Abstract
When optimizing cell culture media for therapeutic antibody production cell lines, it is very important to monitor parameters such as IgG concentration, glucose and lactate levels, and oxygen consumption in culture medium in an automated high-throughput manner to ensure the desirable growth conditions. In this report, assays for measuring human IgG, glucose, and lactate concentrations, and oxygen consumption were automated using the Biomek NXP Span-8 workstation, and analyzed on the PARADIGM Detection Platform. The human-IgG homogeneous time-resolved fluorescence (HTRF) (All trademarks are the property of their respective owners.) assay from Cisbio Inc. (Cisbio US, Bedford, MA) was automated in a 384-well plate format and detected on the PARADIGM with a HTRF Detection Cartridge. Glucose and lactate assays were automated in a 96-well plate format and detected on the PARADIGM with a Fluorescence Intensity Cartridge. Oxygen consumption was determined by the 96-well calibration-free OxoPlate (All trademarks are the property of their respective owners.) (PreSens Inc., PreSens - Precision Sensing GmbH, Regensburg, Germany) and detected by the PARADIGM with a MultiMode Detection Cartridge.
The data for human IgG, glucose, lactate, and oxygen consumption assays automated on the Biomek NXP demonstrate the accuracy and precision obtained using the automated protocols on the PARADIGM Detection Platform, with the user-friendly and assay-specific cartridges for easy, quick, and accurate detection. Using this as a model, one would be able to adapt these approaches to high-throughput media optimization and cell monitoring.
Introduction
IgG concentration, glucose, and lactate levels, and oxygen consumption in culture medium are important parameters in cell cultivation, which directly or indirectly affect the results for both antibody or specific protein production and other cell-based assays. With the increasing demands to contain development costs at the same time to speed up the information acquisition during multifactorial experimentation, scale down mode is more and more favored by pharmaceutical industry. We are providing a high-throughput analytics system that will allow hundreds of microbioreactors to be run in parallel, enabling factorial experiments of large scope for cultivation analysis and optimization. The experiments were performed in an automated high-throughput manner on our Biomek workstation.
The Biomek NXP Laboratory Automation Workstation is a flexible liquid-handling workstation, which can accurately handle pipetting, dilution, dispensing, and integration. The Span-8 with a 360° rotating gripper configuration provides maximum flexibility. The Biomek System Software is an easy-to-use software program that directs all instrument and accessory functions. It provides the ability to customize liquid-handling parameters to import and export methods, to use icon-driven Step Palettes to create and configure methods with drag-and-drop ease, and to define new deck layouts and labware to make changing configurations fast and easy (Fig. 1).

Biomek NXP Span-8 Automation Workstation.
The PARADIGM Detection Platform (Fig. 2)is capable of detecting multiple formats of plates ranging from 6 to 1536 wells, with multiple detection modes, such as absorbance, fluorescence intensity, and fluorescence polarization. The cartridges are designed for specific applications or assays including Cisbio homogeneous time-resolved fluorescence (HTRF) a assay, invitrogen GeneBLAzer assay, PerkinElmer's AlphaScreen assay, and Promega MultiTox-Fluor Multiplex Cytotoxicity assay.

PARADIGM Detection Platform.
Human-IgG Assay
The human-IgG assay uses the HTRF technology. It is a competitive immunoassay between human IgG and the XL665-conjugated human IgG. As shown in Figures 3, when cryptate-conjugated antihuman Fc binds with XL665-conjugated human IgG, it induces fluorescence resonance energy transfer, which can be detected at 665 nm. The competitive binding of nonlabeled human IgG to antihuman Fc-cryptate conjugate will reduce the fluorescent signal proportionally to the nonlabeled human-IgG concentration. The IgG level can be calculated by comparing the fluorescence ratio of 665 to 620 nm with a standard curve of known IgG concentrations.
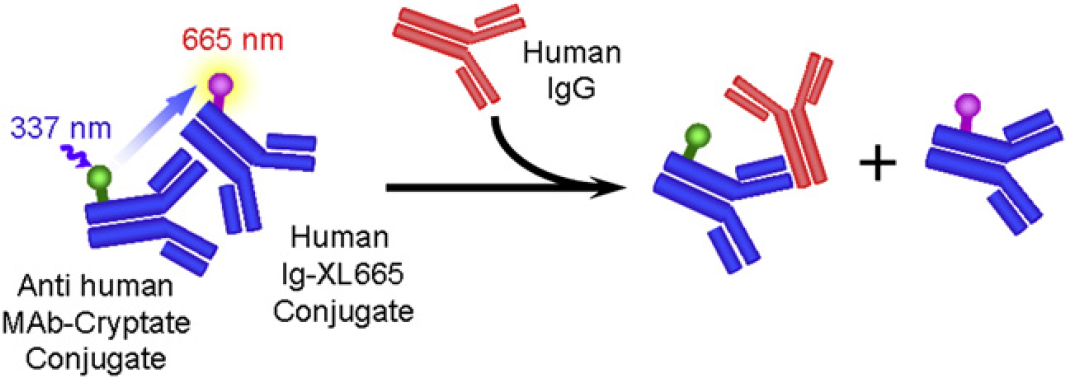
Principle of homogeneous time-resolved fluorescence human-IgG assay.
The human-IgG assay using Cisbio's human MAb screening kit calibrators was automated on the Biomek NXP Span-8 with gripper automation workstation. Calibrators and controls were serially diluted along with stimulation buffer (as shown in Fig. 4) in a 96-well plate; 10 μL from each dilution was transferred into a 384-well plate followed by the addition of 5 μL human-IgG-XL665 and 5 μL antihuman-IgG Fc-cryptate with a final reaction volume of 20 μL.
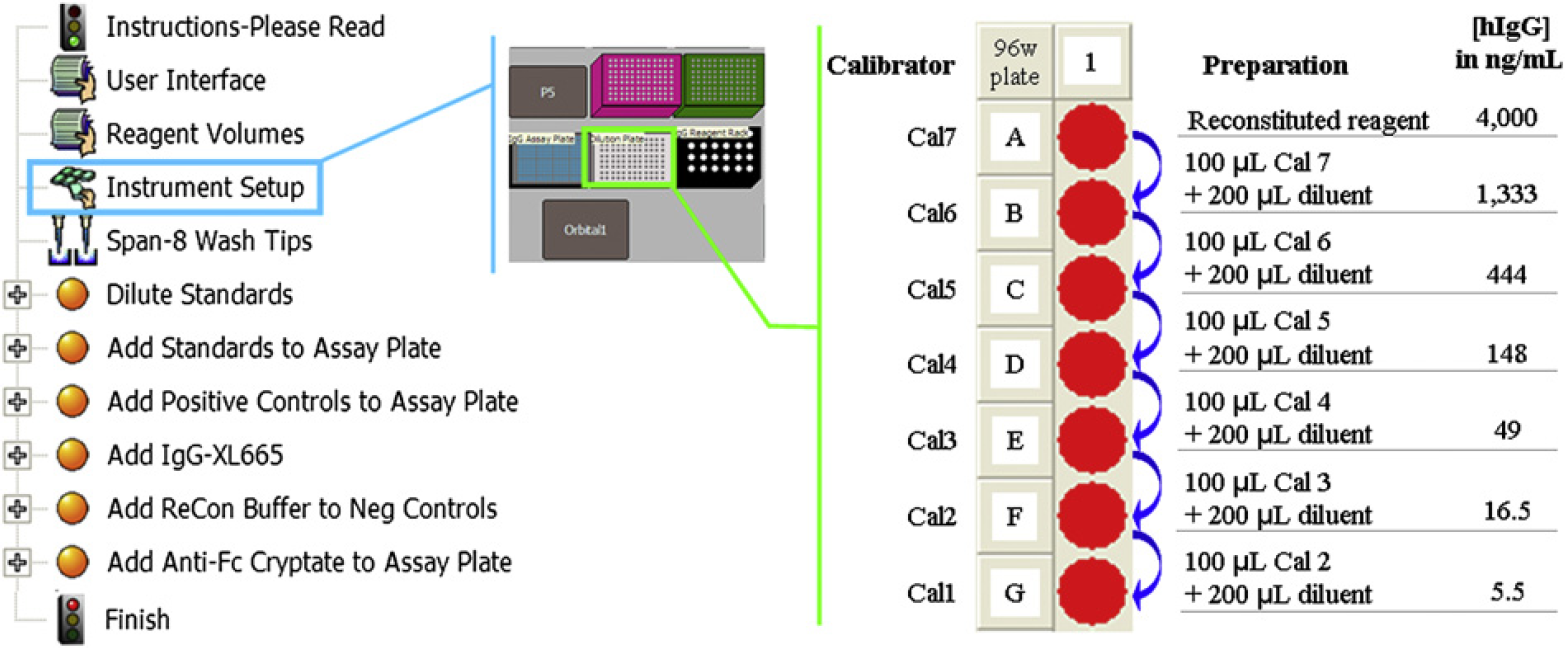
The automation method (left), the deck layout (middle), and serial dilutions of calibrators (right) on Biomek NXP Span-8 automation workstation.
After 3-h incubation in the dark at room temperature, signals were detected at 620 and 665 nm with a 337 nm excitation light source using HTRF cartridge on the PARADIGM. Calibration curves were generated with ratios of signal intensities at 665 to 620 nm, as shown in Figures 5.
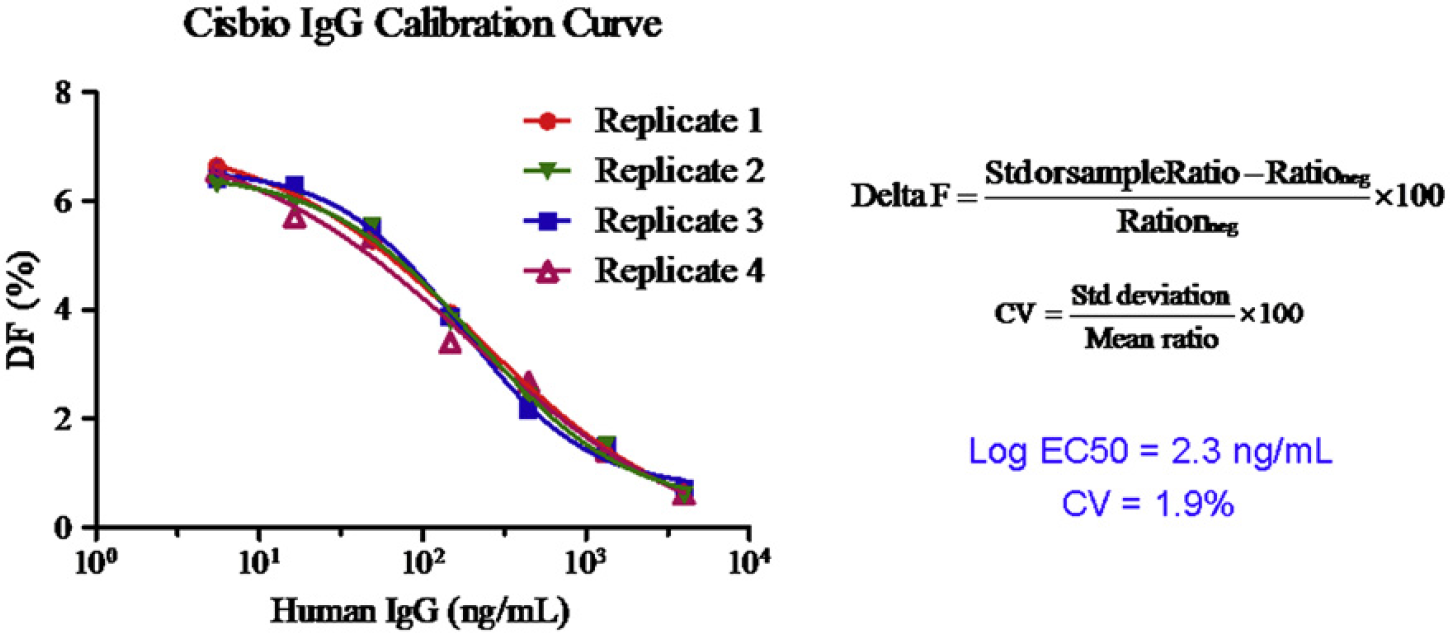
Human-IgG assay standard calibration curves using human MAb screening kit from Cisbio. The Log EC50 of the human IgG is 2.3 ng/mL and the coefficient of variation (CV) is defined as the ratio of the standard deviation to the mean is 1.9%.
Glucose and Lactate Assays
The glucose and lactate assay kits from BioVision Co. (Cat# K606-100 and K627-100, BioVision Corporate Headquarters, Mountain View, CA) were used in the method automated on the Biomek NXP Span-8 in a 96-well plate format at a final reaction volume of 100 μL. Signals of the assays were determined on the PARADIGM.
On the Biomek NXP Span-8 workstation, the glucose standard solution was diluted to 0.01 nmol/mL and 0, 2, 4, 6, 8, 10 μL of it was added into each well to generate glucose standard points of 0, 0.2, 0.4, 0.6, 0.8, 1 nmol/well. Volumes were adjusted to 50-μL/well with the glucose assay buffer. A glucose reaction mix with the glucose probe and the reaction enzyme were added into each well with a final reaction volume of 100 μL. After 30-min incubation in the dark at room temperature, fluorescent signals of the reactions were determined on the PARADIGM with an FI detection cartridge. Results of a representative standard curve are illustrated (Table 1 and Fig. 6).
PARADIGM FI detection cartridge readings of the glucose standards
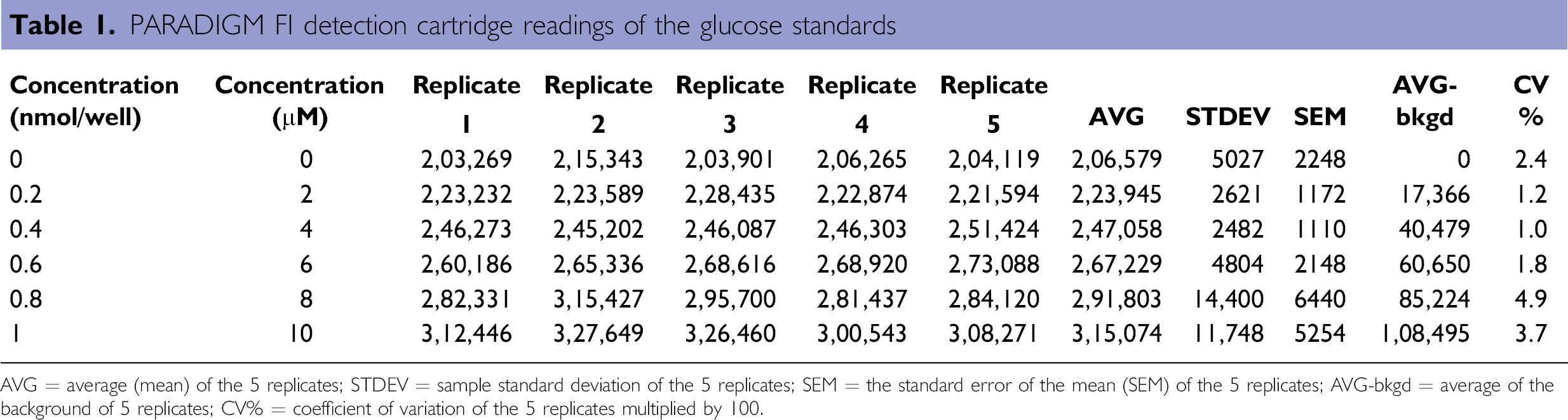
AVG = average (mean) of the 5 replicates; STDEV = sample standard deviation of the 5 replicates; SEM = the standard error of the mean (SEM) of the 5 replicates; AVG-bkgd = average of the background of 5 replicates; CV% = coefficient of variation of the 5 replicates multiplied by 100.
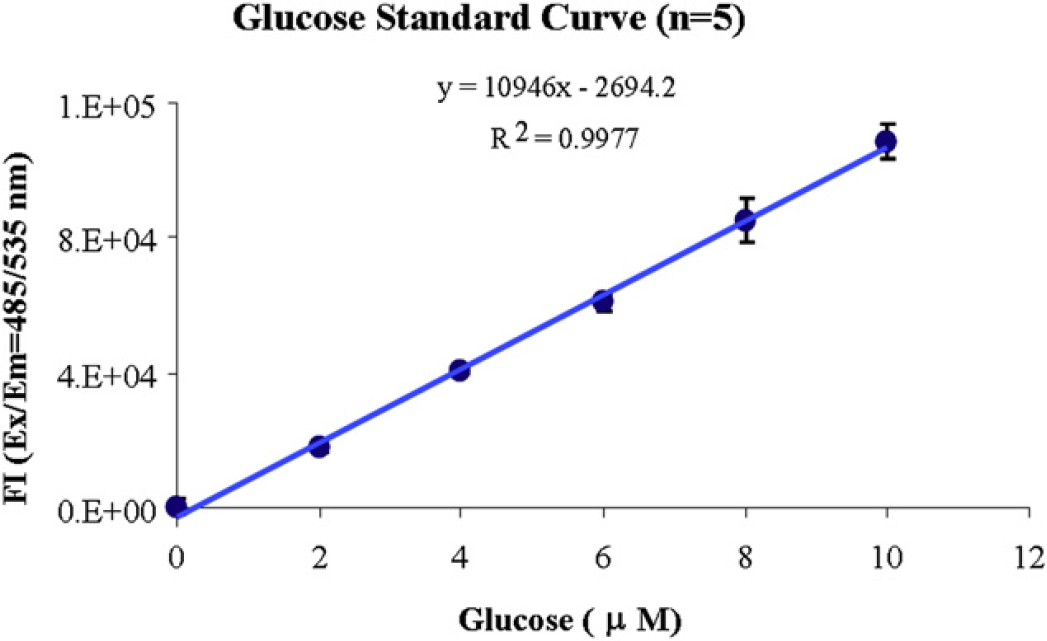
Glucose standard curve. The average percentage CV is around 3.0%.
With a similar procedure described above in the glucose assay, lactate standard points were generated at 0, 0.2, 0.4, 0.6, 0.8, 1 nmol/well with addition of 50 μL lactate reaction mix, which includes the lactate probe and the reaction enzyme with a final reaction volume of 100 μL. After 30-min incubation in the dark at room temperature, fluorescent signals of the reactions were determined on the PARADIGM with a multimode detection cartridge. Results of a representative standard curve are illustrated (Table 2 and Fig. 7).
PARADIGM multimode detection cartridge readings of the lactate standards
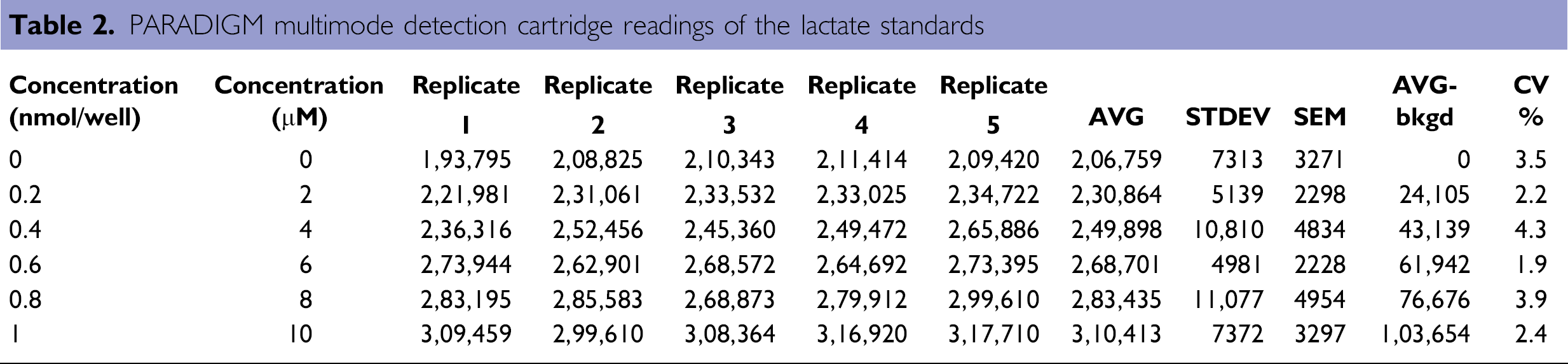
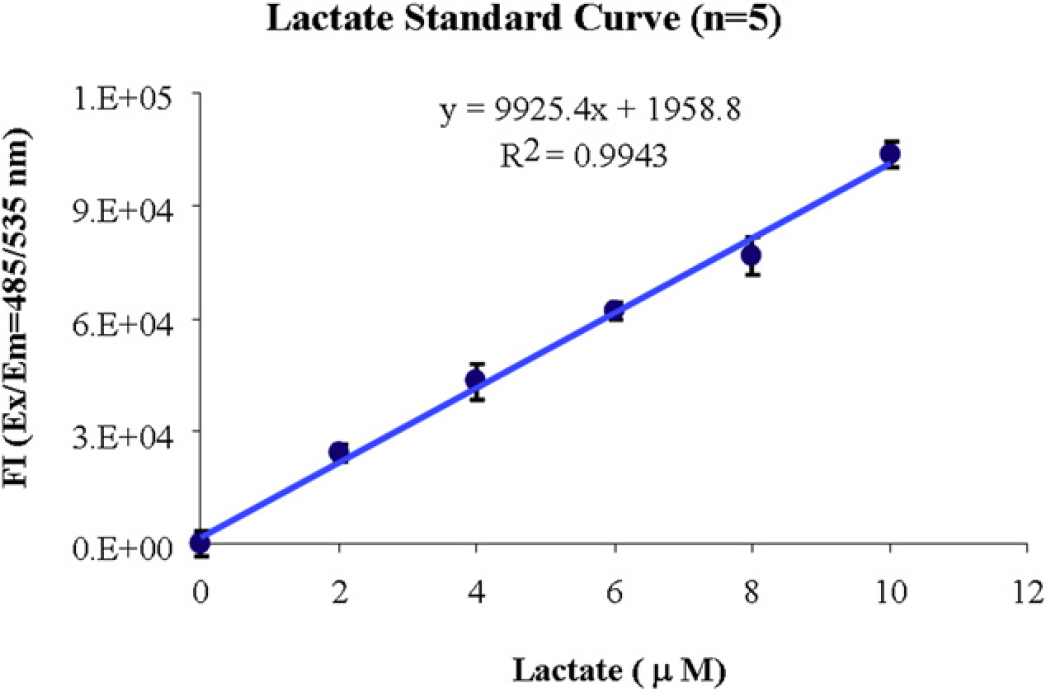
Lactate standard curve. The average percentage CV is around 2.5%.
Oxoplate Oxygen Assay
Oxygen consumption is a very important parameter when optimizing the growth condition in culture medium. The Oxo-Plate from PreSens Inc. is able to measure the oxygen level with integrated oxygen sensors. It is a sterile polystyrene 96-well round-bottom microplate with integrated oxygen sensors in every well. Signals of the sensors can be measured with a fluorescence reader. Each sensor contains two different dyes as an oxygen indicator and a reference. Signal intensity of the oxygen indicator dye (I indicator) corresponds to the concentration of dissolved oxygen in the medium filled in the well. Fluorescence intensity of the reference dye (I reference) is independent of the oxygen concentration. The ratio of I indicator to I reference (I R), corresponding to the concentration of dissolved oxygen, was calculated based on the calibrated standards.

Before measuring oxygen concentrations in the samples, a two-point calibration of the plate reader needs to be performed. Oxygen-free water (cal 0, signal I R as k 0) and air-saturated water (cal 100, signal I R as k 100) were used in the calibration. Oxygen-free water was achieved by dissolving 1 g of sodium sulfite (Na2SO3) in 100 mL water, and air-saturated water was obtained by shaking a sealed vessel filled with 100 mL water rigorously for 2 min. The percentage of air saturation (pO2) was calculated using eq 2:
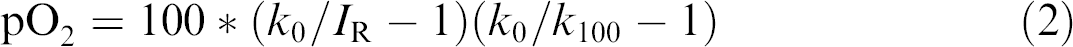
To test this assay, various densities of Escherichia coli bacterial cells (DH5a) (from 1.2 × 105 ∼ 9.7 × 106 CFU/mL) were dispensed into the OxoPlate using Biomek NXP Span-8 at a final volume of 200 μL. Each well was overlaid with 50 μL mineral oil to prevent air diffusion. The plate was then incubated in the PARADIGM at 37 °C and programmed for 15 min-interval timed readings inside of PARADIGM. Using the multimode detection cartridge with a 585 nm/635 nm ex/em filter set for indicator dye and a 535 nm/595 nm ex/em filter set for reference dye, the oxygen consumption pO2([%] air saturation) was calculated using eq 1.
As shown in Figures 8, the higher the density of bacterial cells, the faster the oxygen was consumed. Excellent assay precision (CV = 9.8%) was obtained even with very low dissolved oxygen levels in the sample of 9.7 × 106 CFU/mL.
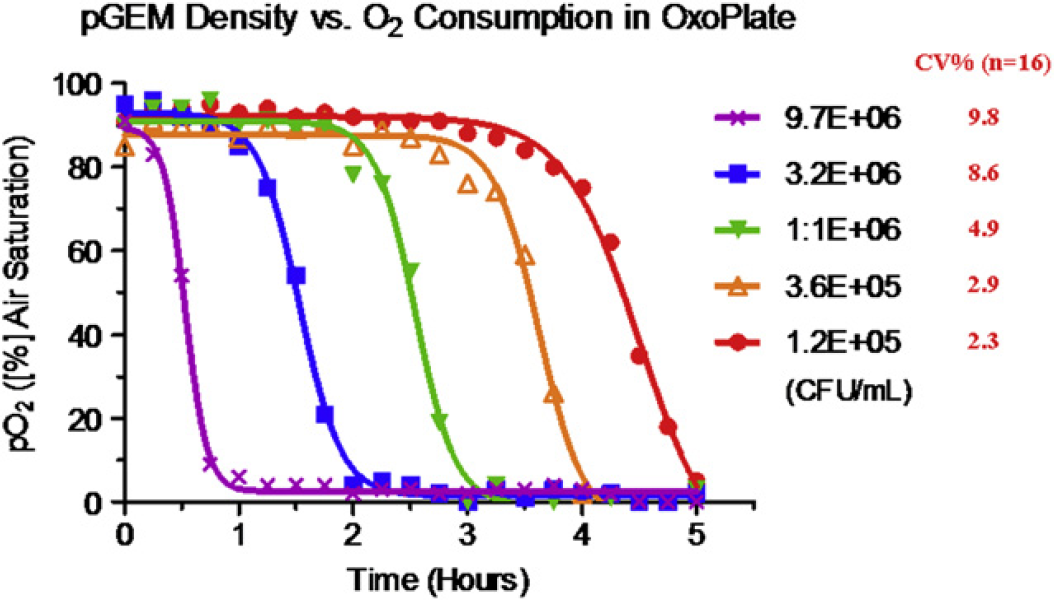
Oxygen consumptions of various densities of DH5 in Oxoplate.
Summary
The Biomek NXP Span-8 automation workstation was successfully used for carrying out human IgG, glucose, lactate, and oxygen consumption assays in an automated higher-throughput manner in 96– or 384-well plates. All those assays are critical tools for cell-culture process optimization. The data demonstrate all the assays have excellent accuracy and precision. The PARADIGM Detection Platform, when combined with automation on the Biomek NXP Span-8, provides a powerful tool for a broad spectrum of high-throughput assays.
Competing Interests Statement: The authors disclose that they are employees of Beckman Coulter.
All trademarks are the property of their respective owners.
