Abstract
The shift from 2D cultures to 3D cultures enables improvement in cell culture research due to better mimicking of in vivo cell behavior and environmental conditions. Different cell lines and applications require altered 3D constructs. The automation of the manufacturing and screening processes can advance the charge stability, quality, repeatability, and precision. In this study we integrated the automated production of three 3D cell constructs (alginate beads, spheroid cultures, pellet cultures) using the Biomek Cell Workstation and compared them with the traditional manual methods and their consequent bioscreening processes (proliferation, toxicity; days 14 and 35) using a high-throughput screening system. Moreover, the possible influence of antibiotics (penicillin/streptomycin) on the production and screening processes was investigated. The cytotoxicity of automatically produced 3D cell cultures (with and without antibiotics) was mainly decreased. The proliferation showed mainly similar or increased results for the automatically produced 3D constructs. We concluded that the traditional manual methods can be replaced by the automated processes. Furthermore, the formation, cultivation, and screenings can be performed without antibiotics to prevent possible effects.
Keywords
Introduction
Cell cultures have a fundamental relevance in biotechnology and medical research. Regarding this, investigation can be made in fields such as basic research, tissue engineering, and drug development to improve regenerative medicine and cancer research.1–3 In the last decade a shift from two-dimensional (2D) cell cultures to three-dimensional (3D) cell cultures has been observed. The in vitro 3D cell cultivation enables the investigation of cell behavior in in vivo similar constructs by mimicking physiological tissue.4,5 There are different 3D systems available on the market. They are distinguishable into scaffold-based and scaffold-free cell constructs. The scaffold-based culture systems use carrier material to support cell growth in 3D dimensions. This can be realized by encapsulation of cells in gels followed by polymerization or cell seeding on acellular matrices.3,6,7 The scaffold-free formation is based on the supremacy of cell–cell interaction over cell–substrate interactions to form cell constructs by self-organization.13,14 The production of 3D cell cultures is primarily performed by manually time-consuming and troublesome methods. Moreover, the automation would be advantageous for the generation of 3D constructs by constant process steps with continual quality, independent of operator skill levels as well as amplified repeatability and precision.4,8–11 However, no published paper covers the field of a fully automated production of 3D constructs.
We addressed this topic by the realization of a unique fully automated process. The Biomek Cell Workstation was modified to perform basic cell cultivation and the consequent formation of different 3D constructs (alginate beads, spheroid cultures, pellet cultures). Subsequently, the quantification was realized by bioscreenings using a high-throughput screening system to evaluate the proliferation (EZ4U assay) and cytotoxicity (adenylate kinase assay).
Formation of 3D Cell Constructs
A variety of methods to cultivate cells in 3D format are known, which depend on cell lines, research, labware, and laboratory equipment. Examples for three 3D cell constructs are alginate beads, spheroid cultures, and pellet cultures, which have the possibility of automated cultivation ( Table 1 ).
Summary of 3D Cell Constructs.
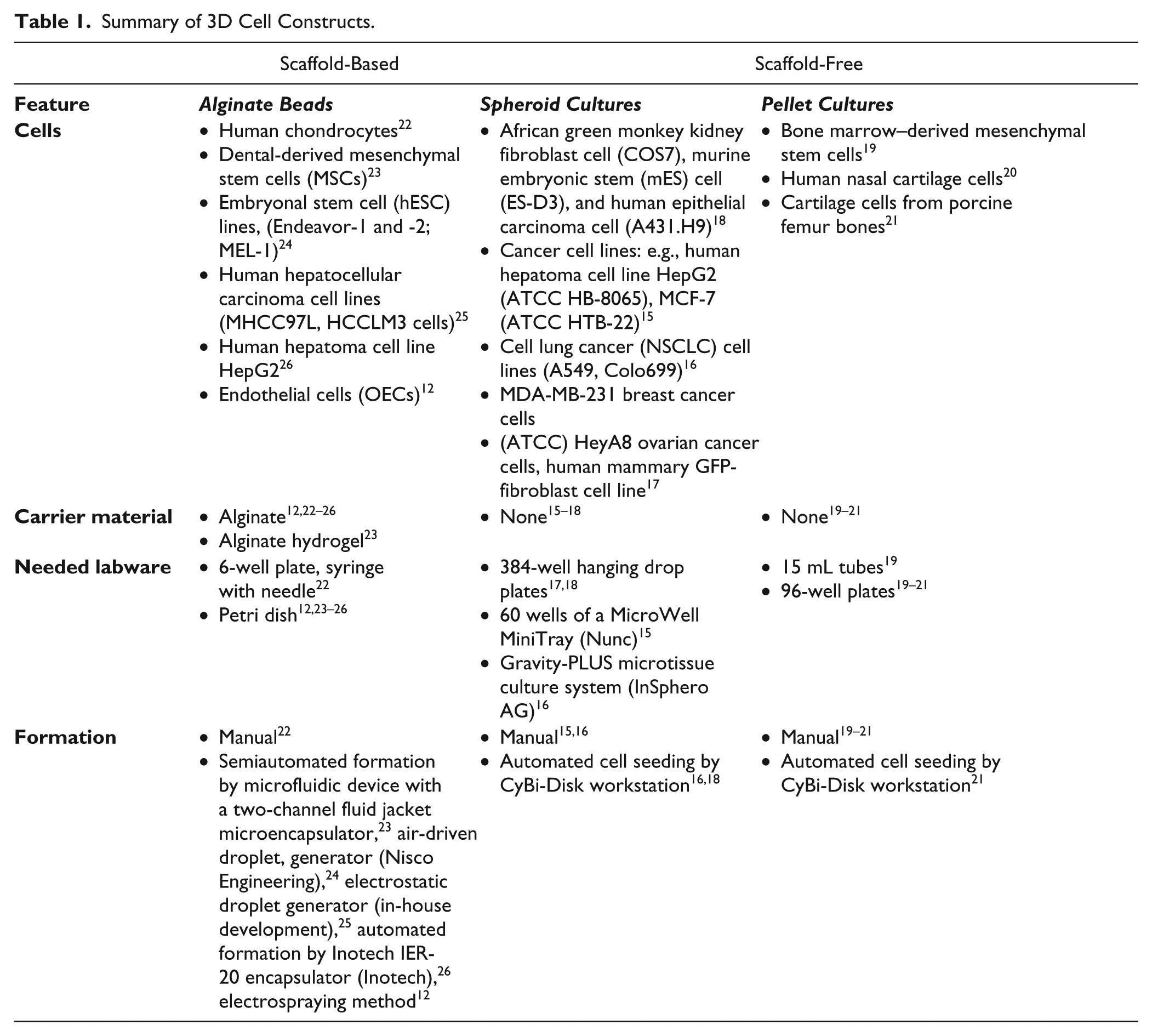
In the case of alginate beads, cells were encapsulated in alginate matrix after the polymerization process. These stable 3D cultures could be cultivated long-term to produce in vivo similar cell constructs. The embedding of cells in alginate matrix showed the highest flexibility for different cell lines. A variety of cell lines were encapsulated: chondrocytes, 12 stem cells,13,14 and hepatocellular carcinoma cell lines.15,16 The alginate bead manufacturing is realized manually in six-well plates 13 or mainly in petri dishes by semiautomated formation processes.17,19–21 Regarding this, the cell detachment processes were manually performed followed by the automated droplet formation. Spheroids are self-aggregated clusters by domination of cell–cell interactions.13,14 In hanging drops, spheroid cultures are formed by cell aggregation in the top of the drop. This 3D cultivation is mainly used for cancer research and does not require carrier material. However, the formation of hanging drops had to be supported by specific labware to enable a higher throughput. Basically, the cells were manually detached. The cell seeding was performed by manual or automated processes with regular liquid handlers.
The 3D pellet cultures are used for stem cell and cartilage research. The pellet formation is realized by high-density cell seeding and bases on predominant cell–cell contacts. The method does not require any carrier material. The pellet formation was mainly manually performed in 15 mL tubes. The semiautomated formation in 96-well plates displayed the basis for high-throughput manufacturing of pellet cultures. 19 Basically, the cells were manually detached and consequently followed the manual or automated cell seeding of cell suspension. The pellets were formed over time.
Material and Methods
System Construction and Design
System Design of the Biomek Cell Workstation
The Biomek Cell Workstation (celisca, Rostock, Germany) was designed for automated cell cultivation ( Fig. 1A ). 4 This system was flexible and configurable, and the devices are vertically arranged (1.49 m/1.64 m/2.63 m). The workstation enabled the cultivation of adherent and suspension cell lines. The automated formation of different 3D cell constructs (alginate beads, pellet cultures, spheroid cultures) was realized by the integration of further devices and automated labware positioners (ALPs).
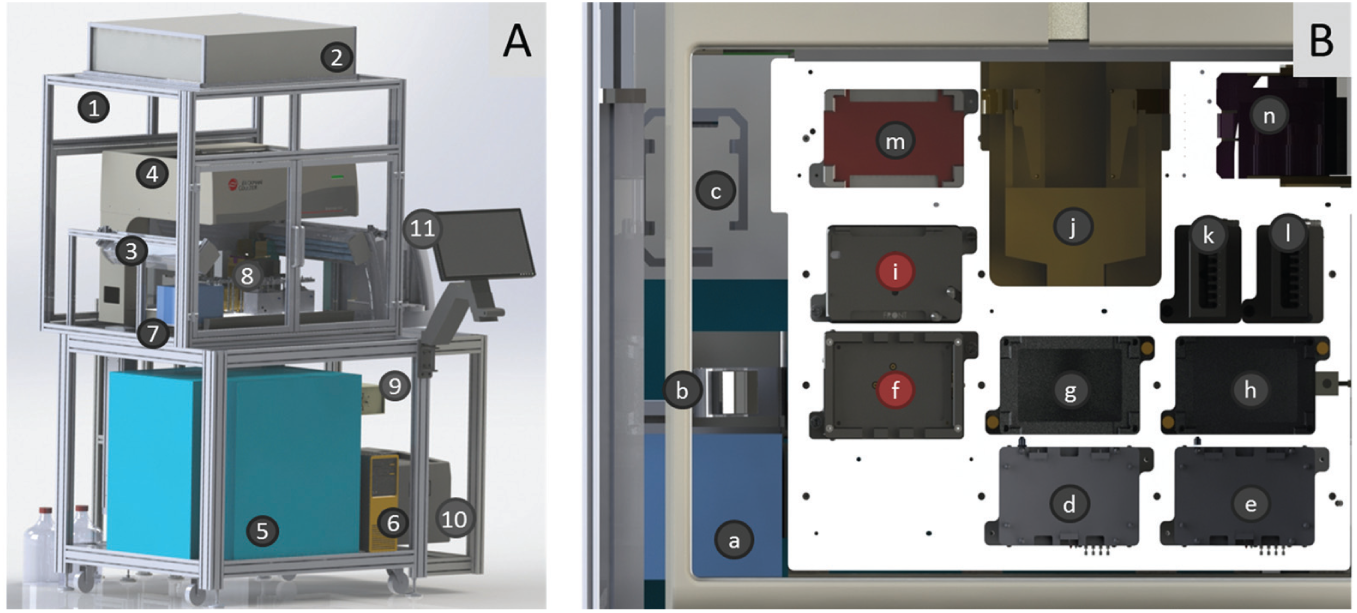
Biomek Cell Workstation and liquid handler deck. (Modified from Reference 27.)
The housing is connected with a HEPA filter (Camfil, Stockholm, Sweden) to support sterile cell handling plus the joined UV lights (Vilber, Eberhardzell, Germany). The Biomek NX (Beckman Coulter, Krefeld, Germany) is integrated in the housing to perform liquid handling with a Span-8 pipetting head associated with steel cannulas. The transfer of cell culture vessels is realized by the integrated gripper. Directly associated with the liquid handler deck are the Vspin centrifuge (Velocity 11, Palo Alto, CA) and the ViCell XR (Beckman Coulter) for cell counting as well as viability tests. The external Cytomat (Thermo Scientific, Schwerte, Germany) is connected with the liquid handler deck about a plate lift for well plates plus flasks and enables the incubation of cells under defined humidity conditions (37 °C, 5% CO2). The port selection valve is equipped with a hose pump and enables sterile solution handling. The cool box (WEMO-Geräte AG, Schlatt, Switzerland) permits the storage of solutions at 4 °C. The computer enables the programming, control, and visualization of the different cultivation steps.
According to
Figure 1B
, the liquid handler deck is connected with the devices and equipped with a variety of automated labware positioners (ALPs) to realize the cultivation processes. These can be active ALPs or only placeholders. The two on-deck incubators (inheco, Martinsried, Germany) enable short incubation periods at 37 °C. The two 3D tilt racks (AIG, Rostock, Germany) simulate manual handling by pivoting, knocking, and tilting (
The changes in the setup of the liquid handler deck have to be integrated to realize the different 3D formations. Two placeholder ALPs have to be changed by a Positive Position ALP ( Fig. 1B, f ) and a Static Peltier Device ( Fig. 1B, i ), which can be variably located at the liquid handler deck.
The Positive Position ALP (Beckman Coulter; Fig. 2A ) is an active ALP. It is placed on position 1 by thumbscrews at the liquid handler deck. This ALP can be used to fix flasks and well plates in a 96-well format at defined positions to realize high-accuracy liquid transfer by the operation modes: clamping, unclamping, and labware detection via sensor. 28 The Positive Position ALP is required for the automated formation of hanging drops to generate spheroid cultures in 384-well Perfecta3D Hanging Drop Plates (3D Biomatrix, Ann Arbor, MI, USA). The cavities have a diameter of only 1.5 mm, and the steel cannulas needed exact positions to form hanging drops.
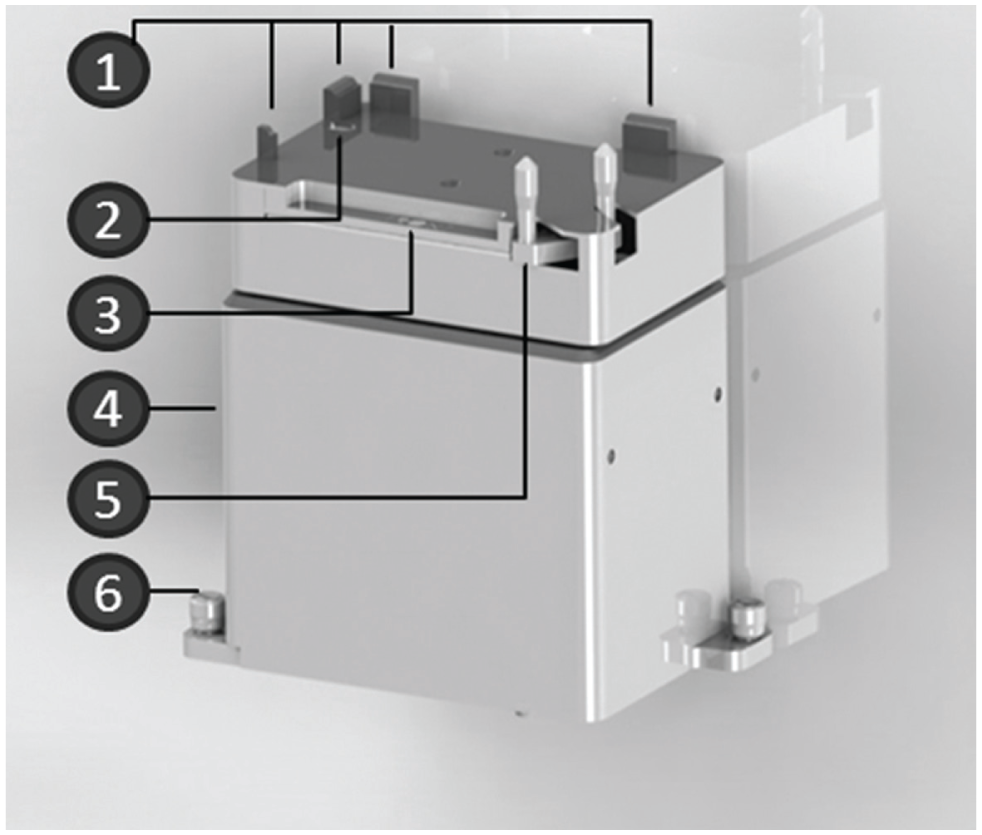
A new integrated automated laboratory positioner (ALP) is needed for 3D cell cultivation.
The Positive Position ALP fixes the labware by the positioning guide and the clamp. The labware transport is realized by the gripper of the liquid handler. The automated handling of this ALP is realized by the Biomek software. The Advanced Manual Control enables the manual control. 28
The Static Peltier Device (PN A36984) enables the heating and cooling of solutions for automated processes by the connected controller. 30 The warming up of the solutions is necessary for the regular cell cultivation in the automated way. Especially, the exact liquid handling of high-viscose alginate is supported in heated conditions. Regarding this, the Static Peltier Device is indispensable for the embedding of cells in an alginate matrix.
This device is placed on position i of the liquid handler deck of the cell culture automation attached with thumbscrews. Different adapters realize the handling of 96- to 384-well plates and modular reservoirs. Regarding this, the black stand made of anodized aluminum enables the heating of solutions in modular reservoirs at the Biomek Cell Workstation. The maximum reachable temperature of the controller is 50 °C, which is required to heat solutions in modular reservoirs up to 37 °C. The temperatures have been proved to be constant in an air-conditioned laboratory environment. The Biomek software realizes the automated processing of the Static Peltier Device. The device is manually controllable without software by the Watlow Controller. 30
Cell Culture Vessels
The Biomek Cell Workstation supports the handling of CELLSTAR AutoFlask (greiner, Frickenhausen, Germany) for automated cell handling. These flasks include a hydrophobic membrane for the gas exchange and a septum for sterile transfer of the solution to realize the horizontal cell handling. 12
The storage of solutions during the cell cultivation processes was realized by modular reservoirs (up to 40 mL, Beckman Coulter) and six glass bottles with air filters (up to 500 mL). The reservoirs were located on the liquid handler deck. The glass bottles are connected with a port selection valve and channel 7 of the liquid handler about a tubing system, which is equipped with a hose pump (IDEX Health & Science, Glattbrugg, Germany) to dispense higher volumes. They are located outside of the housing and can be stored in the cool box (
Fig. 1A
,
Software
Two software types are required for the automated cell handling. The specific liquid handling and calculating steps were programmed by the Biomek software. The SAMI software is used to combine the whole processes in association with the different labware and liquid handling steps. Furthermore, the SAMI software enables scheduling process steps.
Cell Cultivation
The cervix carcinoma cells (HeLa) were manually expanded in 75 cm² cell culture flasks (Sarstedt, Nümbrecht, Germany) at 37 °C and 5% CO2. The cells were cultivated in Dulbecco’s modified Eagle’s medium (DMEM, Sigma Aldrich, Seelze, Germany) supplemented with (1%) and without penicillin/streptomycin (P/S; Sigma Aldrich) and 10% fetal bovine serum (FBS, Sigma Aldrich). The cells were split by a wash step with 5 mL saline buffered with phosphate (PBS; Sigma Aldrich) and treatment with 2 mL trypsin/EDTA (Sigma Aldrich) followed by an incubation period of 4 min at 37 °C. The detachment process was stopped by neutralization with DMEM. The cells were seeded into new 75 cm2 flasks.
The primary chondrocytes were provided by the Depart-ment of Orthopaedics, Biomechanics and Implant Technology Research Laboratory of University Rostock. After the defrosting, the cells were cultivated in Dulbecco’s modified Eagle’s GlutaMax medium (DMEM-GlutaMax; Life Technologies, Darmstadt, Germany) supplemented with 10% FBS, 1% amphotericin B, and 0.5% ascorbic acid, as well as with or without 1% P/S (all acquired from Sigma Aldrich). The cells were manually expanded in a 75 cm2 cell culture flask in a clean room, and in passage two were seeded in specific flasks.
The cell lines were expanded within 75 cm2 flasks for the manual processes and CELLSTAR AutoFlasks (greiner) for automatically performed procedures.
Manufacturing Processes of Alginate Beads, Spheroid Cultures, and Pellet Cultures
Manual Formation
First, the cells were detached by removing the supernatants (15 mL), followed by the washing step using PBS (5 mL; Sigma Aldrich). The cells were treated with enzyme (trypsin, Sigma Aldrich) at specific incubation periods (HeLa cells, 4 min; chondrocytes, 10 min) and stopped with DMEM (HeLa cells) and DMEM-GlutaMax (chondrocytes) with 10% FBS. The cell suspensions were collected in modular reservoirs or tubes. After the cell counting using the ViCell XR (Beckman Coulter) and calculating of the cell numbers, the 3D formations followed.
The production of alginate beads started with the centrifugation (2250 rpm, 5 min) of the cell suspension by the Vspin and the embedding of cells (2 × 106 cells/mL) in 1.2% alginate (Sigma Aldrich). The alginate beads with HeLa cells were formed by dropping alginate–cell solution dropwise into a 100 mM calcium chloride (CaCl2) solution (200 µL/well; Sigma Aldrich) within a 96-well plate using a disposable syringe with cannula. The beads were washed three times with 0.9% sodium chloride (NaCl, Sigma Aldrich) and cultivated with 200 µL/well DMEM with 10% FBS, with 2 mM
For the pellet formation, the chondrocytes were centrifuged (2250 rpm, 5 min) and resuspended in fresh media with growth factors (0.5 × 106 cells/200 µL) as well as transferred into a 96-well plate (200 µL/well). The growth factors were dexamethasone (final concentration, 100 nM; Sigma Aldrich), ascorbic acid (final concentration, 50 μg/mL; Sigma Aldrich), rh IGF-1 (final concentration, 50 ng/mL; RD Systems, Wiesbaden, Germany), rh TGF-β1 (final concentration, 50 ng/mL; Tebu-bio, Offenbach, Germany), and ITS Premix (complete medium to ITS in a 100:1 ratio; BD Bioscience, Heidelberg, Germany). Subsequently, a shaking period (0.5 h) was performed using a thermomixer (37 °C, 300 rpm) (Eppendorf, Hamburg, Germany).
The spheroid cultures with HeLa cells were formed in hanging drops using the Perfecta3D Hanging Drop Plates (3D Biomatrix). Regarding this, the diluted cell suspension (40,000 cells/30 μL) was particularly transferred into every second cavity of the plate by a one-channel 200 μL Eppendorf pipette.
The consequent incubation of the 3D cell cultures was performed in the Cytomat (37 °C, 5% CO2). The media change was performed two times a week.
Automated Formation
In general, the bases for the formation of 3D cell constructs were adherent cells, which had to be detached ( Fig. 3 ). The Biomek Cell Workstation enabled the automated cell cultivation and detachment of different adherent cell lines (e.g., cervix carcinoma cells, chondrocytes). The cells were cultivated in AutoFlask, which were needed for the automatic cell handling. These flasks were located in the Cytomat. These cell culture flasks were transferred to the liquid handler deck via the lift and positioned on the 3D tilt rack by the gripper of the liquid handler. The monolayer was washed with 5 mL of PBS. The cells were detached by trypsin during an incubation period in the on-deck incubators (4 min, HeLa cells; 10 min, chondrocytes). The detachment process was stopped by adding cell culture medium (6 mL at the production of alginate beads; 4 mL at the spheroid cultures, pellet cultures) transferred by the liquid handler. After pivoting, the cell suspensions were transferred into modular reservoirs. The 3D formation started after the cell counting via the ViCell XR and calculation of the cell amount. The general process steps for the 3D formation are visualized in Figure 3 .
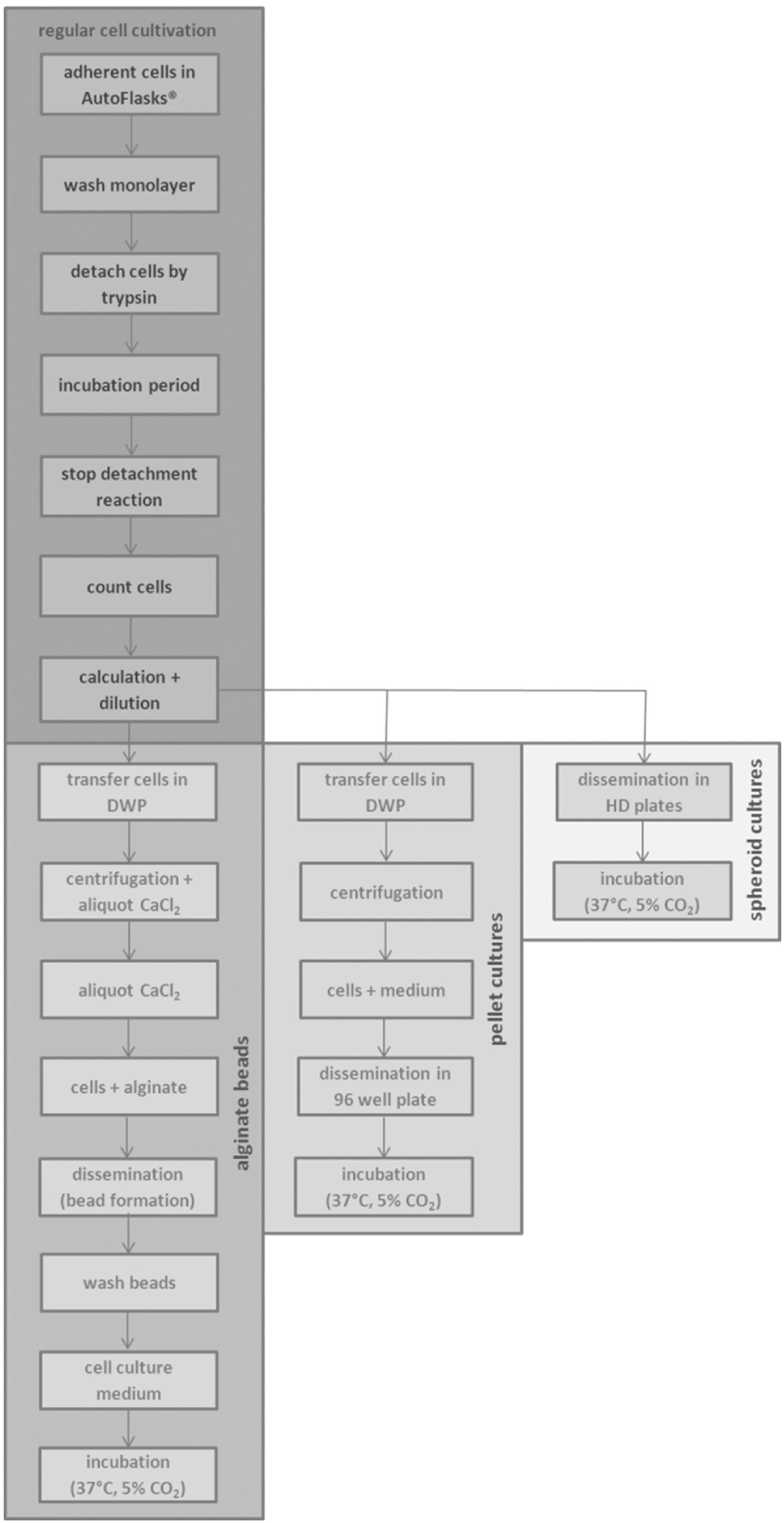
The schematic overview of the cell handling of 3D cell cultures.
The required solutions for the alginate bead formation were positioned on the liquid handler deck ( Fig. 1B : alginate [f], CaCl2 [f], NaCl [f], medium [f], isopropanol [h], water [h]). One AutoFlask and detachment process is needed to produce alginate beads in a 96-well plate. The cell suspension was aliquoted into the deep-well plate (DWP, 1 × 106 cells/well) after calculating the cell count to warrant the exact cell number. Inn parallel, the same volume of water was transferred into the counterweight. The deep-well plates (greiner) were centrifuged (1150 rpm, 5 min) via the Vspin. In parallel, the Biomek NX liquid handler transferred CaCl2 solution into the 96-well plate (200 µL/well). After removing of the supernatant, the pellets were resuspended in 1.2% alginate (500 µL/well). The alginate bead formation was executed by dropping one bead per well from a continual height of 1.5 cm. After a polymerization period of 10 min, the beads were washed three times with 0.9% NaCl (1 × 200 µL, 2 × 130 µL) before the addition of medium (200 µL/well) by the liquid handler.
The required solutions for the pellet formation were located on the liquid handler deck (medium with growth factors, isopropanol, water). To perform the pellet formation in a 96-well plate, the detachment process was required eight times. After calculating the cell number to guarantee the exact cell count, the cell suspension was transferred into a deep-well plate (5 × 105 cells/well) and water was pipetted into the counterweight. After centrifugation via the Vspin (2250 rpm, 5 min), the supernatant was removed by the liquid handler. The pellets were resuspended with medium plus growth factors (200 µL/well) and transferred into a 96-flat-bottom plate.
The spheroid cultures were formed in 384-well Perfecta3D Hanging Drop Plates. Concerning this, two AutoFlasks and detachment processes were needed to form 196 hanging drops at the plates. After the calculation, the cell solution was diluted and transferred into the specific plate (40,000 cells/30 µL).
After the 3D formations the well plates were transferred backward into the Cytomat (37 °C, 5% CO2). The 3D cell cultures were analyzed on days 14 and 35 by a high-throughput screening system (celisca).
Screening Processes
The screening processes were manually and automatically performed. The manual methods were realized by skilled laboratory staff with Eppendorf pipettes. The automated procedures were performed using a high-throughput screening system and the Biomek FX liquid handler to investigate the 3D cell constructs ( Fig. 4 ).
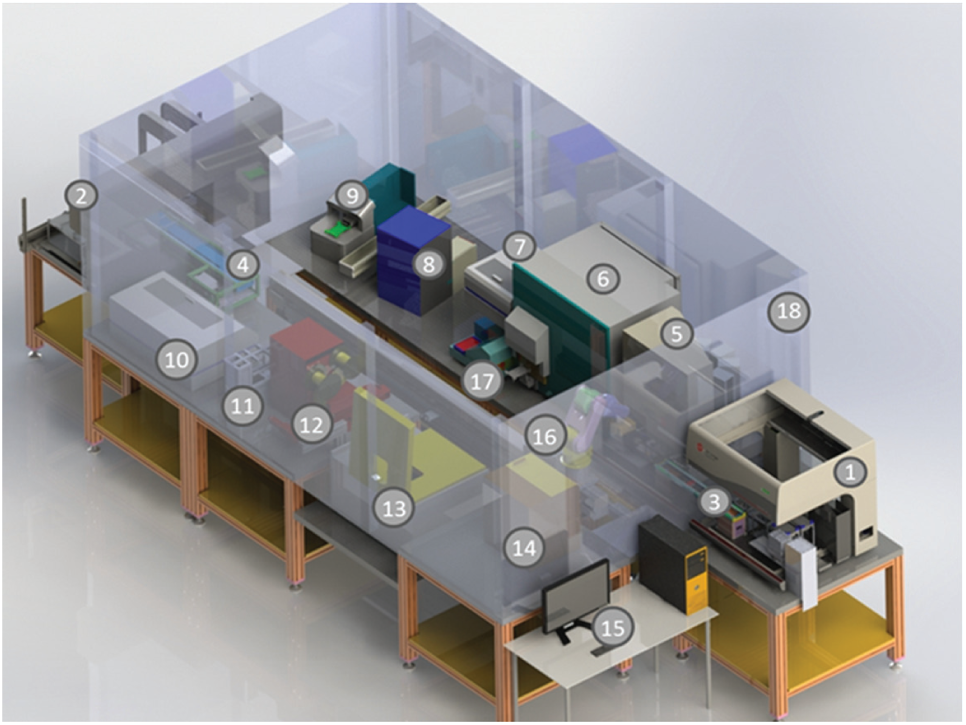
Components of the high-throughput screening system.
Instrumentation: High-Throughput Screening System
The high-throughput screening system is constructed of a large number of components to cover different screening processes in 96-well plate formats. Outside the housing the liquid handler (Biomek FX, Biomek NX; Beckman Coulter) and the computer are located. The Biomek FX is equipped with a 96-well pipetting head and integrated gripper, and the liquid handler deck includes 15 passive placeholder ALPs, a loading station for disposables, a wash station, and a refill reservoir (all purchased from Beckman Coulter). The Biomek NX is equipped with a Span-8 pipetting head for disposables and integrated gripper, and the liquid handler deck includes five placeholder ALPs (Beckman Coulter), three VARIOMAG Teleshake ALPs (Thermo Scientific), and a trash and wash station (Beckman Coulter). The liquid handlers are associated with the devices within the housing about shuttles and the rail-mounted robot Motoman (Yaskawa, Fukuoka, Japan). Integrated in the housing are three microplate readers (PHERAstar Plus, FLUOstar galaxy reader, NOVOstar microplate reader; BMG Labtech, Orthenberg, Germany) for the verification of absorbance, luminescence, and fluorescence. Also integrated in the housing are the Cytomat hotel (Kendro, Langenselbold, Germany) to store disposable boxes and the Cytomat incubator (37 °C, 5% CO2; Thermo Fisher Scientific) for well plates. The barcode reader and printer enable the identification of cell culture plates. The covering and recovering of well plates with foil can be realized by the plate sealer and peeler (HJ-Bioanalytik, Monchengladbach, Germany). The lid station enables the handling of lids. A plate washer realizes washing steps. Ultimately, a centrifuge (Sigma, Osterode, Germany) is integrated in the system. The processes and devices are manageable by the Biomek software and SAMI software. 29
EZ4U Cell Proliferation Assay
The EZ4U assay kit (Biomedica, Vienna, Austria) was used to evaluate the cell proliferation. Hereof, living cells reduce tetrazolium salt to colored formazan.
At the manual bioscreening, the supernatants of the 3D cell cultures were initially removed. Then fresh media (250 µL) was added followed by the reagent (25 µL) with a consequent incubation period of 3 h at the Cytomat (5% CO2, 37 °C). The supernatants were transferred into a new 96-well plate, and the absorbance was detected using the PHERAstar device at 450 nm.
The automated processes started with the transfer of the labware (well plate, disposables) to the Biomek FX liquid handler by the Motoman and positioning on the deck by the gripper. The reagent and fresh media were manually positioned on the liquid handler deck within a low-volume reservoir. Initially, the supernatants were removed followed by the addition of fresh media (250 µL/well) and reagent (25 µL/well). Then, the Motoman moved the well plate to the Cytomat (5% CO2, 37 °C) for an incubation period of 3 h. Subsequently, the plate with the 3D cell cultures, a new 96-well plate, and fresh disposables were moved to the Biomek FX liquid handler deck using the Motoman. The supernatants were transferred into a new well plate by the Biomek FX, and the absorbance (450 nm) was measured by the PHERAstar.
Adenylate Kinase Cytotoxicity Assay
The adenylate kinase cytotoxicity kit (Promokine, Heidelberg, Germany) was used to evaluate the cytotoxicity at manual and automated procedures.
In the manual processes, the liquid transfer was realized by an eight-channel Eppendorf pipette. First, supernatants (100 µL) of the 3D cell cultures were transferred into a white 96-well plate. Then the reagent (100 µL) was added. After an incubation period of 5 min at 20 °C, the luminescence was detected using the PHERAstar device.
For the automated performance, the labware (well plate and disposables) was transferred to the Biomek FX liquid handler with a 96-well pipetting head by the Motoman. The gripper of the liquid handler positioned the labware on the deck. After loading the disposables, the supernatants (100 µL) were transferred into the well plate followed by the reagent (100 µL). The luminescence was detected by the PHERAstar after 5 min incubation on the deck (20 °C).
Statistical Analysis
The statistical analyses were performed by determination of the mean value and the standard errors. Significances were analyzed by the Student
Results and Discussion
We adapted the automated formation of three different 3D constructs (alginate beads, spheroid cultures, and pellet cultures). We embedded cervix carcinoma cells in the alginate matrix to form microtissues similar to tumors over time. The 3D formation by encapsulating cells in alginate can be used for most varieties of cell lines. These include human chondrocytes, 22 dental-derived mesenchymal stem cells (MSCs), 23 embryonal stem cell (hESC) lines (Endeavor-1 and -2; MEL-1), 24 human hepatocellular carcinoma cell lines (MHCC97L, HCCLM3), 25 human hepatoma cell line HepG2, 26 and endothelial cells (OECs). 12 Spheroid cultures are primarily formed from cancer cell lines: human hepatoma cell line HepG2 (ATCC HB-8065), MCF-7 (ATCC HTB-22), 15 cell lung cancer (NSCLC) cell lines (A549, Colo699), 16 MDA-MB-231 breast cancer cells, HeyA8 ovarian cancer cells, and human mammary green fluorescent protein (GFP)–fibroblast cell line. 17 Furthermore, we also manufactured 3D constructs in the form of spheroid cultures with cervix carcinoma cells to produce in vitro tumors without carrier material. Spheroids can also be formed by different cell lines, such as African green monkey kidney fibroblast cells (COS7), murine embryonic stem (mES) cells (ES-D3), and human epithelial carcinoma cells (A431.H9). 18 The pellet cultures are exclusively used to produce cartilage tissue. Regarding this, we used human cartilage cells for the 3D formation. Nevertheless, the literature supports pellet formation consisting of bone marrow–derived mesenchymal stem cells, 19 human nasal cartilage cells, 20 and cartilage cells from porcine femur bones. 21
The 3D constructs were generated with and without carrier material for different applications and cell lines. Regarding this, spheroid cultures15–18 and pellet cultures19–21 are 3D cell formats without carrier material. This is realizable by the domination of cell–cell contacts over cell–matrix bonds. 13 Instead, the bead formations needed different matrices to generate 3D constructs. These are alginate12,22–26 and alginate hydrogel. 23 But biocompatible sodium alginate was used in the present study.
The different 3D constructs needed different labware and devices for formation. The formation of different 3D cell constructs was realized by the flexible Biomek Cell Workstation. This cell culture automation enables a fully automated cell culture process that includes cell detachment followed by 3D manufacturing. Regularly, alginate beads were formed in a six-well plate using a syringe with needle 22 and petri dish.12,23–26 However, this labware did not support the automated bead formation in a high-throughput manner. Regarding this, we adapted a new method to produce alginate beads in a 96-well plate to support the subsequent bioscreening. In detail, the alginate bead production was performed by resuspending HeLa cells in 1.2% alginate matrix and dropping this solution 1.5 cm from the top with a speed of 208 µL/s in CaCl2 solution using the Biomek NX liquid handler. The speed of 208 µL/s is the optimized rate to form beads with the Biomek Cell Workstation. The liquid transfer is realized by steel cannulas with a syringe pump associated with a tubing system filled with system fluid (distilled water). However, published papers support manual cell detachment followed by automated bead formation with different alginate concentrations using encapsulators or droplet generators.17,19–21 Concerning this matter, Coward et al. produced HeLa cell–alginate beads by dropping a cell–alginate solution in a stirred solution (0.204 M CaCl2 in 0.15 M NaCl buffer) with a syringe pump of an Inotech IER-20 encapsulator (Inotech, Dottikon, Switzerland) at a flow rate of 5 mL of cell suspension/min using a 200 mm nozzle vibrating at 1295 Hz. 17
Kim et al. manufactured the beads in a petri dish with human embryonic stem cells (hESCs) by dropping a cell–alginate (1.1%) suspension into a 100 mM CaCl2/10 mM HEPES solution by an air-driven droplet generator (Nisco Engineering, Zurich, Switzerland) at a flow rate of 8 L/mL by a pressure of 100 kPa. 19 Xu et al. formed alginate (1.5%) beads with encapsulated hepatocellular carcinoma cells (HCCs) by an electrostatic droplet generator (in-house development). 20 Furthermore, Moshaverinia et al. semiautomatically produced alginate (2%) hydrogel beads within TGF-b1 (growth factor) with dental-derived mesenchymal stem cells (MSCs) using a microfluidic device with a two-channel fluid jacket microencapsulator plus micropipette by shearing off cell–alginate solution in soybean oil and pooling in CaCl2 solution. 21 The semiautomated bead formation with endothelial cells (OECs) and growth factors (vascular endothelial growth factor [VEGF], hepatocyte growth factor [HGF]) with the electrospraying procedure was investigated by Kim et al. The device realized the formation using a high-voltage source associated with a syringe pump and stainless-steel nozzle, as well as a syringe needle (28 gauge) from NanoNC (NNC-ESP 200, Seoul, South Korea). 12
The manually produced spheroid cultures were realized by different labware: 60 wells of a MicroWell MiniTray (Nunc) 15 and Gravity-PLUS microtissue culture system (InSphero AG). 16 However, the automated formation of HeLa cell–spheroid cultures in this paper was realized by cell seeding using the Biomek NX liquid handler in 384-well hanging drop plates. This is supported by Tung et al., who also produced uniformly sized spheroid cultures of different cell lines (African green monkey kidney fibroblast cells [COS7], murine embryonic stem [mES] cells [ES-D3], and human epithelial carcinoma cells [A431.H9]) in 384 hanging drop array plates. But the hanging drop formation was performed by the CyBi-Disk workstation. 18
Normally, the pellet formation is performed manually in 96-well plates, which was the basis for automated solutions.19,20
In this respect, Ibold et al. formed pellet cultures of cartilage cells from porcine femur bones in 96-well tissue culture plates by manual cell detachment and automated formation by the cell seeding using a CyBi-Disk workstation for liquid handling. 21 We adapted the fully automated pellet formation of human cartilage 3D constructs using the Biomek NX for the liquid transfer and cell seeding.
Evaluation of the Proliferation
EZ4U Assay
The proliferation was automatically detected by the EZ4U assay ( Fig. 5 ) after manual and automated 3D formation on days 14 and 35. The constructs were cultivated in media with (+) or without (–) antibiotics. The used antibiotics were penicillin/streptomycin (P/S).
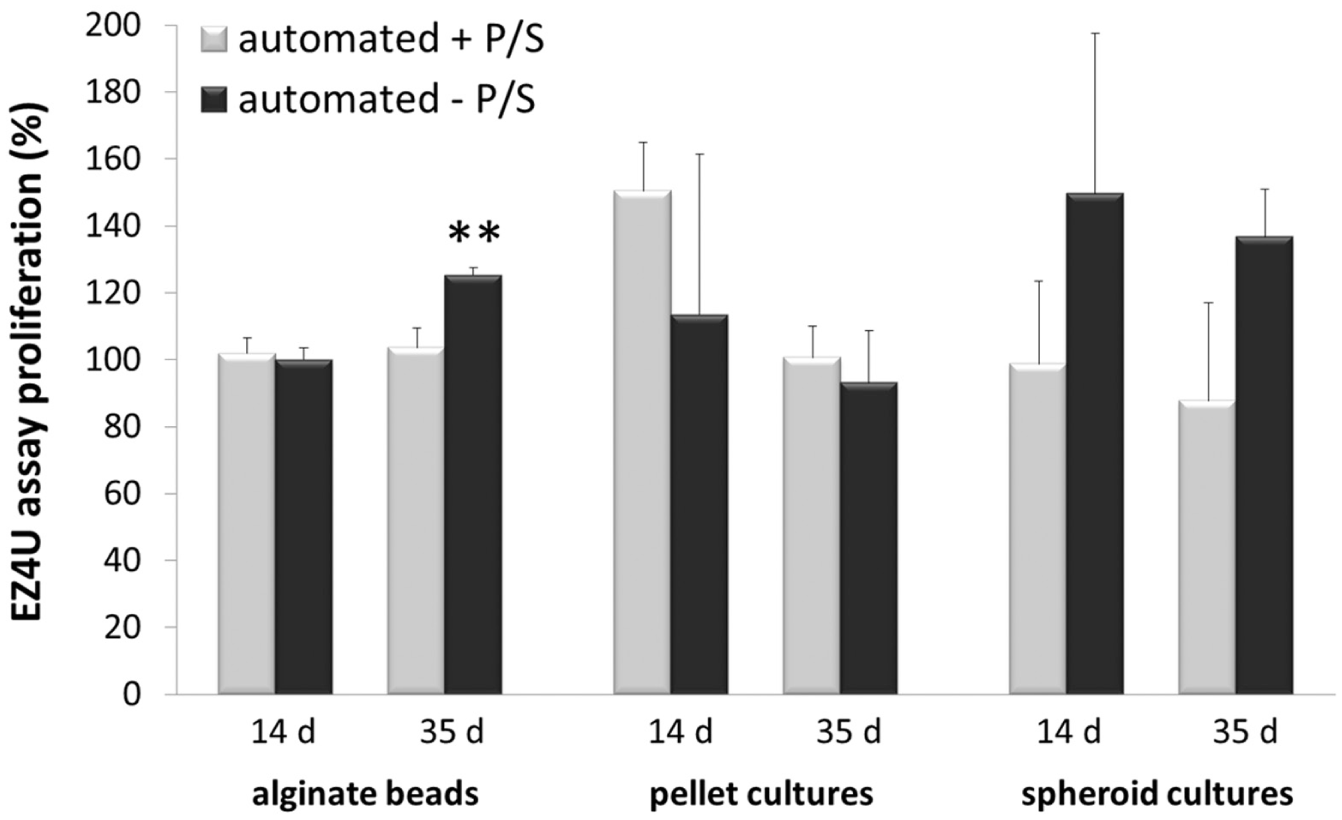
The percentage different proliferations of automatically produced 3D cell cultures (±P/S) detected by the EZ4U assay on days 14 and 35.
The reaction is based on the reduction of tetrazolium salt to colored formazan, with consequent detection of the absorbance using the EZ4U assay (Biomedica). The manually produced 3D cultures (± antibiotic) were set as controls at 100% on the specific detection dates. Figure 5 visualizes the different proliferations of automatically produced alginate beads, spheroid cultures, and pellet cultures in the specific medium (±P/S) compared to the manually performed 3D cultures (controls).
The proliferations of manually and automatically fashioned beads were mainly similar (day 14, ±P/S; day 35, +P/S). However, on day 35 the proliferation of automatically produced cells encapsulated in alginate (–P/S) was significantly (
At both detection times the proliferations of automatically performed spheroids cultivated in media with antibiotics (+P/S) were slightly decreased by about 6.8% (±P/S) compared to manually produced spheroids (controls). However, spheroids in media without antibiotics (–P/S) were increased by about 43.3% against the controls. Regarding this, antibiotics cause a minor reduced proliferation at automatically processed spheroid cultures.
The proliferations of automatically manufactured pellet cultures were mainly increased compared to the control by about 50.5% (day 14, +P/S), 13.4% (day 14, –P/S), and similar (day 35, +P/S). This reflects the more careful cell processing using the automated system. Instead, the proliferation of pellet cultures (automated, –P/S) was decreased by about 7% on day 35.
Evaluation of the Toxicity
The cytotoxicity was detected by the adenylate kinase kit (PromoKine, Heidelberg, Germany). The ubiquitous adenylate kinase (AK) is released by damaged cells and measurable in the supernatant.
The manually shaped 3D cultures (± antibiotic) were set as controls at 100% on the detection dates (days 14 and 35). Figure 6 visualizes the different proliferations of automatically produced alginate beads, spheroid cultures, and pellet cultures cultivated in the specific medium (±P/S) compared to the manually performed 3D cultures (controls).
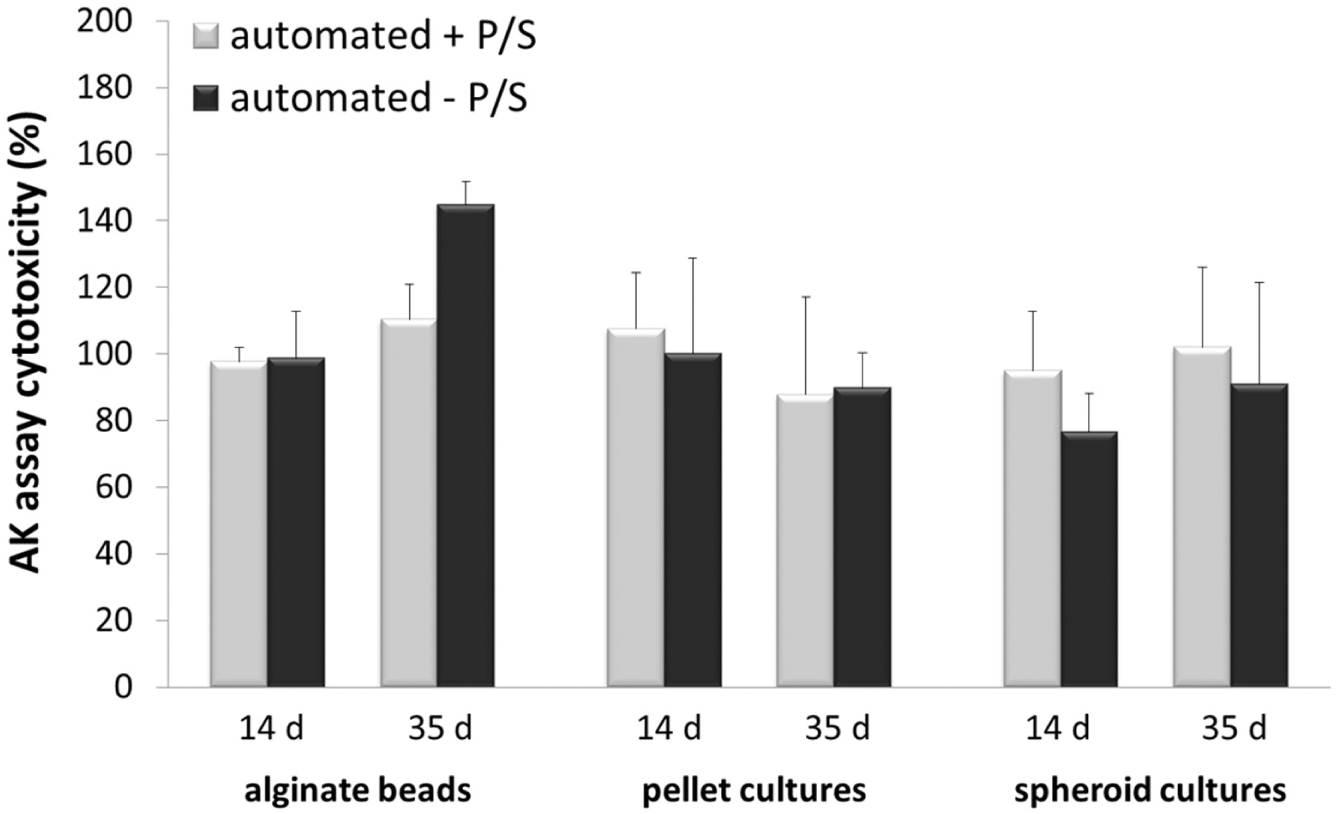
The percentage different cytotoxicities of automatically produced 3D cell cultures (±P/S) detected by the adenylate kinase assay on days 14 and 35.
The percentage cytotoxicity of automatically produced alginate beads was reduced about 1.9% (±P/S) on day 14 in contrast to day 35, with increased toxicities of about 10.4% (+P/S) and 44.9% (–P/S) associated with the controls (100%).
The automatically manufactured spheroid cultures showed mainly decreased cytotoxicities (percentage) of about 5.1% (day 14, +P/S), 23.5% (day 14, –P/S), and 9% (day 35, –P/S) compared to spheroids (day 35) in media without antibiotics (–P/S), with a higher toxicity of 2% compared to the manually produced cultures (controls, 100%).
On day 14, the percentage cytotoxicity of automatically shaped pellet cultures was increased by about 7.5% (+P/S) and similar (–P/S) compared to the manually performed pellets (control, 100%). However, the cytotoxicity of pellets (automated produced) was decreased about 11.3% (±P/S) on day 35.
The mostly reduced toxicity of the automatically processed 3D cell cultures displays a more gentle cell handling using the Biomek Cell Workstation. Thus, the manual 3D formation might cause more shear stress associated with a higher cytotoxicity.
The automation of 3D cell cultivation is a benefit for tissue engineering and compound screening. Within a short time, a similar tissue construct can be generated for the investigation of new drugs or the defect repair. Nevertheless, the manual 3D formation needed a high skill level and a lot of practice compared to the automated systems. Even after training processes, the manufacturing results might vary greatly between different technicians regarding implementation and handling. Instead, the Biomek Cell Workstation with its specific programs enables automated cell cultivation and 3D formation with similar procedure runs for the whole test series, independent of skill level. After a short training period, all technicians should be in the position to produce the 3D formation automatically. Furthermore, during the automated manufacturing processes technicians can implement other research work.
In view of the performance, the working/operation time of the manual processes is about 6 h. Regularly, a workday of laboratory personnel is 8 h. The loss of 2 h is calculated because of breaks, lack of concentration, and other operations. The Biomek Cell Workstation can be operated for up to 24 h/day, including 1 h for solution exchange and possible maintenance/cleaning steps. The production processes are separated in cell detachment methods and 3D formations within 96-well plates. In detail, seven-well plates with alginate beads can be produced automatically and six plates can be manufactured in the manual process. The projected spheroid cultures per workday are 14 plates via the automatic process and 8 plates via the manual one. The automatically manufactured pellet cultures are three-well plates during a workday, compared to two-well plates for the manual process.
Regarding this, the automated 3D formations increase the number of produced 3D cultures per day. 31 Cleaning steps during the automated production are required to guarantee sterile conditions and prevent cross-contamination. Furthermore, the removing and resuspending of solutions has to be implemented more slowly for a low shear stress during liquid handling steps by the Biomek NX liquid handler and steel cannulas.
The proposed automation system is useful for laboratories, which are constantly faced with a high amount of cell culturing processes. The return on investment for typical laboratories in Europe, North America, or Japan is approximately 2 years. In addition, industry in Germany and other European countries is challenged by an increasing deficit of specifically skilled scientists and lab personnel. Also, here the system can be a useful addition for laboratories.
Conclusion
In this work we compared the unique fully automated production (cell detachment and formation) of different 3D cell culture constructs (alginate beads, spheroid cultures, pellet cultures) by the Biomek Cell Workstation with the traditional manual processes. Changes on the liquid handler deck (Static Peltier Device, Positive Position ALP) and the adjustable programming of cell cultivation processes enabled the flexible manufacturing of alginate beads and spheroid cultures with cervix carcinoma cells, which can be used for compound screenings as well as tumor research. Furthermore, the automated manufacturing of pellet cultures containing human chondrocytes can support biomaterial research or tissue engineering. Consequently, through a high-throughput screening system, bioscreening methods can be adapted to the specific applications and investigations. The manual procedures can be replaced by the automated techniques mainly because of the decreased cytotoxicity and amplified proliferation.
Footnotes
Acknowledgements
We thank the Federal Ministry of Education and Research (BMBF Germany) for the financial support (FKZ: 03ZIK021, 03ZIK022, 03Z1KN11). Furthermore, we thank Mrs. Grit Koch (University of Rostock) for the excellent technical assistance.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: We thank the Federal Ministry of Education and Research (BMBF Germany) for the financial support (FKZ: 03ZIK021, 03ZIK022, 03Z1KN11).
