA transfer vector and Gene Specific Primers of L. sericata Netrin-A.

Transformation of pFastBac HTA to DH10Bac
According to the manufacturer’s instructions, the pFast-Netrin-A construction plasmid was transformed into DH10Bac (Invitrogen, USA) competent cells (Bac-to-Bac Baculovirus Expression System ThermoFisher, USA). DH10Bac cell includes two vectors Bacmid and a helper vector that facilitates the transposition of the gene into the Bacmid structure. After transformation the solution was then cultured on LB agar plate containing tetracycline (10 μg/ml), kanamycin (50 μg/ml), gentamycin (7 μg/ml), IPTG (50 μg/ml), and X-gal (100 μg/ml) for an overnight. Colony-PCR was performed on white colonies with pUC/M13 and Netrin-A gene-specific primers (GSP) (Table 2). Then the plasmid was extracted from the positive colony (expected size was shown in PCR assay) by the method mentioned in the molecular cloning book for Bacmid.45,46 The plasmids were stored at 4°C for the next stage.
Sf9 cell culture and transfection
At first, the frozen Sf9 cells containing dimethyl sulfoxide (DMSO) were taken from the nitrogen tank. It was then cultured in 7 ml Grace’s insect medium (Gibco, USA) with 20% fetal bovine serum (FBS) and 1% pen/strep antibiotics in a 25 cm cell culture flask (Thermo Fisher, UK). The cells were stored in a 27°C incubator for 3–5 h. To remove DMSO, the previous Grace’s insect medium was discarded, and then 7 ml of fresh Grace’s insect medium with 10% FBS and 1% pen/strep antibiotics were added to proliferate these cells.
For transfection, eight steps were conducted as follows:
A total number of 8 × 105 cells in the log phase were seeded overnight onto a six-well plate with complete Grace’s medium (10% FBS and 1% pen/strep).
The Grace’s medium was wholly removed, and 2.5 ml of fresh medium unsupplement was added to each well on Sf9 cells.
A volume of 8 μl Cellfectin® II (Thermo Fisher Scientific, USA) with 100 μl unsupplement Grace’s medium mixed in a 1.5 ml sterile microtube was gently vortexed.
Exactly 1 μg of Bacmid plasmid (DNA baculovirus) mixed with 100 μl unsupplement Grace’s medium was slowly mixed.
The microtubes of stages 3 and 4 were gently pulled out and kept at room temperature for 15 min.
The solution of step 5 was slowly added drop by drop to step 2 and incubated at 27°C for 3–5 h.
The surface medium was discarded, and 2 ml complete Grace’s medium was added.
After 72 h, the cytopathic effects were checked. However, the transfected Sf9 cells were checked daily with an inverted microscope to find signs of cellular infection (the cytopathic marks).
After this observation on the viral infection of cells, they were harvested. The harvested cells were then centrifuged at 12,000g for 5 min, collected, and stored at 4°C. They were protected from light. This solution became the P1 viral stock used in the next steps for large-scale expression. Furthermore, P2-P3 viral stocks were prepared according to the manufacture’s instruction (Invitrogen, USA). Overall, 120 ml (cells) were used for the large-scale expression. When the viral infection titer of cells reached its maximum, and the time of high protein expression was maximized, these cells were harvested and centrifuged, then re-suspended with 5 μl PBS 1×, and centrifuged. Finally, the cells’ pellet and supernatant were analyzed using the SDS-PAGE method to determine if the protein was secreted or expressed in the cell membrane. These steps were essential to select the proper cell purification method in the future.
SDS-PAGE, Western blot analysis, and recombinant protein purification
Transfected Sf9 cells were harvested by Cell Scraper size 25 cm (Sigma Aldrich Co.) and then centrifuged for 5 min at 12,000g, supernatant stored at 4°C. The cell pellets were suspended by 50 μl sterile PBS and then freeze-thawed in liquid nitrogen 4–6 times. In the next step, SDS-PAGE was done based on the relevant protocol to detect the expected size Netrin-A protein on 12% acrylamide gel. After confirmation of the expression of the desired protein in the expected band size, the presence of His-tag was evaluated by performing the western blot assay based on the given protocol. Following large-scale recombinant protein production, the cell lysate was purified according to the Ni-NTA agarose in accord with the Invitrogen protocol.
Bradford test
The concentration of Netrin-A was then calculated by Bradford protein assay. Bradford and bovine serum albumin (BSA) were used in this method at different concentrations as a standard. The standard curves were plotted at concentrations of 62.5, 125, 250, 500, and 1000 μg/ml by measuring the absorbance at 595 nm. Excel 2010 was used to draw the curve and prepare the linear regression formula.
Determination of Netrin-A level by enzyme-linked immune sorbent assay
Netrin-A recombinant level was measured using a commercially available human ELISA kit with cat number NBP2-76771 from Novus Biologicals company (US).
Results
Cloning of Netrin-A gene
Netrin-A was successfully amplified by TA cloning. Three white colonies were selected, and colony-PCR was performed with pUC/M13 and GSP. As expected, the band sizes of 1250 bp with specific primers and 1450 bp with universal primers were shown (Figure 1). The positive colony was cultured in 5 ml LB broth (with ampicillin), plasmids were extracted, and then double digested with BamH1 and EcoR1 restriction enzymes.
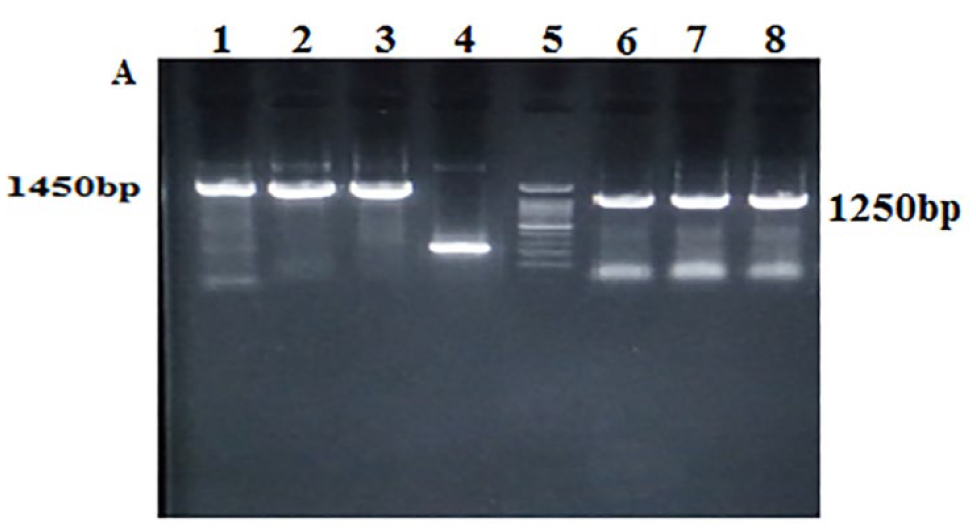
The results of TA cloning were confirmed by M13 primers (wells: 1–3), well 4 self-ligation, well 5 ladder of 100 bp, and confirmed by GSP wells 6-8.
Ligation of L. sericata Netrin-A and linearized PfastBac HTA plasmid
Digested fragments of Netrin-A (Figure 2(a)) and linearized pFastBac HTA vector were ligated and successfully transformed into DH5α competent cells. The pFastBac HTA-Netrin-A construct was confirmed with colony-PCR using universal primers of pFastBac HTA and GSP primers of Netrin-A (Figure 2(b)). The expected band sizes were 1650 bp and 1250 bp, respectively. The positive colony was cultured in LB broth, and the extracted plasmid was stored at 4°C to the next step. The sequencing results of the pFastBac HTA transfer vector showed that the placement of the L. sericata Netrin-A gene in the pFastBac HTA transfer vector was correctly performed. Sequencing operations with universal primers (PfastF and PfaR) were performed by Bioner Company (South Korea).

(a) Enzymatic cleavage of the pTG19 plasmid and isolation of the Netrin-A gene coding region with the restriction enzymes BamH1 and EcoR1; Lane 1- Digested plasmid pTG19 (cut), Lane 2- 100 bp marker (Cat. No. PR901644), Lane 3- Undigested plasmid pTG19 (uncut). (b) The results of constructing pFast HTA recombinant with L. sericata Netrin-A gene. Lane 1- Confirmed by pFastHTA universal primers, Lane 2- Confirmed by GSP of Netrin-A, and Lane 3- DNA ladder of 100 bp.
Recombinant Bacmid
The pFastBac HTA-Netrin-A construct was transformed into DH10Bac Escherichia coli, a desirable gene transposed into the Bacmid and disrupted the lacZ gene; therefore, the white colonies were evaluated. After 48 h, colony PCR was performed with universal pUC/M13 and GSPs primers. The expected band size was observed at 3680 bp (2430 bp + 1250 bp) since when the gene is transposed into the Bacmid vector, 2430 bp is added to the size of the cloned fragment according to the Invitrogen Bac-to-Bac protocol (Figure 3). Finally, one of the positive clones was selected for Bacmid plasmid extraction and transfection into the Sf9 cell line of the next step.

The results of colony PCR to investigate the pFastBac transfer vector transposition into Bacmid DNA in DH10Bac E. coli, Lane 1- Blank, Lane 2- The 3680 bp band expected in case of correct transposition, Lane 3- Marker ladder (Pub. No. MAN0013004) 1 kb.
Sf9 Transfection and expression of Netrin-A protein
The Sf9 cells were transfected with 1 ng recombinant Bacmid using a liposome-mediated method to produce recombinant baculovirus. Transfected cells were then observed by inverted microscopy before and after (24, 48, and 72 h) infection with recombinant baculovirus. The Sf9 cell cytopathic effect was observed 24, 48, and 72 h post-exposure. The symptoms of infected cells included enlarged nuclei and condensed cytoplasm (Figure 4). Based on bioinformatics analysis with Oligo 7.0, the neutrino sequence is expected to encode a 45 kDa protein. According to the Bacmid protocol, the baculovirus vector adds about 7 kDa to the target protein, which in quantitative and qualitative evaluations, an exactly 52 kDa protein was observed (Figure 5).

(a) Control (normal cells of Sf9), (b) Infected cells of Sf9 after 72 h.

SDS-PAGE and Western bloting (a) Lane 1: SDS-PAGE on infected cells indicated a 52 kDa band, Lane 2: Control (Bacmid plasmid), and Lane 3: Protein marker. On protein purification, (b) Lane 1: SDS-PAGE exhibited the size band, Lane M: Protein marker. (c) Lane 1: Western blot of protein purification on elution 1, and Lane M: Protein marker.
Purification of Netrin-A
Netrin-A was purified using the hybrid purification method based on the Invitrogen Bac-to-Bac protocol, using Ni-NTA affinity chromatography, since this protein was expressed in the cell membrane. The expression of the Netrin-A protein of the cell lysate was confirmed by western blot analysis revealing a band of about 52 kDa (Figure 5(a)). The purified proteins were analyzed by 12% SDS–PAGE (Figure 5(b)) and western blot analysis (Figure 5(c)).
Determination of the concentration of recombinant protein produced
The Bradford protein assay measured the concentration of Netrin-A. After standardization with BSA protein as described in the previous section, the concentration of recombinant Netrin-A protein was measured in four replicates at 450 nm wavelength with a spectrophotometer. Finally, based on the regression formula obtained from Excel software (Figure 6), the concentration of recombinant protein was calculated to be 48.8 μg/ml.

Regression line based on BSA standard to obtain regression line formula and calculate Netrin recombinant protein concentration (48.8 μg/ml.).
Determination of netrin-A level by ELISA
The regression curve (standard) of the best fit was drawn using Excel software (version 2013) based on the company’s manual, and then the OD of netrin samples in elutions 1 and 2 were calculated at a wavelength of 450 nm. The concentration of the recombinant netrin-A sample was also measured in four repetitions and its average was calculated, and based on the obtained formula, its concentration was calculated as 62.5 pg/mL.
Discussion
In this study, the recombinant Netrin-A protein was expressed in the BEVS. L. sericata Netrin-A protein with 1250 nucleic acids and 404 amino acids had a molecular weight of 45 kDa. Expression of this protein (http://tools.thermofisher.com/content/sfs/manuals/bactobac_man.pdf) in the BEVS added 3–7 kDa to it. Finally, the Netrin-A protein with 52 kDa was obtained in the quantitative and qualitative evaluation. In a previous study conducted by Alipour et al. 11 in 2019, such cases were reported in recombinant collagenase protein. 11 With the advancement of biotechnology, searches are underway to identify and generate miraculous medicinal products extracted from insects. They can be used as suitable models to produce many effective biocompatible compounds for treating diseases by manufacturing industries due to their fast and easy breeding and adaptation to the environment. These insects include bees, scorpions, ants, flies, and other beneficial insects.11,43,47
Maggot therapy utilizes sterile larvae of L. sericata in wound healing. 48 The present study achieved the expression and production of the Netrin-A protein from L. sericata as a candidate in wound healing using the BEVS. The Netrin-A had a molecular formula of C3378H5352N1012O1042S56. The number of its incorporated atoms was 10,840. Its highest percentage (8.4%) of amino acid frequency belonged to Serine. The protein sequence of this enzyme had 53.6% and 99.49% similarity (sequence identity) with the human Netrin-1 and Netrin-A of Lucilia cuprina, respectively, using the protein BLAST (blastp suite) online software from the NCBI. The observed band of expressed protein was about 52 kDa using SDS-PAGE analysis. Based on the In Silico calculations, the recombinant protein content of L. sericata Netrin-A was estimated to be 45 kDa, which increased to 52 kDa after CDS transfection of the target gene into Sf9 cell due to post-translational modification. The increase in protein size was consistent with the expression vector manufacturer’s instructions.
In a recent study, this band size increase also showed in the recombinant collagenase protein expressed by the baculovirus system. 11 In another survey of luciferase, an increase in the size of the recombinant protein after transfection into Sf9 cells was evident. 49 The concentration of Netrin-A, measured by the Bradford protein assay, was 48.8 μg/ml. Some studies have been done in the field of expression and recombinant production of netrin types, but so far no study has been done in the insect cell line using the baculovirus expression vector to produce netrin recombinant protein, and this study reports the production process of netrin in the insect cell line for the first time. In the study conducted in 2000, researchers used pcDNA4Myc/HisA to express netrin-1 in HEK293T cells. 50 Also, in 1999, Netrin-3 was cloned into the mammalian expression vector pCEP4 (Invitrogen) and transfected to 293 EBNA cells. 51 In another study in 2021, netrin-1 was expressed in human cells (HEK-293) and the concentration of netrin-1 recombinant protein was calculated to be 0.0625 μg/ml, Compared to this article, the concentration of netrin recombinant protein in the insect cell is much higher. Of course, in the mentioned study, to increase the protein concentration, they were able to significantly increase the method by increasing doxycycline and using the Sleeping Beauty transposon system. 52 Further, Recombinant collagenase protein was also expressed and produced with BEVS for L. sericata larvae. According to the SDS-PAGE analysis, the size of the collagenase protein band was 52 kDa. Alipour et al. reported the concentration of collagenase protein to be 68.8 μg/ml in 2019. 11 The results of both studies were almost similar and in concordance. This finding was possibly due to using a standard baculovirus system to express both of these proteins. 11
In recent years, the application of maggot therapy and the use of sterile larvae of L. sericata has been welcomed due to the resistance of wounds to various antibiotics and people’s prejudice to traditional and natural therapies. 53 ES factors of L. sericata larvae are a natural source of effective ingredients in wound healing, such as proteases, AMPs, 14 growth factors, and thousands of unknown substances effective in wound healing and other diseases. 54 A distinct stalemate is how blowfly larva interacts with microbiomes at the molecular interface. 14 Whether Netrin-A spatiotemporal production is under the collective influence of microbial interactions and larval L. sericata immune genes remains to be unraveled.
The Netrin-A is one of the secretory proteins in the green blowfly larvae of L. sericata. It has many diagnostic as well as biotherapeutic applications in the biomedical sciences. It can be used for wound healing, anti-inflammatory, hepatic ischemia repair, and improvement after liver transplantation. A biomarker in diagnosing renal ulcer, ulcerative colitis, heterogeneous diseases affected by gastrointestinal nerves, burns, inflammatory bowel disease has been noted.53,55–57 Netrin plays a role similar to growth factors, one of the most important proteins involved in the formation of blood vessels by proliferation and their cell migrations, and plays a regulatory role in replacing blood vessels. This mechanism causes the expansion and distribution of the vascular system, and better appreciation can help new blood vessels germinate among ulcerated tissues. In organs such as the legs, where blood flow is low, this protein plays an essential role in preventing tissue death and amputation. 58
In severe wounds that cause damage to parts of the peripheral nerves, the injury does not heal spontaneously and requires special treatment to heal the damaged nerve successfully. In these cases, the most common treatment globally is nerve autografts. Since this method is associated with many limitations, one of the proposed methods for wound healing is the use of proteins that direct axonal conduction and ultimately cause wound healing. It provides a good fit for axon growth and wound healing. In this study, Sf9 cells and BEVS were used to produce Netrin-A. Many prokaryotic and eukaryotic cells are used as hosts to make these proteins. Prokaryotic cell usage is more straightforward, yet this expression system’s most crucial problem is to produce eukaryotic proteins which need post-translational modification (folding, glycosylation, phosphorylation, etc.). This prokaryotic system is not able to perform this. Therefore, eukaryotic class cells such as insect class cells (Sf9, Sf21, and High 5 cells) are used to generate these proteins. The Sf9 class cells (which are from the ovaries of S. frugiperda) can change after translation. These cells grow readily in culture media, free from any compounds of animal origin, and have solid viral security. Insect cell-BEVS is a powerful tool for producing large-scale recombinant proteins. 58 Its medico-pharmaceutical industry uses only basic and theoretical information (bioinformatics). Not much research has been done on making these compounds through chemical synthesis or biotechnology due to their production and biosynthesis’s complexity and high costs. Most recently, alloferon peptides, essential insect-derived peptides in the early detection and suppression of cancer cells and tumors and viral diseases, which are synthesized by chemical methods, were initially isolated from the blue blowfly larvae of Calliphora vicina.43,58 However, the gap of this research in the discussion of extraction and production of effective biological compounds from the larval secretions of Calliphoridae flies, especially L. sericata (a species used in larval therapy) due to the presence of enzymes and peptides effective in wound healing, deemed to be challengeable. Therefore, considering the importance of this issue and the use of insects, especially blowflies, in the production of bio-medicinal compounds, this study aimed to use molecular biotechnology methods and the Sf9 expression system to generate the recombinant protein, Netrin-A, obtained from the larval secretions of L. sericata for use in the medical sciences.
Conclusions
The expression and production of netrin-A in the insect cell using the baculovirus expression system is a very valuable method because the action of post translation modification is carried out in the recombinant protein and the function of the product is preserved. In addition, the use of blowflies as suitable physiological and ecological research models can be contemplated due to their easy breeding, mass production, high bioavailability, and resistance to vulnerable factors related to their necrophagy. The use of these insects in medicine has been considered by many researchers.43,59 Therapeutic proteins are the most prominent new class of products in the biopharmaceutical industry.
Footnotes
Acknowledgements
All those who helped implement this project should be thanked. The respected staff of Shiraz Larval Therapy Center, the respected experts of the Insectarium, and the laboratory staff at Shiraz School of Health are acknowledged. The authors appreciate the Vice-Chancellor for Research and Technology of the Shiraz University of Medical Sciences (SUMS) (Grant No. 98-01-04-19596) and the National Institute for Medical Research Development (NIMAD) (Grant No. 957300) for their helpful contribution and financial support. Futher, the study with ethics code IR.SUMS.REC.1399.1155 is registered at SUMS. This manuscript emanates partly from the PhD thesis of our student, Ms. Masoumeh Bagheri, in the Biology and Control of Disease Vectors group.
Author contributions
The idea, H.A.; methodology, H.A. and M.B.; validation, all authors; formal analysis, all authors; investigation, all authors; resources H.A.; data curation, H.A. and M.B.; writing – original draft preparation, H.A. and M.B.; writing; all authors; review and editing, H.A. and M.D.M.; visualization, all authors; supervision, H.A. and M.D.M.; project administration, H.A. and M.D.M.; All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shiraz University of Medical Sciences: (Grant No. 98-01-04-19596)(21693 new) and the National Institute for Medical Research Development (NIMAD) (Grant No. 957300) granted to the corresponding author, Dr. H. Alipour.
Ethics approval
Ethical approval for this study was obtained from *NAME OF ETHICS COMMITTEE OR INSTITUTIONAL REVIEW BOARD (APPROVAL NUMBER/ID)*.
Shiraz University of Medical Sciences
IR.SUMS.REC.1399.1155
Or
Ethical approval for this study was waived by *NAME OF ETHICS COMMITTEE OR INSTITUTIONAL REVIEW BOARD* because *REASON FOR WAIVER*.
Or
Ethical approval was not sought for the present study because *REASON*.
Or
Not applicable.
Informed consent
Written informed consent was obtained from all subjects before the study.
Not applicable
***
Or
Verbal informed consent was obtained from all subjects before the study.
Or
Written informed consent was obtained from legally authorized representatives before the study.
Or
Verbal informed consent was obtained from legally authorized representatives before the study.
Or
Informed consent was not sought for the present study because *REASON*.
Or
*OTHER DETAILS*.
Not applicable
Or
Not applicable.***
Trial registration
*NAME OF TRIAL REGISTRY: TRIAL REGISTRATION NUMBER*
Or
This randomized clinical trial was not registered because *REASON*.
Or
Not applicable.***
