Abstract
Acoustophoresis is currently in a state of transition from the academic laboratories, moving into the biomedical laboratories and industries. Clear areas of interest are seen in clinical diagnostics and therapeutics, where new approaches to cell handling and purification are emphasized as highly potent areas. This article outlines some of the basic unit operations of acoustophoresis, where applications as cell washing, binary separation, free-flow acoustophoresis, and affinity acoustophoresis are highlighted. The most recent steps to move acoustophoresis into clinical and preclinical applications are also presented.

Introduction
Acoustic forces have been used for decades to manipulate particulate matter, although the major efforts have targeted larger chamber and batch-processing systems. 1 These devices mainly used the acoustic force to agglomerate particles into larger clusters, which eventually sedimented and clarified the surrounding medium. Key areas of application were acoustically enhanced cell sedimentation in cell culturing/fermentation-based processing systems to enable efficient removal of a cell-free supernatant, not having to involve mechanical filters or centrifugation processes. 2,3 With the introduction and development of microelectromechanical systems (MEMS) techniques, new possibilities emerged to reproducibly manufacture acoustic resonators of few of single wavelength dimensions, operating at frequencies (typically the range: 1–10 MHz) that are well suited for cell manipulation by means of acoustic standing wave forces. The precise design and the well-defined fluidic conditions, which are the consequences of microscale structures, that is, laminar flow, allowed improved spatial control of fluid transport and precise definition of acoustic resonant structures, and hence precision in the manipulation of the solid content in a suspension. 4
Particles or cells present in an acoustic standing wave field will experience a radiation force, which will cause them to migrate either to the pressure node or the antinode of the standing wave (Fig. 1). The direction of the movement depends on their inherent physical properties, such as the density,
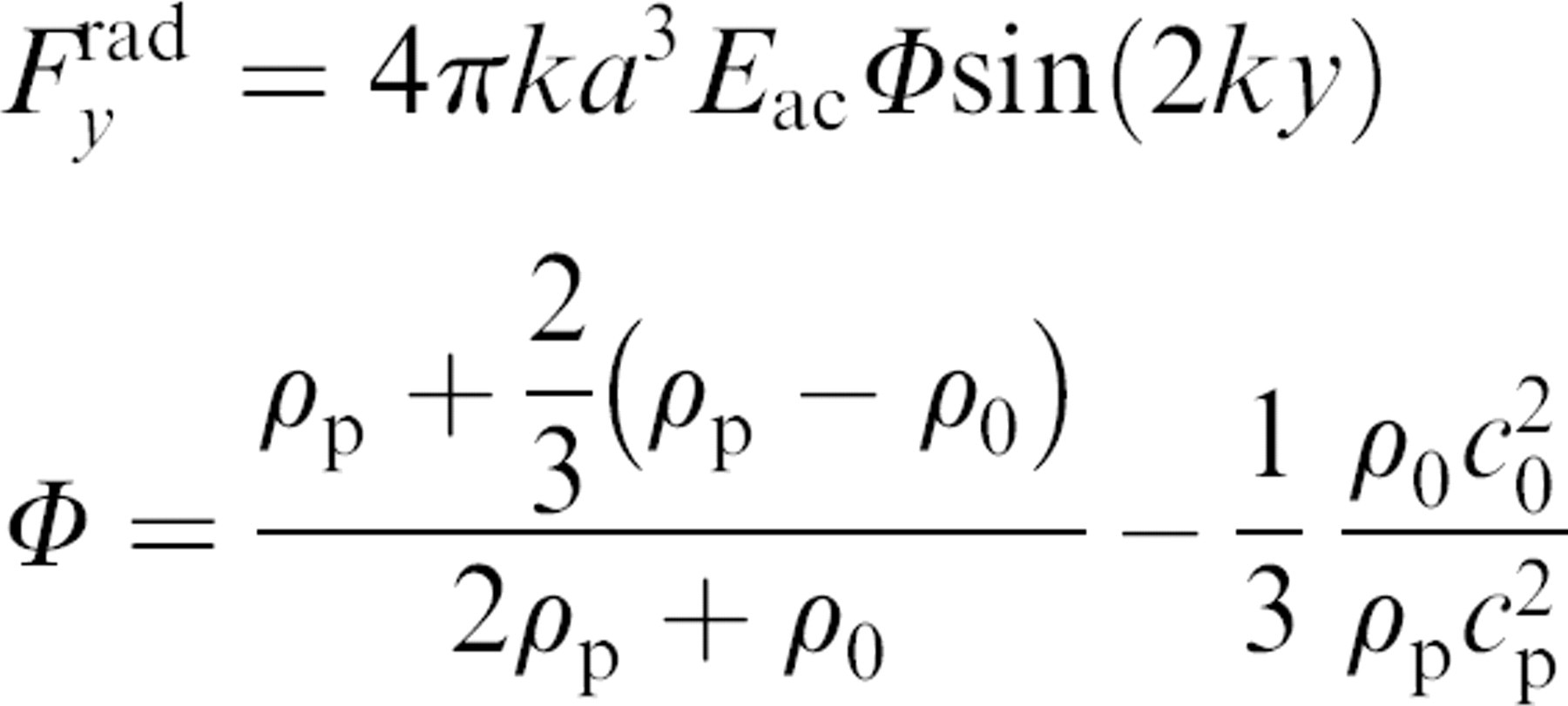
(A) Particles are affected by a radiation force when exerted to an acoustic standing wave. (B) The force is proportional to the size of the particle, resulting in a larger force on the purple bead and smaller force on the green beads. (C) This causes the larger beads to reach the pressure node faster than the smaller ones; (D) however, given enough time in the force field all particles gather at the pressure node eventually.
Although multiple nodes can be well defined and operated in microfluidic resonators, 5 –7 most microacoustofluidic systems use a single pressure node standing wave system where a pressure node is present in the center of the channel and pressure antinodes are present along the channel walls. The advantage of this setup is the ease of dividing the flow using flow splitters. This will generally result in a center branch for concentrated sample with two other branches for excess fluid transport from each side of the channel. One of the earlier designs with this configuration was designed by Hawkes and Coakley in which they used a layered stainless steel resonator. 8 Despite having a flow channel in the microscale domain, the device itself was not fabricated using standard MEMS techniques, but rather conventional machining techniques. The first silicon-integrated acoustophoresis structures were reported in the early years of the past decade. 9,10 They were both designed with a flow channel in silicon, having a glass lid anodically bonded to seal the flow through resonator.
The combination silicon–glass works very well acoustically for two reasons. Firstly, the electrochemical bond between the silicon and glass provided by the anodic bonding process is a tight and reproducible bond, free of adhesives such as glue that can distort the flow channel and hence affect the intended design. Glue also has the tendency to absorb acoustic energy, which lowers the conversion efficiency from the actuator to the channel resonance. Also, irreproducible glue layer thickness strongly influences the resonator performance and reproducibility. Secondly, both glass and silicon have quite high difference in density compared with the liquid in the channel, which in turn gives a good reflective index that is suitable for defining an acoustic resonator solely by its geometrical design. The crystalline structure of silicon allows structures of very precise dimensions and high-aspect-ratio vertical walls through anisotropic etching, which is suitable for standing wave acoustics. However, recently it has been showed that the vertical walls are not a prerequisite for the standing wave resonances to work, as glass channels that have a semicircular cross section obtained in the isotropic wet-etch process have proven to have the equal performance to the vertical wall silicon resonators. 11 Both simulations and confocal microscope measurements confirm similar behavior in glass channels as in silicon channels.
The advantage of an all glass-based acoustophoretic chip is a simplified fabrication process and substantially less expensive material costs. Figure 2 shows the trifurcation outlet of a wet-etched glass-based acoustophoresis chip.

Flow channel isotropically wet etched in glass. The cross section is not rectangular but semicircular. However, it has been shown that vertical walls are not a prerequisite for acoustic standing waves to form.
Applications
Blood Washing in Major Surgery
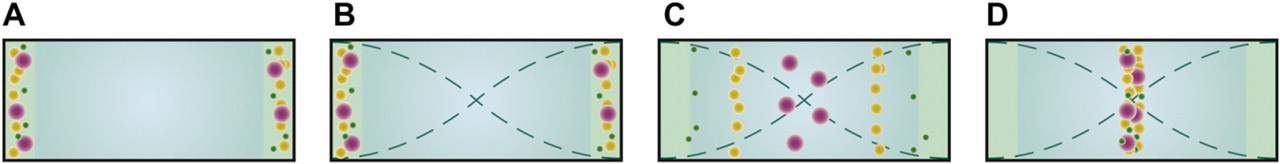
A most fundamental acoustophoresis structure is a straight separation channel with one inlet that ends in a trifurcation. When actuated in a half wavelength, standing wave mode particles will gather in the center of the channel and in combination with the laminar flow, a single band of particles will form (Fig. 3A). The entrained particles will exit through the central branch, whereas the cell-free medium will exit through the side branches. A concentrated particle suspension is accomplished because carrier medium has been removed through the side branches. Besides operating as a concentration device, the design can be used to separate particles, that is, if two particle types display different sign on the acoustic contrast factor (Fig. 3B). These circumstances were used by Petersson et al., 12 where lipid particles were separated from human erythrocytes. They targeted the need for efficient removal of lipid microemboli contaminating shed blood recovered during cardiothoracic surgery. Unless the lipid particles were removed from the shed blood before blood retransfusion, they would cause emboli in all major organs, being especially critical for the brain. Data on the recovery of erythrocytes of 95% and a lipid depletion of 95% opened up the possibility of developing acoustophoresis for autologous blood washing before retransfusion. 13 To increase volume throughput, a multichannel chip with eight parallel channels driven with the same ultrasonic transducer was demonstrated. 14
(A) Green particles are focused by the acoustic standing wave. As particle free medium is removed to the sides, a concentration of the beads is accomplished. (B) Particles with different acoustic contrast factor can easily be separated from each other as the yellow particles with negative contrast factor moves toward the pressure antinodes at the channel walls, whereas the red particles with positive contrast factor gather at the central pressure node. (C) The magnitude of the acoustic radiation force is size dependent, resulting in the larger purple particles focusing faster toward the pressure node than the smaller green ones. By tuning the flow rates such that the green particles do not have the time to travel to the pressure node, a size fractionation is enabled.
Since the acoustic radiation force scale with the size of the particle, it is possible to use this to fractionate particles by size. By adding two extra inlets to the previous trifurcation channel, the input sample can be laminated toward the channel sidewalls (Fig. 3C). 15 When the particles subsequently are exposed to the standing wave, the largest particles will move toward the central pressure node much faster than the small particles. This was used by Thevoz et al. for cell cycle synchronization of mammalian cells. 16 The acoustic radiation force acted stronger on the larger cells (e.g., those in G2/M and S phases) and directed them to the central stream more rapidly compared with the smaller cells (e.g., those in the G1 phase). This enabled them to receive a fraction with 84% synchronized cells in the G1 phase.
Thrombocyte Depletion
Dykes et al. 17 demonstrated the use of acoustophoresis to separate platelets from apheresis product. In the preparation of peripheral blood progenitor cells from apheresis product, platelets are an unwanted side effect when the apheresis instrument continuously separates the buffy coat from the rest of the blood. Two negative aspects follow from the platelet contamination of the apheresis product: First, the donor loose platelets, and secondly platelets interfere substantially with the subsequent immunomagnetic separation process used to extract the blood progenitor cells from the collected apheresis product. Acoustophoresis can be used to solve this problem using a similar chip design as in Ref. 15, where sample enters the channel at the side inlets and clean buffer enters centrally, Figure 3C. The smaller platelets were left practically unaffected by the acoustic standing wave and proceeded with the laminar flow along the channel sidewalls, whereas the larger leukocytes and the few erythrocytes present in the apheresis product were focused in the center of the channel and exited through the central outlet. Results showed a median of 98% of the leukocytes recovered, whereas 89% of the platelets were depleted.
Circulating Tumor Cells
Enumerating circulating tumor cells in clinical samples from cancer patients have in recent years become an important diagnostic tool of metastatic state of cancer and also most importantly the number of circulating tumor cells per blood volume can be monitored early during treatment as an indicator of ineffective or successful treatment. The golden clinical standard for measuring the number of circulating tumor cells is done by immunoaffinity extraction of EpCAM positive cells in peripheral blood and subsequent staining protocols to identify metastatic cells by fluorescence imaging. 18,19 More recently, microchip approaches that use EpCAM–immunoaffinity interaction in large surface area flow through chips have been proposed as alternatives to the Veridex system. 20
Circulating tumor cells can also be extracted from blood by means of acoustophoresis, where the differing acoustophysical properties of circulating tumor cells are explored as the basis for separation from other peripheral blood cells. A first effort to capitalize on this aspect was recently reported by Augustsson et al. 21 and Magnusson et al., 22 where the basic setup for cell washing, Figure 3C, was used to extract circulating tumor cells from blood cells. The larger cancer cells are acoustophoretically driven into the center stream by the radiation force, whereas white and red blood cells do not migrate as far in the acoustic field and thus exit through the side outlets. A most important fact in this respect is that the cell separation is done without any labeling and hence no bias in the selection of circulating tumor cells is induced in regard to epitope-specific binding. It was also shown that cells that pass through the acoustic separator did not display any change in viability, and thereby the route to cell culturing after separation is open.
Cell Sample Processing for Flow Cytometry
The acoustophoresis configuration in Figure 3C is especially well suited for removing dissolved substances, that is, medium exchange of the suspended cells. The cells enter via the side inlets and a clean buffer is delivered in the center flow path of the acoustophoresis channel. When actuating the chip, cells are translated orthogonally to the flow into the clean buffer solution in the channel center. This has recently been explored in an application targeting pretreatment of cell samples before flow cytometry analysis. 23 When analyzing cells using flow cytometry, the use of fluorescent labeling is a standard procedure. However, the labeling process always involves excess fluorescent label, which does not bind to any cells and contributes to increased background fluorescence. If such free label can be removed, the sensitivity in analyzing the weakly stained cells could be increased. Commonly this can be accomplished by introducing additional manual centrifugation steps, at the cost of increased analysis time and risk of sample-handling errors. By acoustically switching the cells into a clean buffer, 99% of the unbound label could be washed away while keeping the cell subpopulation intact. Additionally, cell debris from the lyzing procedure of the red cell lysis of the blood sample could be decreased as most of the debris are too small to be affected by the radiation force.
Bacteriophage Selection
Generally, there is a size limit to when a particle becomes affected by the radiation force of about a micrometer. Below that limit, the force is so weak that it cannot counteract other fluidic forces such as acoustically induced streaming of the fluid. One way of getting around this problem is to use affinity beads that bind to the small particle or molecule that is desired. This was used by Persson et al. to facilitate automation of phage display selection. 24 Phage display is a routine technique to select bacteriophages from large phage libraries that genetically encode for a vast variety of affinity binders, each expressed at the surface on a given bacteriophage. A well-designed library in principle holds affinity binders for every target molecule possible. By exposing the library to, for example, microbeads coated with an antigen for which a good affinity binder is sought and subsequently extract and wash the beads, a bead surface bound population of bacteriophages that encode for good affinity binders against the antigen is accomplished. By expanding the captured population and subsequently repeating the selection process, a highly specific set of bacteriophages against the target antigen can be extracted and expressed recombinantly for industrial production. A key step in this process flow is the successful extraction (low loss and low background contamination) of the beads with the captured bacteriophages. This is commonly done by centrifugation and/or magnetic capturing of the beads while removing the supernatant—a process that is repeated a number of rounds for sufficient reduction of background. Many times the loss of beads in this case results in a bacteriophage population with lost potential binders.
Acoustophoresis have recently been shown to successfully perform this bead extraction and washing process with a good yield of specific affinity binders. An antibody against a grass pollen allergen was desired and 2.8-μm microbeads were coated with the Phl p 5 allergen and subsequently incubated with a phage library. The bacteriophage/bead suspension was injected via the side inlets into an acoustophoretic wash chip (Fig. 3C) and beads with bound bacteriophages were translated into the clean buffer solution in the center, whereas the unbound bacteriophages were too small to be affected by the acoustic force and remained in the flow along the sidewalls, leaving the chip via the side outlets. To increase the wash efficiency, the acoustophoresis chip was designed such that the washed beads suspension for the first wash step entered into a second identical wash step before a purified bead fraction was recovered. The extracted bacteriphages were subsequently tested in an ELISA assay against Phl p 5 showing an increase in assay sensitivity after one acoustophoretic wash sequence of about three orders of magnitude. This matches well what was accomplished if the corresponding manual wash procedure was performed. Clonotypes obtained after only one selection round displayed a highly specific readout when matched to related allergens. The acoustophoretic phage display separation method offered the same performance as conventional techniques, although the processing is done in a continuous flow and in minutes, without any sample-handling intervention or sample loss. The proposed acoustophoretic phage display extraction platform opens the route to rapid and qualitative development of new affinity binders, for example, diagnostic immuno assays and multiplex immuno microarray assays.
Separation of Complex Particle and Cell Mixtures
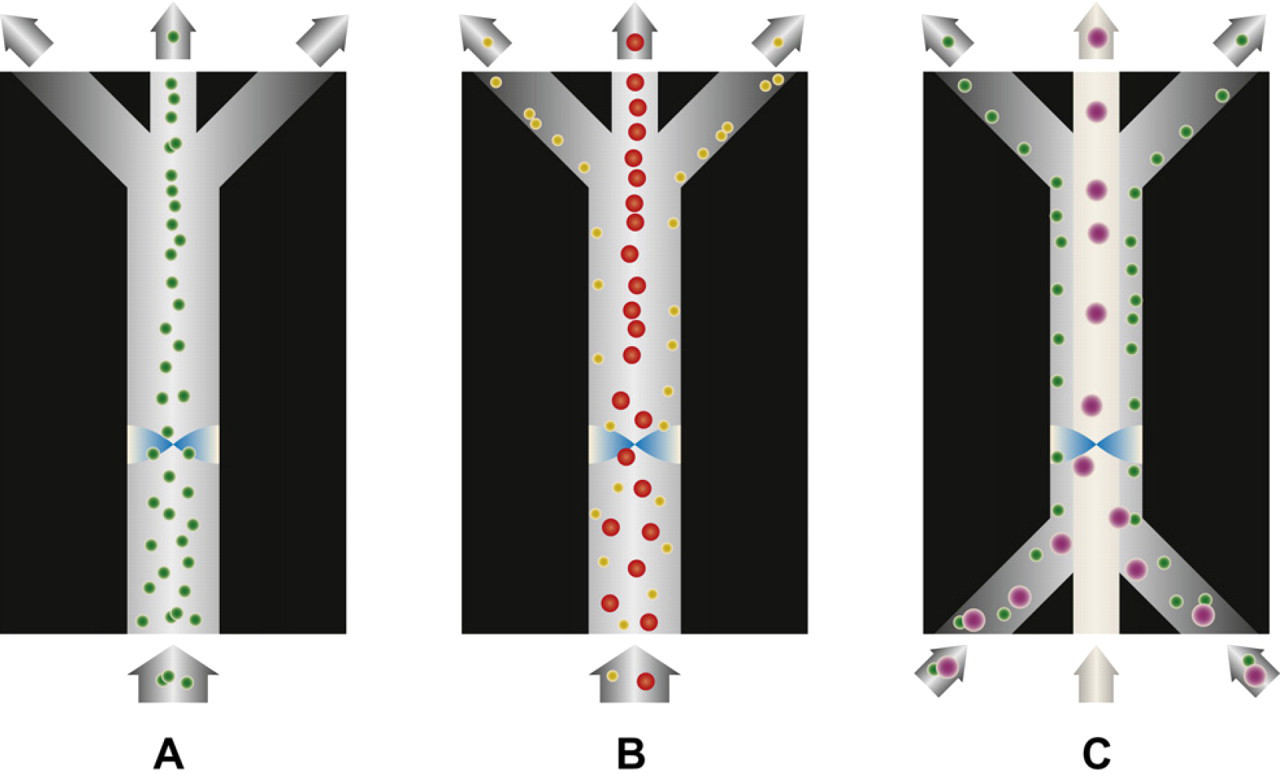
To fractionate a suspension containing multiple particles of different sizes, a modified chip design with multiple outlets was reported by Petersson et al. 25 As the particles are affected by the primary acoustic radiation force, a symmetrical size gradient is created with the largest particles reaching the center of the acoustophoresis channel before the center exit and the smallest remain along the sidewalls. By tuning the flow rates such that only the largest particles actually reach the pressure node, it is possible to collect that fraction at the center outlet. Because of the laminar flow, the following flow splitter will divert the second largest particles and the process is repeated. Ideally, all particles end up in a fraction of their own size, Figure 4. This mode of operation was called free-flow acoustophoresis, FFA. FFA was used for separating blood components into samples containing enriched fractions of erythrocytes, leukocytes, and platelets. 25
Fractionation of multisize particle suspension. The flow rate and the acoustic radiation force are balanced such that only the largest particles exit through the central outlet, the second largest to the next outlet, and so on. The fractionation is possible because of the size dependency of the radiation force.
Acoustophoretic Sample Preparation for Biomarker Discovery
Analogous to the described affinity bead-based bacteriophage selection, molecular species can also be selected and purified in a continuous flow format, using the acoustophoretic wash strategy as in Figure 3C. Augustsson et al. 26 showed that a dissolved color compound (Evans blue) could continuously be removed from a bead population (0.05% wt) to 99.995%. As can be expected, it was also shown that the wash efficiency was reduced linearly with increasing bead concentrations. This was explained by the convective transport of the contaminant in the stagnant boundary layer around the bead into the clean buffer zone. In that paper, this mode of bead washing was also capitalized on in an application of decomplexing proteomic samples before mass spectrometry. Examples of specific phosphopeptide extraction from a complex protein digest background was reported, using metal oxide affinity capture beads (MOAC). After the acoustophoretic washing, the analytes captured by the MOAC beads were eluted and detected by MALDI mass spectrometry. The setup opens up the development for automated process flows for proteomic sample processing before MALDI readout without having to transfer samples in several steps by means of pipetting.
Plasmapheresis for Online Diagnostics
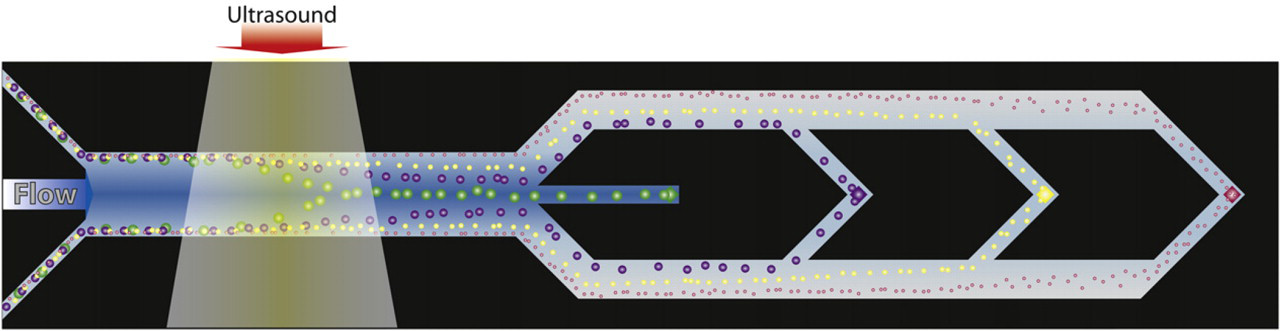
Acoustophoresis lends itself very well to deplete suspensions of particles or cells as long as the concentration of the solid content is not too high. A rule of thumb is to work at particle concentrations well below 10% per volume. However, in accordance with most microfluidic systems that handle particles, acoustic systems are also challenged by clogged systems and occluded channels when the particle concentration increases. This makes for instance plasmapheresis from whole blood where the solid content is up to 50% of the volume, a challenging task. However, by introducing a longer acoustophoresis channel, allowing the cells to be increasingly focused along the channel in combination with sequential outlets placed in the channel bottom perpendicular to the flow, blood cells can gradually be removed, reducing the solid content load on the separation system. 27 As the bottom outlets are placed in the channel center where the cells were focused, a sequential removal of acoustically concentrated cells was accomplished while an increasing fraction of pure plasma was received along the sidewalls, which grow toward the center as more cells are removed. In the final trifurcation outlet of the chip, the few cells that are left exit through the center outlet while cell free plasma can be collected through the side branches, Figure 5. Cell counting of the plasma showed that the number of cells present was below the clinically defined cell concentration limit recommended for transfusion by the Council of Europe, proving that the plasma matched clinical grade.
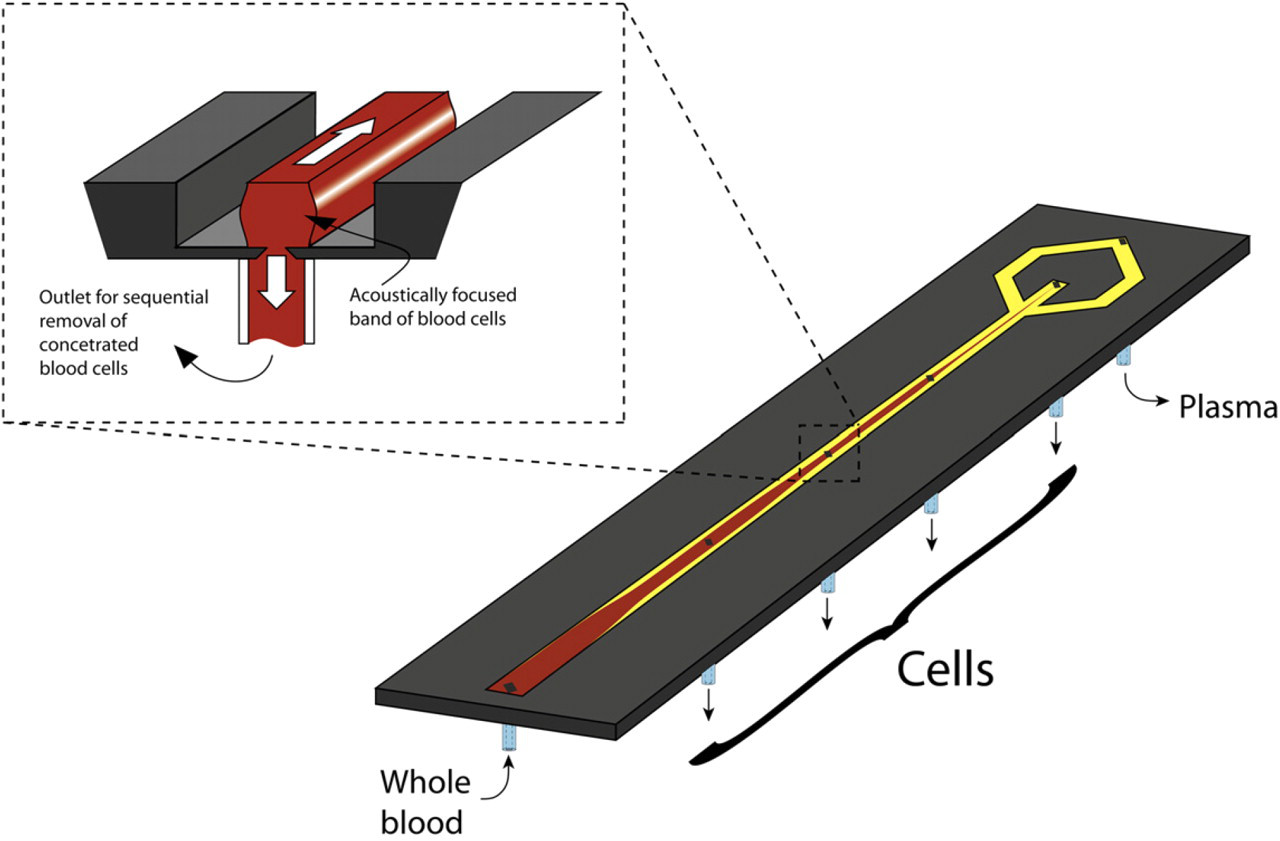
Acoustophoretic plasmapheresis principle. The acoustically focused cells are removed through a sequential exit holes located in the center bottom of the channel. The cell concentration is thus gradually lowered until cell free plasma is obtained through the side branches of the trifurcation.
The clinical performance of the generated plasma was also shown by processing blood samples that contained various levels of prostate-specific antigen, the most widely used biomarker for prostate cancer. It was shown that the acoustophoretically extracted plasma delivered consistent data when compared with routine clinical analysis of the blood samples.
Outlook
Acoustophoresis offers a robust and high-throughput cell processing and is in principle independent of ionic concentration and pH why a more generic approach to system design can be followed in life science applications when compared with most other microfluidic cell separation techniques.
Acoustophoresis offers new means to process and handle cells in an efficient way without inducing any cell damage, where buffer exchange, cell washing, size separation, affinity separation, and concentration are all fundamental modalities of acoustophoresis.
Especially clinical applications in cell handling are foreseen to emerge from the ongoing research in the field, where both diagnostic and therapeutic tools are anticipated. Current clinical developments within our own group are executed in our research environment CellCARE (www.cellcare.lth.se), where stem cells, blood components, and tumor cells are key target areas. We currently also see a rapidly increasing interest from industry, and commercial products have already entered the market as reflected in the acoustic flow cytometer, Attune, by Life Technologies.
Footnotes
Acknowledgments
Financial support is acknowledged from Swedish Research Council (VR 621-2010-4389), Vinnova Innovationer för Framtidens Hälsa—CellCARE, Formas—TvärLivs—Dnr.: 222-2010-413.
