Abstract
The 96-well microplate is a ubiquitous tool in the laboratory; its use is so extensive that in a limited number of situations it can be restrictive. Consider the situation where 96 samples need analysis or a downstream process in which the 96-well format leaves no space for additional standards or controls in the upstream 96-well processing. Consequently, plates are split or sample number reduced thereby incurring additional cost for plates, reagents, standards, controls, sample tracking, data files, and time to analyze the entire plate. A simple solution is proposed with the development of a companion 8 times 13-array microplate. The 104-well microplate was developed within the American National Standards Institute/Society for Biomolecular Science standards as to plate geometry and dimension, including well spacing (9 mm) with the exception that the columns have been shifted 4.5 mm to the left to accommodate the 13th column. The extra column allows for additional standards/controls without modifying chemistry, incorporating additional plates or changing to a 384-well plate. We show negligible difference (−0.0003 optical density) when comparing mean absorbance readings in 96- and 104-well format. We demonstrate use of the 104-well plate in a 96-well environment by incorporating it in an enzyme-linked immunosorbent assay on a standard liquid handler. Results from the assay show no difference between formats (y = 1.039x − 0.004, r = 0.997). Although the 104 plate was not created to supplant the 96-well standard, we conclude that the 104 plate can be incorporated into the 96-well environment without significant change in existing systems.
Introduction
Microplates, an essential tool of the laboratory, have changed little over the last 60 years. The first “microplate” was made from polycarbonate sheet measuring 8 × 95 × 130 mm with 6-mm diameter wells machined in an 8 × 12 array. These trays were developed by Gyula Takatsy as part of a microtitering system and became commercially available in a resource limited post-World War II Hungary. 1 Evolution of the contemporary disposable microplate was a result of independent work by John Liner, John Sever, Nelson Cooke, and others through the late 1950s. 2 Since that time, numerous companies have developed plate styles and format each independent of the other. Dimensional inconsistencies between plates from the various manufactures made their incorporation into automated platforms difficult. In the late 1990s, the Society for Biomolecular Sciences (SBS) began standardizing microplates. By 2004, microplate standards were finalized through the American National Standards Institute (ANSI) for geometry and dimensions to enhance their integration into automated instrumentation. 3 –6 Today, the 96-well microplate is ubiquitous and has been used for sample archiving, 7,8 reagent handling, 9 extraction, 10 –12 and analysis. 13,14 The acceptance of the microplate is such that when assays are developed, its limits are second nature. For instance, consider an assay where 96 samples or compounds are archived, processed, or analyzed. To accommodate the need for standards and controls, the sample number is either reduced, split to multiple plates, standards run on a second plate, or chemistry scaled down for a higher density well plate. This incurs extra costs of additional plates, reagents, standards, controls, sample tracking, and time to complete a 96-sample assay.
We present the development of an 8 × 13 array, a 104-well microplate to answer these concerns. The extra column increases the plate capacity by 8% allowing for additional standards or controls without modifying chemistry, multiple plates, plate well density, or multiple data file manipulation. The plate was developed within the ANSI/SBS standards as for plate footprint, height, and flange dimensions. We present the functionality of the 104 plate in a clinical laboratory using a semiquantitative ELISA (enzyme-linked immunosorbent assay) Helicobacter pylori assay as a model.
Methods
Samples
Analyzed clinical H. pylori assay plates were ansalyzed using both 96-well and 104-well formatted plates. Deidentified clinical serum and ethylenediaminetetraacetic acid (EDTA) plasma samples originally submitted for H. pylori testing were handled according to a global ARUP protocol under Institutional Review Board #7275.
The 104-Well Plate
The 104-well plate was developed within ANSI/SBS standards, with the exception that the orientation well (A1) was placed 9.88 mm from the plates left edge, leaving enough room for a 13th column (Fig. 1). The clinical H. pylori assay was performed in Nunc's 12-position frame containing MaxiSorp Immuno Modules (aka, eight-well strips) (Thermo Scientific; Waltham, MA). For this comparison, a similar frame was designed in SolidWorks (Dassault Systems Solid-Works Corp; Concord, MA), which holds 13 modules and was 3D printed out of acrylonitrile butadiene styrene (ABS) at 0.18 mm resolution on a Stratasys Dimension 3D Printer (Phoenix Analysis & Design Technologies; Tempe, AZ) 15 (Fig. 1).

Dimensions of the 104-well microplate based on ANSI/SBS standard definitions. A. A standard commercial 96-well plate and the 104-well plate are shown side by side. The 104-well plate was 3D printed with standard external dimensions of 127.76 × 85.48 × 14.35 mm. Differences between the two plates are shown. Placement of column 1 for a standard plate is 14.38 mm from the left edge. Column 1 of the 104-well plate has been shifted 4.5 mm so that it is located 9.88 mm from the left edge, which allows for the 13th column. Well and plate dimensions, spacing, and geometry are the same for both plates. B. The 3D-printed frame designed to hold 13 of Nunc's Immuno Modules. These modules (aka 8-well strips) fit internal to the frame on 9-mm centers. The corner notch is visible for plate orientation. SBS, Society for Biomolecular Science.
Liquid Handling
The 104-well plate was tested on the Freedom EVO liquid handler (Tecan; Durham, NC) equipped with standard Tecan accessories and software. It has a LiHa (Tecan, Durham, NC) pipetting arm for independent 8-channel pipetting, a MCA96 (Tecan, Durham, NC) pipetting arm for 96-well pipetting, a HydroFlex plate washer (Tecan, Durham, NC), and a Safire microplate reader (Tecan, Durham, NC). Software control of the HydroFlex used HydroControl V2.0 and the Safire 2 used Magellan V6.4. Overall programming of the Freedom EVO and integration of its auxiliary devices was controlled with Freedom EVOware V2.2. With the exception of the 104-well frame, standard consumables were used to perform the H. pylori assay.
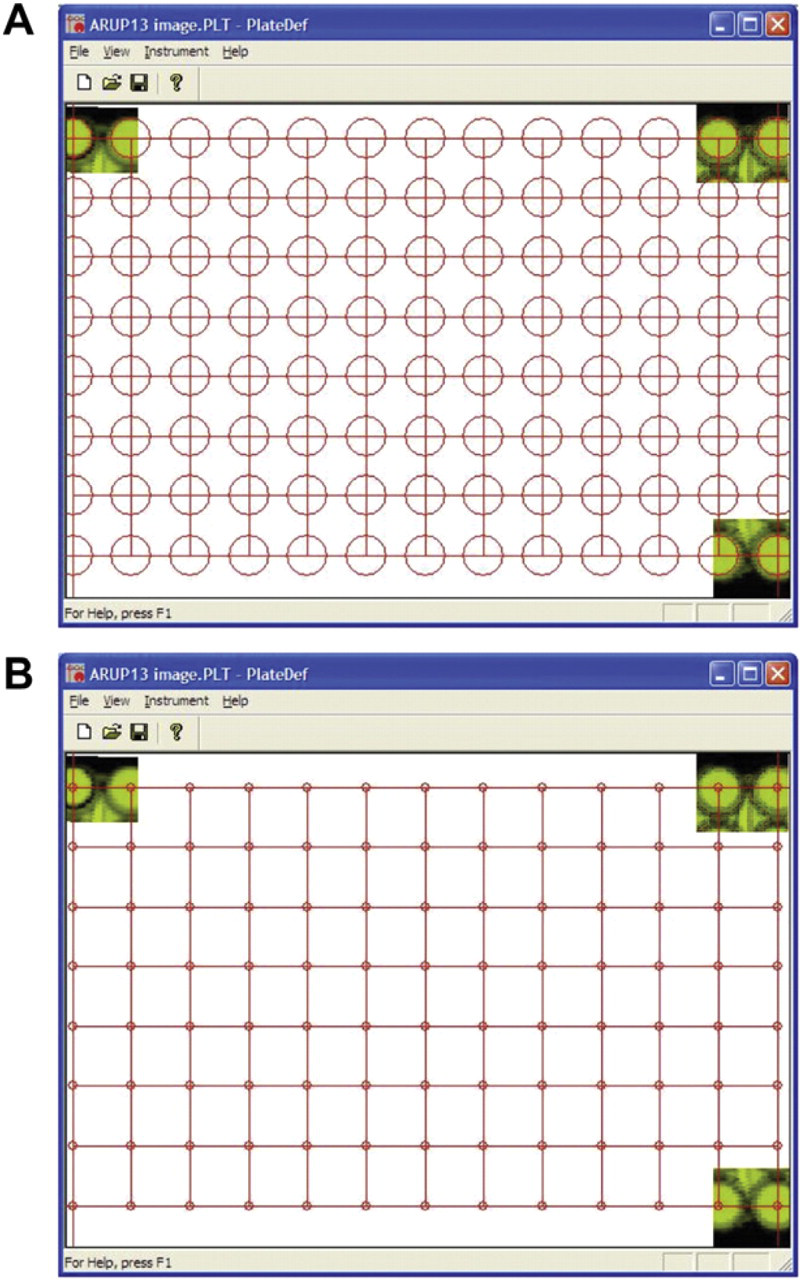
Tecan script modifications. Screen shots of the PLT file used to set the positioning of the absorbance optics on the Safire 2 for the 104-well plate. A. When well centers were defined with 6-mm circles, absorption measurements were not obtained. The truncated circles failed to define the well centers. B. When the centers were defined with a 1.5-mm circle, absorption measurements were obtained. The circles define the center of the well not the reading area.
Absorbance readings of the 104-well plate were compared with those of the standard 96-well plate on the Safire 2 . A diluted solution of, “Range A” (Artel; Westbrook, ME), with an absorption value of 0.5 optical density (OD) at 730 nm was distributed across 13, 8-well Immuno Modules at 200 μL per well. These modules were read in both 96- and 104-well formats. To reduce variation after reading the 96-well plate, the modules in columns 1–6 were transferred to their corresponding positions in the 104 plate and the modules in columns 7–12 were transferred to columns 8–13. Column 7 received an additional module filled with dye but was not included in the statistical analysis, which was performed in Excel (Microsoft; Redmond, WA).
To assay for H. pylori antigen; patient serum, calibrators, and controls were initially diluted 1:101 in 150 mM sodium chloride, 5 mM Tris, 45 mM Tris HCl, 0.05% ProClin 300 (Sigma-Aldrich; St. Louis, MO), 0.05% Tween 20, and 3% StabilCoat pH 7.5. Then, 100 μL of the dilution was transferred to the wells and incubated at room temperature for 35 min. The wells were washed 4 times in 350 μL of wash buffer, then 100 μL of Immunoglobulin G-anti-horseradish peroxidase (IgG-HRP) conjugate (Jackson ImmunoResearch Laboratories; West Grove, PA) was added to each well and incubated at room temperature for 35 min. The wells were washed again as previously stated, then 100 μL of 3,3',5,5'-Tetramethylbenzidine (TMB) substrate solution was added to each well and incubated at room temperature for 30 min. After incubation, 100 μL of 1N sulfuric acid was added to each well to stop the reaction. Absorbance was measured for all wells at 450 nm with 650 nm used as a reference wavelength within 5 min of addition of the sulfuric acid.
Daily clinical assay plates were obtained for end point absorbance analysis over 5 days consisting of 344 patient samples. After completion of the assay, absorbance readings were obtained on the ESP600 (The Binding Site; San Diego, CA). The plate was then removed and the Immuno Modules reformatted into the 104-well frame for reading on the Safire 2 . All absorbance measurements were completed within 5 min of stopping the reaction.
When the H. pylori assay was performed on the Freedom EVO, standards and controls were placed in column 1 with contiguous samples from column 2 through 13. The absorbance measurements were captured using the Magellan software and exported in a tab delineated Excel compatible file. Data from the Excel spreadsheet were transferred to Soft-Max Pro V.4.8 (Molecular Devices; Sunnyvale, CA) for calculation of the H. pylori antibody concentration in ELISA values (EVs). The software is capable of analysis in 96- or 384-well formats; for analysis of the 104 formatted data, the 384-plate definition was used.
Results/Discussion
The absorbance profile across the 96- and 104-well plates using the Artel dye is shown in Figure 3. Similar patterns are seen in both plate formats. The mean (standard deviation) of the absorbance across the 96-well plate was 0.5864 (0.006) OD and across the 104-well plate was 0.5861 OD (0.006); a difference of −0.0003 OD between the plates. The modules were then transferred back to the 96-well frame and read the second time. The second reading of the 96-well plate had an average absorbance of 0.5826 (0.006) OD with a difference between the two 96-well readings of 0.0039 OD.
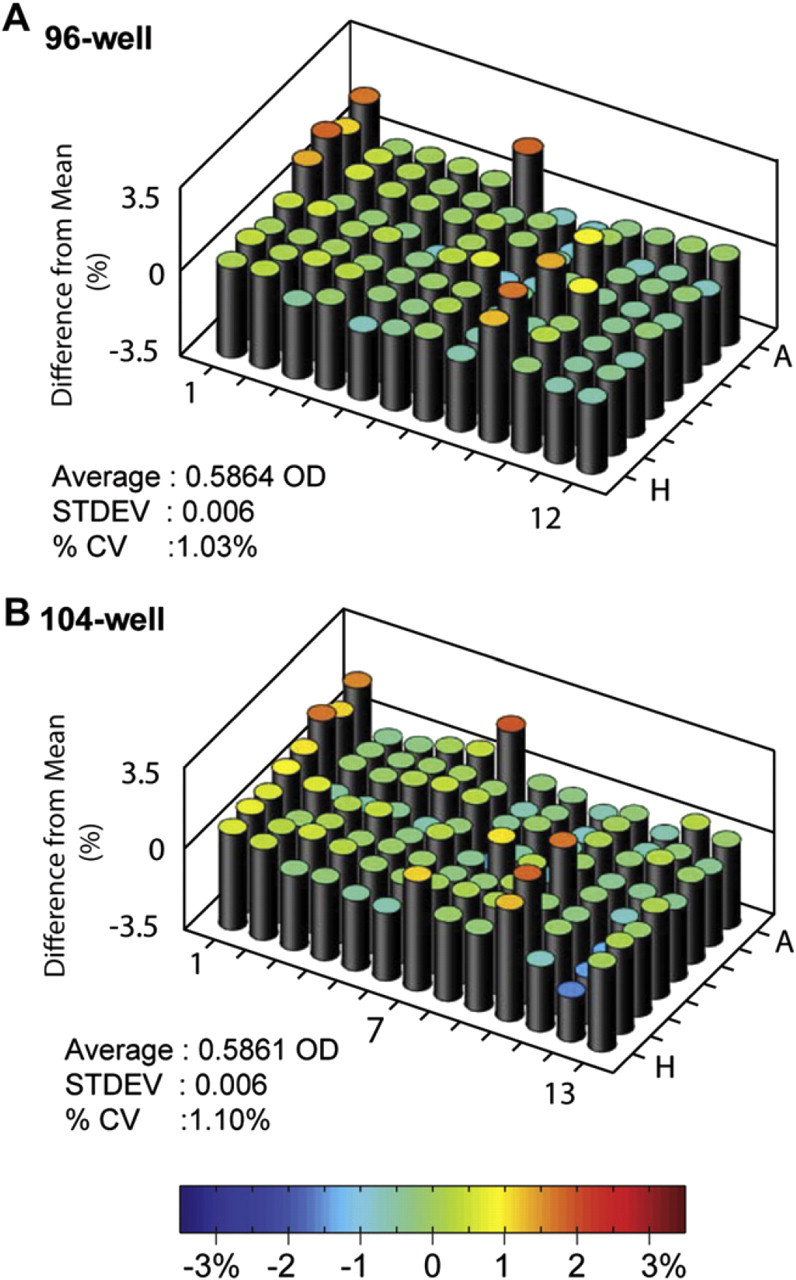
The observed absorbance characteristics of 96-well and 104-well microplates. The percent difference between the observed absorbance and the plate average is shown for both the 96-well and the 104-well plates. A. The 96-well plate had a mean absorption of 0.5864 OD within a range of 0.5683 OD (-1.02%) to 0.5966 OD (1.81%). B. The 104-well plate had a mean absorption of 0.5861 OD within a range of 0.5675 OD (-1.56%) to 0.6017 OD (1.86%). Column 7 was not used in statistical analysis.
Absorbance readings from the completed clinical H. pylori assays on the ESP600 in 96-well format and those obtained on the Safire 2 in 104-well format also show no difference between the two reading formats (Fig. 4a). No bias was observed between the two formats (m = 1), and the offset was minimal (b = 0.004) with excellent correlation (r = 0.9998).
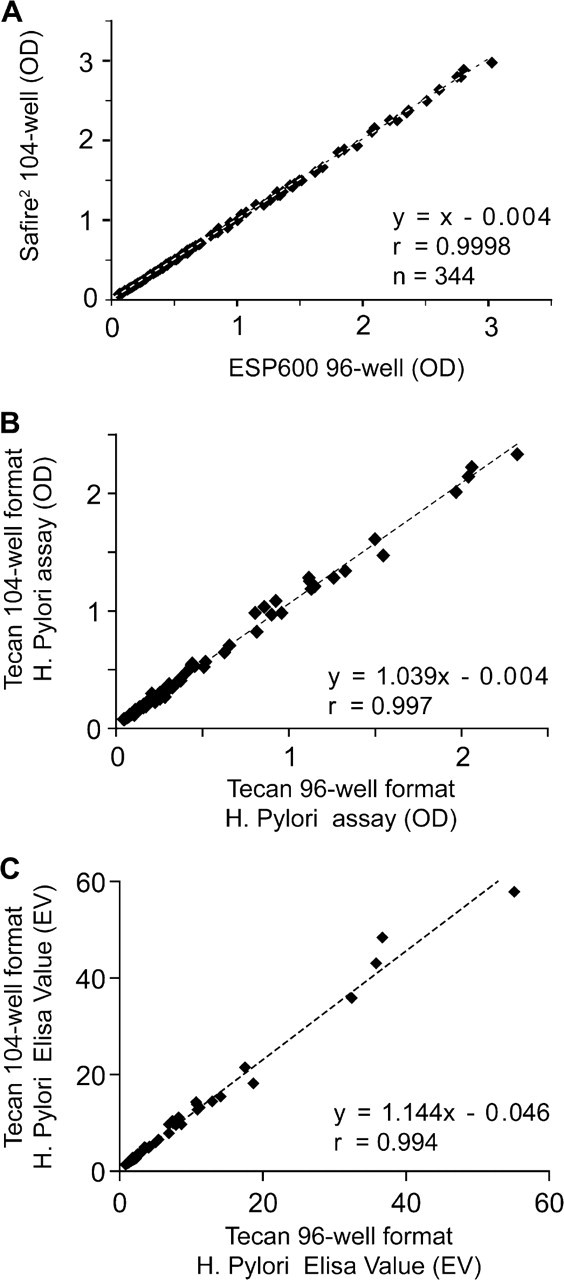
Comparison of the 96-well and 104-well with the Helicobacter pylori assay. Comparison of absorption and ELISA value results for the H. pylori assay. A. The absorbance comparison in OD values of completed assay plates obtained from the ESP600 in 96-well format and that in 104-well format on the Safire 2 . Little difference is observed between data sets as shown with a slope of 1 and minimal offset of 0.004. The correlation between the two methods is excellent with an r value of 0.9998. B. Correlation comparison for the H. pylori assay ran simultaneously in both 96- and 104-well format on the Freedom EVO. Some bias is observed in the absorbance comparison with a slope of 1.039 and an offset of −0.004, the methods still correlate well with an r value of 0.997. C. The calculated ELISA value for H. pylori reflects a similar pattern as the absorbance results but with more bias for the 104-well plate with a slope of 1.144 and an offset of 0.046. The correlation is good with an r value of 0.994.
Results of the H. pylori assay ran simultaneously in both 96 and 104 formats are compared in Figure 4b. Correlation of the absorbance between the two methods had an r value of 0.997 with minimal bias (m = 1.039) and offset (b = −0.004). When the absorbance values of the two methods were converted into EVs, a similar correlation (r = 0.994) was seen (Fig. 4c).
The traditional response to having insufficient well density is to transfer the test to the next highest plate type, for example, a 96- to a 384-well plate, or to use an additional plate of the same size. For many procedures, this is a proper response. However, in some situations these options are not ideal. For instance, our laboratory routinely tests an excess of 20,000 patient samples per month for Vitamin D 1, 25-diOH. This assay consists of solid-phase extraction followed by an 125I-based radioimmunoassay (RIA). The samples are initially prepared on a liquid handler and then extracted simultaneously along with the standards and controls in 96-well–formatted 1-mL columns. The samples are returned to the liquid handler where they are split into two duplicate racks of tubes for the RIA. The RIA procedure introduces two more controls into the assay; total count and nonspecific binding. Two options now exist to incorporate these controls and to keep the process automated. The initial sample count could be reduced by two for the extraction freeing up space in the racks used for the RIA. Alternatively, the extraction sample count can be left unchanged and the number of racks doubled for the RIA. The first option was undesirable because it increased the number of runs required to process the same number of samples, in our situation nearly a full day's work for every 30. The second option was also undesirable because it doubled the RIA tube racks to four, thereby increasing the complexity of the downstream manual manipulations and chances of human error. The 104 format was developed out of a need to simplify this process, 96 samples could now be processed and analyzed with the addition of standards and controls without breaking the initial plate map.
The 104 plate was developed within the ANSI/SBS standards to maintain automation compatibility. Most contemporary instrumentation is not restricted mechanically. By modifying one rule, additional well formats can become compatible within the ANSI/SBS system. Obvious extensions of the 104 plate include similar 96-well plate extensions to 384- and 1536-well plates, and 416- and 1664-well formats, respectively. Within the microplate footprint, other well variations are possible. The maximum array size without changing the well size is 9 × 14 or 126 wells. The wells would be positioned close to the outer limits of the plates; 3 mm of the top/bottom and 2 mm left/right. Most instruments, either automated or manual, are designed for either 8- or 12-position processing; making plates differing in both standards difficult. Other workable options include plates of mixed formats. For instance, a single plate could consist of a column of 8 wells or 2 large wells in addition to the adjacent traditional 96, 384, and 1536 wells. The plate could also consist of alternating columns of 96- and 384-formatted wells (8, 32 × 6 arrays). The final plate design being driven by the needs of the test not plasticware availability.
Although commercial alternative formatted consumables do not exist, custom hardware can be easily created. For example, pipette tip racks were made to hold three sets of 8 × 13 arrayed disposable tips, freeing deck space used by the second tip storage location. Custom-arrayed tube racks retaining microtubes can be made and used as a deep well-plate alternative. Microplates for absorbance, fluorescence, and luminescence assays can be configured from commercially available 8-well strips and custom-made frames. Standard SBS compliant components can be integrated to form custom-formatted consumables.
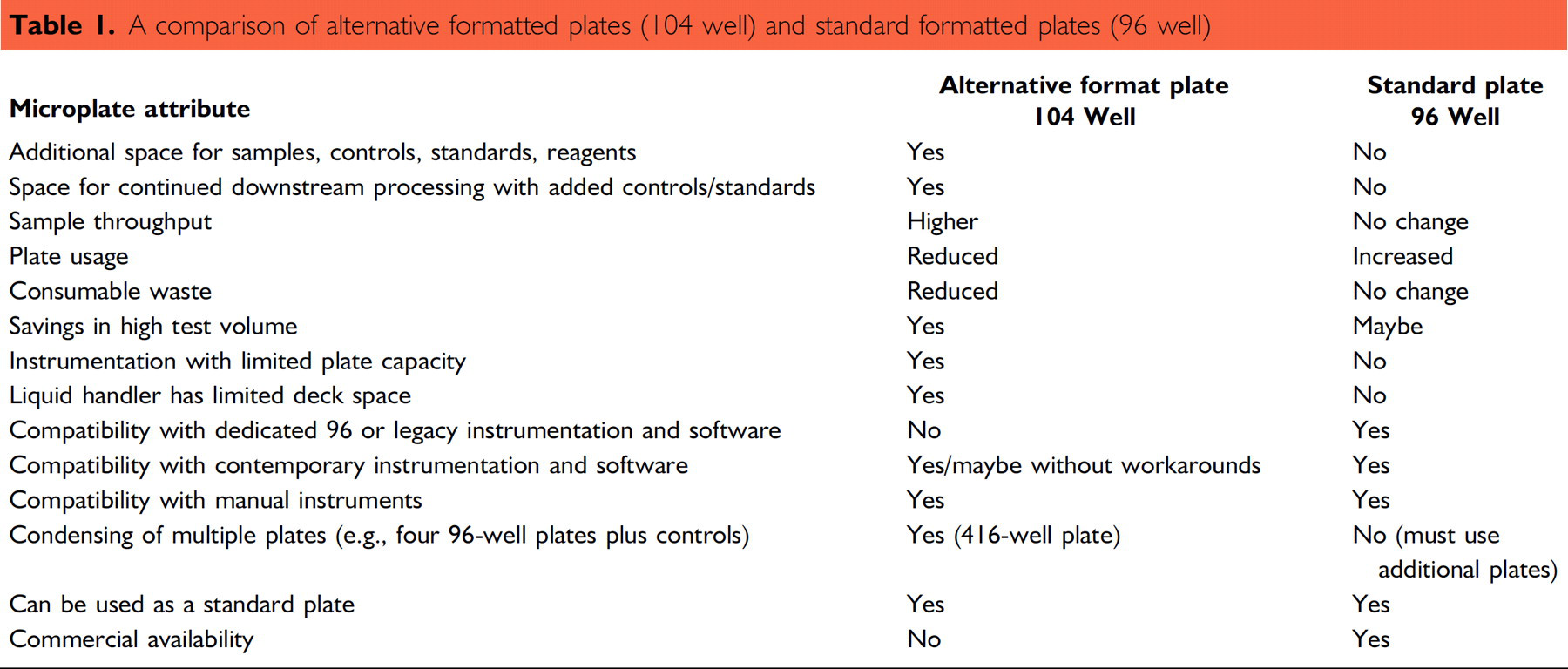
The 104-well format was not developed to supplant the 96-well standard, but to simplify a laboratory processes making it more efficient. By extension, we have demonstrated that an alternative plate can be incorporated into a routine microplate environment. The additional column eliminated the need for a second plate of standards keeping the absorbance readings all in one file for analysis. The extra wells would be a benefit in experiment setup, providing a location for common buffers, reagents, or samples to be held before dispersal across the plate. The 96-well portion of the plate would become the source plate from which PCR, HPLC, or other assays can be started. Table 1 provides a brief summary and comparison of the alternative formatted plates and the standard plates.
A comparison of alternative formatted plates (104 well) and standard formatted plates (96 well)
In conclusion, we have shown that an assay can be performed completely in 104-well format without impacting the results. Contemporary instrumentation can process alternative formatted plates without major problems, either mechanically or in software. The use of alternative formats may have an impact on the throughput of clinical assays, screening, and general laboratory processes.
Acknowledgments
We wish to thank Carl Wittwer of University of Utah School of Medicine and Richard Cavicke of Tecan for manuscript review; Jacob Durtschi of ARUP Laboratories for 3D graph generation; Daniel Leach, Mark Torresan, and Kelli McCoy of Tecan for program development in support of the 104-well plate on the Freedom EVO.
Competing Interests Statement: The authors certify that they have no relevant financial interests in this article.
